Abstract
Objective:
We designed a prospective experiment to explore the whole promoter region of fibrinogen B β (FGB) with the intent to detect all single nucleotide polymorphisms (SNPs) in this region and their impact on the susceptibility of aseptic lower extremity superficial thrombophlebitis (STP).
Methods:
The experiment was conducted prospectively and randomized. Thirty-four idiopathic STP patients and 50 healthy people were recruited, respectively, as case and control groups. The whole FGB promoter was gene sequenced.
Results:
From upstream to downstream of the promoter, a totally 6 SNPs—rs1800789 (−1420G/A), rs2227389 (−993C/T), rs1800791 (−854G/A), rs1800790 (−455G/A), rs1800788 (−249C/T), and rs1800787 (−148C/T) were detected via gene sequencing. A statistical difference was found in the −455A allele (rs1800790) between the two groups (p < 0.05, odds ratio = 0.368, 95% confidence interval: 0.093 − 0.852), while no statistical difference as for the other five SNPs (all p > 0.05).
Conclusions:
The 455A allele may be a protective locus for the onset of STP. However, due to the low incidence of idiopathic STP, multiple verification tests are still needed.
Introduction
Superficial thrombophlebitis (STP) is characterized by the formation of a thrombus in superficial veins, inducing occlusion and inflammation along the veins (Di Nisio et al., 2018; Nasr and Scriven, 2015). For a long time, STP has been considered a self-limiting and benign disease (Czysz and Higbee, 2025). However, it can progress to deep venous thrombosis (DVT) and pulmonary embolism (PE) (Di Minno et al., 2016). The causes of STP are related to factors such as intimal injury, varicose veins, and infections. Some cases might be associated with tumors (Mouton et al., 2009), such as migratory STP and chest wall STP. Sometimes in the early stages of malignant disease, STP is the only symptom (Sobreira et al., 2008). Some patients experience STP due to abnormal coagulation function (Tait et al., 2012), for example, lacking antithrombin III, abnormal protein S and protein C, or increased fibrinogen concentration. Fibrinogen is mainly synthesized by hepatocytes and acts as the core protein in the coagulation system. Following activation of coagulation, thrombin proteolytically cleaves fibrinogen to form fibrin. Some studies have shown that an increase in plasma fibrinogen will lead to an increase in the incidence of cardiovascular diseases and thrombotic events. Fibrinogen is a 340 kDa glycoprotein, which is encoded by three structural genes (FGA, fibrinogen B β [FGB], and FGG) (Mosesson, 2005). Previous studies indicated that synthesis of β chain may be a rate-limiting factor in the production of human fibrinogen (Dobson et al., 2024; Roy et al., 1990), thereby affecting plasma fibrinogen levels and thrombotic events. We had made this assumption more than once that polymorphisms in FGB might influence the transcription of FGB and the synthesis of the entire fibrinogen molecule, thereby influencing the onset of STP. Due to the fact that most of the discovered FGB polymorphisms are located in the promoter, we thus developed the idea of fully sequencing the whole promoter, once for all, to explore all SNPs (known or unknown) in this region.
Methods
Study design
From January 2022 to December 2022, we randomly enrolled 34 idiopathic aseptic lower extremity STP cases and 50 healthy people in the present study. All patients in the STP group were the first time of onset without any treatment before admission, including 18 males and 16 females, aged (55.10 ± 14.79) years old. The control group was composed of 50 healthy people (19 males and 31 females), aged (51.70 ± 10.81) years old. Ultrasonographic evaluation and blood testing data were collected in both groups. Meanwhile, residual blood samples from both groups were recollected for gene sequencing. This study was supervised under the Ethics Committee of the First Affiliated Hospital of Kunming Medical University (Approval number 2021L56). Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Inclusion criteria of the case group:
Diagnosis of aseptic lower extremity STP by duplex ultrasonography Age ranging from 18 to 80 years old Without heart, kidney, liver, thyroid diseases; without tumor, without rheumatoid immune disorders, or metabolic disorders Without trauma, oral contraceptive use, and pregnancy Without a COVID-19 infection within 6 months Excluding recent severe infections such as sepsis and systemic infection, procalcitonin detection is negative (< 0.05 ng/mL) to exclude severe infection
Inclusion criteria of the control group:
Healthy physical examination population without history of thrombotic events such as venous thromboembolism (VTE) and STP Age ranging from 18 to 80 years old Without heart, kidney, liver, thyroid diseases; without tumor, without rheumatoid immune disorders, or metabolic disorders Without oral contraceptive use and pregnancy Without a COVID-19 infection within 6 months Excluding recent severe infections
Gene sequencing
The promoter contains the core region and the regulatory region. The core region produces basal-level transcription, and the regulatory region can respond to different environmental conditions and make corresponding adjustments to gene expression levels. The promoter covers a wide range, which usually includes ∼2000 bp upstream and 200 bp downstream of the transcription start site. Some binding sites for transcription factors within the transcription regions also belong to the promoter range. Due to research limitations, we only focused on the core region. First, we entered the Genebank database of the National Biomedical Network in the United States to search for all sequences of human FGB, with the website http://www.ncbi.nlm.nih.gov/. The results showed a total of 8878 bp for the FGB gene. Meanwhile, there was much software for promoter prediction, but no one could accurately predict the precise range of it. After reviewing the literature, six SNPs and their adjacent fragment sequences in the promoter were identified. They were: −148C/T(rs1800787), −249C/T (rs1800788), −455G/A (rs1800790), −854G/A (rs1800791), −993C/T (rs2227389), and −1420G/A (rs1800789). The transcription start site, that is, the “+1” position, was then determined, using these six sites as reference. Following the principle of “2000 bp upstream and 200 bp downstream of the transcription start site” to contract the scope of investigation, a target sequence of ∼3001 bp was obtained. But this scope was still large for us.
After comprehensive prediction (including Promoter 2.0 software), it was confirmed in the end that the rough range of the FGB promoter was 1560 bp (−1525 to~+34). Primer sequences were designed and evaluated using Primer Premier 5.0 and Oligo6.0. After that, it was sent to University of California Santa Cruz (UCSC) (website: http://genome.ucsc.edu/) to confirm the length and accuracy of the amplification sequences. Primers were synthesized by the Shanghai Biotech company. The primers used in the Polymerase Chain Reaction (PCR) are shown as follows.
Forward: 5′-ACGTCACTAAAATAAAATCCTGCTA-3′
Reverse: 5′-GAAGCTCAAGAAACCATCC-3′
Peripheral blood samples or residual blood samples were recollected and stored at −80°C. GeneAmp® PCR System 9600 (Applied Biosystems, USA) was used for the polymerase chain reaction, which was set up to run the following program: 94°C for 3 min, 94°C for 40 sec, 58°C for 40 sec, and 72°C for 10 min, followed by 32 cycles of 72°C for 10 min, 15°C for 30 min. The 1560 bp PCR products were temporarily placed at 4°C for electrophoresis detection or −20°C for long-term preservation (Fig. 1). After that, the Sanger dideoxy chain termination method was used to complete gene sequencing from both positive and negative directions to explore the whole promoter (Figs. 2–7). Using the American Applied Biosystems PRISM-3730 sequencer, each sequencing direction could sequence ∼1000 bases. Therefore, the 1560 bp promoter could be fully covered. After the automatic sequencing of the sample, an “ab1” format file was generated. The SeqMan software in the DNAStar package was used to open the file, with positive and negative sequences being spliced together. The contaminated sequences were automatically or manually removed, and SNPs (including known and unknown) were read and recorded into SPSS 27.0 (SPSS Inc., Chicago, IL).
PCR products of FGB promoter (1560 bp). FGB, fibrinogen B β.
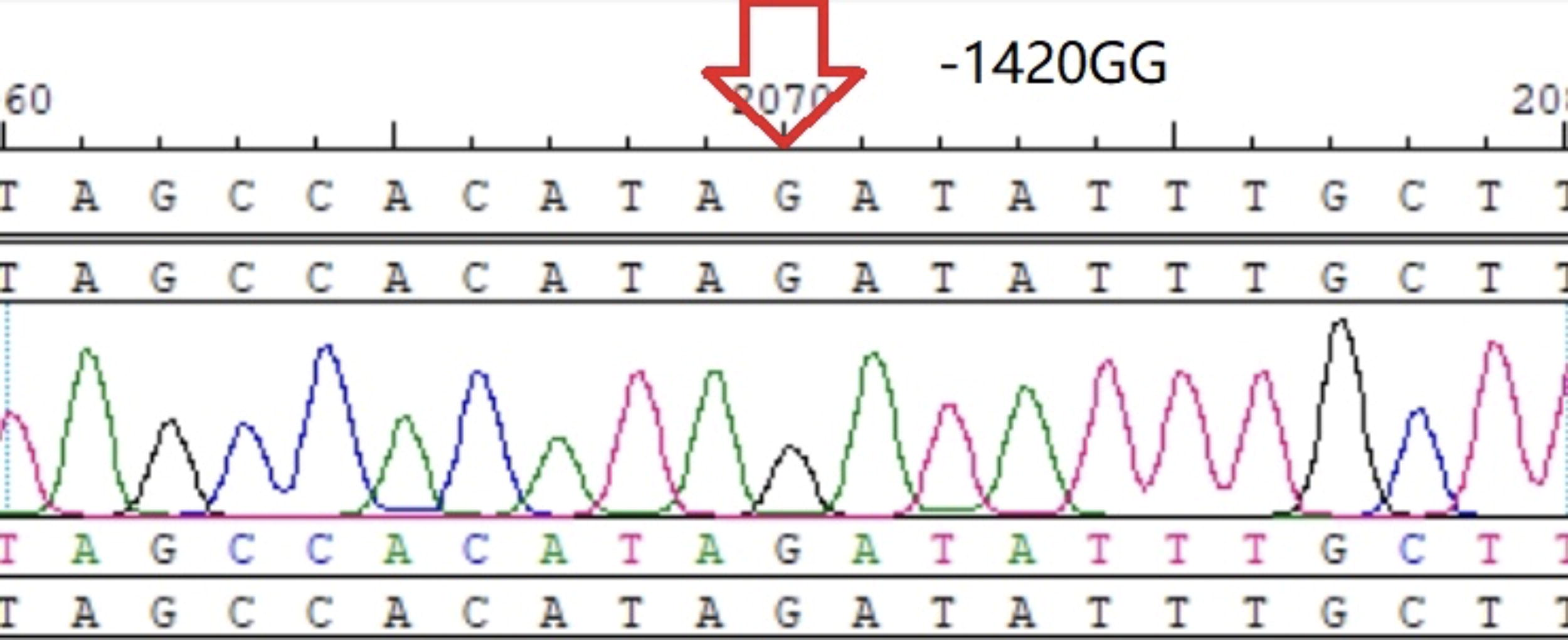
−1420GG homozygote.
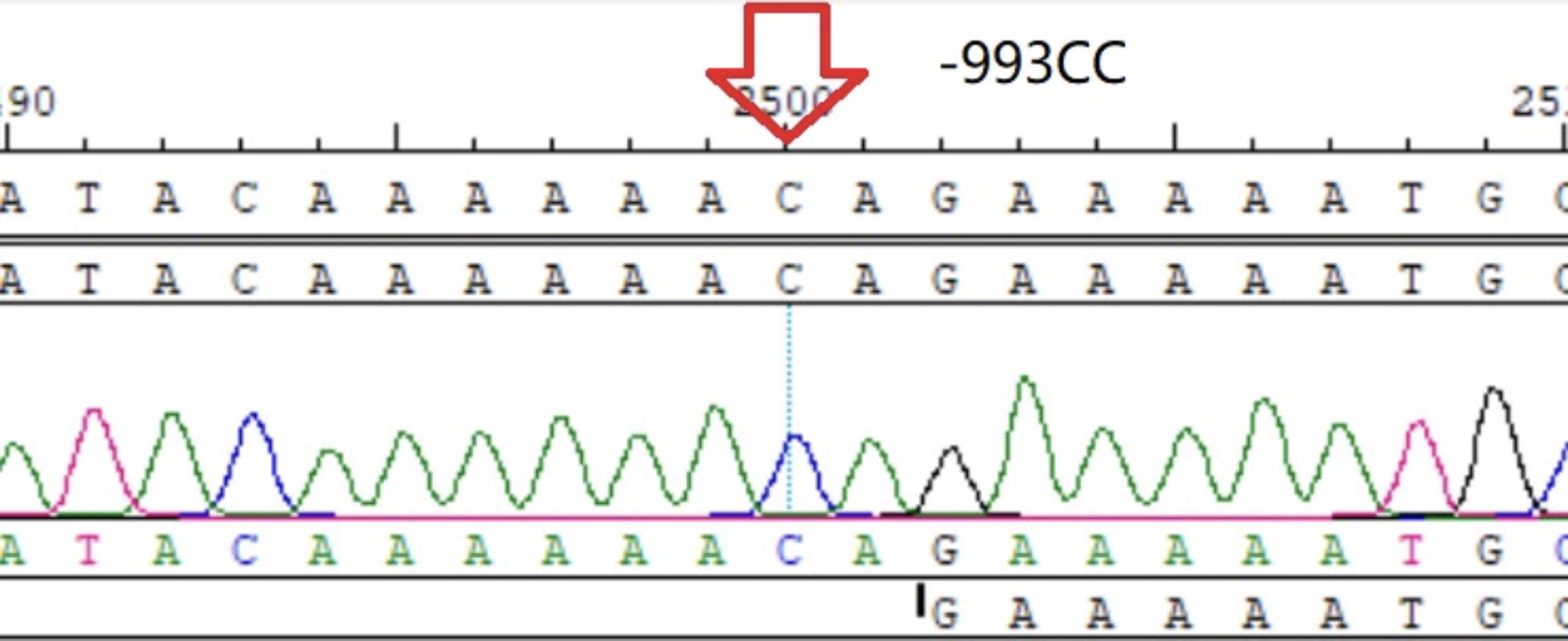
−993CC homozygote.
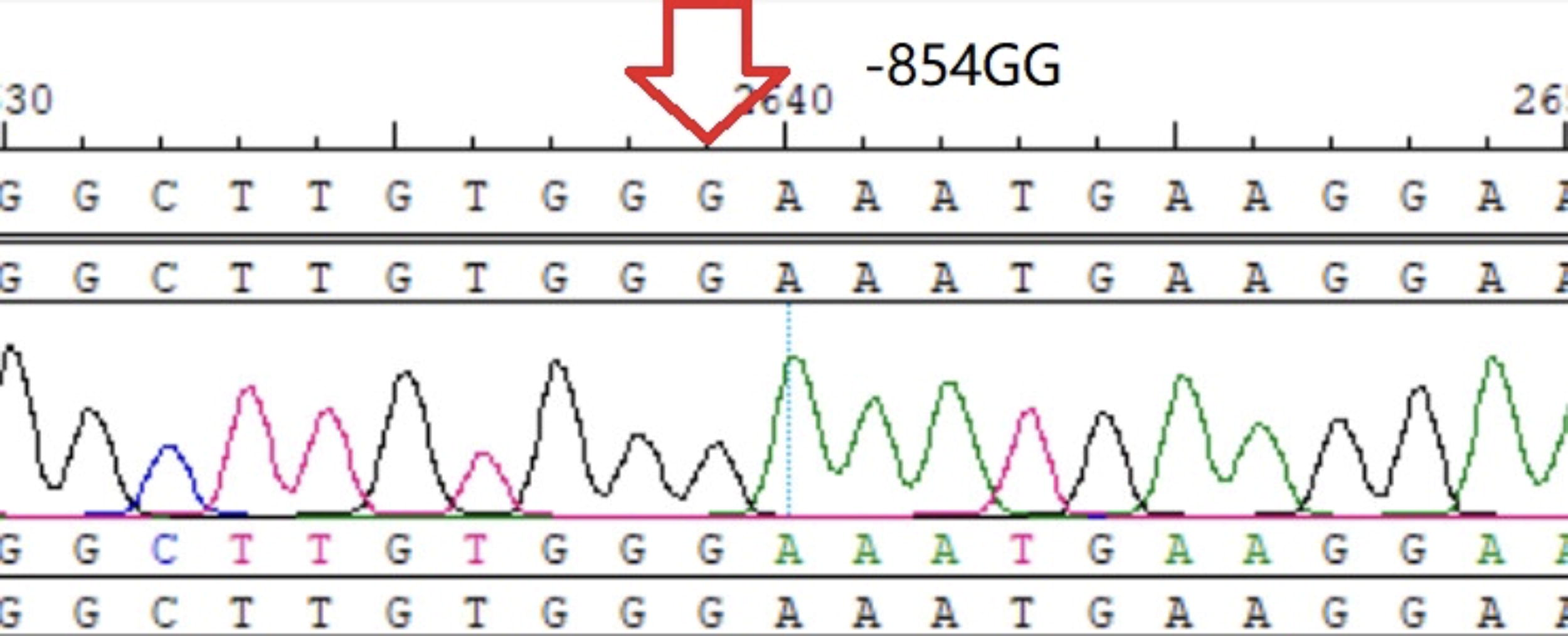
−854GG homozygote.
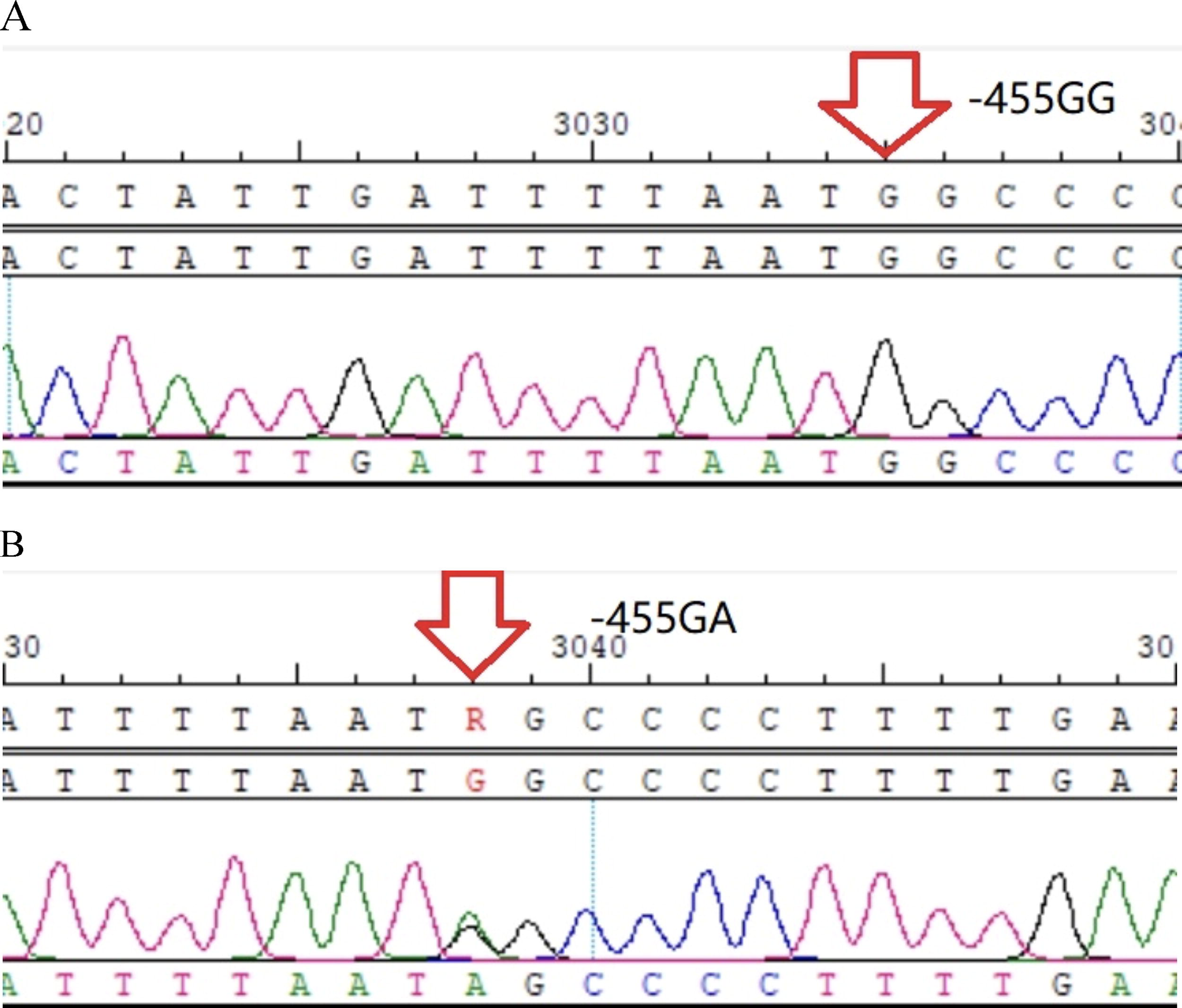
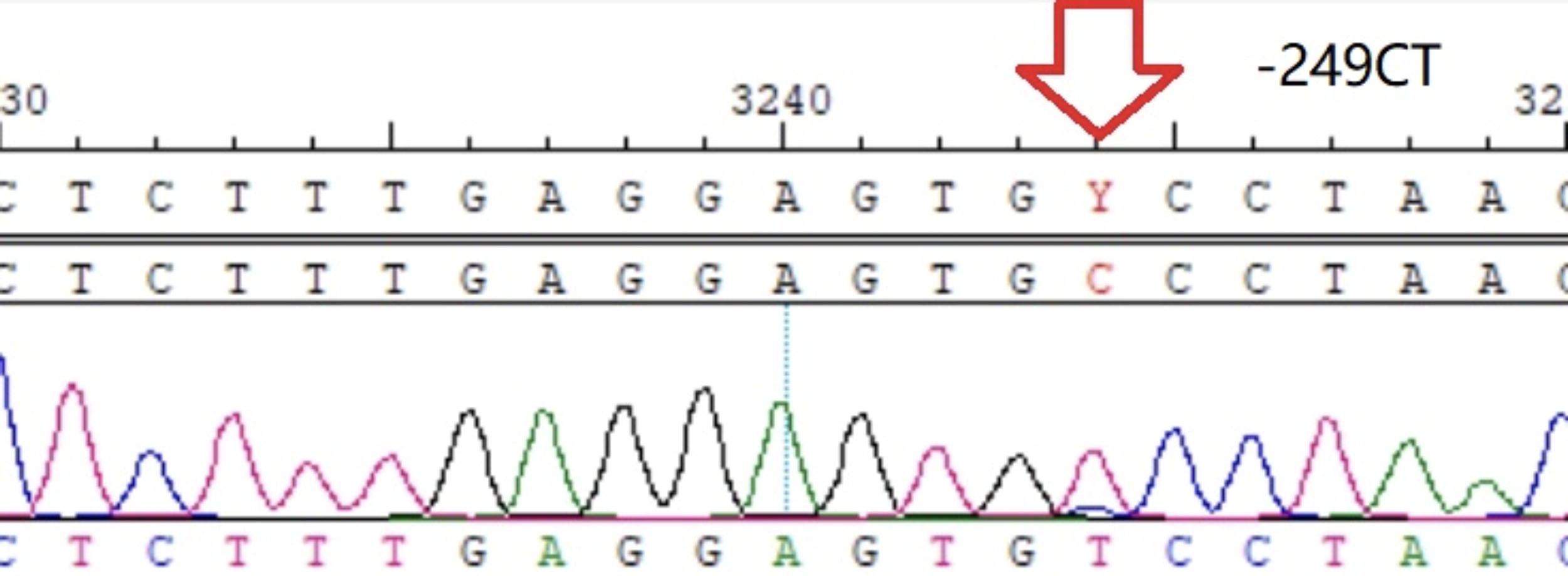
−249C/T heterozygote.
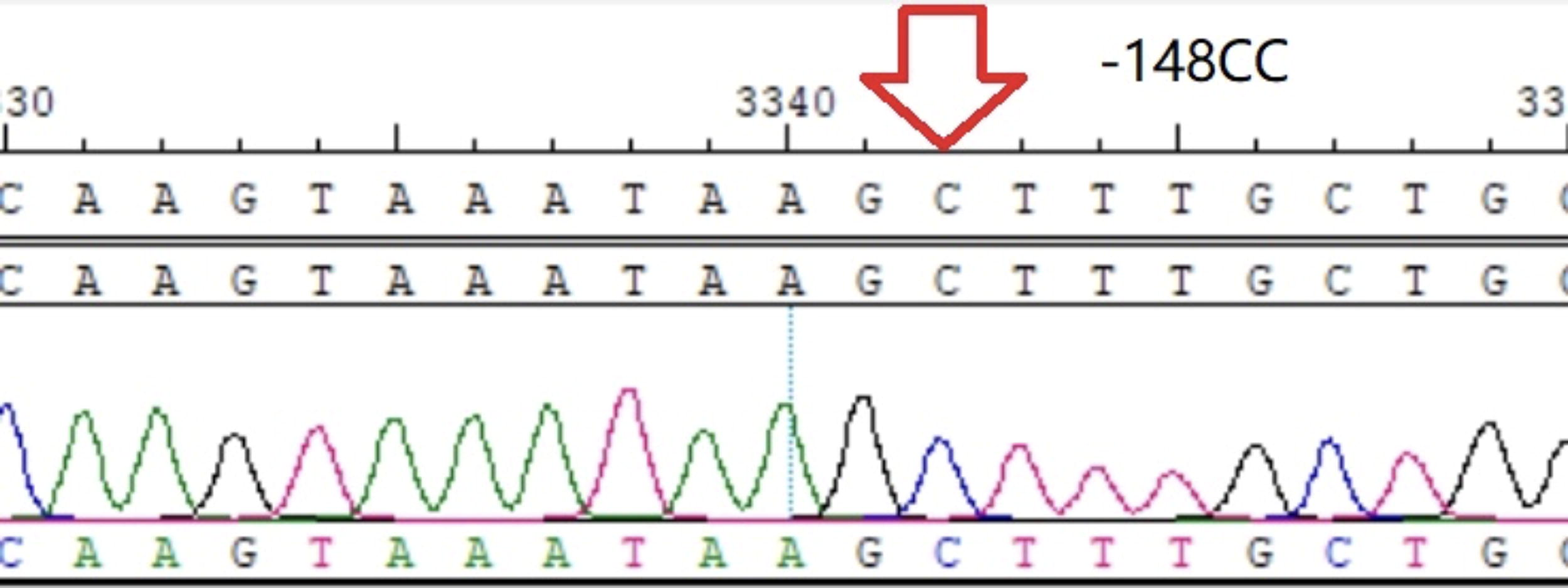
−148CC homozygote.
Statistical analysis
Wilcoxon rank-sum test or t test were used when necessary. Hardy–Weinberg equilibrium was performed to compare the gene balance. Odds ratio (OR) and 95% confidence interval (CI) were adopted to express the differences of genotypes and allele frequency. p < 0.05 indicated a statistical difference.
Results
General information
The monocyte count, fibrinogen degradation products (FDP), and D-dimer levels in the STP group were statistically higher than those in the control group (all p < 0.05). Except for the self-explanatory results caused by STP, the other indicators in both groups had good balance (all p > 0.05) (Table 1).
General Information of Study Cohort
ALT, alanine aminotransferase; APTT, activated partial thromboplastin time; AST, aspartate aminotransferase; CREA, creatinine; FDP, fibrinogen degradation products; FG, fibrinogen; HGB, hemoglobin; LYMPH, lymphocyte; MONO, monocyte; NEUT, neutrophils; PLT, platelet; PT, prothrombin time; RBC, red blood cell; STP, superficial thrombophlebitis; TT, thrombin time; UREA, urea nitrogen; WBC, white blood cell.
Indicates the presence of statistical differences, i.e. p < 0.05.
Hardy–Weinberg equilibrium test
Most of the SNP loci reached Hardy–Weinberg genetic balance (p values > 0.05) except β−249C/T in the STP group (p < 0.05) (Table 2).
Hardy–Weinberg Equilibrium Test in STP Group and Control Group (n = 84, STP = 34, Control = 50)
SNPs, single nucleotide polymorphisms.
Indicates the presence of statistical differences, i.e. p < 0.05.
Gene/allele frequency of the FGB promoter
A statistical difference was observed in the gene/allele frequency for −455A/G between the two groups (p < 0.05, OR = 0.368, 95% CI: 0.093 − 0.852) (Table 3).
Single Factor Logistic Analysis in Gene/Allele Frequency of FGB Promoter (n = 84, STP = 34, Control = 50)
FBG, fibrinogen B β.
Indicates the presence of statistical differences, i.e. p < 0.05.
Plasma fibrinogen level difference between −455G and−455A loci
There was no statistical difference concerning fibrinogen content between −455G and −455A loci after the Wilcoxon rank-sum test (all p > 0.05) (Table 4).
Fibrinogen Content of −455G and −455A Loci
Discussion
STP is a special kind of VTE. Traditional beliefs suggest this disease is related to infection, trauma (including intravenous injection), varicose veins, tumors, autoimmune diseases (such as Behcet’s disease, antiphospholipid syndrome), and so on, as described by Virchow’s triad. In fact, not all STP cases can be well explained by Virchow’s triad. Genetic risk factors are often profound and hidden beneath clinical manifestations and available examinations. We are short of understanding about how genetic background can affect STP. Meanwhile, the onset of STP is frequently influenced by various external factors, such as environment, comorbidities, and oral contraceptives. Therefore, in our study, we tried to collect “pure” idiopathic STP cases as much as possible, excluding secondary cases caused by external factors. This strategy naturally minimized the sample size. However, it increased statistical power from another perspective by diminishing external risk factors to a large extent and obtaining relatively “pure” samples. It should be noted that in order to reduce the influence of region and ethnicity on gene polymorphism, both the case and control populations were from the local Han population in Yunnan province.
Fibrinogen serves as a core protein for hemostasis in the coagulation system. Fibrinogen is also an acute phase reactive protein that is produced in large amounts and released during inflammatory and stress responses. Meanwhile, fibrinogen can also induce the adhesion between endothelial cells and neutrophils through intercellular adhesion molecule-1, thereby causing damage to the venous intima and valve (Tsakadze et al., 2002). In the past few decades, FGB has increasingly become a hotspot of cardiovascular/cerebrovascular diseases research (Rallidis et al., 2010; Luo et al., 2019; Lee et al., 2008). However, the extent to which these genes can affect the onset of STP is still unknown. Previous studies of FGB were mostly based on restriction enzyme digestion (PCR) methods, which were often time-consuming and frustrating, usually demonstrated in the form of some single nucleotide polymorphism (SNP). We always have the eagerness to fully sequence the whole promoter of FGB to compensate for the shortcomings of previous “fragmented” research (Komitopoulou et al., 2006; Li et al., 2019). Considering the fact that COVID-19 infection might be a high-risk factor and would have some uncertain impact on thrombotic events (Nalbandian et al., 2021; Poor, 2021), volunteers who had suffered from COVID-19 within 6 months were excluded from the present study.
Blood test indicators for STP
With the onset of STP, innate immunity activation and inflammatory reaction are usually localized and self-limiting rather than systemic inflammatory response syndrome. The white blood cell, neutrophil counts, and most blood test indicators were still within the normal range. It only showed a noticeable rise in monocytes for the STP group. Some studies showed that in the acute phase, inflammasomes could induce proliferation of monocytes/macrophages and promote thrombotic inflammation, which ultimately led to VTE through the pyroptosis pathway (Potere et al., 2023). Meanwhile, in order to avoid excessive thrombosis and accumulation of fibrinogen, fibrinolysis is accelerated, leading to an increase in d-dimer and FDP levels. Levels of monocytes, D-dimer, and FDP in the STP group were higher than those in the control group, but they were still within the physiological range, and these diversities were self-explanatory and related to STP.
Hardy–Weinberg equilibrium test
There may be significant differences in genotype between different regions and races (Camilleri et al., 2004). This study mainly focused on aseptic lower limb STP, excluding individuals with trauma, malignant tumors, rheumatic and immune diseases will minimize the influence of external factors. At present, there is little research on the correlation between FGB and venous thrombotic diseases, and most of them are based on Caucasian/African/Caribbean populations. In this study, we selected the local Han population in Yunnan province as the research object. From the Hardy–Weinberg test, only in the STP group did the −249C/T locus not comply with the law of genetic balance (p < 0.05), while all other gene loci met genetic balance (p > 0.05). Hardy–Weinberg equilibrium is the foundation of population genetics research, but it may be influenced by multiple factors (Schaid et al., 2006). Common ones include: (1) Population stratification, including population migration, coupled with geographical and cultural reasons (Wigginton, et al., 2005). (2) Mutation is another potential force that alters the frequency of alleles. For example, the frequency of a certain allele that determines a disadvantageous trait is gradually eliminated through multiple generations of inheritance, which is obviously beneficial for population evolution. (3) Linkage disequilibrium: For independently assigned loci in random mating, nonlinked genes have a 50% chance of recombination, while linked genes tend to reach equilibrium at a slower rate and take longer to reach equilibrium. In addition, the sample size selected for this study was not large enough, and the statistical power for the −249C/T locus might be low; confounding factors such as gene-environment interactions could also complicate genetic balance, causing Mendelian randomization principles to be ineffective and resulting in biased outcomes. Possible confounding factors such as immobilization, potential tumors or rheumatic immune diseases were easily overlooked and sometimes hard to detect during medical history collection. It was difficult to collect large amounts of “pure” idiopathic STP cases in the real world, and it seemed unrealistic to attempt to achieve Hardy–Weinberg equilibrium for the −249C/T locus by increasing the sample size in a short period of time. Animal experiments might be another way to improve Hardy–Weinberg equilibrium results, however, STP did not seem to be a universal disease for animals. Moreover, it was difficult and unnecessary to replicate this human disease into an animal model. Therefore, it was not determined yet whether this locus might have a potential effect on STP.
FGB promoter polymorphism and STP
SNP differs from mutation (Edwards et al., 2007); mutation refers to the frequency of gene variation being less than 1% in a population. When the frequency of genetic variation is greater than 1%, it is called an SNP. SNP accounts for more than 90% of genetic polymorphism in the human genome. At present, most of the FGB-associated SNPs have been discovered in their promoter. Therefore, we conducted a thorough sequencing of the whole promoter. Most of the research concerning FGB is focused on cardiovascular and cerebrovascular diseases (Gu et al., 2014), but few on STP. Through binary logistic regression analysis, we found that −455A was a protective locus for the onset of STP. Since the absence of genetic research on STP, we could only refer to previous VTE studies. Koster (Koster et al., 1994) found that −455G/A heterozygotes (AA+AG) were more likely to decrease VTE by 40% than the wild-type homozygotes (GG). However, Raymond believed there was no statistical difference in the frequency of the −455G/A allele between VTE and control groups (Camilleri and Cohen, 2005). Studies indicated that −455G/A was adjacent to the homeostatic element of interleukin-6, which could promote FGB gene transcription and fibrinogen synthesis (Morozumi et al., 2009). However, in our study, we did not detect any statistical difference concerning fibrinogen content between the −455G and −455A loci after the Wilcoxon rank-sum test. The reason might be that there are many factors that could affect plasma fibrinogen level. In addition to polymorphisms, factors such as fibrinogen consumption during thrombosis, fibrinolysis may all have a direct impact on it.
Similar to research on DVT, PE, and other thrombotic events, the process of STP was dynamic, and the critical point of onset was difficult to confirm. However, our research can roughly reflect some deep-seated factors beneath the manifestations and traditional concepts of STP.
Footnotes
Authors’ Contributions
R.L.: Data collection and gene sequencing. S.H.: Research design, writing—original draft, and project administration. J.X.: Methodology and translation. S.D.: Statistical analysis. H.G.: Duplex and conceptualization. All authors: Final approval of article.
Author Disclosure Statement
The authors declare no competing interests.
Funding Information
Supported by the Applied Basic Research Foundation of Yunnan Province [2019FE001(−216)].
