Abstract
Abstract
Introduction
The primary aim of this study was to evaluate the safety of performing, in an office, the NovaSure endometrial ablation procedure after Essure micro-inserts had been placed, and to determine if the effectiveness of either procedure was altered by this treatment sequence.
Materials and Methods
Institutional Review Board (IRB) approval was obtained prior to study initiation. Although this was a retrospective study, the authors desired an IRB board to review study collection methods for validity and good clinical practice. Data for this retrospective study were obtained from a single physician in a private obstetric and gynecologic practice in southern Indiana from July 1, 2008 to December 31, 2009. Medical charts of women who had previously undergone planned Essure followed by NovaSure procedures in two separate in-office sessions were reviewed.
All patients had menorrhagia and desired permanent sterilization. Because this treatment sequence is outside of current labeling* informed consent was obtained for each procedure and included a discussion of NovaSure use after Essure placement. If a patient elected to proceed with the NovaSure ablation prior to the 3-month Essure confirmation hysterosalpingogram (HSG), additional counseling was provided and informed consent was obtained regarding the possibility of an inadequate Essure confirmation. 1 All patients were scheduled for a 3-month Essure confirmation HSG after placement of the Essure micro-inserts regardless of ablation timing. The interval between the Essure and subsequent NovaSure procedure was typically scheduled to be 14 days apart. Adjustments to the time interval were made based on patient request or insurance considerations, or delayed until a diagnosis of menorrhagia was noted with the cessation of hormonal medications after the Essure HSG confirmation test.
Patients received oral misoprostol, ondansetron, diazapem, oxycodone, 60 mg intramuscular (IM) or 30 mg intravenous (IV) ketorolac, and lidocaine paracervical blocks prior to each procedure. Patients were offered IV midazolam and/or fentanyl prior to both the Essure and NovaSure procedures to achieve mild conscious sedation.
During the Essure procedure, an attempt was made to leave 1–2 trailing coils from each ostium after micro-insert deployment. If the Essure procedure was difficult, patients were required to undergo a 3-month HSG confirming satisfactory occlusion prior to proceeding with NovaSure ablation. Transvaginal ultrasound was performed on all patients prior to the NovaSure ablation procedure, and, when ultrasound was obtained after the placement of Essure micro-inserts, confirmation of appropriate placement and retention was documented.
Follow-up telephone contact and/or an office visit was attempted for all patients 2–7 days following each in-office procedure to assess for any adverse events. In addition, a follow-up outcome questionnaire was administered, via telephone or direct patient contact, 3–24 months after completion of both procedures, to assess for patient satisfaction and perceived effectiveness with each procedure. Patients were asked to rate their satisfaction with the Essure and NovaSure procedures on a scale of 1 (very dissatisfied) to 5 (very satisfied). Patients were also asked to: (1) state if they were relying on Essure; and (2) quantify their menorrhagia symptoms pre- and post-NovaSure by describing the number of tampons or pads soaked during a menstrual cycle before and after the procedure.
Patient inclusion was determined from a procedure log maintained by nursing staff. The electronic medical records of all eligible patients were reviewed by a nurse–practitioner and a physician. Data collection included demographic information, anesthesia type, bilateral placement success, ablation treatment time, and adverse events as assessed by telephone and/or direct patient contact and questionnaire results.
The trial was conducted in accordance with good clinical practice guidelines. All data were monitored by QST Consultation, LTD (Allendale, MI) for validation.
Results
From July 2008 to December 2009, 117 patients were identified as having undergone planned Essure micro-insert placement followed by NovaSure endometrial ablation in the office. The average age of subjects was 38 (range 24–52 years). The population was predominantly Caucasian (70%) with only 3% documented as being of African-American or Hispanic ethnicity, and with 20% being of no stated ethnicity. In addition, private insurance coverage was noted for 99% (116/117) of the patients in this study.
The median interval between the two procedures was 14 days (range 0–236). Conscious sedation was offered to all patients prior to each procedure. During the Essure procedure, 70% (82/117) of patients requested IV midazolam and/or fentanyl, whereas 99% (116/117) of patients undergoing NovaSure requested IV medications. One patient elected to have general anesthesia for the NovaSure procedure after experiencing a vasovagal reaction during the in-office Essure procedure. Another patient experienced a vasovagal reaction in the office with introduction of the hysteroscope through the cervix, resulting in cessation of the Essure procedure. On the same day, she elected to undergo general anesthesia for the Essure followed by the NovaSure procedure in a single session in the operating room.
Bilateral placement on first attempt was achieved in 114/117 (97.4%) of subjects, and intentional unilateral placement on first attempt was achieved in 3/117 (2.6%). Two patients received intentional unilateral placements because of proven salpingectomy status, and 1 was noted to have a unicornuate uterus at the time of the procedure. This was also demonstrated by HSG, completed the day of procedure, and later confirmed with magnetic resonance imaging (MRI). The median Essure procedure time (scope in–scope out) was 2 minutes (range 1–30 minutes), and an average of two trailing coils was observed (range 0–5). NovaSure treatment time averaged 99 seconds.
All patients were scheduled for a 3-month post-Essure HSG confirmation test and 83/117 (71%) patients returned for testing. Of the 83 patients who returned for HSG testing, 71 underwent HSG after completion of the NovaSure procedure, whereas 12 completed the HSG test prior to completion of the NovaSure procedure. In the patient group who underwent confirmation HSG after completion of the NovaSure procedure, the findings were satisfactory placement and bilateral occlusion in 67/71(94%) patients. In the remaining four (4/71) patients, 2 had satisfactory placement and unilateral patency, 1 patient requested cessation of the HSG test because of pain, but satisfactory location was confirmed, and 1 patient was noted to have satisfactory placement of Essure inserts but had intrauterine synechiae preventing an adequate HSG confirmation test. In the patient group that underwent HSG prior to the NovaSure ablation procedure, 12/12 patients were noted to have satisfactory placement and bilateral occlusion. A total of 81/83(98%) confirmation HSGs at 3 months were successfully completed in this study (Fig. 1). Both of the patients with tubal patency diagnosed at 3 months were unable to obtain a successful follow-up HSG confirmation because of subsequent intrauterine synechiae preventing an adequate study, although appropriate retention and projection of Essure inserts were noted during the examination. After discussion of the results with a physician, all 4 patients with unsuccessful 3- or 6-month HSG confirmation examinations elected to rely on the Essure inserts for contraception with no reported pregnancies in this cohort.
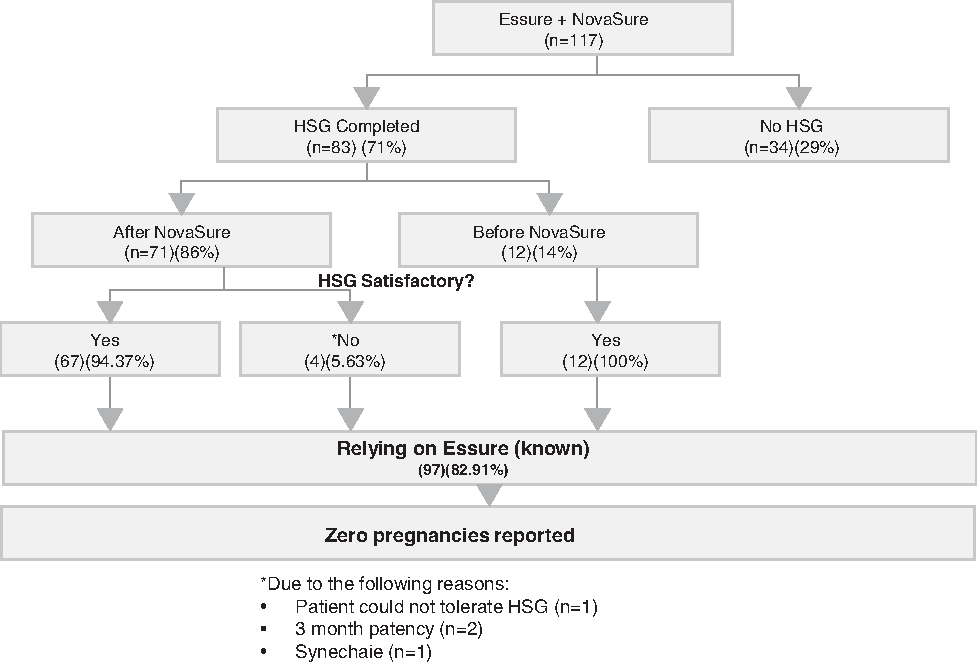
Hysterosalpingogram (HSG) follow-up and results. “No” response to “HSG Satisfactory?” was for the following reasons: patient could not tolerate HSG (n=1); 3-month patency (n=2); and synechaie (n=1).
Outcome questionnaire results for both the Essure and NovaSure procedures are presented in Table 1. A total of 83% (97/117) of subjects completed the outcome questionnaire. With respect to the Essure procedure, 92% (89/97) reported being “very satisfied” and 8% (8/97) reported being “satisfied,” and no patient reported being “dissatisfied.” All patients said that they would recommend the Essure procedure to other women. All 97 patients reported that they were relying on the Essure inserts for contraception from 6 to 24 months with a total of 754 months of reliance and no reported pregnancies occurring in this cohort. With respect to the NovaSure procedure, 86% (83/97) reported being “very satisfied,” 11% (11/97) reported being “satisfied,” 1% (1/97) reported being “dissatisfied,” 1% (1/97) reported being “very dissatisfied,” and 1% (1/97) had no opinion. All but 1 patient said they would recommend the procedure to others. In this study after NovaSure ablation, outcome data were obtained for 97 women, with an average follow-up of 9.4 months (Fig. 2). Fifty-nine of 97 (61%) reported “no bleeding,” 13/97 (13%) reported “occasional spotting,” 22/97 (23%) reported a decrease in menstrual flow, and 3/97 (3%) reported no bleeding reduction. In addition, 2/97 (2%) underwent hysterectomy because of persistent pelvic pain, and 1/97 (1%) required treatment for hematometra (Fig. 3).
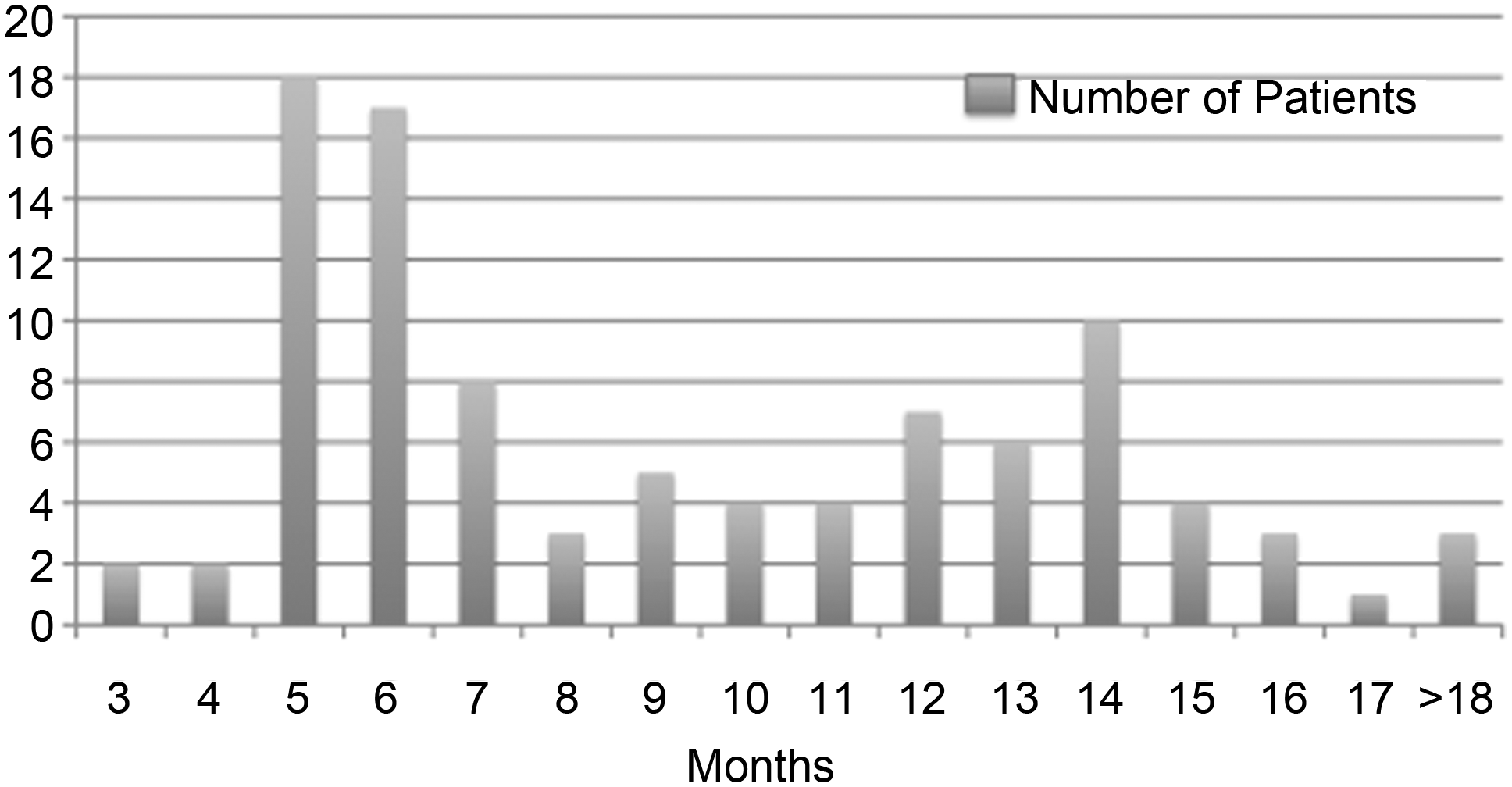
Number of patients per months of follow-up after NovaSure.®
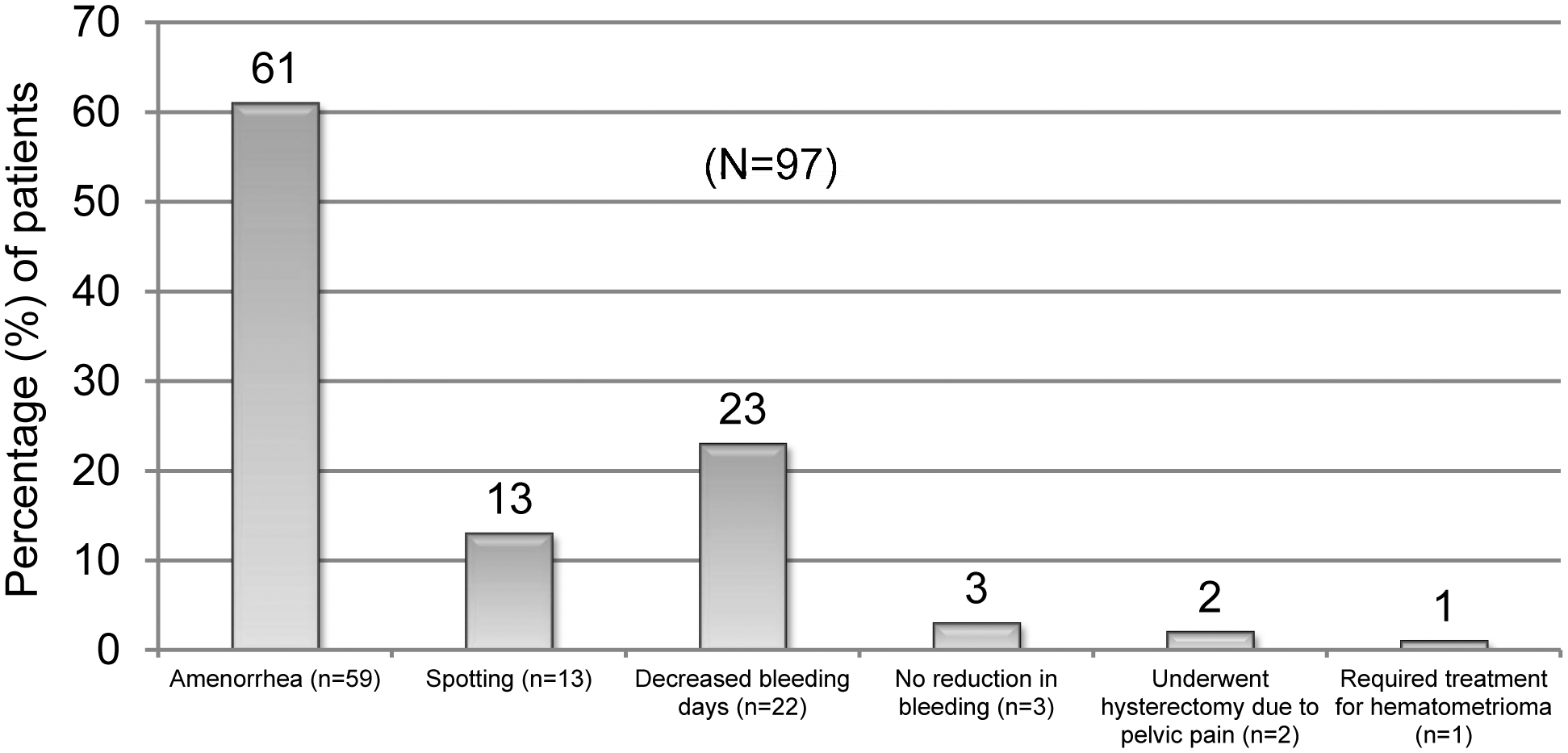
NovaSure® effectiveness.
Adverse event data, collected by telephone and/or direct patient contact, was obtained on 100% (117/117) of patients after the Essure procedure, and on 95% (111/117) of patients after the NovaSure procedure; 6 patients were lost to follow-up after completion of both procedures. In a total of 228 procedures for which follow-up could be obtained, 8 adverse events were noted for an overall adverse event rate of 4% in this cohort (Fig. 4). Two patients underwent hysterectomy for continuing chronic pelvic pain and both patients were diagnosed with endometriosis at the time of surgery. Two vasovagal events occurred, one during hysteroscope insertion and the other during an Essure procedure. Both patients elected to complete one or both procedures in the operating room under general anesthesia. One patient experienced severe dysmenorrhea 10 months after her NovaSure ablation and was diagnosed with a hematometra. She experienced resolution of her dysmenorrhea after treatment with hysteroscopic resection of intrauterine synechiae in the office under ultrasound guidance. One luteal phase pregnancy was diagnosed despite a negative urine pregnancy test at time of Essure placement but prior to the NovaSure procedure. This patient elected to terminate the pregnancy and subsequently presented for the NovaSure procedure. One patient complained of pelvic pain for 1 week after the Essure procedure, but the pain resolved spontaneously without further treatment. One patient who underwent combined Essure and NovaSure in the operating room was hospitalized with a severe headache, nausea, vomiting, and dizziness within 1 day of her procedures. Her symptoms resolved within 24 hours and she was discharged to go home with no further complications.
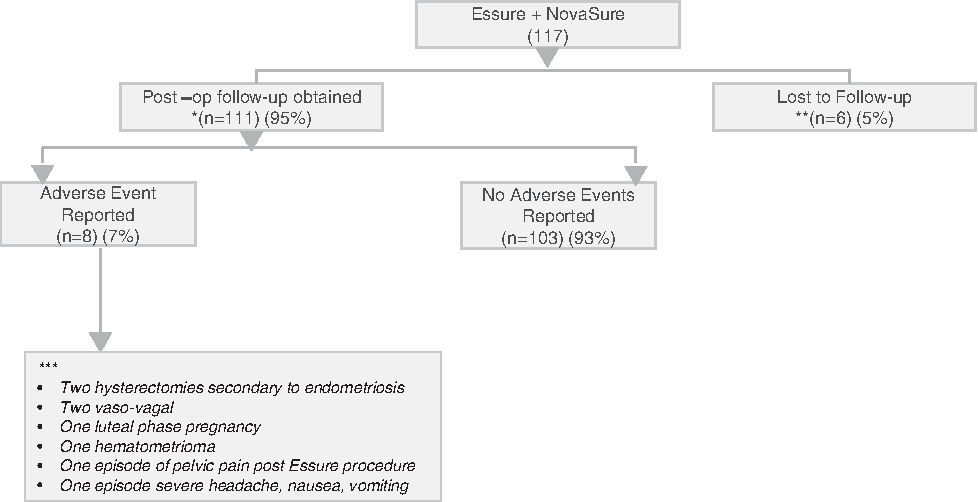
Adverse events data. *Fourteen patients had telephone contact 1 week post procedures; 97 completed an additional follow-up (outcome survey). **No patient contact following NovaSure® procedure. ***No hospitalizations secondary to thermal injury to contiguous pelvic organs.
Discussion
In this cohort, no observed adverse events occurred with respect to hospitalization for injury or thermal damage to contiguous pelvic organs. In addition, there were no reported pregnancies, micro-insert perforations, or expulsions/displacements caused by adherence to the NovaSure mesh, nor was there any malfunctioning of the NovaSure equipment as a result of the Essure devices.
Despite these findings, several concerns remain when considering the performance of a NovaSure procedure when Essure micro-inserts have been previously placed in a patient:
(1) Could radiofrequency energy and/or heat transfer along the Essure insert increase the likelihood of surrounding bowel injury over use of NovaSure ablation alone? (2) Will the Essure inserts cause malfunction of the NovaSure device? (3) What is the probability that a properly placed Essure insert will become dislodged or damaged because of adherence to the NovaSure mesh after completion of the ablation procedure? (4) Will the sequence of Essure followed by NovaSure procedure decrease the effectiveness of either procedure?
When considering heat transfer along the Essure micro-insert during the NovaSure procedure, research evaluating safety and feasibility of these combined procedures demonstrates that heat transfer to the Essure coils during the NovaSure procedure does not differ from that seen with other ablation technologies such as Thermachoice or HydroThermal Ablation (HTA), which are currently approved for use after Essure placement by the United States Food and Drug Administration (FDA).7–9 Garza et al. presented data regarding the use of NovaSure ablation after placement of Essure micro-inserts at the 2008 American Association of Gynecologic Laparoscopists (AAGL) meeting. 9 In this report, 13 women scheduled to undergo total abdominal hysterectomy had insertion of the Essure micro-insert in one fallopian tube using the contralateral tube as a control. The NovaSure procedure was then performed and thermal imaging was used to monitor serosal temperatures throughout the procedure. Specimens were then analyzed for thermal injuries to all tubes. During the NovaSure treatment cycle, mean serosal temperatures did not exceed 40°C and were similar between the Essure implanted and control tubes. The micro-insert did not significantly alter the maximum thermal injury depth (implanted 6.3±1.7mm and non-implanted 6.3±1.1mm, p=0.989). No primary serosal thermal injury emanating from the micro-inserts was identified. The micro-inserts were also found not to alter the primary NovaSure ablation to the intrauterine cavity. Interestingly, Garza et al. and Valle et al. evaluated both Thermachoice and HTA in a similar fashion,8,10 technologies currently approved by the FDA for use after placement of the Essure micro-inserts and satisfactory confirmation HSG. 1 They found that serosal temperatures remained<45°C during the Thermachoice and HTA procedures, and that no thermal injury was observed in fallopian tubes implanted with Essure micro inserts.8,10 The thermal distribution and heat injury down the fallopian tube with an Essure micro-insert in situ appear to be similar among the Thermachoice, HTA, and NovaSure procedures and between implanted and non-implanted tubes.
Second, no malfunctioning of the NovaSure device was detected during energy application in this study. No reports appear in the Manufacturer and User Facility Device Experience (MAUDE) database reporting this event, and no peer-reviewed or non-peer-reviewed presentations could be found, through a Pub-Med search in February of 2011, reporting malfunction of the NovaSure device caused by an Essure micro-insert. However, further bench studies may be warranted to definitively address this concern.
Third, during this study no adverse events related to dislodgement or damage to a previously placed Essure micro-insert were observed. It is important to note that in this study, the physician attempted to place the Essure micro-inserts with only 1–2 trailing coils, and no more than 5 trailing coils were observed in this cohort. This would presumably decrease the likelihood of direct contact of a micro-insert to the NovaSure mesh, thus decreasing the probability that an insert would become displaced. However, Saunders reported his experience in a non-peer-reviewed report with 118 patients who had undergone the NovaSure procedure after Essure micro-inserts had been placed. 11 In Saunders' report, 2 patients experienced adverse events with removal of an Essure micro-insert after the insert had become adherent to endometrial tissue attached to the NovaSure mesh. Both patients underwent second procedures to replace the removed micro-insert with success and with no reported pregnancies. 11 Based on this information, use of an alternative ablation technology to NovaSure may be prudent in patients with more than 5 trailing coils seen on pre-ablation hysteroscopy.
Finally, the performance of a NovaSure ablation when Essure micro-inserts are present did not appear to alter the effectiveness of either procedure in this study. Outcomes for NovaSure ablation in this study population were consistent with labeling and outcomes reported for this technology by other investigators.12–14 Additionally, no known Essure failures/pregnancies have been reported by patients in this study and 96% of 3-month confirmation HSGs could be successfully obtained with no unsatisfactory device placements.
In this study, the performance of NovaSure after Essure does not appear to impede proper functioning of either device, increase risk for bowel injury, or decrease the effectiveness of either procedure. However, a significant effort was made to insure that the micro-inserts were properly positioned within the tubal lumen prior to proceeding with NovaSure. If transvaginal ultrasound did not demonstrate satisfactory micro-insert retention and location, patients were instructed to wait until they had completed a satisfactory 3-month HSG prior to ablation. It is unclear whether a micro-insert that is perforated through the tubal lumen into the abdominal cavity would pose a risk for bowel injury. This circumstance was not encountered in this cohort, and further studies are warranted to evaluate the risk in the case of micro-insert perforation. Therefore, if tubal perforation is detected or suspected, the safe use of NovaSure could not be determined. Therefore, caution should be exercised when considering NovaSure ablation in these patients.
Limitations of this study include retrospective study design, single investigator, homogeneity of the population, and lack of pathologic or histologic studies to confirm clinical findings. However, the sample size was relatively large, with a small number of patients lost to follow-up.
In this retrospective medical chart review, Essure hysteroscopic sterilization followed by NovaSure in women who desire permanent birth control and reduction of menorrhagia was found to be safe, effective, and well tolerated. Patients reported high rates of satisfaction with both procedures. Although this study supports the safe use of NovaSure after Essure micro-insert placement, further research is necessary.
Footnotes
Acknowledgments
The authors thank Conceptus, Inc. for providing funding for the IRB process and data analysis for this study. The authors also thank Jeffery Russell, and Mark Levie for assistance in editing this manuscript.
All authors have given written permission to be named.
Disclosure Statement
Cindy Basinski received no financial compensation for any aspect of this study. She has served as a paid consultant for the Essure product manufactured by Conceptus, Inc. and the NovaSure product manufactured by Hologic, Inc. Jamie Burkhart and Jamie Johnson have no disclosures.
*
During the first year of patient procedures, use of NovaSure after Essure placement was labeled under warnings in the Essure instructions for use (IFU): Sometime in the year 2009, the U.S. Food and Drug Administration (FDA) changed labeling for use of NovaSure after Essure placement to a contraindication in the NovaSure IFU but a warning in the Essure IFU. Since that time, Hologic and Conceptus have introduced data successfully to the FDA to warrant a reversal of the label change. This change occurred in early March. The use of NovaSure after Essure placement is again included in the warning sections of both NovaSure and Essure IFUs and is no longer included in the contraindication section of either IFU.
