Abstract
Background:
The main objective of this prospective, multicenter study (REVEAL-CP) was to test children with cerebral palsy-like signs and symptoms for raised 3-
Methods:
Patients were identified in pediatric secondary and tertiary care hospitals through database searches and personal communication. 3-OMD concentrations from Guthrie card tests were determined using liquid chromatography/mass spectrometry. If 3-OMD was raised, cerebrospinal fluid analysis and
Results:
In total, 166 patients were enrolled in this study. The median age was 8 years. Sixty-six patients (39.8%) had a diagnosis of cerebral palsy, with the most common type being “mixed” (
Conclusions:
A Guthrie card test for 3-OMD is a recognized screening technique for AADCd. If universal newborn screening for this metabolic disease is not available, children with signs and symptoms of a movement disorder should be investigated for AADCd.
Introduction
Aromatic
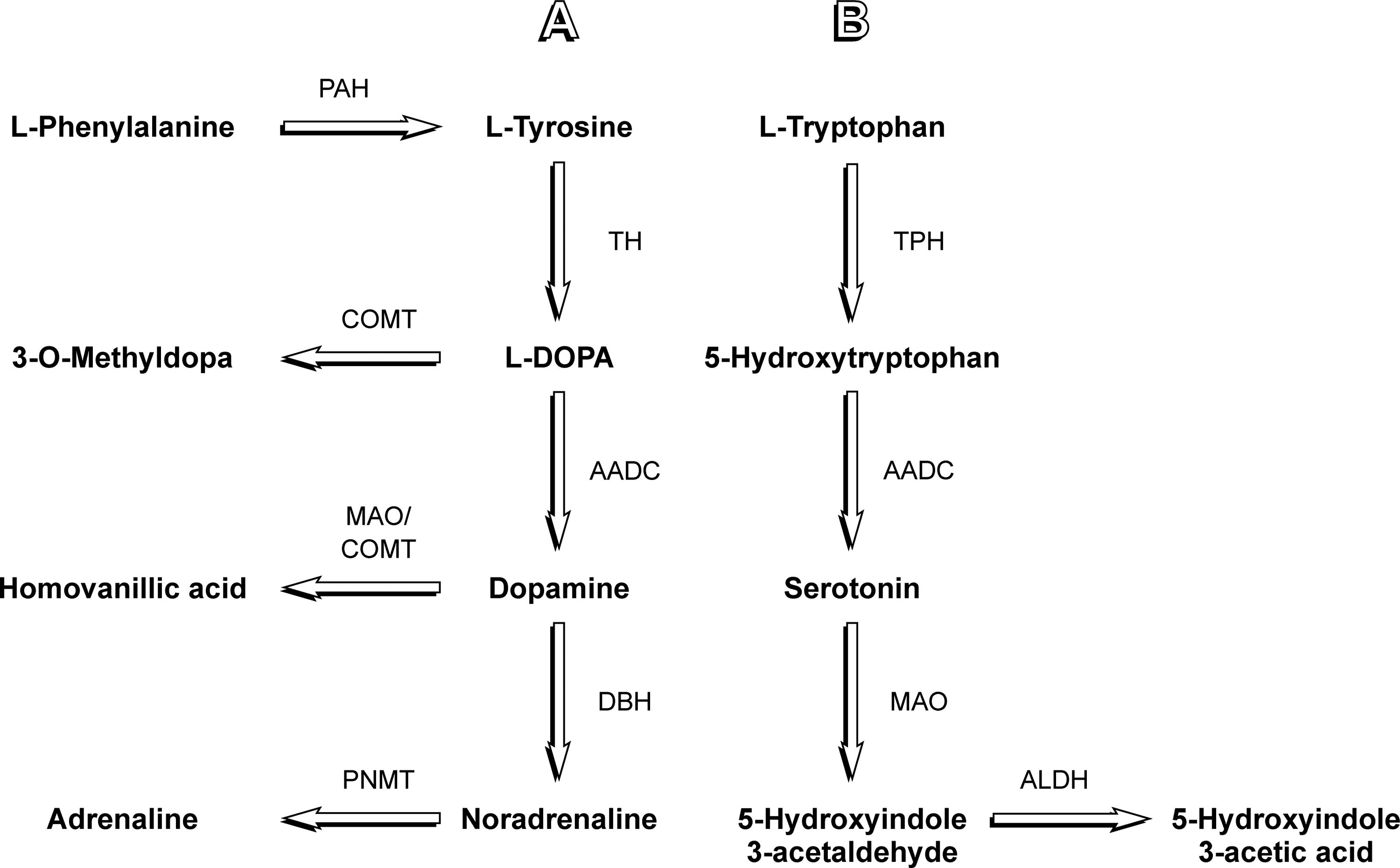
Diagram depicting relevant steps of dopamine
Methods
General
This study was sponsored by
Patient and public involvement
Patients or the public were not involved in the design, conduct, reporting, or dissemination of this research.
Ethics and consent
Before patient enrollment, research ethics committee approval was obtained in each participating country: the USA (SSU00111601), France (2019-A03014-53), Germany (EA2/057/20), Italy (219/2020; 58/20—CE; 0790/2020, 2021/489/DdP; 9/CE Medea; 2164/2020), and the UK (IRAS 274582, 20/LO/0597). Written consent was obtained from each patient and/or their legal guardian.
Patient selection
Patients of all ages who had signs and symptoms of CP or another movement disorder of unknown etiology were eligible to participate in this study. In practice, predominantly children and young people from secondary and tertiary pediatric hospital departments were recruited. Patients with periventricular leukomalacia, hypoxic-ischemic encephalopathy, cerebral infarction, encephalitis, cortical malformations, neuronal migration defects, spinal cord lesions, hydrocephalus or known AADCd, and those taking carbidopa/
Statistics
Descriptive statistics (the mean, standard deviation [SD], median, and range of patient characteristics) and the statistical analysis software IBM SPSS version 29.0.2 was used to analyze this large case series of an at-risk population.
3-OMD test
A capillary or venous blood sample was obtained from each patient and dripped onto a CentoCard (Centogene GmbH, Rostock, Germany). The filter paper cards were sent by surface mail to the Centogene laboratory in Rostock (Germany) for analysis. A detailed description of the methodology applied to measure 3-OMD blood levels with liquid chromatography/mass spectrometry has been published by Rizzi et al. (2023).
The method was validated in line with recommendations made by the College of American Pathologists and the Clinical Laboratory Improvement Amendments. A non-age-specific cut-off of >1000 nmol/L 3-OMD concentration was chosen based on blood samples from 110 healthy controls (upper limit of 95% confidence interval) and 80 patients with genetically determined AADCd.
DDC sequencing
The
Bioinformatic analysis
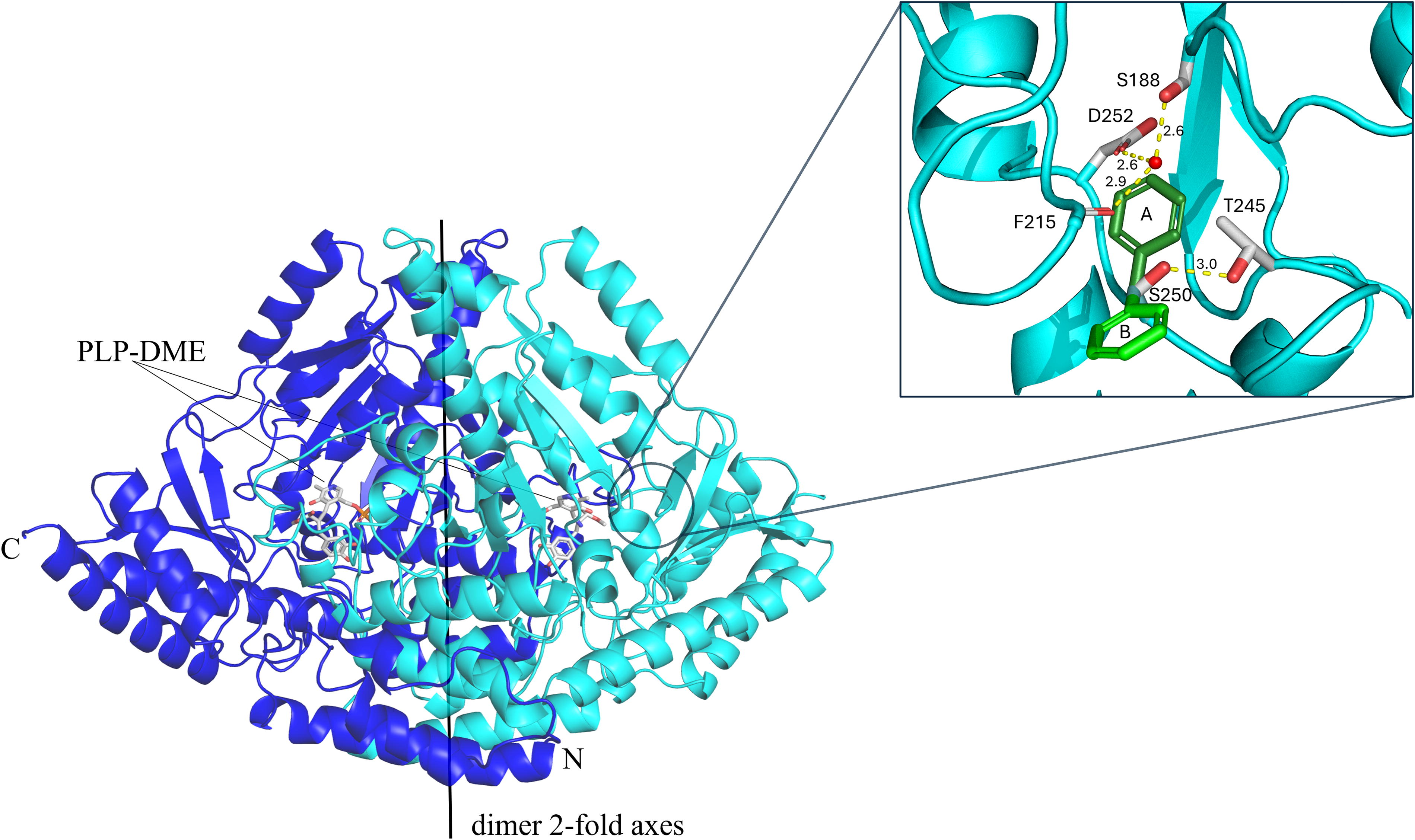
Analysis of the localization of the Ser250 residue and its substitution to phenylalanine was carried out by
Results
Patient cohort
In total, 166 patients were recruited during the 16-month-long study period. Eighty-nine patients (53.6%) were from Italy, 49 (29.5%) from the UK, 13 (7.8%) from France, 8 (4.8%) from Germany, and 7 (4.2%) from the USA. Ninety-seven patients (58.4%) were males and 69 (41.6%) were females. The mean age of the patient cohort was 9.5 years (SD, 11.63) and the median age was 8.0 years (25th percentile, 2.0; 75th percentile, 13.0; minimum, 0.0; maximum, 66.0). One hundred and 59 patients (95.8%) were 25 years or younger. One hundred and 32 patients (79.5%) were Caucasian; 11 (6.6%) Asian; 6 (3.6%) Black or African American; and 17 (10.2%) multiple, other, or undetermined. Sixty-six patients (39.8%) had a diagnosis of CP. Their mean age at diagnosis was 1.7 years (SD, 2.75; median, 0.5 years). The most common type of CP was “mixed” (
Signs and Symptoms of REVEAL-CP Study Patients (
3-OMD, 3-
Child with AADCd
This Caucasian boy was born after an uneventful pregnancy and delivery at term. He is the offspring of nonconsanguineous Italian parents. In the first few weeks of life, he was found to have remarkable irritability and generalized hypotonia. On examination, at the age of 11 months, he presented with severe global developmental delay, axial hypotonia, lack of postural reactions, mild distal dystonia of the limbs, hypokinesia, hypomimia, bilateral ptosis, oculogyric crises, and poor eye fixation. In addition, irritability, excessive salivation, hyperhidrosis, feeding and swallowing difficulties, gastroesophageal reflux, and faltering growth were observed. DBS analysis showed an increased 3-OMD concentration of 9910 nmol/L (normal range, ≤1000 nmol/L). Investigation of his cerebrospinal fluid (CSF) revealed a pattern characteristic of AADCd with neurotransmitter metabolite levels as follows (normal ranges are reported in brackets): homovanillic acid (HVA) 90 nmol/L (236-867 nmol/L); 5-hydroxyindole-3-acetic acid (5-HIAA) 26 nmol/L (97-367 nmol/L); HVA/5-HIAA ratio 3.46; 3-OMD 1843 nmol/L (<50 nmol/L); 5-hydroxytryptophan (5-HTP) 114 nmol/L (<10 nmol/L); and 3-methoxy-4-hydroxyphenylglycol (MHPG) 12 nmol/L (47-81 nmol/L). All other neurometabolic tests were within normal limits. Magnetic resonance imaging of the brain demonstrated a mild delay in white matter myelination in the frontal region bilaterally. Electroencephalography showed slow wave activity in the posterior regions. The child was initially prescribed the type-B monoamine oxidase inhibitor selegiline, pyridoxine (4-methanol form of vitamin B6), and the dopamine agonist pramipexole, which led to partial clinical improvement. Later, he underwent treatment with intraputaminal eladocagene exuparvovec (data not yet published).
Gene and protein analyses
Discussion
In this prospective study among patients with signs and symptoms of CP or mimic-CP, one child with AADCd (0.6%) was identified through selective screening using a Guthrie card test for 3-OMD. Although this percentage seems low, it is significantly higher than the prevalence of AADCd reported in the general European population (1/116 000) (Himmelreich et al., 2019). Rizzi et al. performed a similar prospective, multicenter study in Italy (Rizzi et al., 2023). 3-OMD blood levels were analyzed from 390 patients with an undiagnosed neurodevelopmental disorder using Guthrie cards, and none of them were found to have AADCd (Rizzi et al., 2023). Hyland and Reott investigated 20,000 CSF samples for neurotransmitter abnormalities and detected 22 new cases of AADCd (Hyland and Reott, 2020). The prevalence of 1/900 in their at-risk population more closely resembles the figure of 1/166 from this study, which as a case series provides low-level evidence. Selective clinical screening (e.g., focused on patients with signs and symptoms of CP, like in the present study, or an early-onset movement disorder in general) may be more effective in intercepting affected patients. The current gold standard for diagnosing AADCd is a combination of
The young patient in this investigation who presented with typical clinical features of AADCd was only diagnosed after enrollment in this study. This demonstrates the challenges clinicians in different healthcare settings face when encountering this rare disease. According to Himmelreich et al., the patient identified in the present study is the tenth patient with a homozygous, pathogenic variant (c.749C>T; p.Ser250Phe), making this the fifth most common variant of AADCd worldwide (Himmelreich et al., 2023). Overall, 5.3% of AADCd genotypes are responsible for 41.4% of all reported patients with this disorder (Fig. 3). Caine and collaborators have developed a knock-in mouse model for AADCd by introducing the same Ser250Phe missense variant in the murine
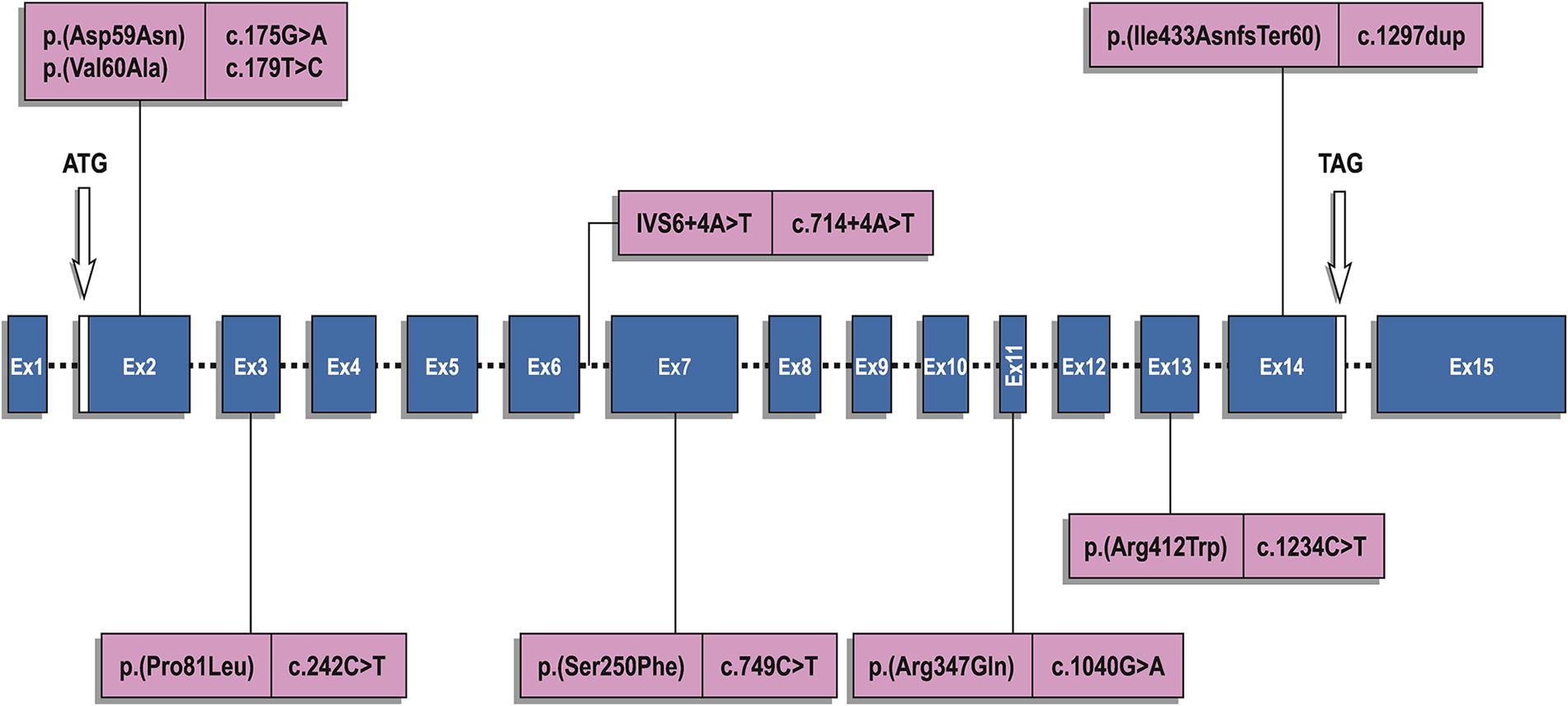
Eight most common
Conclusion
In conclusion, this study provides evidence that 3-OMD DBS screening using Guthrie card tests targeted at patients with a movement disorder of unknown etiology can assist in identifying those with AADCd in an economic fashion and with a higher yield than in the general population. In addition, bioinformatics technology applied to one proband has enriched our understanding of the molecular pathogenesis of AADCd.
Footnotes
Acknowledgments
The authors are grateful to the young people and their carers who participated in this study. The authors would like to thank the healthcare professionals and administrative staff who were involved in this study, especially Jo-anna Allen, Kapil Arya, Guja Astrea, Eleonora Bonaventura, Peter Blomqvist, Alessandro Capuano, Carla Carducci, Claudia Carducci, Tamsin Chambers, Giovanni Cioni, Edward Dabrowski, Ian Davidson, Christian De Goede, Laura Demuth, Andrew Duckworth, Tim Ellison, Timothy Feyma, Robert Flamini, Peta Heslop, Keith Hyland, Angela Kaindl, Allen Kristensen, Michael Kruer, Warren Marks, Andrea Martinuzzi, Daniella Mascarenhas, Marie Monaghan, Thomas Opladen, Neil Parlett, Phillip Pearl, Vinay Penematsa, Mark Rance, Agathe Roubertie, Annamaria Russo, Roberta Scalise, Michael Shrader, Marie-Aude Spitz, Marc Steder, Micheal Taylor, Antonio Trabacca, and Suresh Vijay.
Authors’ Contributions
Conceptualization: S.J., P.L., and C.W. Investigation: E.-M.S., R.B. (identification of the patient with AADCd), V.G., A.K., S.A., M.B., M.P., and V.L. Supervision: S.J., P.L., and E.F. Data curation: S.A., M.B., M.P. Formal analysis: M.B., M.P., V.L. (CSF analysis for the patient with AADCd), and E.L. Writing—original draft preparation: E.-M.S. (lead), R.B., M.B., and M.P. Writing—review and editing: all authors.
Author Disclosure Statement
V.G. has participated in scientific advisory boards, scientific symposia, and teaching initiatives for Biogen, Italfarmaco, Novartis, Pfizer,
Funding Information
The study was sponsored by PTC Therapeutics MP, Inc. (Warren, NJ; PTC-MA-AADC-402). This research was supported in parts by the Italian Ministry of Health, Ricerca Corrente 2021, and the IRCCS Fondazione Stella Maris.
