Abstract
Parental care is among the most profound behavior expressed by humans and other animals. Despite intense interest in understanding the biological basis of parental behaviors, it remains unknown how much of parenting is encoded by the genome and which abilities instead are learned or can be refined by experience. One critical factor at the intersection between innate behaviors and experience-dependent learning is oxytocin, a neurohormone important for maternal physiology and neuroplasticity. Oxytocin acts throughout the body and brain to promote prosocial and maternal behaviors and modulates synaptic transmission to affect neural circuit dynamics. Recently we developed specific antibodies to mouse oxytocin receptors, found that oxytocin receptors are left lateralized in female auditory cortex, and examined how oxytocin enables maternal behavior by sensitizing the cortex to infant distress sounds. In this study we compare oxytocin receptor expression and function in male and female mice. Receptor expression is higher in adult female left auditory cortex than in right auditory cortex or males. Developmental profiles and mRNA expression were comparable between males and females. Behaviorally, male and female mice began expressing parental behavior similarly after cohousing with experienced females; however, oxytocin enhanced parental behavior onset in females but not males. This suggests that left lateralization of oxytocin receptor expression in females provides a mechanism for accelerating maternal behavior onset, although male mice can also effectively co-parent after experience with infants. The sex-specific pattern of oxytocin receptor expression might genetically predispose female cortex to respond to infant cues, which both males and females can also rapidly learn.
Introduction
Oxytocin is a peptide hormone involved in many physiological processes and especially important for maternal behaviors and pair bond formation.1–7 Oxytocin is primarily synthesized in the hypothalamic paraventricular nucleus (PVN) and supraoptic nucleus and secreted into the body from hypothalamic nerve terminals constituting the posterior pituitary.6,8–12 Hypothalamic neurons also project and release oxytocin throughout the brain,3,4,6,10–21 although it remains unclear how a peptide important for water homeostasis and milk ejection is also involved in regulation of neural circuit function and social cognition for parenting behavior. A long literature of studies in voles and other mammals indicate that the pattern of oxytocin receptor expression is an important factor related to social and parental behaviors,2–5,16–32 but it has largely remained unclear which cells or synapses express oxytocin receptors and are directly modulated by oxytocin.
Thus it has been challenging to relate the physiological effects of oxytocin receptor signaling to behavioral consequences. This confounds attempts to understand and optimize if or how oxytocin might be used in humans to improve social behavior, including therapeutic treatments for psychiatric conditions such as autism spectrum disorders or postpartum depression.6,33–38
To understand how oxytocin influences parental behaviors—or social cognition more broadly—requires robust animal models of maternal and paternal interactions. A remarkable series of studies in rodents has demonstrated that oxytocin can enable or induce pair bond formation and maternal behavior, either when directly infused into the brain or systemically injected.18,19,22,25 Parental behavior such as retrieval of isolated pups, nest building, and nursing are relatively straightforward to document and reliably expressed by experienced maternal mammals.
In contrast, paternal behavior in male rodents is less clear, with reports of infanticide or neglect unless males are mated, cohoused with experienced females, and/or have undergone physiological changes to steroid hormone or hypothalamic peptide systems, for example, cells expressing galanin or vasopressin.39–47 Male mice and other mammals, including humans, express oxytocin receptors and experience heightened levels of plasma oxytocin during some social interactions.6,16,48–52 However, it remains unclear whether there are important differences in the organization of the oxytocin system in females and males, and how oxytocin might be involved in establishing paternal behavior.
We recently generated new antibodies specific to the mouse oxytocin receptor, to determine where and when oxytocin receptors are expressed with increased precision and help understand the functional consequences of oxytocin receptor activation for synaptic transmission and neural signaling. 21 We showed that both endogenous oxytocin (released optogenetically) and exogenous oxytocin (either injected systemically or directly infused into left auditory cortex) could enable the initial expression of pup retrieval behavior. In pup-naive virgin female mice cohoused with an experienced dam and litters, oxytocin accelerated retrieval onset and increased the overall number of female virgins expressing this alloparenting behavior. 18
In this study we now use these antibodies to compare the pattern of oxytocin receptor expression in males to females, throughout development and in adults. We also examine changes in gene expression and parental behavior after oxytocin supplements, contrasting sex-specific functional effects that might depend on differential receptor expression in males and females. We focus on the auditory cortex given the importance of this brain area for learning to recognize the behavioral significance of mouse pup isolation distress calls. 18
Materials and Methods
All procedures were approved under NYU IACUC protocols. In this study we report results from a total of 134 female and 55 male C57BL/6 mice; of which 43 males and 23 females were not previously described in our past studies of oxytocin receptor expression and function.18,21
Immunohistochemistry
OXTR-2 antibodies were generated and validated as previously described.18,21 For immunohistochemical analysis with light microscopy, wild-type or oxytocin receptor knockout C57BL/6 mice were anesthetized using intraperitoneal (IP) injection (0.1 mL per 10 g) of a ketamine–xylazine mixture containing 15 mg/mL ketamine and 5 mg/mL xylazine in 0.9% sodium chloride solution. Mice were perfused intracardially with a solution of heparin (1000 U/mL) and PBS to prevent clotting, followed by 40 mL per mouse of freshly prepared 4% paraformaldehyde in 10 mM phosphate buffer. The brains were carefully removed and postfixed in 4% paraformaldehyde for 2 h at 4°C and then cryoprotected overnight in 30% sucrose at 4°C. The brains were then embedded in a cryoprotectant medium (Tissue-Tek, Optimum Cutting Temperature medium, VWR) and stored at −80°C until sectioned. Coronal 16 μm sections were cut on a cryostat and collected on Superfrost Plus glass slides (Fisher Scientific).
Sections were washed in PBS and blocked for 2–3 h at room temperature in PBS containing 0.2% v/v Triton X-100 and 5% v/v normal donkey serum. The blocking solution was aspirated, and the sections were incubated with oxytocin receptor primary antibody diluted in block to a concentration of 1 μg/mL. Sections were incubated for 2 days at 4°C in a moist chamber. Sections were washed with PBS (3 × 15 min at room temperature) in a staining jar and incubated for 1–2 h at room temperature in Alexa Fluor-conjugated secondary antibodies diluted 1:500 in PBS. Any unbound secondary antibody was washed with PBS (3 × 15 min at room temperature), and sections were incubated for 15 min at room temperature with a DAPI solution (1:10,000 stock diluted in PBS) for nuclear staining.
After a final rinse, the slides were coverslipped using Fluoromount G (Southern Biotechnology Associates, Inc.). The brains of wild-type and knockout animals were processed together to minimize any confounding factors, and parallel sections from knockout animals served as controls. As a control, omission of primary antibody eliminated immunofluorescent labeling from that channel.
Slides were examined and imaged using a Carl Zeiss LSM 700 confocal microscope with four solid-state lasers (405/444, 488, 555, 639 nm) and appropriate filter sets. The distribution and number of immunoreactive cells in each section were determined by taking images of wild-type and knockout sections under the same laser power output, pinhole aperture, and gain. Images were collected and saved for manual counts by two to five independent blinded observers. Maximum intensity projections of image stacks are shown, each representing at least six to eight distinct optical planes.
Next-generation sequencing of total RNA
Frozen brains from six animals in each age group (three male, three female) were sectioned in the coronal plane (rostral to caudal) on a sliding microtome and viewed through a surgical microscope.21,53 As areas targeted for sampling became visible, they were extracted using a sterile tissue punch or curette of a size appropriate to the brain region. Samples of auditory cortex were obtained using a 0.5 mm diameter punch, with the ventral edge beginning ∼1 mm dorsal to the rhinal fissure. Samples of medial geniculate body (MGB) were harvested with a curette after using a microdissecting scalpel to circumscribe its perimeter, and the microdissection procedure was designed to exclude the lateral geniculate nucleus and adjoining nuclei dorsal, medial, and ventral to the MGB. The extreme rostral and caudal poles of the MGB were largely excluded from these samples. Punches from homologous areas of both hemispheres were combined in sterile tube containing 400 μL of TRIzol, homogenized for 45 s using a mechanized sterile pestle, flash frozen on dry ice, and then stored at −80°C.
For each TRIzol lysate, 100 μL of Reagent Grade Chloroform (Fisher Scientific; S25248) were added. Samples were centrifuged for 3 min on a desktop centrifuge to fractionate the aqueous and organic layers. After centrifugation, the resulting aqueous layer was carefully removed and transferred to 2.0 mL Sarstedt tubes (Sarstedt; 72.694) were run on the QIAsymphony using the QIAsymphony RNA Kit (Qiagen; 931636) and protocol RNA_CT_400_V7, which incorporates DNAse treatment. Before each run, the desk was uv-irradiated using the programmed cycle. The resulting RNA was eluted to 100 μL of RNase free water and stored at −80°C in 2.0 mL Sarstedt tubes until use. Samples were initially quantitated using a Qubit RNA assay. Additional analyses of purity and quantitation of total RNA were performed using a NanoDrop spectrophotometer (Thermo Scientific) and Agilent RNA 6000 Pico chip (Agilent) according to the manufacturer's protocol using reagents, chips, and ladder provided in the kit.
RNA-seq was performed by the Vanderbilt Technologies for Advanced Genomics core (VANTAGE) as previously described.21,53 Total RNA was isolated with the Aurum Total RNA Mini Kit. All samples were quantified on the Qubit RNA assay. RNA quality was verified using an Agilent Bioanalyzer. RNA-seq data were obtained by first using the Ribo-Zero Magnetic Gold Kit (Human/Mouse/Rat) (Epicentre) to perform ribosomal reduction on 1 μg total RNA following manufacturer's protocol. After ribosomal RNA (rRNA) depletion, samples were purified using the Agencourt RNAClean XP Kit (Beckman Coulter) according to the Epicentre protocol specifications. After purification, samples were eluted in 11 μL RNase-free water.
Next, 1 μL ribosomal depleted samples were run on the Agilent RNA 6000 Pico Chip to confirm rRNA removal. After confirmation of rRNA removal, 8.5 μL rRNA-depleted sample was input into the Illumina TruSeq Stranded RNA Sample Preparation Kit (Illumina) for library preparation. Libraries were multiplexed six per lane and sequenced on the HiSeq 2500 to obtain at least 30 million paired end (2 × 50 bp) reads per sample.
RNA-seq data went through multiple stages of quality control as previously described. 21 Normalized read counts were averaged over all samples for each age (P7, P14, P21, adult) and brain region (auditory cortex, auditory thalamus). ANOVA with Tukey post hoc testing was used to screen for significant differences in expression between ages for each area and gene.
Mass spectrometry
Dams and male C57BL/6 mice between 2–4 months of age were used for injection of oxytocin and saline (N = 12 mice total, 3 of each sex for each condition). For both sexes, the left auditory cortex was injected with 10 μM oxytocin and the right auditory cortex with 0.9% saline as vehicle control, under general anesthesia with 2% isoflurane. The injection volume for oxytocin and the vehicle was 1 μL over a period of 15 min (66 nL/min), respectively. After a total incubation time of 30 min, animals were decapitated, and the brain was extracted and flash frozen in liquid-nitrogen cooled isopentane in OCT and stored overnight at −80°C. Brains were sectioned using a cryostat. Sections at a thickness of 10 μM encompassing the auditory cortices on both hemispheres were mounted on glass slides with a polyethylene naphthalate membrane (PEN) (26 × 76 mm; Leica) and stored at −80°C.
Laser capture microdissection was performed using a LMD6500 microscope (Leica). Oxytocin, as well as vehicle injected auditory cortices, was collected separately in 0.5 mL PCR tubes. Protein extraction was performed using a RIPA buffer (1% (v/v) Triton X-100, 1% (v/v) SDS, 50 mM Tris-HCL pH 7.4, 500 mM NaCL, 1 mM EDTA) and 10 × aspiration using a U-100 Insulin Syringe (28G1/2, Becton Dickinson) for each sample. The samples were centrifuged at 16,900 g to remove cellular debris and PEN particles. Samples were denatured using 4 × Laemmli buffer (250 mM Tris-HCL pH 6.8), 8% SDS, 40% glycerol, 8% beta-Mercaptoethanol, and 0.02% bromophenol blue (Boston BioProducts) and run on 12% SDS-PAGE for subsequent mass spectrometry analysis.
Approximately 20–50 μg of each sample was concentrated by running briefly on 15% SDS/PAGE gels. Gels were washed 3 × in ddH2O for 15 min each and visualized by staining overnight with GelCode® Coomassie blue reagent (Pierce). The bands were excised from each gel, cut into slices, and reduced with DTT and alkylated with iodoacetamide. In-gel digestion was performed using mass spectrometry grade trypsin (Trypsin Gold; Promega, Madison, WI) at 5 ng/μL of 50 mM NH4HCO3 digest buffer. The resulting peptides were desalted using Sep-Pak tC18 1 cc Vac Cartridge (Waters, #WAT03820).
Tandem mass tag (TMT) labeling and the remaining proteomics protocol were carried out as previously described 54 with some modifications. Peptides were resuspended in 18 μL acetonitrile, and 262 μL of 0.2 M HEPES buffer pH 8.5 were added to each. TMT10plex amine reactive reagents (5 mg per vial) (Thermo Fisher Scientific) were resuspended in 1024 μL of anhydrous acetonitrile, and 24 μL of each reagent were added to each sample (TMT label:peptide [w/w] = 6:1) and mixed briefly on a vortexer. The mixture was incubated at room temperature for 1 h, quenched by the addition of 25 μL of 5% hydroxylamine for 15 min, and then acidified by the addition of 30 μL 10% formic acid. A small aliquot from each reaction was desalted on a Stage Tip manually packed with Empore C18 High Performance Extraction Disks (3 M; St. Paul, MN) and eluted peptide solutions partially dried under vacuum then analyzed by LC-MS/MS with a Q Exactive High Field Orbitrap mass spectrometer. 55 The data were searched in MaxQuant using its corresponding TMT label as variable modifications on N-terminus and lysine. The percentage of peptides with either N-terminal or lysine TMT labels was calculated, representing the labeling efficiency in each channel. To ensure that equal amounts of labeled peptides from each channel were mixed together, a two-step mixing strategy was used; in the first step, a small (∼5 μL) and identical volume of peptides from each channel was mixed and analyzed, and the value of the median ratio (defined by the median of the ratios of all peptide intensities of one channel over their corresponding peptide average intensities of all channels) for each channel was determined as the correction factor. In the second step, the 7-plex mixed channels were prepared by adjusting their volume using the correction factors. In this way, a median ratio ranging from 0.9 to 1.1 was achieved. The mixture of reaction products from seven TMT channels was desalted on a Sep-Pak tC18 1 cc Vac Cartridge. Eluted peptides were dried and stored at −20°C. We prepared one 7-plex mix of peptides from two female mice and one male mouse and a second one that contained peptides from two male mice and one female mouse, each contributing right and left side of brain samples, annotated as 7-plex 1 and 7-plex 2, respectively. The seventh TMT channel in each mix was used as the reference channel consisting of a pool of all peptide samples.
TMT-labeled peptides were dissolved in 90% acetonitrile with 0.1% TFA. The TMT peptide mix was briefly centrifuged and injected on the HILIC column composed of TSK gel amide-80, 4.6 mm ID × 25 cm long, with 5 μm beads from TOSOH Bioscience, LLC. Peptides were eluted over a 65 min HPLC gradient with 300 μL flow rate. Peptides were collected every 4 min. Approximately 16 fractions were generated; 10% of each fraction was analyzed for quantitative analysis of the total proteome, and the remaining material was subjected to phosphopeptide enrichment step using Titanium dioxide chromatographic media (TiO2 beads; GL Sciences, Inc., Japan) as previously described. 54
Online chromatography was performed with a Thermo Easy nLC 1000 UPLC system (Thermo Fisher Scientific) coupled online to a Q Exactive HF with a NanoFlex source (Thermo Fisher Scientific). Analytical columns (∼30 cm long and 75 μm inner diameter) were packed in-house with ReproSil-Pur C18-AQ 3 μM reversed-phase resin (Dr. Maisch GmbH, Ammerbuch-Entringen, Germany).
The analytical column was placed in a column heater (Sonation GmbH, Biberach, Germany) set to a temperature of 45°C. Peptide mixtures were loaded onto the analytical column with buffer A (0.1% formic acid) at a maximum back pressure of 300 bar; peptides eluted with a 2-step gradient of 3% to 40% buffer B (100% ACN and 0.1% formic acid) in 180 min and 40% to 90% B in 20 min, at a flow rate of 250 nL/min over 200 min using a 1D online LC-MS2 data-dependent analysis method as follows: mass spectrometry data were acquired using a data-dependent top-10 method, dynamically choosing the most abundant not-yet-sequenced precursor ions from the survey scans (300–1750 Th).
Peptide fragmentation was performed using higher energy collisional dissociation with a target value of 1 × 105 ions determined with predictive automatic gain control. Isolation of precursors was performed with a window of 1 Th. Survey scans were acquired at a resolution of 120,000 at m/z 200. Resolution for HCD spectra was set to 60,000 at m/z 200 with a maximum ion injection time of 128 ms. The normalized collision energy was 35. The “underfill ratio,” specifying the minimum percentage of the target ion value likely to be reached at the maximum fill time, was defined as 0.1%. We excluded precursor ions with single, unassigned, or seven and higher charge states from fragmentation selection. Dynamic exclusion time was set at 30 s.
All data were analyzed with the MaxQuant proteomics data analysis workflow (version 1.6.0.1) with the Andromeda search engine.56,57 The type of LC-MS run was set to “Reporter ion MS2” with “10plex TMT” as isobaric labels for Q Exactive MS2 data. Reporter ion mass tolerance was set to 0.01 Da, and the false discovery rate set to 1% for protein, peptide spectrum match, and site decoy fraction levels. Peptides were required to have a minimum length of eight amino acids and a maximum mass of 4600 Da. MaxQuant was used to score fragmentation scans for identification based on a search with an allowed mass deviation of the precursor ion of up to 4.5 ppm after time-dependent mass calibration. The allowed fragment mass deviation was 20 ppm. MS2 spectra were used by Andromeda to search the UniProt mouse database (16,729 entries).
Enzyme specificity was set as C-terminal to arginine and lysine, and a maximum of two missed cleavages was allowed. Carbamidomethylation of cysteine was set as a fixed modification, and N-terminal protein acetylation, deamidated (N, Q), and oxidation (M) were set as variable modifications. The reporter ion intensities were defined as intensities multiplied by injection time (to obtain the total signal) for each isobaric labeling channel summed over all MS/MS spectra matching to the protein group as previously validated. 57 Following MaxQuant analysis, the protein and peptide .txt files were imported into Perseus (version 1.6.0.2) software, which was used for the statistical analysis of all the proteins identified. The basic statistics used for significance analysis was the moderated t-statistics. 58 Benjamini–Hochberg correction was used to calculate the adjusted p-values.
Parental behavior
Two- to four-month-old C57BL/6 pup-naive virgin males, pup-naive virgin females, and dams were used for cohousing and analysis of pup retrieval and nest building. Male mice were raised with male littermates and had normal social interactions.43–45 Male and female mice were bred on-site and weaned at P10, then housed in a separate environment not containing any dams or pups before cohousing. Virgin mice (either one male or one female) were then cohoused with a dam and litters ranging from P0-10. Dams were initially prescreened to ensure that they retrieved pups; ∼1% of dams did not retrieve pups, and these animals were not used for cohousing. Naive virgins were initially prescreened for retrieval or pup mauling before cohousing; <30% of naive female virgins retrieved at least one pup or mauled pups during prescreening, while none of the males retrieved pups and only one male mauled pups before cohousing. These animals were excluded from subsequent behavioral studies.
Pup retrieval testing was performed as before, 18 in a similar manner for cohoused males or females. Testing included two parts: a habituation period and ∼10 retrieval trials per testing session. During the habituation period, the animal was exposed to a novel behavioral arena (38 × 30 × 15 cm) with bedding and cotton balls for nest material. After 5–20 min (20–30 min on first testing session per animal), several pups from the cohousing home cage were introduced into the arena and placed in one corner together with cotton balls as a nest. In each retrieval trial, one pup was removed from the nest and placed in a different corner. Each trial lasted for 2 min and time to retrieval scored; if the animal did not retrieve after 2 min, the pup was put back in the nest, and another trial was initiated with a new pup. After ten trials were completed, the adult male or female was injected IP with oxytocin (20–50 μM in saline) or saline alone at a volume of 0.3 mL per injection; animals were given 5–10 min to recover and retested for another 10 trials. Animals were then placed with the pups back in to the cage to continue cohousing.
Retrieval testing was performed at 1 h, 3 h, 6 h, 12 h, 24 h (1 day), 36 h, 48 h (2 days), 72 h (3 days), 96 h (4 days), 120 h (5 days), and 144 h (7 days). Animals in the oxytocin group received IP injections of oxytocin at each of these testing timepoints; animals in the saline group received IP injections of saline at each testing timepoint. We used an ultrasonic microphone (Avisoft) to verify that isolated pups vocalized during testing. Nest building by males was also monitored during each entire testing session. We defined nest building as animals biting and bringing the cotton balls near the pups as nesting material. Power analysis was performed to determine sample size for statistical significance with a power of β:0.7; these studies required at least six animals. Fisher's two-tailed exact test was used for comparing numbers of animals retrieving in each group as these data were binomial.
Results
Oxytocin receptor expression in left auditory cortex is higher in females than males
We first asked how oxytocin receptor expression differs between male and female mouse auditory cortex. Auditory cortical activity is required for maternal responses to ultrasonic pup isolation calls, 18 indicating that this brain area might be especially sensitive to oxytocin modulation and important for processing social vocalizations. Previously we generated novel and specific antibodies to the mouse oxytocin receptor 21 and identified an unusual left lateralization of oxytocin receptor expression in female mouse auditory cortex that might promote neural plasticity for recognizing the behavioral meaning of pup calls. 18 However, little is known about the oxytocin receptor expression profile in male cortex, or the relation of oxytocin receptor signaling to male mouse paternal behavior.
We used the most specific antibodies we generated, OXTR-2, to label oxytocin receptors in male mouse left and right auditory cortex (Fig. 1a). We also verified that OXTR-2 was specific for oxytocin receptors in male oxytocin receptor knockout mouse brain (Fig. 1a). OXTR-2 expression was not significantly different across hemispheres in male auditory cortex (Fig. 1b; male left vs. right OXTR-2+ cells: p = 0.866), in contrast to the significant left lateralization of OXTR-2 expression in dams and virgin females (Fig. 1b; OXTR-2+ cells in left vs. right auditory cortex of dams: p = 0.027 and virgin females: p = 0.001). Interestingly, this lateralization was due to a higher absolute level of OXTR-2+ cells in female left auditory cortex than female right auditory cortex or either hemispheres in males (Fig. 1c). In other words, male left and right auditory cortex had a similar (and nonzero) level of oxytocin receptor expression as female right auditory cortex. Consequentially, this suggests that oxytocin might have some function in male auditory cortex and additionally provides an opportunity to probe the functional significance of left-lateralized receptor expression in female auditory cortex.
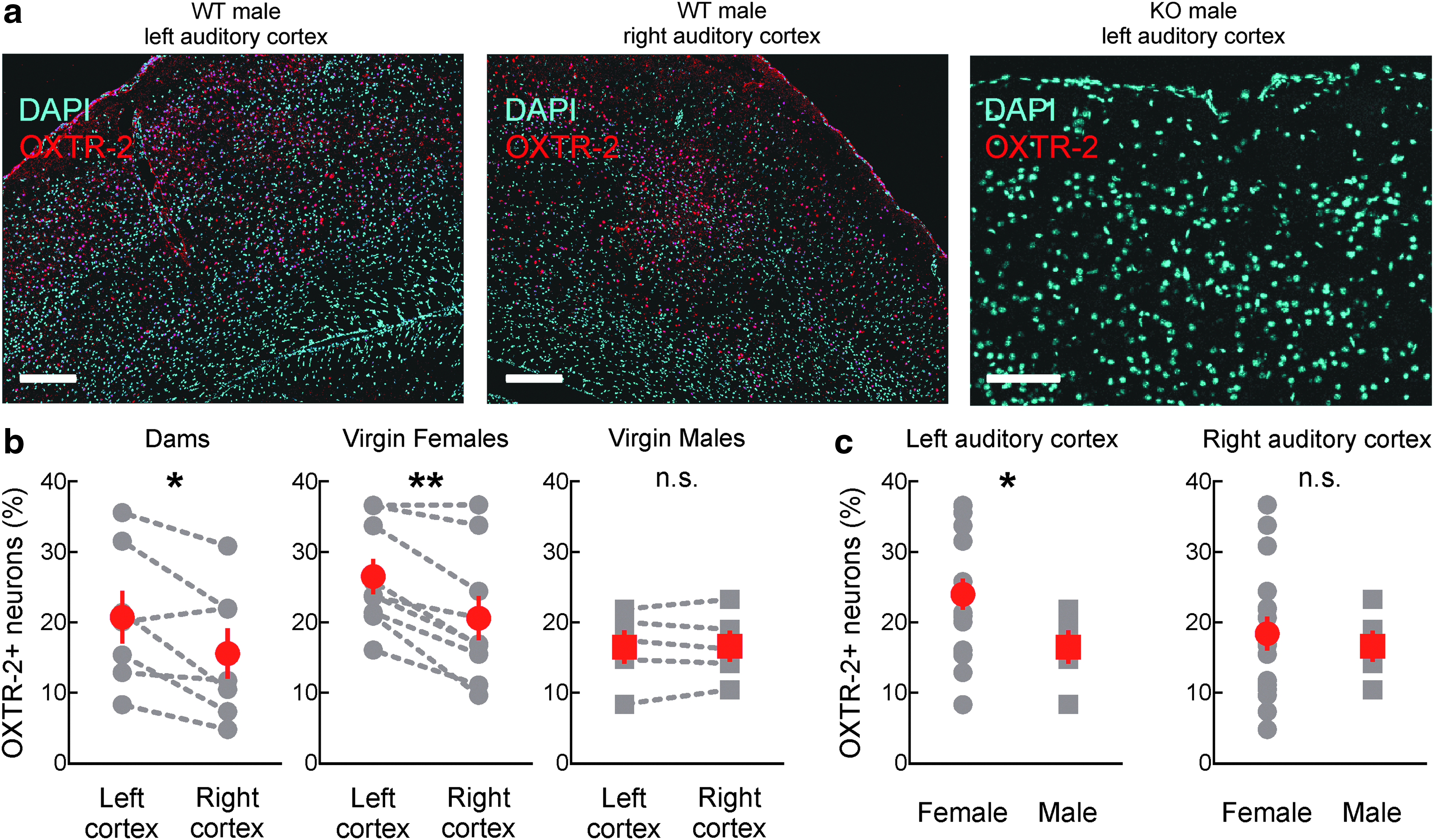
Oxytocin receptor expression is higher in female mouse left auditory cortex than right or male auditory cortex.
Cortical oxytocin receptor lateralization in females is developmentally regulated
Next we wondered if the difference in male and female left auditory cortex oxytocin receptor expression was present at birth or emerged over development. Previous studies have shown that oxytocin receptor expression in mouse cortex is highest during the second postnatal week, increasing from birth and then subsequently decreasing to substantially lower levels. In parallel, receptor expression begins highest in superficial and deep cortical layers and then increases in layer 4.7,21,59 Using OXTR-2 antibodies in tissue sections from animals at the second postnatal week, we found a similar pattern in males. Specifically, the relative fraction of cells expressing oxytocin receptors was much higher in the young brain and comparable between males and females (Fig. 2a).
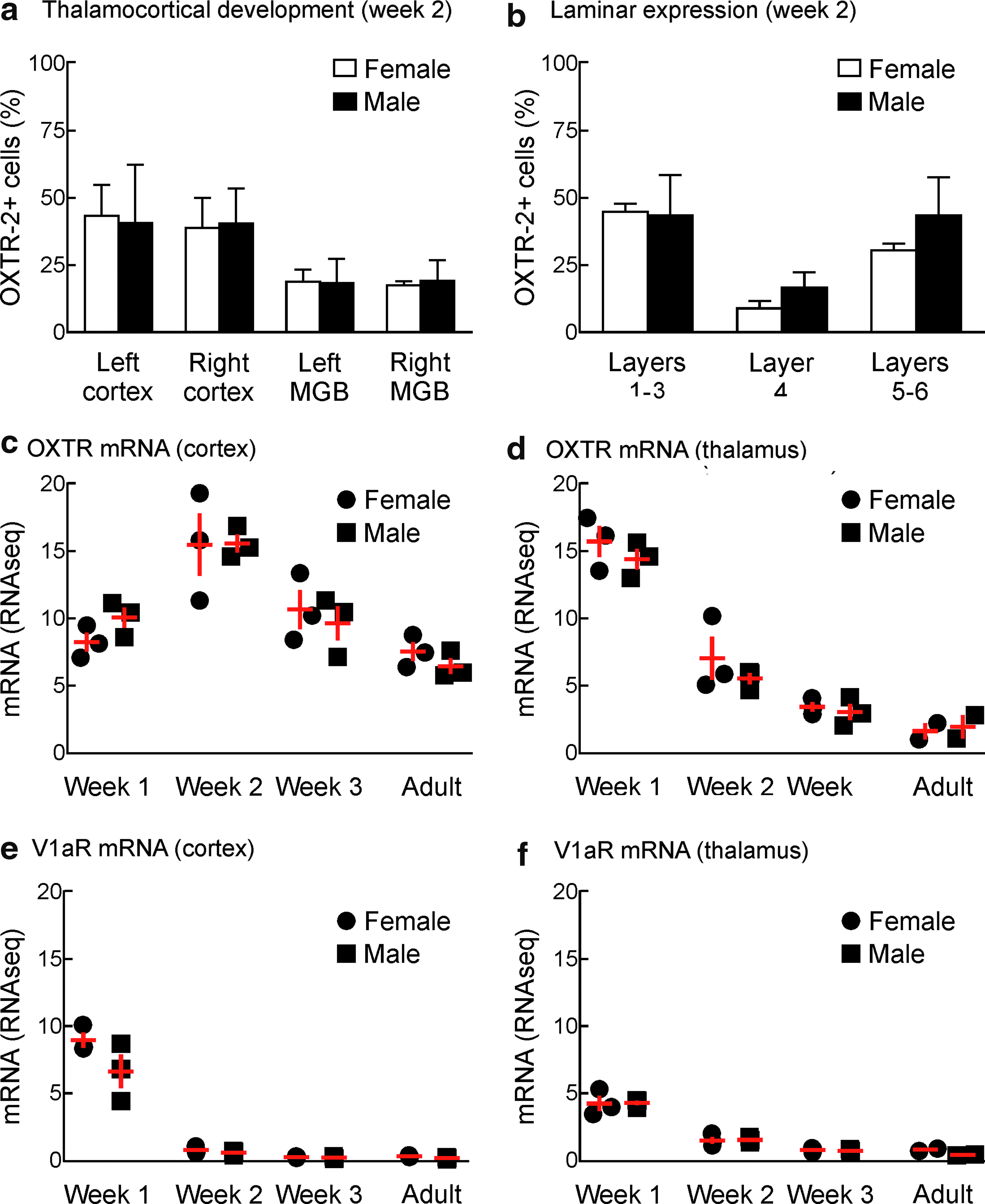
Asymmetric cortical oxytocin receptor expression in females emerges over postnatal development.
The laminar organization of receptor expression was also similar in females and males at the second postnatal week, with layer 4 having the fewest number of cells expressing oxytocin receptors (Fig. 2b). This indicates that the lateralization of oxytocin receptor expression evident in the adult female auditory cortex is not apparent at earlier ages when receptor expression levels are highest. Instead, over the first postnatal month, receptor expression drops in male and female left and right auditory cortices, but decreases less in female left auditory cortex compared to greater decreases in female right auditory cortex and male cortex across hemispheres.
We also performed next-generation sequencing to describe the profiles of oxytocin receptor mRNA in male and female auditory cortex (Fig. 2c) and auditory thalamus (Fig. 2d) over postnatal development. When pooled across hemispheres, there was no significant difference between males and females in terms of the developmental trajectories of receptor mRNA levels. In both males and females, relative mRNA levels were lower in the cortex during the first postnatal week, increased during the second postnatal week, and then decreased to adult levels thereafter (Fig. 2c). In contrast, in the thalamic MGB, receptor mRNA levels were initially high in the first postnatal week and then declined (Fig. 2d). These patterns of neurohormone receptor mRNA regulation were specific for oxytocin, as quantification of mRNA for the vasopressin V1a receptor revealed almost zero expression beyond the first postnatal week in cortex (Fig. 2e) and thalamus (Fig. 2f). Therefore, while oxytocin can cross-react and signal through vasopressin receptors in other systems, this is unlikely to occur in the auditory cortex due to lack of vasopressin receptor expression. We also note that while receptor expression depends on mRNA, other cellular factors (e.g., related to mRNA/protein degradation and/or posttranscriptional regulation) are also important for determining how mRNA abundance leads to functional expression of oxytocin receptors.60,61
Proteomic effects of oxytocin
Previously we have examined the physiological effects of oxytocin on synaptic transmission in the auditory cortex and other brain areas important for social behavior.18,21 However, little is known about the signaling systems engaged by G protein-coupled oxytocin receptor activation in the brain. To document the functional consequences of oxytocin receptor signaling, we performed tandem mass tag mass spectrometry and total, as well as phosphoproteomic, analysis.54–58 This is an unbiased approach that can reveal changes in protein expression levels and phosphorylation events across the proteome. We infused oxytocin directly into the auditory cortex of male and female adult mice in vivo, prepared tissue samples with laser microdissection for mass spectrometry, and compared the effects of elevating oxytocin levels in left female versus male auditory cortex.
We first examined changes in total gene expression induced by oxytocin infusion. Out of 3571 proteins, changes in protein levels were reliably quantified for 2696 proteins for which we had multiple samples. While changes in protein level were minimally different in oxytocin-treated male left versus saline-treated right auditory cortex (Fig. 3a), we noticed a strong asymmetry in females, with many more proteins upregulated in left auditory cortex after oxytocin infusion (Fig. 3b; points above the dashed unity line). In total, the expression of 367 proteins was significantly different between female and male left auditory cortex (Fig. 3c and Table 1), including synaptic vesicle proteins such as synaptophysin and VGLUT-2, and components of signal transduction pathways such as PKA.
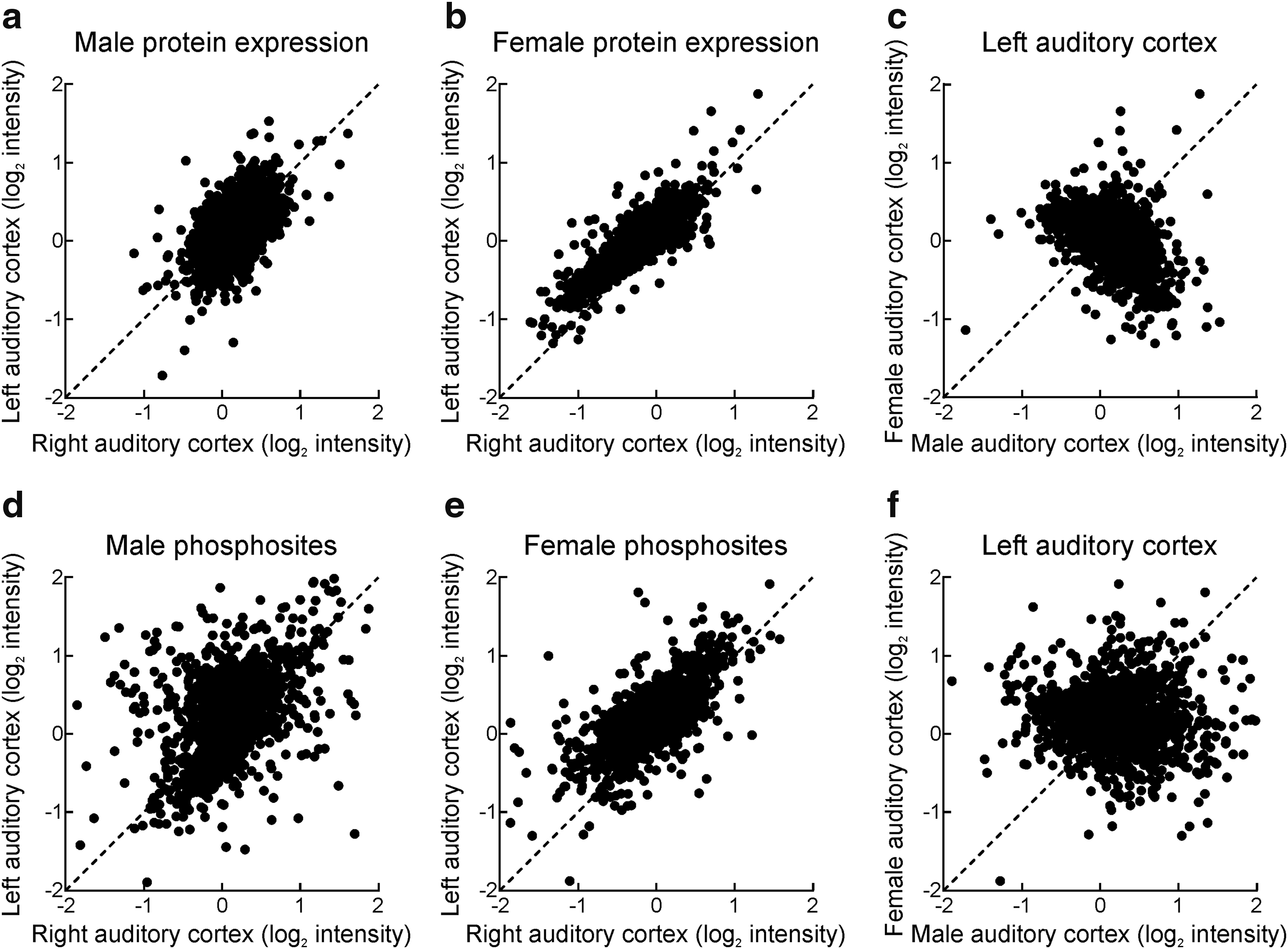
Proteomic analysis of adult mouse auditory cortex after oxytocin treatment.
List of Proteins Differentially Expressed in Female Versus Male Left Auditory Cortex After Oxytocin Infusion
We then examined phosphorylation events occurring after oxytocin receptor activation. We examined 1325 out of a total of 1794 serine, threonine, and tyrosine phosphosites identified in the combined samples and focused our analysis on 468 phosphosites identified in the complete sample set, because these 468 sites were quantified in all of the replicates. As with total protein expression, phosphorylation occurred symmetrically in male left versus right auditory cortex (Fig. 3d), but was asymmetrical in females such that more phosphorylation occurred in female left than right auditory cortex (Fig. 3e). After Student's t-test with Benjamini–Hochberg correction, there were eight sites with statistically-significant differences in phosphorylation (p < 0.05) between female and male left auditory cortex, including a reduction in phosphorylation of female CaMKIIβ subunits and an increase on clathrin heavy chain 1 (Fig. 3f and Table 2). Thus there are molecular and functional differences between oxytocin receptor activation in left female auditory cortex and other auditory cortical circuits in right female cortex and in males. This lateralized program of phosphorylation and protein expression might relate to the overall higher expression level of oxytocin receptors in female left auditory cortex and also might be related to the heightened plasticity in this brain region for infant vocalizations. 18
List of Phosphosites Differentially Phosphorylated in Female Versus Male Left Auditory Cortex After Oxytocin Infusion
Oxytocin supplements enhance female but not male parental behavior
Given these anatomical and molecular differences, we asked whether oxytocin had similar effects on males and females for enhancing parental behavior. After weaning, mice were isolated from pups and raised to adulthood. Individual adult pup-naive males or females were screened to ensure that they did not initially retrieve pups and then cohoused continuously with litters and dams that were verified to reliably retrieve pups. Retrieval abilities and other types of parental behavior were assessed at regular intervals over a week of cohousing, with some animals receiving systemic oxytocin after each testing session and other animals receiving saline injections.
About half of all male C57BL/6 mice cohoused with experienced dams began retrieving pups after several days (Fig. 4a). Individual male mice began retrieving at various times, with a minority retrieving after <6 h of cohousing (∼15%), and other males requiring more cohousing to begin expressing this behavior. In contrast to the effects of oxytocin treatment on female mice, 18 oxytocin injections did not affect the time course of retrieval. Males receiving oxytocin (Fig. 4a, red) and males receiving saline injections (Fig. 4a, black) began expressing retrieval behavior at approximately the same rates.
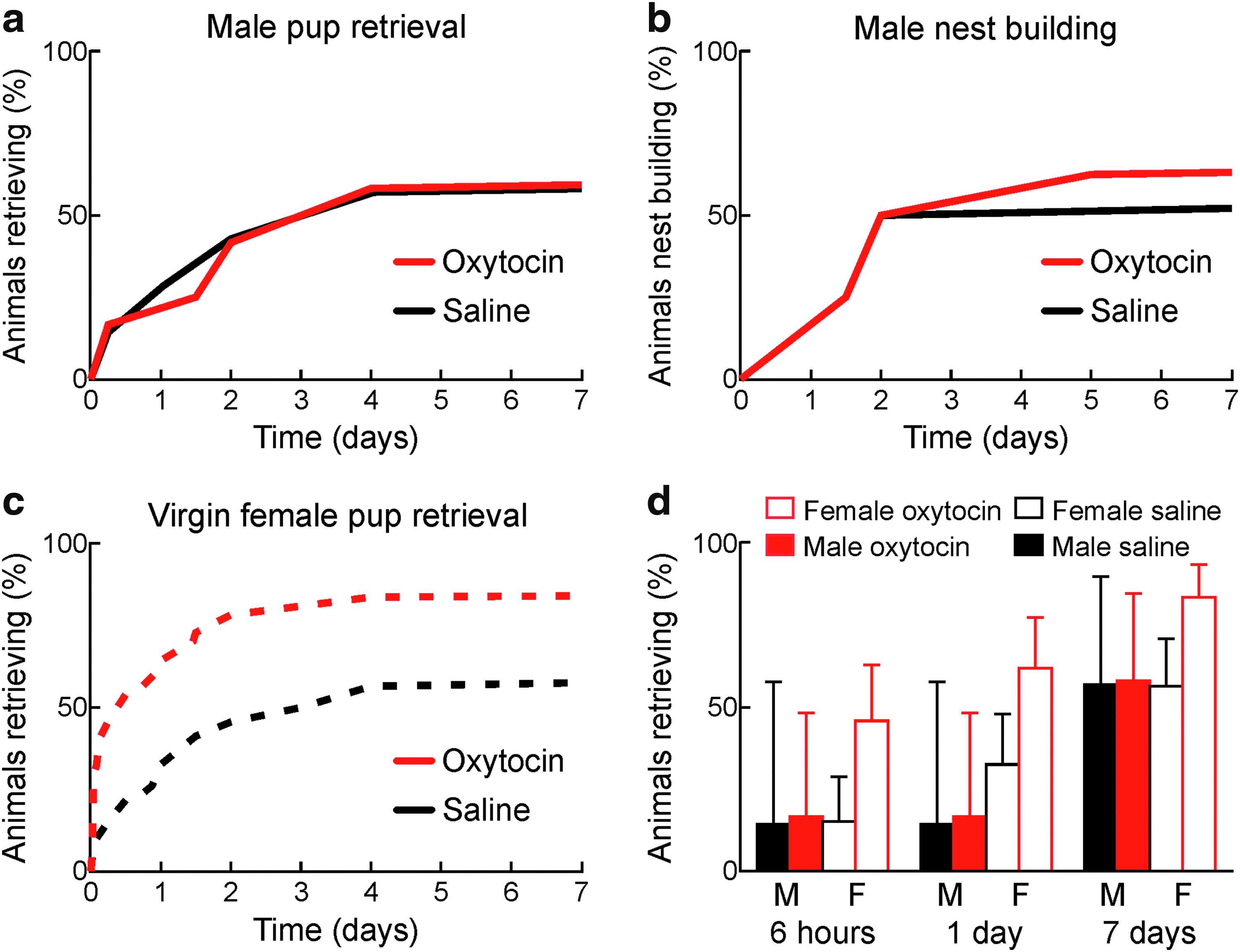
Oxytocin does not accelerate onset of parental behaviors in male C57BL/6 mice.
In general, male C57BL/6 mice expressed many of the same parenting behaviors as cohoused virgin female mice. We observed that individual cohoused males began crouching and grooming pups around the times that they began reliably retrieving pups during retrieval testing, as with cohoused virgin females. We quantified the rate at which males began collecting bedding and nest building when placed in the retrieval test chamber with pups. Oxytocin-injected and saline-injected males both began nest building at similar rates, closely matching the time course of retrieval behavior onset (Fig. 4b). Males cannot nurse pups, but another important difference we noticed between cohoused males and females is that the dams and cohoused male mice quickly mated soon after cohousing began. Mating may provide important contributions to the onset of male parenting behaviors, even when these behaviors are first expressed considerably later in time after mating as previously described by Wu et al. 44
After days of cohousing, retrieval behavior was expressed similarly in males and saline-injected females. The major effect of oxytocin was to accelerate this process in female cohoused virgins (Fig. 4c), with no effect in males. Saline-injected females began expressing retrieval slightly earlier than males; after 24 h of cohousing, the number of females retrieving was double that of males (Fig. 4d, “1 day”). However, after a week, these fractions were equivalent, with about half of all males and saline-injected females retrieving (Fig. 4d, “7 days”). Thus male mice can express parental behaviors in a similar manner as females, although some females can express these behaviors with less maternal experience, as well as remain sensitive to oxytocin supplements that increase and accelerate the rate of behavioral expression.
Discussion
In this study we examined the expression patterns and functions of oxytocin receptors in mouse auditory cortex. We showed that adult female mouse left auditory cortex has more cells expressing oxytocin receptors than male cortex or female right auditory cortex. Previously we observed in female mouse auditory cortex that the relative number of OXTR-2+ cells increased from the first to the second postnatal week in both left and right hemispheres, but then decreased from postnatal week 2 to adulthood, decreasing more in right female auditory cortex than left auditory cortex. 21 In other words, receptor levels were initially at a moderate level in early postnatal females and males, peaked during the second postnatal week, and declined to an equivalent degree in males and female right auditory cortex, leaving receptor expression levels higher in female left auditory cortex.
This is consistent with the cortical developmental profile of receptor expression first described by Hammock and Levitt, 59 with the additional finding of the hemispheric differences in females. As we reported in this study, OXTR-2 labeling was similarly high in postnatal week 2 males and females, but decreased to adult levels in parallel with a reduction in oxytocin receptor mRNA levels. These adult levels are therefore less decreased in female left auditory cortex compared to female right or male auditory cortex of either hemisphere. This was the sole identifiable difference between males and females in terms of peptide modulation of central auditory system structures examined here, with little oxytocin receptor expression or mRNA in adult auditory thalamus, and virtually no detectable vasopressin V1a receptor mRNA in either cortex or thalamus of older mice.
The specific pattern of receptor expression should confer certain properties on oxytocin signaling in the mammalian brain. Previous reports of the distributions of oxytocin receptors have relied on autoradiography or in situ hybridization.5–7,23,24,26 These studies have been important for determining which brain structures express oxytocin receptors. Recently we developed the OXTR-2 antibodies used in this study and found in female left auditory cortex that the majority of cells expressing oxytocin receptors were cortical inhibitory interneurons,18,21 similar to a study in transgenic animals demonstrating oxytocin receptors in somatostatin interneurons of mouse prefrontal cortex. 30
This agrees with the functional effects of oxytocin receptor activation for modulating synaptic transmission. Specifically, in adult auditory cortex, olfactory piriform cortex, PVN, and hippocampal CA1, oxytocin has been found to reduce GABAergic transmission.18,21,62 This disinhibition of principal cells in each area leads to increased excitability, promotes LTP induction, and in PVN might underlie the phenomenon of oxytocin-induced oxytocin release.6,18,21,63 In the central auditory system, auditory cortical neurons in experienced maternal animals but not pup-naive animals can reliably respond to pup call sounds,18,64–66 and activity in the left but not right auditory cortex is required for experienced mother mice to respond to infant distress sounds for pup retrieval.18,67 In naive female auditory cortex, the reduction of inhibition produced by oxytocin should make the cortical network much more responsive to incoming inputs such as pup calls that co-occur with heightened periods of oxytocin release. Repetitive presentation of the sensory signals in the disinhibited state would then induce enduring representational plasticity for pup cues 18 through mechanisms of NMDA-receptor-dependent LTP, 21 making maternal recognition of pup distress much more rapid and reliable. We speculate that the extensive amount of protein regulation and phosphorylation observed in this study in the left auditory cortex after oxytocin treatment might be related to expression of long-term synaptic modifications. The functional effects and interrelations between these genes will require further investigation, particularly in terms of lateralization in the female cortex.
We hypothesize that the left lateralization of oxytocin receptor expression in female auditory cortex confers a selective advantage for recognizing conspecific vocalizations, especially infant distress calls. This might manifest as circuit-level specializations on the left (and possibly right) hemispheres, consistent with hemispheric parcellation of specific auditory functions in human left versus right temporal lobes from imaging studies 68 (Hickok and Poeppel 2007). A left-lateralized preference for underlying pup call modulation rhythms might also lead to selectivity for or perceptual categorization of pup calls in the left auditory cortex presented at the natural call rates (∼3–8 Hz); this would only be the case if this pup call rhythm or bout rate was the predominant acoustic feature used by experienced co-carers for recognizing distress calls, as opposed to strictly their component ultrasonic frequencies. Furthermore, asymmetries in inhibitory neuron circuit composition might convey differential temporal sensitivities to the left and right sides; we note that most oxytocin receptors are expressed on cortical interneurons and regulate GABAergic transmission.18,21,30,62 Oxytocin receptor activation in left auditory cortex might lead directly to changes in protein expression and phosphorylation through mechanisms downstream of G protein-coupled receptor signaling or indirectly lead to changes in activity-dependent gene regulation after increased excitability or LTP induction.
Why might oxytocin enhance retrieval onset in females but not males? Additionally or instead, it is also possible that other factors contribute to this sex specificity of oxytocin action. First, oxytocin may have little to do with emergence of paternal behavior in males. For example, pair bonding in adult voles displays a similar sex-specific enhancement by oxytocin supplements. Exogenous oxytocin was found to increase pair bonding in females but had little effect in males, which might instead be regulated by vasopressin. 69 In male C57BL/6 mice, galanin neurons might be more important for initiating these behaviors after cohousing and mating. 44
Second, native release of endogenous oxytocin during or after social interactions and mating might be higher or lead to saturating activation of oxytocin receptors in males, but occur at more moderate levels in females. 70 However, measurement of endogenous oxytocin in amygdala during social interest behavior revealed similar peptide levels in male and female rats, indicating that central release of the hormone may not play a major role in sex-specific differences to oxytocin modulation. 71
Third, blood–brain barrier permeability could be more permissive in females than males, 72 enabling systemic oxytocin to have more access to the female central nervous system. Finally, peripheral treatment with oxytocin might bind to peripheral oxytocin receptors, which are expressed by many tissues and cell types outside of the brain. 6 Activation of peripheral receptors might indirectly affect central activity, perhaps further stimulating endogenous oxytocin release to enhance parental behavior.63,73 Little is known about the potential for these processes to provide mechanisms for oxytocin signaling throughout the organism, and it remains unclear how peripheral oxytocin administration affects neural activity to influence social cognition in any mammalian species.
Sex-specific differences in regional oxytocin receptor expression are consistent with the major role of this hormone in maternal physiology. In rat forebrain, Dumais et al. 74 found that most brain areas examined had lower oxytocin receptor radiolabel binding in females than in males. These regions included ventromedial hypothalamus (VMH), hippocampal CA1, the medial preoptic area (MPOA), nucleus accumbens, the bed nucleus of the stria terminalis (BNST), and medial amygdala. In agreement, our characterization of oxytocin receptor expression with OXTR-2 labeling 21 revealed that many of these same areas had higher expression levels in female mice (dams or virgins) than in males, consistent with these areas serving as a distributed network regulated by oxytocin, likely important for social and maternal behavior. One interesting difference is that while estrus led to higher oxytocin receptor radiolabel binding densities in female rats in most subcortical areas examined (including MPOA, VMH, BNST, and nucleus accumbens),5,74 we found that estrus did not affect oxytocin receptor expression in female mouse auditory cortex. 21 Regional differences in oxytocin receptor expression and regulation of receptor levels during estrus are believed to be due, in part, to a hormone response element upstream from the oxytocin receptor transcription initiation site. 10 This suggests that perhaps during postnatal development or puberty, steroid hormone signaling helps pattern receptor expression across different brain areas and in a sex-specific manner; such a mechanism might also be important for emergence of left-lateralized oxytocin receptor expression during postnatal development in female mice.
More studies are also required to understand the cellular mechanisms and functional significance of oxytocin receptor lateralization in female auditory cortex. In general, it is unknown how larger scale neural organizational features (such as retinotopy in the visual system or tonotopy in the auditory system) relate to the functions of those systems, although it seems reasonable to expect that such organization might improve detection and recognition of important sensory signals such as pup distress calls, by clustering cells together to ensure a more coherent network-level response from cells with similar robust tuning properties.75,76 Given the sparseness of axonal projections from hypothalamic oxytocin neurons throughout the brain,6,12,18,21 enhanced density of cells expressing oxytocin receptors, located together in a local cluster, might also facilitate oxytocin modulation especially if this system relies on volume transmission. Finally, left-lateralized oxytocin receptor expression in female auditory cortex is reminiscent of some features of human speech and language representation.68,77,78 Understanding the mechanisms and consequences of asymmetrical cortical pup call processing might therefore provide some information about the biological basis of human language abilities.
Conclusion
Female and male mice both can express parental behaviors such as pup retrieval within hours to days after initial contact with pups and experienced mothers. Females naturally exhibited maternal behaviors somewhat earlier than males, and exogenous oxytocin augmented the speed and relative number of females but not males displaying parental behaviors. As the only apparent difference between oxytocin receptor expression in adult males and females was a higher level of receptor expression in female left auditory cortex, we suggest that this left lateralization might provide a biological basis for oxytocin to enhance maternal behavior.
Footnotes
Acknowledgments
This work was funded by NICHD (HD088411 to R.C.F.), NIDCD (DC009635 and DC012557 to R.C.F., DC012527 and DC015388 to T.A.H.), NINDS (NS21072 to M.V.C.; T32 NS086750 fellowship to M.M.; P30 NS050276 to T.A.N.), NIMH (T32 MH019524 fellowship to B.J.M.), NCRR (S10 RR027990 to T.A.N.), a McKnight Scholarship (R.C.F.), a Pew Scholarship (R.C.F.), a Sloan Research Fellowship (R.C.F.), a Howard Hughes Medical Institute Faculty Scholarship (R.C.F.), and a Skirball Institute Collaborative Research Award (M.V.C. and R.C.F.). The authors thank I. Carcea, M. Jin, N. Lopez, E. Xh. Morina, and N. Zaika for comments, discussions, and technical assistance and C.A. Loomis and the NYU School of Medicine Histology Core for assistance with anatomical studies.
Author Disclosure Statement
No conflicts of interest exist.
