Abstract
Background:
Non-small cell lung cancer with brain metastasis (NSCLCBM) is normally observed in advanced-stage patients. Bevacizumab has shown to improve survival in the first-line treatment of metastatic brain NSCLC when added as a bolus plus irinotecan. However, a better understanding of the molecular mechanism is required to further drive progress in this field.
Methods:
A total of 155 patients were selected, including 42.10% with Kirsten rat sarcoma viral oncogene homolog (Kras)-mutant tumors. Of the 155 patients, 62.04% had developed brain metastasis (BM). Seven functional single-nucleotide polymorphisms (SNPs) in the Kras gene were extracted from the HapMap SNP database and were used for genotyping. The haplologit command in Statistical Software for Data Science (STATA) was used to model the association between haplotypes and case status. A Cox analysis was used to evaluate the prognostic value of the SNPs.
Results:
Among the patients treated with combination regimens, recurrence after local treatment was more frequent in those with two types of Kras mutations (odds ratio [OR] = 2.033 [0.5015-4.2552], p = 0.009). Among the patients with untreated BM, overall survival was shorter than that of patients with Kras mutations according to univariate analysis (OR = 5.130 [1.240-41.012], p = 0.033).
Conclusions:
Kras mutations have a predictive role for BM recurrence and outcome in patients with NSCLC treated with bevacizumab combination regimens.
Introduction
Lung cancer is the most commonly diagnosed malignancy and has been the leading cause of cancer-related deaths for decades (Herbst et al., 2018). In 2018, lung cancer affected 2,093,876 new cases, accounting for 11.6% of all cancers (Bray et al., 2018). Most (∼85%) patients with lung cancer are those with non-small cell lung cancer (NSCLC) (Molina et al., 2008). Due to the lack of evident symptoms, NSCLC is usually diagnosed at advanced stages, and its prognosis is generally poor (Postmus et al., 2017). NSCLC with brain metastasis (NSCLCBM) is normally observed in advanced-stage patients. The mean overall survival (OS) time for patients with lung cancer brain metastasis (BM) is ∼4.5 months with standard whole-brain radiation therapy and 4-11 weeks in untreated patients (Gaspar et al., 1997; Ampil et al., 2007).
Although improvements in brain imaging and systemic treatment for lung cancer treatment greatly prolong the survival time of patients with NSCLCBM, efficient early diagnosis and treatment methods are still limited. For the cancer BM stage, there is still a lack of efficient biomarkers for tumor cell growth, metabolism, drug resistance, and tumor recurrence to assist diagnosis and treatment (Baek et al., 2018; Franchino et al., 2018).
In the past decades, Kirsten rat sarcoma viral oncogene homolog (KRAS) protein has been considered a switch in cancer cell signal transduction. It plays an oncogenic role in regulating cancer cell proliferation, migration, and epithelial-to-mesenchymal transition (Fernández-Medarde and Santos, 2011; Tímár, 2014). Specifically, Kras mutations always lead to differential induction of downstream signal transduction cascades and specific drug sensitivity (Ghimessy et al., 2020). For example, KRAS G12C mutations always lead to higher extracellular signal-regulated kinase (ERK) 1/ERK2 phosphorylation and improved efficacy of the mitogen-activated protein kinase kinase inhibitor, selumetinib (Li et al., 2018). The role of Kras mutations in lung cancer was investigated in a recent study, and these mutations in combination with Lkb1 copy number variation could predict BM in NSCLC (Zhao et al., 2014).
Traditionally, surgical resection, combined with radiation therapy and palliative chemotherapy directed toward symptom palliation was used as standard care to treat NSCLCBM (Gállego Pérez-Larraya and Hildebrand, 2014; Kotecha et al., 2018). NSCLC patients with epidermal growth factor receptor (Egfr) mutations are a special group of individuals. The treatment for patients carrying a Egfr mutation is different compared with that for other NSCLC patients (Eberhard et al., 2005). The use of EGFR tyrosine kinase inhibitors was regarded as a new approach toward Egfr mutant carrying NSCLC patients with BM, with reported response rate of 70% and median progression-free survival (PFS) of ∼1 year (Rosell et al., 2012). The bevacizumab+erolitinib is considered a second-line treatment in patients with nonsquamous NSCLC (Cortinovis et al., 2017). However, the effect of this treatment is largely limited to the Egfr mutant status (Yu and Fan, 2019).
In recent years, irinotecan was considered an alternative treatment for NSCLC beyond second-line treatment (Kumar and Wakelee, 2006). Topoisomerase I (TOPO1) is the downstream target of irinotecan. Some researchers have reported that patients harboring EGFR mutations have abnormal TOPO1 activity due to increased Topo1 mRNA levels, which resulted in these patients benefiting from irinotecan, but the effect of irinotecan was not limited by the Egfr mutations (Sakai et al., 2012). Irinotecan- or bevacizumab-based chemotherapy has already been shown to be effective against advanced NSCLC (Yang et al., 2015; Ascha et al., 2019). Bevacizumab, a recombinant humanized monoclonal antibody directed against vascular endothelial growth factor (VEGF), has also been approved by the United States Food and Drug Administration to treat nonsquamous NSCLC and has been shown to improve survival in first-line treatment of nonsquamous NSCLCBM when added to bolus plus irinotecan (Wills et al., 2017).
The combination of bevacizumab and irinotecan has also been used to treat advanced NSCLC, which overexpresses Ercc1 (Takemoto et al., 2019). As a receptor tyrosine kinase, the EGFR-related downstream signaling pathway is also regulated by KRAS. There are few reports on the investigation of Kras mutations in bevacizumab-based metastatic lung cancer treatment (Garassino et al., 2013; Ghimessy et al., 2019). In this study, we explored the effect of Kras mutation polymorphisms on the safety and efficacy of patients with nonsquamous NSCLCBM treated with bevacizumab combination regimens.
Materials and Methods
Clinical sample collection
A total of 155 patients with nonsquamous NSCLC were admitted at Hebei Hospital from August 2015 to March 2017. Among these patients with NSCLC, 88 (40 females and 48 males, 32-71 years) who were diagnosed with NSCLCBM were enrolled in this study. This study passed the Review Board of the hospital. The inclusion criteria were as follows: (1) patients who were newly diagnosed, (2) patients with complete treatment and survival follow-up, and (3) patients who were diagnosed with NSCLCBM by pathological examination and medical imaging. Patient treatment, recurrence, and survival status information were all well documented. The exclusion criteria were as follows: (1) patients with recurrent NSCLC, (2) patients receiving therapies performed before admission, (3) patients with other clinical disorders, and (4) patients with other cancers and tumors of unknown origin.
All the patients were informed of the experimental principle, and they all signed informed consent forms. The protocols were approved by the Ethics Committees of the Affiliated Hospital of Hebei University (No. AHHU20190321). Clinical information is provided in Supplementary Table S1. The flow chart of subjects enrolled and exlcued in the study was shown in Supplementary Figure S1.
According to clinical findings, such as imaging information, histopathological examinations, and other laboratory indicators, the 88 patients were staged according to the American Joint Committee on Cancer (AJCC) guidelines (8th edition). Based on their conditions, 37 and 51 patients were treated with surgical resection in combination with radiotherapy and bevacizumab combined with bolus plus irinotecan after surgical resection, respectively.
From the day of admission, all the patients were followed up every 2 months through phone calls or outpatient visits. The last visit was conducted in August 2020. Moreover, 3 mL peripheral blood from the patients was extracted before and after treatment, and patients who died of causes unrelated to NSCLC and patients without BM tumor were excluded from the survival analysis. Finally, 15 patients were excluded from the study. Analysis of PFS was performed in this study. The design of the study is shown in the flow diagram according to STARD (www.equator-network.org/reporting-guidelines/stard/).
Polymorphism selection
The public HapMap single nucleotide polymorphism (SNP) database (www.ncbi.nlm.nih.gov/) and SNP information (http://snpinfo.niehs.nih.gov/) were used to select functional SNPs in the Kras gene. The selection criteria were as follows: minor allele frequency ≥5%, linkage disequilibrium coefficient r2 < 0.8, and previously selected SNPs located in codons 12, 13, and 61. All SNPs have been reported to be associated with lung cancer.
Genomic DNA isolation and genotyping
Blood DNA was isolated from the blood using the EasyPure Blood Genomic DNA kit (Transgene). The detailed steps followed the manufacturer's instructions. The primers were designed according to the six selected SNPs from the HapMap SNP and SNP information database. Genotyping was performed using the SNPshot method.
Haplotype analysis
The assembled haplotype was estimated separately in the NSCLCBM patient cohort by case status. The haplo.stats package in R was used to estimate the posterior probabilities of haplotypes. The haplologit command in STATA was used to model the association between haplotypes and case status, as in a previous study (Marchenko et al., 2008).
Statistical analyses
All data were analyzed using the SAS software (version 9.0; SAS Institute, Cary, NC). Kaplan-Meier (K-M) curves were used to assess the cumulative probability of BM. The K-M plotter was used to plot survival curves, which were compared using the log-rank test. Univariate and multivariate analyses were performed using the Cox proportional hazards model to investigate the influence of genotypes on the BM risk, and the analysis was adjusted for age, sex, smoking status, and other related factors. The χ2 test was used to evaluate the differences in gene mutation frequency between the groups. Statistical significance was set at p < 0.05.
Results
Clinical characteristics of the study population
The clinical characteristics of the 88 patients with nonsquamous NSCLCBM and 67 patients with nonsquamous NSCLC without BM were assessed in this study. After establishing the diagnosis and obtaining informed consent from the patients, the 51 patients with nonsquamous NSCLCBM were found suitable for treatment with bevacizumab combination regimens (bevacizumab bolus + plus irinotecan) (56.3% of the enrolled patients had a smoking history, 100% had Karnofsky Performance Status score >80 and Zurich Pituitary Score >2, and 73.5% had body mass index <25 kg/m2). The other 37 patients with NSCLC with both brain and other site metastases received standard surgical resection and radiotherapy. The median age was 55 (range, 33-71) years. Regarding the sex ratio, 45 male (51%) and 43 (48%) female patients were enrolled.
Moreover, 53 (60%) patients had stage III or IV disease according to the AJCC tumor staging criteria. Seven (7.9%) patients were lost to follow-up at the end of the study. Cox analysis was performed to evaluate the association between clinical characteristics and BM. The univariate and multivariate results showed that the risk of BM was significantly higher in patients with AJCC stage III/IV disease (p = 0.002) and tumor-node-metastasis stage III/IV disease (p = 0.005) (Table 1).
Clinical-Pathological Characteristic in Nonsquamous Non-Small Cell Lung Cancer Patients and Their Association with Brain Metastasis
p < 0.05.
HR, hazard ratio; CI, confident interval.
Association between Kras SNP and BM risk
In total, seven SNPs were identified for genotyping in the cohort (Fig. 1). Three SNPs were located in codon 61 of Kras, and the four remaining SNPs were located in codons 12 and 13. We performed SNP genotyping of the seven SNPs in all patients with NSCLC with hetero- and homozygous characteristics. The highest mutation rate in patients was observed in rs121913240 (Fig. 1). Three SNPs (rs121913535, rs17851045, and rs397517040) did not differ between patients with or without BM (Table 2). Two SNPs (rs121913240 and rs121913529) were characterized by the highest occurrence rate in the NSCLCBM.
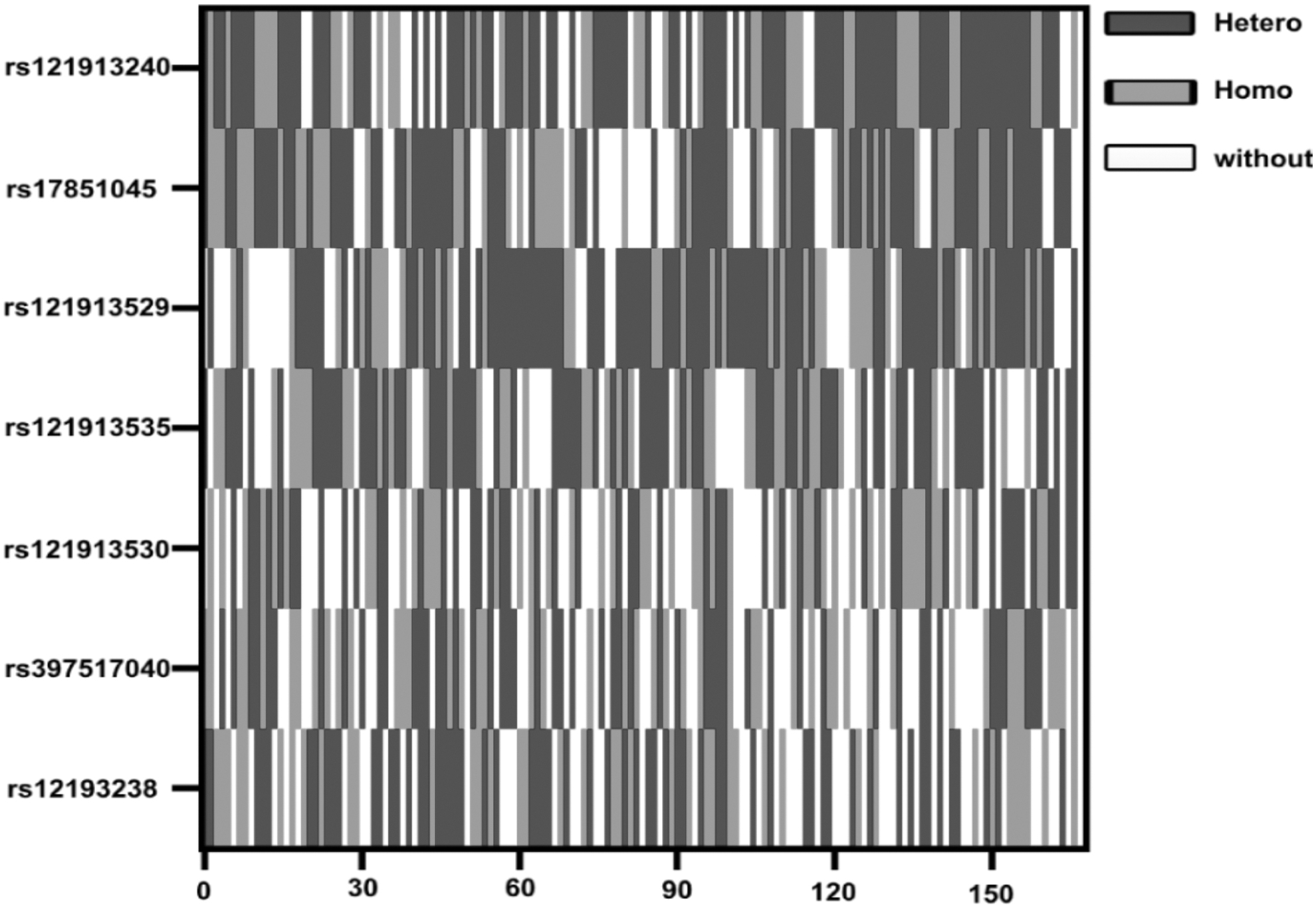
Association between Kirsten rat sarcoma viral oncogene homolog SNPs and brain metastasis risk. Alterations in seven candidate polymorphisms in all patients with NSCLC. The data were analyzed for the heterozygous and homozygous genotypes. The x-axes indicate the 155 subjects enrolled in the study; the y-axes indicate the rs number of each SNP. “Hetero” indicates the subject with heterozygous sites; “Homo” indicates subject with homozygous sites; “without” indicates subject without the SNPs. NSCLC, non-small cell lung cancer; SNP, single nucleotide polymorphism.
Correlation Between Different Genotypes of Genes in KRAS and Brain Metastasis
Multivariate analyses in this table were adjusted for age, sex, smoking status, Karnofsky Performance Status, body mass index, tumor histology, TNM stag.
SNP, single nucleotide polymorphism.
Furthermore, cumulative survival analysis was performed to compare the two SNPs that contributed to the prognosis of patients with NSCLCBM. The patients with SNP rs121913240 had worse prognosis than those with SNP rs121913529 (Fig. 2A, B). In addition, Cox model analysis revealed that patients with NSCLCBM with heterozygous SNP (rs121913240) displayed a closer association with BM risk (Table 2).
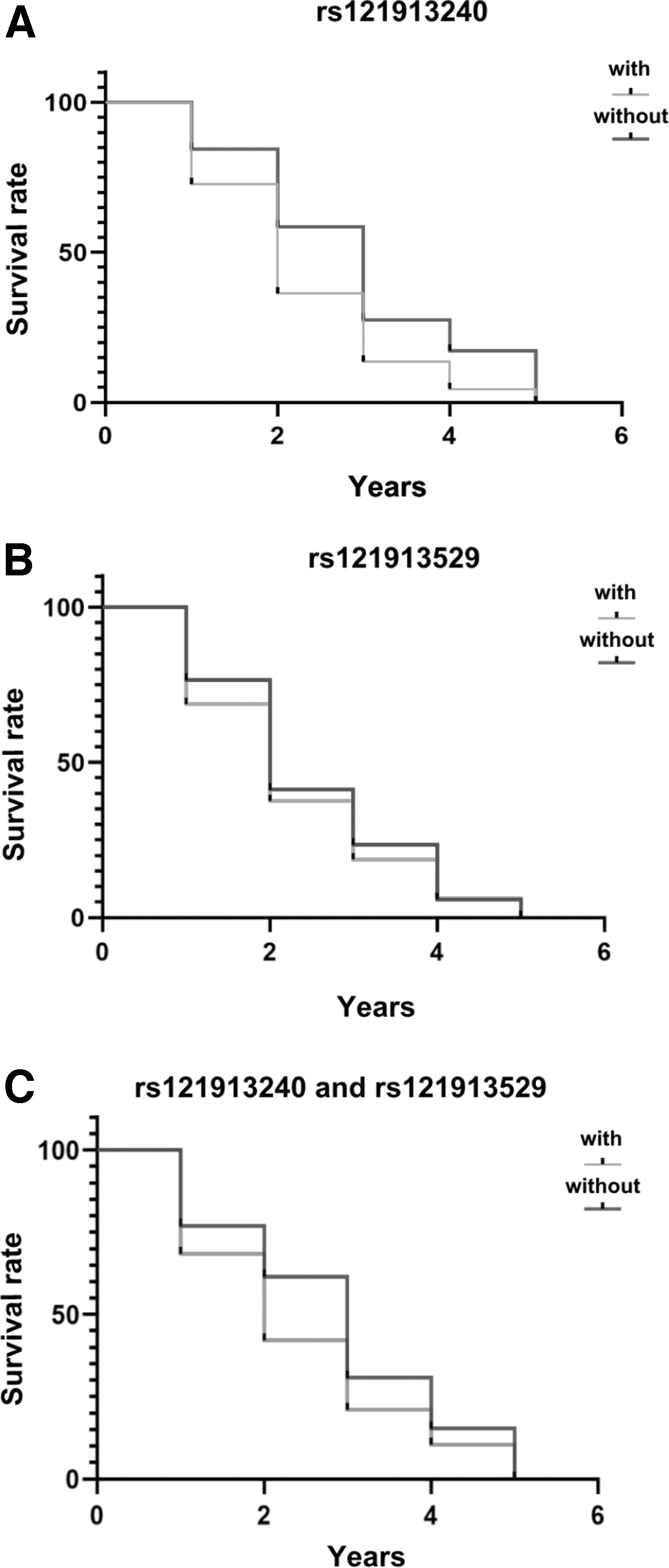
Combined effect of polymorphisms on the outcome of bevacizumab combination regimen treatment. Kaplan-Meier analysis of the cumulative probability of brain metastasis in patients with non-small cell lung cancer according to the following unfavorable genotypes:
Combined effect of polymorphisms on bevacizumab combination regimen treatment outcome
Considering that SNPs rs121913240 and rs121913529 preferably co-occur in the same NSCLCBM sample, we defined the two SNPs as a haplotype to identify their contribution to bevacizumab combination regimen treatment outcome. Forty-six patients who received the bevacizumab combination regimen treatment and 35 patients who did not receive the bevacizumab-based chemo treatment underwent genotyping. K-M analysis results indicated that the hazard ratio (HR) index in patients with the haplotype was significantly higher than that in patients with one or no SNPs for the survival years (rs121913529:5 years odds ratio [OR] = 0.5%, p = 0.009; rs121913240:5 years OR = 0.37%, p < 0.01) (Fig. 2A, B). In particular, the patients who carried the haplotype displayed shorter survival than their counterparts (OR = 0.28%) (Fig. 2C).
Association among clinical features, clinical index, and risk SNPs in BM
In this study, the correlation between clinical features and SNPs rs121913240 and rs121913529 was explored. The results of Cox analysis showed that patients with BM exhibiting N2/N3 stage lymph node metastasis had a significantly higher tendency of SNP rs121913240 (Fig. 3A, p < 0.05), whereas T4 stage patients had a higher tendency to correlate with SNP rs121913529 (Fig. 3B, p < 0.05). In our previous study, the carcinoembryonic antigen (CEA) and squamous cell carcinoma (SCC) antigen, which are considered classical poor prognosis biomarkers detected in serum, were significantly downregulated in patients who received bevacizumab combination treatment. In this study, a relatively higher expression of CEA and SCC antigen was also highly correlated with SNPs rs121913240 and rs121913529 in the bevacizumab-treated patient group (Fig. 3C, p < 0.05).
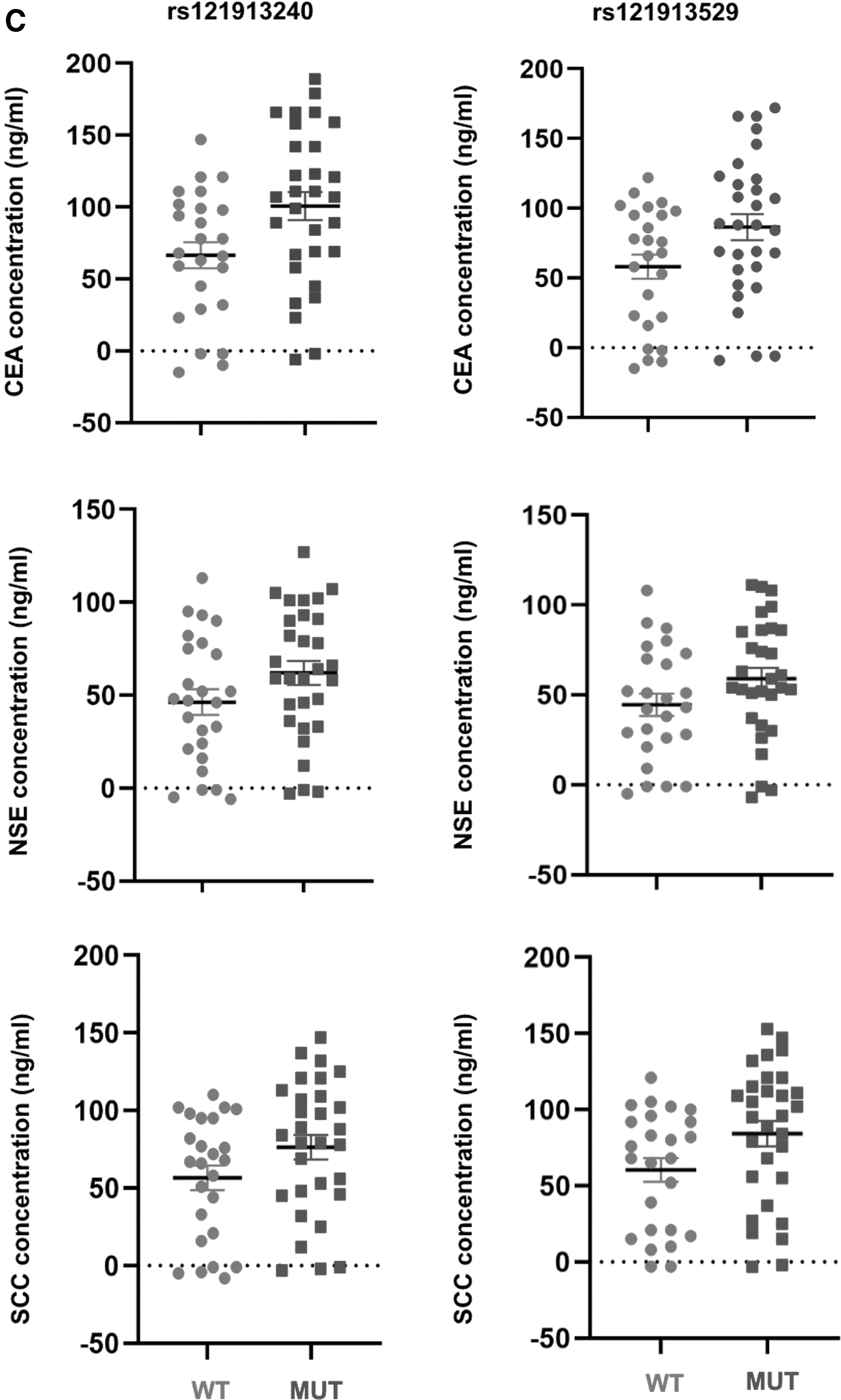
Association among clinical features, clinical index, and risk SNPs in brain metastasis. Forest plot of association between variable clinical characteristics and different genotypes of rs121913240
Discussion
To the best of our knowledge, nonsquamous NSCLCBM always causes high mortality and serious neurological symptoms. According to the phase III Eastern Cooperative Oncology Group, bevacizumab in combination with erlotinib was considered a second-line treatment in patients with nonsquamous NSCLC (Sandler et al., 2006; Marchenko et al., 2008). Despite the combination therapy significantly prolonging PFS and improving the response rate, some patients respond poorly to this therapy (Herbst et al., 2005).
Kras is considered a hub gene in many pathways, including Vegf and Egfr. It regulates many cell functions (Besse et al., 2015). To date, there have been no effective drugs designed to target RAS (Ghimessy et al., 2020). Kras mutations in tumors indicate many malignant phenotypes, such as chemoresistant and brain metastatic phenotypes and phenotypes involved in vascular formation (Chin et al., 1999; Fernández-Medarde and Santos, 2011; Wiesweg et al., 2019). In this study, we first investigated the genotype and frequency of occurrence of Kras SNPs in patients with nonsquamous NSCLC with and without BM. Two SNPs were identified as BM at-risk SNPs. Furthermore, the heterozygote of SNP rs121913240 displayed a closer correlation with the BM cohort. This result potentially indicates that SNP rs121913240 has a higher HR for NSCLCBM.
In addition, we compared the OS of patients with nonsquamous NSCLC who received bevacizumab in combination with erlotinib and those who did not. The two SNPs had a higher co-occurrence frequency in patients with nonsquamous NSCLCBM who received bevacizumab in combination with erlotinib. Combined SNPS can largely enhance the clinical prediction capability. The co-occurrence of SNPs rs121913240 and rs121913529 can create a haplotype that strongly indicates a high HR for NSCLCBM. Patients with haplotypes have a worse prognosis when they receive bevacizumab treatment. Furthermore, we found that the haplotype distribution was not closely correlated with any clinical characteristics of NSCLCBM. However, this haplotype was highly correlated with a relatively higher expression of CEA and SCC antigen by Cox analysis.
The role of Kras mutation spectrum in NSCLC has been reported in a previous study. Zhao et al. found that Lkb1 copy number alternation in combination with Kras mutations predicted BM in NSCLC (Zhao et al., 2014). G12V, G12A (Wei et al., 2019), and G12D (Fiala et al., 2016) KRAS mutations have been reported as poor prognostic biomarkers for patients with colorectal cancer receiving bevacizumab. Even in NSCLC, G12D mutation in KRAS indicated a potentially worse response to bevacizumab plus platinum-based chemotherapy (Bruera et al., 2015). Mutations in codons 12, 13, and 61 of Kras are normally considered oncogenic mutations (Chaft et al., 2013).
In our study, we identified a novel SNP haplotype that contains mutations in codons 12 and 61. This haplotype partially indicates sensitivity of patients with a BM response to bevacizumab in combination with erlotinib. One limitation of this study is its relatively small sample size, which may limit its universality for clinical use. To improve the clinical value, a combination study of this SNP haplotype and other known biomarkers or genetic mutations for NSCLC and the cellular functional role of the SNP haplotype in tumor BM should be clarified in future studies.
Footnotes
Authorship Confirmation Statement
All authors read and approved the final article.
Availability of Data and Materials
The analyzed data sets generated during the study are available from the corresponding author upon reasonable request.
Authors' Contributions
Z.S. made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; Z.S. took part in drafting the article or in revising it critically for important intellectual content; G.R. and X.W. performed the collection of samples and clinical data. Z.S. and G.R. participated in data analysis. H.L. contributed to the experimental design and the review and revision of the article and provided final approval of the version to be published.
Ethics Approval and Consent to Participate
The present study was approved by the Ethics Committee of the Affiliated Hospital of Hebei University. The study was conducted in accordance with the World Medical Association Declaration of Helsinki. All patients provided written informed consent before their inclusion in the study.
Author Disclosure Statement
The authors report that there are no competing interests to declare.
Funding Information
This study was supported by the Affiliated Hospital of Hebei University. The study was supported by the Foundation Project of Affiliated Hospital of Hebei University in 2018 (no. 2018Q003).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
