Abstract
The aim of this study was to investigate the correlation between Wnt1/β-catenin expression and the clinicopathologic features and prognosis of patients with esophageal squamous cell carcinoma (ESCC). The mRNA and protein expression levels of Wnt1/β-catenin genes in 70 ESCC and 15 adjacent noncancerous paraffin-embedded samples were determined by real-time quantitative polymerase chain reaction and immunohistochemical staining. The mRNA expression level of Wnt1/β-catenin in ESCC was significantly higher than that in the adjacent noncancerous tissues (1.9934 ± 1.9888 vs. 0.8863 ± 0.665, p = 0.0184; 0.2854 ± 0.1298 vs. 0.0128 ± 0.0158, p = 0.0000, respectively), and the overexpression of Wnt1/β-catenin mRNA was aggressively associated with lymph node metastasis and advanced pathological stage (p < 0.0001). The protein expression level of Wnt1/β-catenin was also significantly higher than that in the adjacent noncancerous tissues (0.3830 ± 0.0947 vs. 0.2721 ± 0.1474, p = 0.0002; 0.2835 ± 0.0844 vs. 0.2352 ± 0.0670, p = 0.0210, respectively); however, the overexpression was not associated with clinicopathologic characteristics. Meanwhile, the protein expression level of Wnt1 had no relevance with that of β-catenin. The overexpression of Wnt1/β-catenin might be an important molecular marker to predict the clinicopathologic stage and prognosis of ESCC, and the level of Wnt1/β-catenin mRNA was conversely correlated with lymph node metastasis and advanced pathological stage. The overexpression of Wnt1/β-catenin mRNA should also predict poor prognosis of ESCC; however, it might not be an independent prognostic factor.
Introduction
T
Materials and Methods
Cases selection
Between June 2002 and December 2003, 70 ESCC and 15 adjacent noncancerous paraffin-embedded samples were collected from Tumor Center of the First Affiliated Hospital of Nanjing Medical University. The features of 70 cases are shown in Table 1. Preoperative examinations including biopsy, barium X-ray, computed tomography, and ultrasonic endoscopy were performed. All patients received no treatment before operation, and all underwent radical resection for ESCC. All samples collected were sent to postoperative pathological examination, according to the American Joint Committee on Cancer (2002) pathological grade of pTNM standards including tumor size, lymph node, and metastasis (Greene, 2002). Clinical follow-up after surgery was based on periodic visit (every 3 months in the first year, every 6 months in the second year, and then yearly until relapse). The follow-up time for survivors ranged from 8 to 62 months (median: 40 months). On the endpoint of May 31, 2009, 4 out of 70 patients were found to be lost in the follow-up. The use of specimens for analyses was approved by the Ethics Committee of Medical Sciences, Nanjing Medical University. The use of study specimens for analyses was approved by the research ethics committee of Nanjing Medical University of Medical Sciences.
AJCC, American Joint Committee on Cancer.
RNA extraction and real-time quantitative polymerase chain reaction
Real-time quantitative polymerase chain reaction (RT-PCR) was performed on paraffin-embedded sections from 70 ESCC patients and 15 adjacent noncancerous samples. Briefly, total RNA was extracted by Recover All™ Total nucleic Acid Isolation (cat. no. 1975; Ambion, Austin, TX). Ten micrograms of Dnase I-treated total RNA was used for reverse transcription with Superscript III (Invitrogen, Carlsbad, CA). An aliquot representing 100 ng of input RNA was amplified by RT-PCR using the TaqMan PCR reagent kit and assay-on-demand gene expression products (FAM/Sybr, Foster City, CA). RNA extracted from the noncancerous lesion of a patient was used as the standard. After reverse transcription, standard cDNA was serially diluted to five standard solutions to prepare the reference curve. The primary design is shown in Table 2. RT-PCR was carried out using the Rotor-Gene 3000 Real-Time PCR kit (Corbett Research, Sydney, Australia) with 10000 × Sybergreen (Molecular Probes, Houston, TX). After reverse transcription, standard cDNA was serially diluted to five standard solutions to prepare the standard curve. The relative amount of cDNA in each sample was measured by interpolation using the standard curve, and then the relative ratio of β-catenin and Wnt1 to β-actin (the housekeeping gene) expression was calculated for each ESCC sample.
Immunohistochemistry
Histopathologic evaluation was performed on 4-μm slides stained with hematoxylin and eosin (Fig. 1). Commercially available rabbit polyclonal antibodies against Wnt1 (H-89, 1:200 diluted; Santa Cruz Biotechnology, Santa Cruz, CA) and β-catenin mouse monoclonal antibody (C19220, 1:200 diluted; Transduction Laboratories, Lexington, KY) were used as primary antibodies. A paraffin section of the ESCC sample was deparaffinized and rehydrated in graded alcohol to water. Antigenic enhancement was performed by submerging in citrate buffer (pH 6.0) and microwaving. Endogenous peroxide activity was quenched by applying 0.3% hydrogen peroxide for10 min, followed by incubation with 1% bovine serum albumin to block nonspecific binding. The primary antibody was incubated for 60 min at 37°C. After washing, the tissue sections were then reacted with the biotinylated anti-rabbit Wnt1 IgG (Vector Laboratories, Burlingame, CA) and anti-mouse β-catenin IgG (Vector Laboratories). The slides were immersed in the prepared diaminobenzidine (DAB) solution. Slides were then counterstained with hematoxylin, dehydrated, placed in xylene, coverslipped, and analyzed by optical microscopy. Each section was evaluated by at least two independent professional pathologists without knowledge of the stage and patient profiles. The distribution of Wnt1 and β-catenin was scored on a semiquantitative scale, and the percentage of tumor cell positive was recorded and divided as follows: negative (<10% tumor cells positive), locally positive (10-50% tumor cells positive), diffusely positive (>50% tumor cells positive).
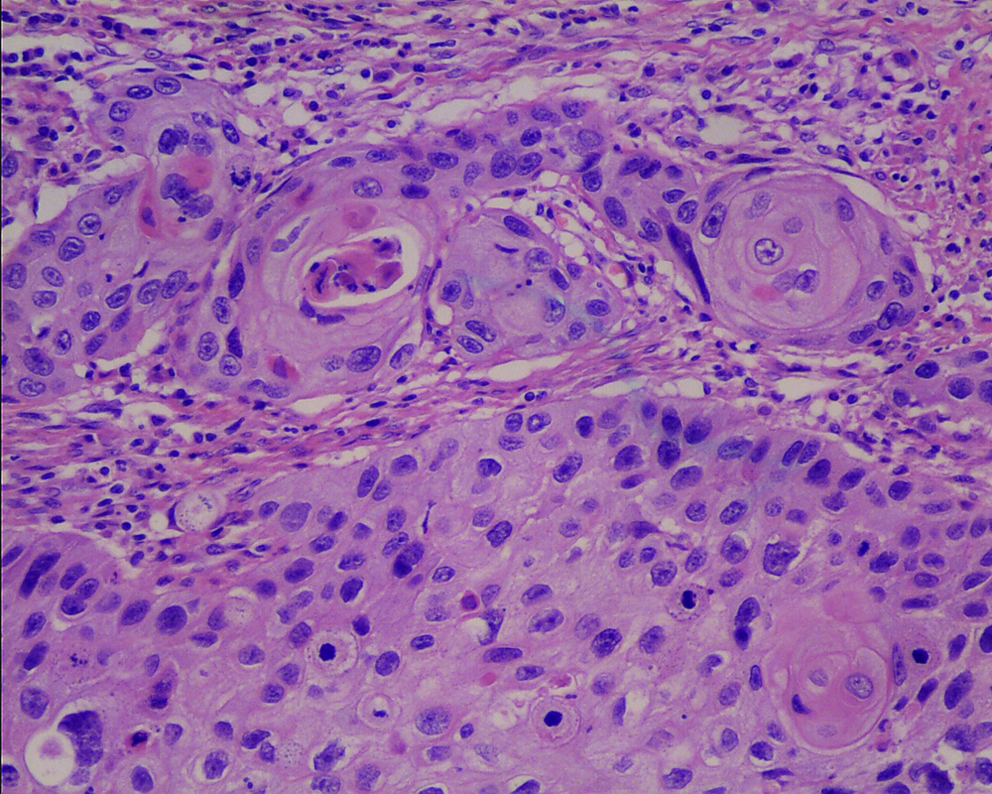
Hematoxylin and eosin staining of ESCC tissues. The tumor cells of cancerous tissues were stained violet in the nuclei and pink in the cytoplasm (× 200). ESCC, esophageal squamous cell carcinoma. Color images available online at www.liebertonline.com/gtmb.
Statistical analysis
Data of characteristics of 70 ESCC were expressed as median and percentage, and data of mRNA, protein expression, and clinicopathologic characteristics were expressed as mean ± standard deviation and analyzed using the Stata v9.0-CYGiSO bin (Computer Resource Center, Atlanta, GA). The significance of differences among groups was determined by Student's t-test and chi-square test or Fisher's exact test. Correlation between Wnt1 and β-catenin protein expression was determined by correlation test. The difference in free survival between groups was analyzed using the Kaplan-Meier curve and log-rank test. The starting point for calculating free survival was the date of surgery, and the endpoint was the date of death. A p-value of less than 0.05 was considered statistically significant.
Results
Wnt1/β-catenin mRNA expression in ESCC
The amplification curve and standard curve of β-catenin and Wnt1 are shown in Figure 2. The posttranscriptional levels of the β-catenin and Wnt1 mRNA expression in ESCC were significantly higher than that in the adjacent noncancerous tissues (p < 0.05), and the overexpression was aggressively associated with lymph node metastasis and advanced pathological stage (p < 0.05) (Tables 3-4), indicating the poor prognosis of patients with ESCC (Fig. 3A, B).
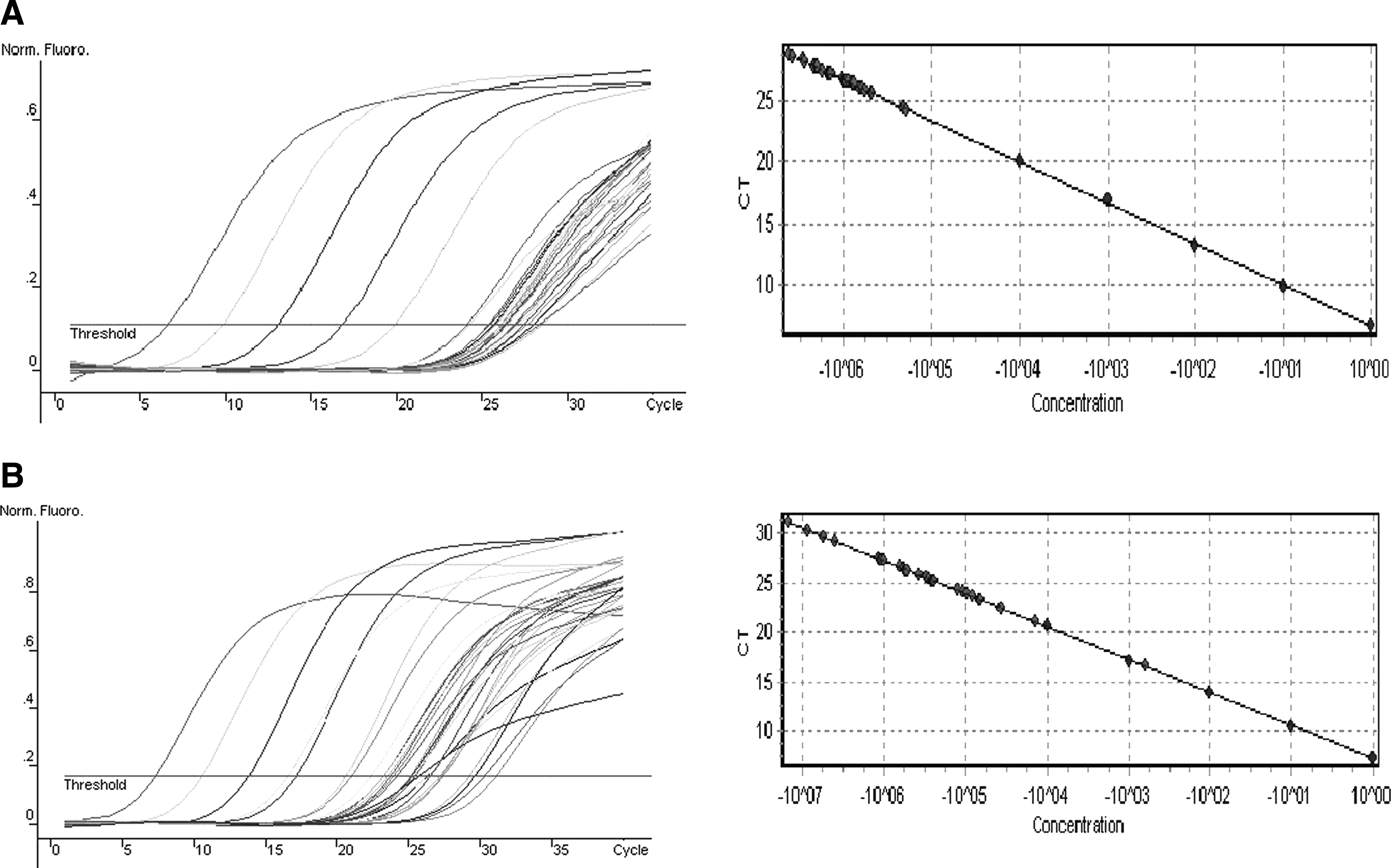
Total RNA was extracted for subsequent reverse transcription, and standard cDNA was serially diluted to obtain five standard solutions (1 × 10−1, 1 × 10−2, 1 × 10−3, 1 × 10−4, 1 × 10−5) for polymerase chain reaction to generate the reference curve. (
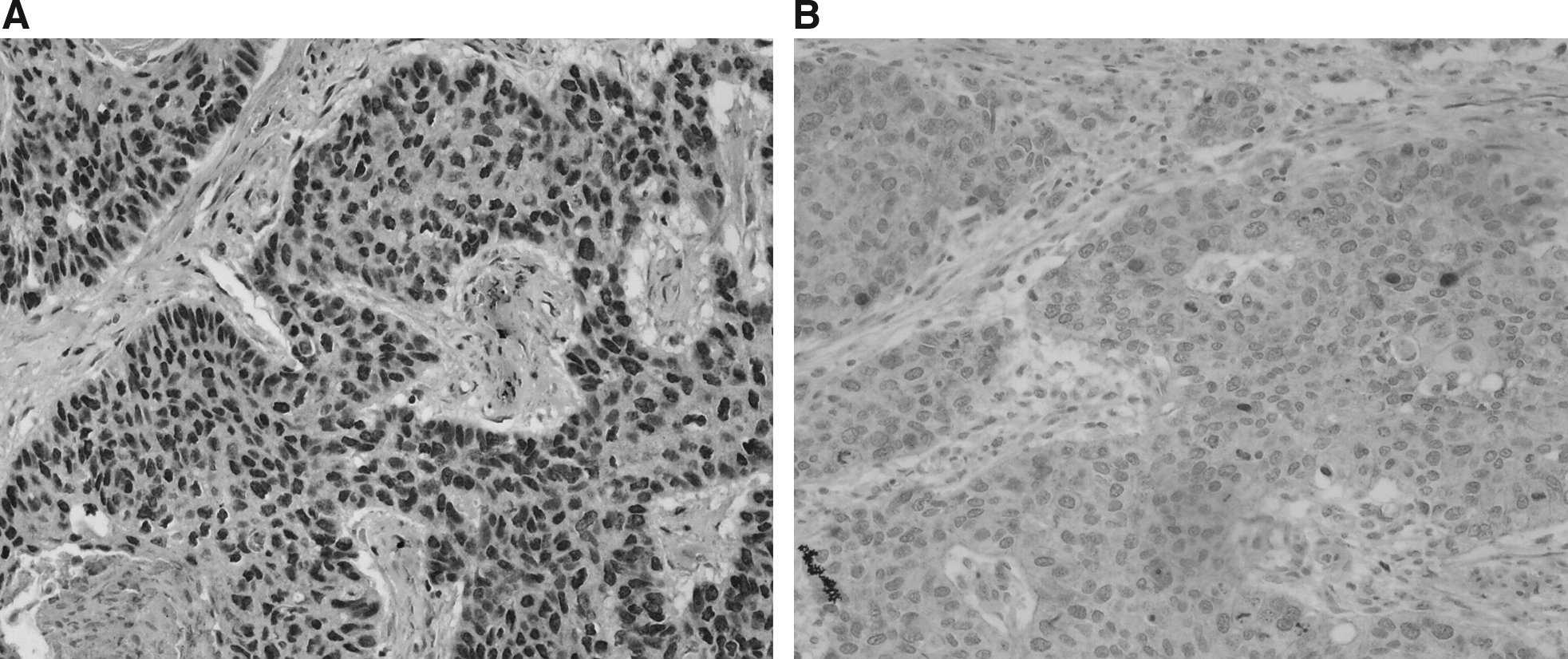
Immunohistochemical staining of esophageal squamous cell carcinoma for Wnt1 (
p1: well-differentiated compared with middle-differentiated; p2: well-differentiated compared with poorly differentiated; p3: middle-differentiated compared with poorly differentiated.
See Table 3 footnote for the definitions of p1, p2, and p3.
β-Catenin and Wnt1 protein expression in ESCC
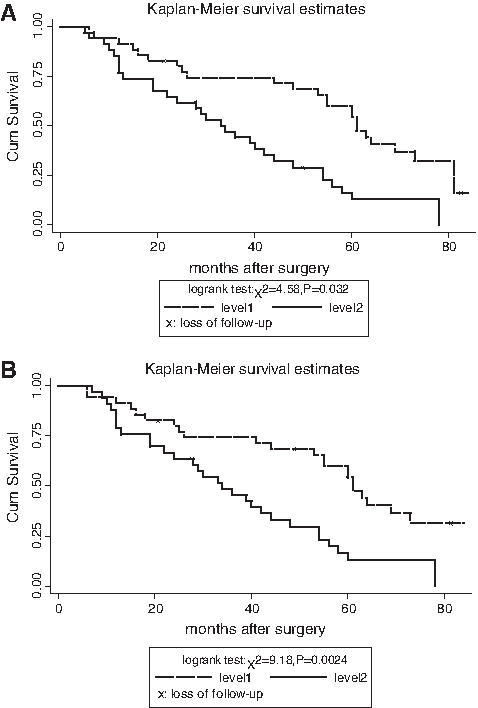
The protein expression levels of β-catenin and Wnt1 in ESCC were all significantly higher than that in the adjacent noncancerous tissues (Fig. 4); however, the overexpression was not associated with lymph node metastasis, advanced pathological stage, and cell differentiation (Tables 3 and 4). According to the median level of β-catenin protein expression (0.2491), 70 patients were divided into high and low expression groups. To the end of the follow-up, the 1-, 3-, and 5-year survival rates of the two groups showed no significant differences, and the comparison by log-rank test were χ2 = 0.7268, p = 0.3972; χ2 = 1.6758, p = 0.3027; and χ2 = 2.1085, p = 0.4460, respectively. Similarly, the median level of Wnt1 protein expression (0.3784) and the 1-, 3-, and 5-year survival rates of the two groups showed no significant differences, and the comparison by log-rank test were χ2 = 0.0901, p = 0.956; χ2 =1.8068, p = 0.613; and χ2 = 2.8068, p = 0.0795, respectively.
(
Relationship between Wnt1 and β-catenin protein expression in ESCC
As shown in Figure 5, the correlation between Wnt1 and β-catenin was not statistically significant (p = 0.6189674).
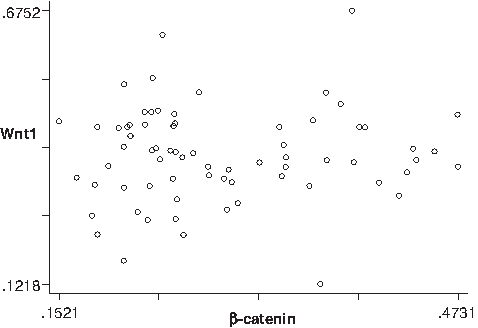
Correlation between Wnt1 and β-catenin protein expression in ESCC; r = 0.0596, t = 0.49953738, p = 0.6189674.
Discussion
According to different mechanisms and roles, Wnt signaling has been divided into typical and atypical pathways, activated by the extracellular Wnt binding the curled transmembrane receptors (James et al., 2008). In the typical Wnt pathway, Wnt binding of the curled transmembrane receptor (Fz) and the related protein family of low-concentration lipoprotein receptor could inhibit the β-catenin degradation complex composed of the adenomatous polyposis coli tumor suppressor gene, scaffolding protein Axin, glycogen synthase kinase 3β, and casein kinase I (Corrigan et al., 2009). Then β-catenin accumulates in the cytoplasm and enters the nucleus, subsequently binding the TCF/lymphoid enhancing factor family to activate Wnt target genes including c-Myc (Fillmore et al., 2009), Cyclin D1 (Egashira et al., 2009; Matsubayashi et al., 2009), vascular endothelial growth factor A (Clifford et al., 2008), and extracellular metalloprotease-7 (Tian et al., 2009), which results in changes in cellular procedures involved in the regulation of cellular proliferation and apoptosis (Dihlmann et al., 2005; Barker and Clevers, 2006; Huang et al., 2006). However, atypical Wnt signaling is implicated in a number of different downstream effectors, which are mediated by the FZD family receptor and receptor complex, such as ROR2 and RYK (Lyu et al., 2008; Winkel et al., 2008; Verkaar et al., 2009), thereby controlling cell movement and organism polarity (Herbst and Kolligs, 2007; Katoh and Katoh, 2007). Investigations have confirmed that Wnt/β-catenin would be a new molecular target for therapy of cancer (Paul and Dey, 2008). So, it is important to explore what role Wnt/β-catenin plays in ESCC.
A previous study had found that β-catenin mRNA expression was obviously abnormal in early ESCC and overexpression was implicated in poor prognosis of ESCC patients (Ji et al., 2007). Without a doubt, the present results proved it once again. Thus RT-PCR and immunohistochemical method were performed to further verify the relationship between Wnt1/β-catenin and the clinical pathology and prognosis of ESCC. At posttranscriptional level, β-catenin mRNA expression in ESCC was significantly higher than that in the adjacent noncancerous tissues (p < 0.05), and its overexpression was aggressively associated with lymph node metastasis and advanced pathological stage (p < 0.05). Confusedly, as shown in the results, although the level of β-catenin protein was significantly higher than that in adjacent noncancerous tissues of ESCC (p < 0.05), no significant relevance was shown with the pathological stage, lymph node metastasis, or cell differentiation. There was also no correlation between level of β-catenin protein and the prognosis, with the 5-year survival rate unaffected (p > 0.05). The possible reasons were as follows: (1) we did not rule out the sample selection bias or information bias; (2) we did not rule out the false-positive results from RT-PCR; (3) β-catenin was not considered as a dependent prognostic factor. So far, the relationship between abnormal expression of β-catenin and prognostic indicators in esophageal cancer remains poorly understood. Nakanishi et al. (1997) reported that β-catenin expression in esophageal cancer cell membrane decreased to 73%, and β-catenin expression was directly correlated with the pathological grade; however, no value in prognosis was observed. Krishnadath et al. (1997) reported that the rate of β-catenin weak expression in esophageal cancer was 72%; meanwhile, there were statistically significant differences between the dysfunction of β-catenin and the pathological grade (p < 0.01) and shorter survival time (p = 0. 01), indicating that the reduced β-catenin expression was correlated with poor prognosis. Otherwise, Osterheld et al. (2002) reported that, with β-catenin abnormal expression rate of 61% (43/70), β-catenin abnormal expression was not correlated with the pathological grade or lymph node metastasis (p > 0.05); however, abnormal β-catenin expression showed better prognosis. Taking into account that the above sample size was smaller and β-catenin protein content in esophageal specimens was determined by an immunohistochemical method, which might be implicated in a certain degree of subjective factors of immunohistochemical criteria, the experimental results were bound to have certain deviations. Concordance was observed between the β-catenin mRNA level and protein level in ESCC tissue by RT-PCR and immunohistochemistry in our center; nevertheless, the changes of both were discrepant with clinical pathological staging and prognosis, and so large sample and multicenter prospective randomized studies on its transcriptional level and protein level should also be further carried out to reduce the impact of bias.
Meantime, this study also showed the consistency of the expression levels of Wnt1 between the mRNA and protein, which were significantly higher than in the adjacent noncancerous tissues (p < 0.05). Combined with the high expression in the mRNA and protein level of β-catenin in ESCC, the results suggested that Wnt1 induced the transcription of β-catenin/TCF in ESCC, which was consistent with the literature (Mizushima et al., 2002). Wnt1/β-catenin plays an important role in the occurrence and development of ESCC, and so it is potentially an important molecular marker to determine disease staging and prognosis of ESCC. Further analysis showed that the overexpression of Wnt1 mRNA in the advanced stage and lymph node samples was directly correlated to the pathological stage and lymph node metastasis (p < 0.05). All these indicated that Wnt1 played an important role to a large extent in the onset rather than in the progression of ESCC. Meanwhile, it was also found that Wnt1 was not related to the tumor cell differentiation (p > 0.05). Therefore, it is reasonable to come to a conclusion that Wnt1 mRNA expression may affect the prognosis of patients with ESCC. But the interesting result was that the expression level of Wnt1 protein did not show correlation with the pathological stage or lymph node metastasis either, simultaneously, which is the same relationship as shown between the expression level of Wnt1 protein and the survival time of patients (p > 0.05). Taking into account that the abnormal expression of other nuclear genes at the protein level may be involved in the progress of ESCC, which may be interfered or in coordination with Wnt1 protein expression, the hypothesis was drawn that Wnt1 was not an independent prognostic factor, besides the possible bias for the smaller sample size.
In addition, there was no correlation between Wnt1 and β-catenin expression in the protein level (r = 0.0596). The prompt may be as following: (1) there are not only Wnt1 but also other Wnt family members that play a role in the process of ESCC; (2) Wnt1 signaling plays a role in ESCC through the typical pathway rather than the atypical, or in some proportion of both channels at the same time; (3) in the translation phase, there are other effects of target gene expression in Wnt pathway and Wnt1 expression may also be regulated by other nuclear factors.
In a word, the level of Wnt1/β-catenin mRNA is conversely correlated with advanced pathological stage and lymph node metastasis. The overexpression of Wnt1/β-catenin mRNA should be involved in poor prognosis of patients with ESCC; however, it might not be an independent prognostic factor, and it should be further verified by experiments with larger samples, more comprehensive follow-ups, and more advanced experimental techniques.
Footnotes
Acknowledgments
This study was supported by the Affiliated Nanjing First Hospital, Nanjing Medical University, and Nanjing Health Bureau (no. ZKX0114). We also appreciate Mr. Qianglin Duan for paper revision.
Disclosure Statement
No competing financial interests exist.
