Abstract
Sex-specific differences exist in innate and adaptive immune responses and are mediated by hormone signaling. Estrogen is able to differentially modulate the development and differentiation of immune cells, including T cells. However, the effect of estrogen on T cell function, especially at concentrations other than physiological, remains controversial and incompletely understood. Immunotherapy is one of the most promising cancer treatments to date with a high probability of future enhancements. The adoptive transfer of genetically modified T cells can mediate tumor regression but there are still many hurdles to enhancing the proficiency of this treatment. This study demonstrates for the first time that one major aspect to consider for designing potent immunotherapies for cancer is the impact of the patient's sex. Herein, using two different Ag-specific T cell groups, we investigated the effect of sex and estrogen in antitumor effector responses, T helper cytokine secretion, and, importantly, on T cell whole polyfunctionality important for memory T cell development and survival. Major differences were observed in T cell function and polyfunctionality between sexes and on E2 treatment. The findings of this study may be critical to understand the results of immunotherapy on different patients and for the enhancement of immunotherapy for cancer.
Introduction
Males and females have measureable differences in their immunological responses to self and foreign antigens (Ags). 1 Some of these differences can be present throughout life and can be due to genetic imprinting or environmental stimuli 1 ; others vary with puberty or reproductive senescence demonstrating an important role for hormones or hormone receptor signaling. Sex-specific immunological differences can effect the incidence of autoimmune diseases and oncologic malignancies, as well as the susceptibility to viral and bacterial infections. While females are significantly more prone to autoimmune diseases, males have a higher incidence of infections and malignancies.2–6 In addition, there are several cancers that are highly sex driven.
Among those impacted by sex include melanoma, the most dangerous form of skin cancer, and hepatocellular carcinoma (HCC), which is the most dangerous form of liver cancer and the fifth leading cause of cancer-related deaths in the United States.7,8 Melanoma and HCC are the only two cancers that still have a rising rate of mortality in the world, and a clear sex-influenced progression. We recently demonstrated significant differences in melanoma tumor growth between male and female mice, dependent on infiltration of specific immune cell populations, including dendritic cells (DCs) and T cells. 9 In human studies, it can be observed that men are 2.5 times more likely to develop melanoma, and 4 times more likely to develop HCC than women of the same age group. 10 Mortality is also heavily sex influenced since men are 55% more likely to die from these cancers than women. 10
Despite the increase in incidence of these cancers, there are now more promising treatments that have been developed, including multiple forms of immunotherapy. 11 The development of checkpoint blockade inhibitors has been a huge step in the fight against cancer, but the most dangerous and difficult aspect to treat on cancer is still distal metastases. Adoptive cell transfer of genetically modified T cells (Ag-specific T cells and CAR T cells) was found to be an effective method of targeting spreading disease.12–14 This technique works very well in some patients, but it still remains unknown why it has little effect treating cancer in others. The effect of sex hormones, especially estrogen, has been studied in cancers, immune responses against infectious diseases, and autoimmunity, however, the impact of estrogen receptor (ER) activation on the effect on antitumor immune responses of T cells is unclear.
We now show for the first time that the effect of the female hormone, 17β-estradiol (E2), at various doses (including physiological levels, 0.5 nM) significantly and differentially impacts the overall functionality of Ag-specific T cells derived from male and female donors. In this study we demonstrate that these differences in functionality may be an important consideration to obtain a successful antitumor effect during immunotherapy. Sex differences are observed on single cytokine secretion and, even more importantly, on the simultaneous production of multiple cytokines or “polyfunctionality” of these Ag-specific T cells. Furthermore, it was shown that E2 stimulation further mediated differences in cell signaling and the ability to mediate antitumor immunity.
Materials and Methods
Cell lines
The T2 cell line (CD3 negative) used as HLA-A2-expressing but Tap-deficient APC was a gift from Dr. Michael Nishimura, Loyola University Chicago, Maywood. T2 cells were maintained in RPMI 1640 medium supplemented with 10% fetal bovine serum, 5% penicillin and streptomycin, and 5% L-glutamine.
Peptides
HCV NS3 (1406–1415): KLVALGINAV and tyrosinase (368–376): YMDGTMSQV were obtained from Synthetic Biomolecules (San Diego, CA). T2 cells were pulsed for 4 h, and DCs were pulsed for 18 h with peptides at 5 μg/mL.
Human buffy coat processing and peripheral blood mononuclear cell isolation
Buffy coats from three different HLA-A2+ male and female human donors were obtained from Key Biologics, LLC (Memphis, TN). One hundred to 150 mL of buffy coats were dissolved in Dulbecco's phosphate-buffered saline (DPBS) and overlayed over Ficoll-Paque (1.077 g/mL) before high speed gradient centrifugation at 800 g for 20 min. Lymphocyte layers were collected and washed in DPBS, an average of 1 × 109 peripheral blood mononuclear cells (PBMCs) were obtained from each donor's buffy coat.
Retroviral transduction of PBMCs
The HCV and tyrosinase-reactive T cells were obtained from the retroviral transduction of HLA-A2+ female and male donor PBMCs.15,16 Briefly, 1 × 108 PBMCs were cultured and expanded in AIM-V medium containing 10% human serum, 100 ng/mL IL-15, 300 IU/mL IL-2, and 50 ng/mL OKT-3. 15 PG13 viral producing cells were used to obtain retroviral supernatants containing the T cell receptor (TCR) retro viral (RV) vectors. After 3 days, activated PBMCs were transduced with 6 × 108 viral particles/mL in Retronectin (Takara Bio USA)-coated 24-well plates (30 μg/mL) at 2 × 106 cells/mL. Ag-specific cells were assessed for transduction via flow cytometry using CD3-APC-Cy7, CD34-Alexa Fluor 700, CD4-PE-Cy7, and CD8-PE (BioLegend) antibodies. Transduced cells were sorted using CD34 MicroBeads (Miltenyi Biotec) to obtain pure transduced T cell populations (>95% CD34+).
Isolation of cytoplasmic and nuclear extracts
2 × 106 Ag-specific T cells from male and female donors were treated with none, 0.5, or 50 nM E2 for 2, 10, 30, 60, or 120 min and nuclear and cytoplasmic protein extracts were obtained using the NE-PER Nuclear Cytoplasmic Extraction Reagent Kit (Pierce, Rockford, IL) according to the manufacturer's instructions. Treated cells were washed twice with ice-cold phosphate buffered saline (PBS) and centrifuged at 500g for 5 min. Cell pellets were resuspended in 100 μL of cytoplasmic extraction reagent I (CER I) by vortexing and incubated on ice for 10 min followed by the addition of 5.5 μL of CER II. They were then vortexed for 5 s, incubated on ice for 1 min, and centrifuged for 5 min at 16000g. The supernatant fraction (cytoplasmic) was transferred to a prechilled tube. The insoluble fraction was resuspended in 50 μL of nuclear extraction reagent and vortexed every 10 min for a total of 40 min. Tubes were centrifuged at 16000g for 10 min, and the resulting supernatant was collected (nuclear fraction).
Western blot analysis
Equal amounts of nuclear and cytoplasmic proteins were loaded onto a sodium dodecyl sulfate/polyacrylamide gel. After electrophoresis, proteins were transferred into a polyvinylidene fluoride membrane (Bio-Rad) and blocked for 1 h with 5% BSA in Tris-buffered saline + Tween-20. After blocking, membranes were incubated overnight with the anti-human ERα primary antibody (Cell Signaling Technologies). After washing, membranes were incubated for 1 h with the horseradish peroxidase-conjugated anti-rabbit IgG secondary antibody (Cell Signaling Technologies). Proteins were detected using an enhanced chemiluminescence kit (Thermo Fisher). All blots were stripped and reprobed with β-actin or Lamin A/C (Cell signaling Technologies).
Amnis image stream
CD34-sorted Ag-specific cells from three separate female and male donors were treated with 0 or 0.5 nM 17β-estradiol for 2 h and then stained with anti-human CD3-PE (BioLegend). Then, they were fixed and permeabilized using the FoxP3 intracellular staining kit (eBiosciences). Cells were blocked with 1 × PBS containing 10% goat serum (Sigma Aldrich) and 0.3 M glycine for 30 min at room temperature, and stained with anti-human ERα-Alexa Fluor 488 (Abcam). DRAQ5 (BioLegend) was added to all cells 15 min before analysis for staining of cell nuclei. Cells were acquired on the Amnis Image Stream X machine at × 60 magnification, using the 488 and 642 lasers and brightfield. Analysis was performed on the IDEAS software, using the colocalization analysis wizard. Templates of software settings were created in IDEAS to standardize analysis.
ER inhibition
Male and female Ag-specific T cells were treated with 0, 0.5, or 50 nM 17β-estradiol in combination with 100 nM of MPP [1,3-Bis (4hydroxyphenyl)-4-methyl-5-[4-(2-piperidinyleth oxy) phenol]-1-H pyrazole dihydrochloride] or PHTPP [4-[2-pheyl-5,7-bis (trifluoromethyl) pyrazole [1,5-a] pyrimidin-3-yl phenol] (Tocris Bioscience) for 2 h previous to coculture with T2 target cells pulsed with irrelevant or cognate antigen. Cocultures were maintained for 2 h in the presence of protein transport inhibitors Golgi Stop and Golgi Plug (BD Biosciences); after that, cells were stained with anti CD3e-APC-Cy7, CD34-Alexa Fluor 700, CD4-PE-Cy7, IL-4-AlexaFluor 647, IFNγ-Brilliant Violet 421, TNFα-Brilliant Violet 711 (BioLegend), and CD8-FITC (eBiosciences) and analyzed via flow cytometry.
Polycytokine flow cytometry
Multicolor flow cytometry was used to create cytokine profiles of 17β-estradiol-treated HCV and tyrosinase-reactive female and male T cells cocultured with pulsed activated PBMC-derived DCs. 17 These 11 different fluorochrome-conjugated antibodies for extracellular and intracellular markers were used: CD3e-APC-Cy7, CD34-Alexa Fluor 700, CD4-PE-Cy7, CD107a-Brilliant Violet 510, IL-4-Alexa Fluor 647, IL-22-PE, IL-17a-Brilliant Violet 570, IFNγ-Brilliant Violet 421, TNFα-Brilliant Violet 711 (BioLegend), CD8a-FITC, and IL-2-PerCP-efluor710 (eBiosciences). The different cytokine profiles were generated by analyzing the data using FlowJo.
First, the specific transduced T cell population was gated using CD3 and CD34. Then, CD8 and CD4 populations were gated and every cytokine and CD107a were gated versus side scatter to obtain the overall frequency of CD8 and CD4 cells producing each cytokine. Using Boolean gates, all the combinations of cytokines simultaneously produced were generated for CD8+ and CD4+ T cells. T cells stimulated with DCs pulsed with irrelevant peptide were used as background to normalize the data by performing a background subtraction in the software Pestle®. Using the software SPICE®, each combination of cytokines and CD107a was obtained and then plotted into pie charts using Microsoft Excel.
ELISPOT and ELISA
Previous to performing the assay, HCV and tyrosinase-reactive female and male T cells were treated with 50, 0.5, or 0 nM 17β-estradiol for 2 h. Then, 5 × 104 target and T cells were cocultured in a 1:1 ratio in an ELISPOT plate coated with anti-human interferon-γ (IFNγ) or anti-human granzyme B antibodies. Cocultures were incubated at 37°C for 20 h, and then, the biotinylated secondary and streptavidin–horseradish peroxidase antibodies were added, and plates were developed using 3amino-9 ethyl carbazole substrate. The amount of cytokine released was measured by counting the spots per well. T2 cells pulsed with irrelevant peptide were used as negative control, and T cells treated with cell stimulation cocktail (PMA/ionomycin) were used as positive control.
Same treatments and coculture conditions were set for the ELISA. After 20 h, coculture supernatants were collected and 100 μL were plated on anti-human TNFα or anti-human IL-4-coated high-protein binding plates. Plates were developed according to the manufacturer (Mabtech).
Statistical analysis
Statistical analysis for differences between group means was performed using unpaired Student's t-test. Data are presented as mean ± SD and values of p < 0.05 were considered statistically significant.
Results
ERs are expressed at equivalent levels on transduced Ag-specific T cells derived from male and female donors
To study the effect of estrogen on the function of Ag-specific T cells, two different sample populations were generated by retroviral transduction of PBMCs with Ag-specific TCRs. The first TCR is reactive to the hepatitis C virus NS3 (HCV:NS3) peptide present in HCC derived from chronic HCV infection, and the second TCR is reactive to tyrosinase peptide present in many cases of melanoma. The TCRs for HCV (1406) and tyrosinase (1883i) were shown to stimulate T cells with specific recognition of these tumor cells and are capable of generating antitumor immune responses in mice and humans.15,16,18 The TCR containing retroviral vector also codes for a truncated version of CD34 (CD34t), which is used for transduction recognition and purification of transduced T cells for further in vitro study (Supplementary Fig. S1A; Supplementary Data are available online at www.liebertpub.com/gg). Transduction of either male or female T cells with the HCV-1406 TCR typically yields ∼25–80% transduction efficiency (Supplementary Fig. S1B). On purification by cell sorting using the CD34 expression as a target, our studies are based on the effect of estrogen on a population of Ag-specific T cells at >95% purity (Supplementary Fig. S1C).
After transduction, the T cells were tested for expression of ERα by western blot. Briefly, purified transduced T cells were cultured in media containing 10% human serum and treated with no estrogen (0), physiological (0.5 nM), or superphysiological (50 nM) concentrations of E2 for 0, 2, 10, 30, 60, and 120 min before lysis and separating cytoplasmic and nuclear fractions to test for ERα expression. Male and female T cells expressed ERα in the cytoplasm, which was not dramatically different between males and females at each time point as demonstrated by normalizing expression to β-actin as a loading control (Fig. 1A, B). ERα protein expression was also not significantly changed between male and female T cells on E2 treatment, but can be observed to be either in the cytoplasm (Fig. 1A) or in the nucleus (Fig. 1B). Lamin protein from the nuclear membrane was used as a source of housekeeping and loading control for measuring proteins in the nucleus (Fig. 1B).
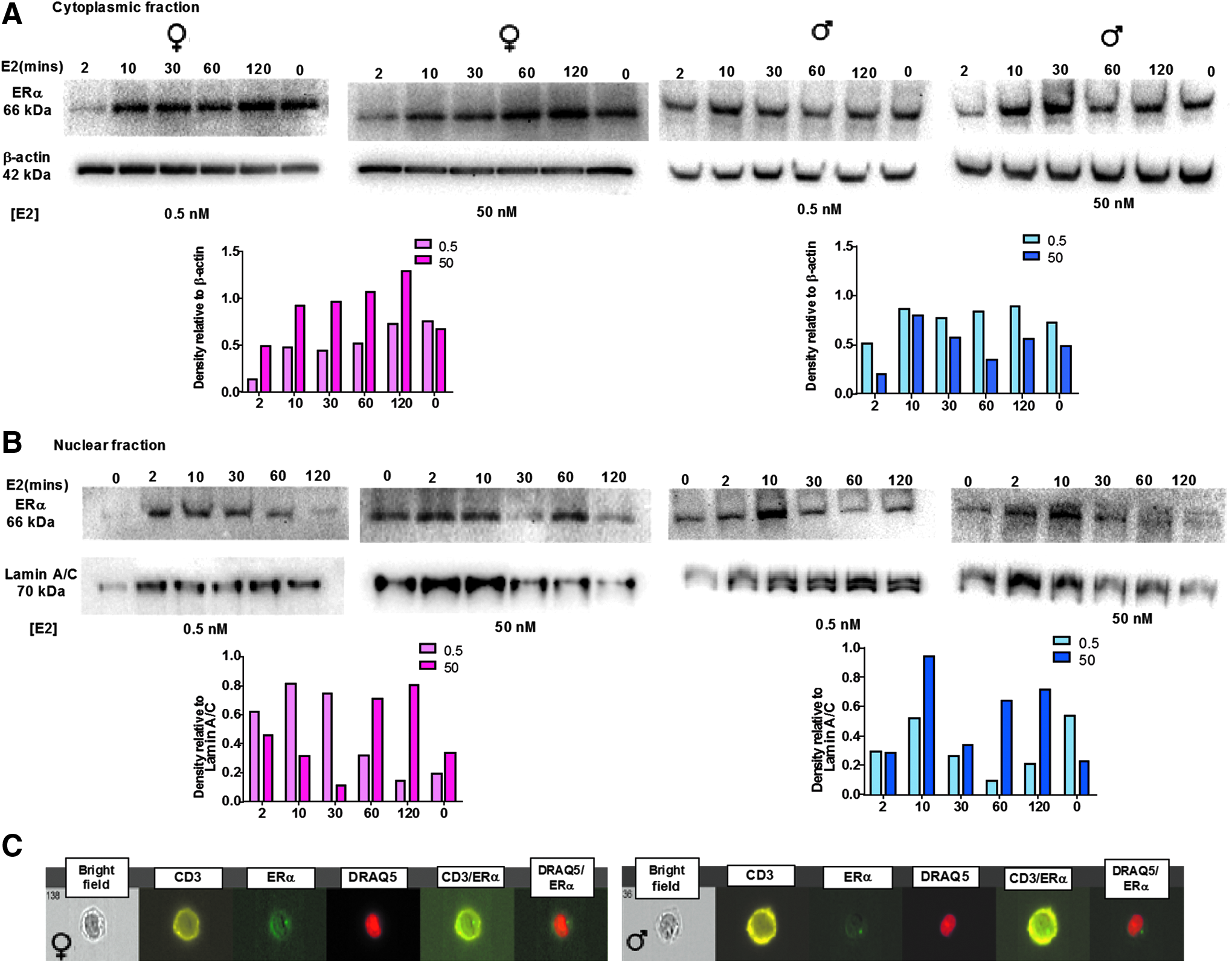
Female and male Ag-specific T cells express the same levels of ERα, translocating into the nucleus on 17β-estradiol treatment. Western blotting of protein extracts from
The time course of E2 treatment revealed that nuclear translocation of ERα on E2 ligation is very rapid, within 2 min (Fig. 1A, B), but it is also transient and protein begins to reaccumulate in the cytoplasm within 10 min and continues for 120 min post-E2 treatment. Furthermore, ERα expression in T cells is predominantly cytoplasmic until ligation. ERα was not detected on cellular membranes in T cells from either male or female donors (Fig. 1C). These data suggest that ER signaling in T cells occurs mainly through the ERα when it is engaged by E2 in the cytoplasm and then translocates to the nucleus.
Specific antitumor effector functions are increased in response to E2 in T cells from females but not males
T cell effector functions, including IFNγ production and release, and granzyme B release are crucial to tumor cell destruction. 19 Therefore, to next determine the effect of estrogen on IFNγ and granzyme B release, Ag-specific T cells from three different male and female donors were treated with no estrogen, physiological (0.5 nM), or superphysiological (50 nM) concentrations of E2 for 2 h before coculture with T2 target cells pulsed with cognate (HCV for TCR-1406 and TRP2 for TCR-1383i) or irrelevant peptide (TRP2 for TCR-1406 and HCV for TCR-1383i) Ags.
After a 20-h coculture, IFNγ and granzyme B secretion in response to Ag stimulation was measured by ELISPOT. Female but not male T cells treated with increasing concentrations of E2 had a measureable and significant increase in IFNγ and granzyme B production as the hormone dosage increased (Fig. 2A–D). However, it is important to point out that although IFNγ and granzyme B production did not increase in response to E2 in males, production and release were already higher on Ag stimulation in the T cells from males (Fig. 2B) compared to females at 0 or physiological levels of E2 treatment. These data indicate that E2 treatment differentially impacts effector functions in T cells from males and females by increasing production of effector molecules in female but not male T cells. Some differences in IFNγ production and detection can likely be attributed to the variance in frequency of the ratio of total CD4 to CD8 between male and female donors. Consistently, female donors had a 1:1 ratio of CD4:CD8 T cells (Fig. 2E), whereas male donors were more closely a 1:3 ratio of CD4 to CD8 T cells (Fig. 2E). The higher frequency of CD8+ T cells in the male donors may account for the more robust IFNγ response regardless of the E2 concentration observed in Figure 2B.
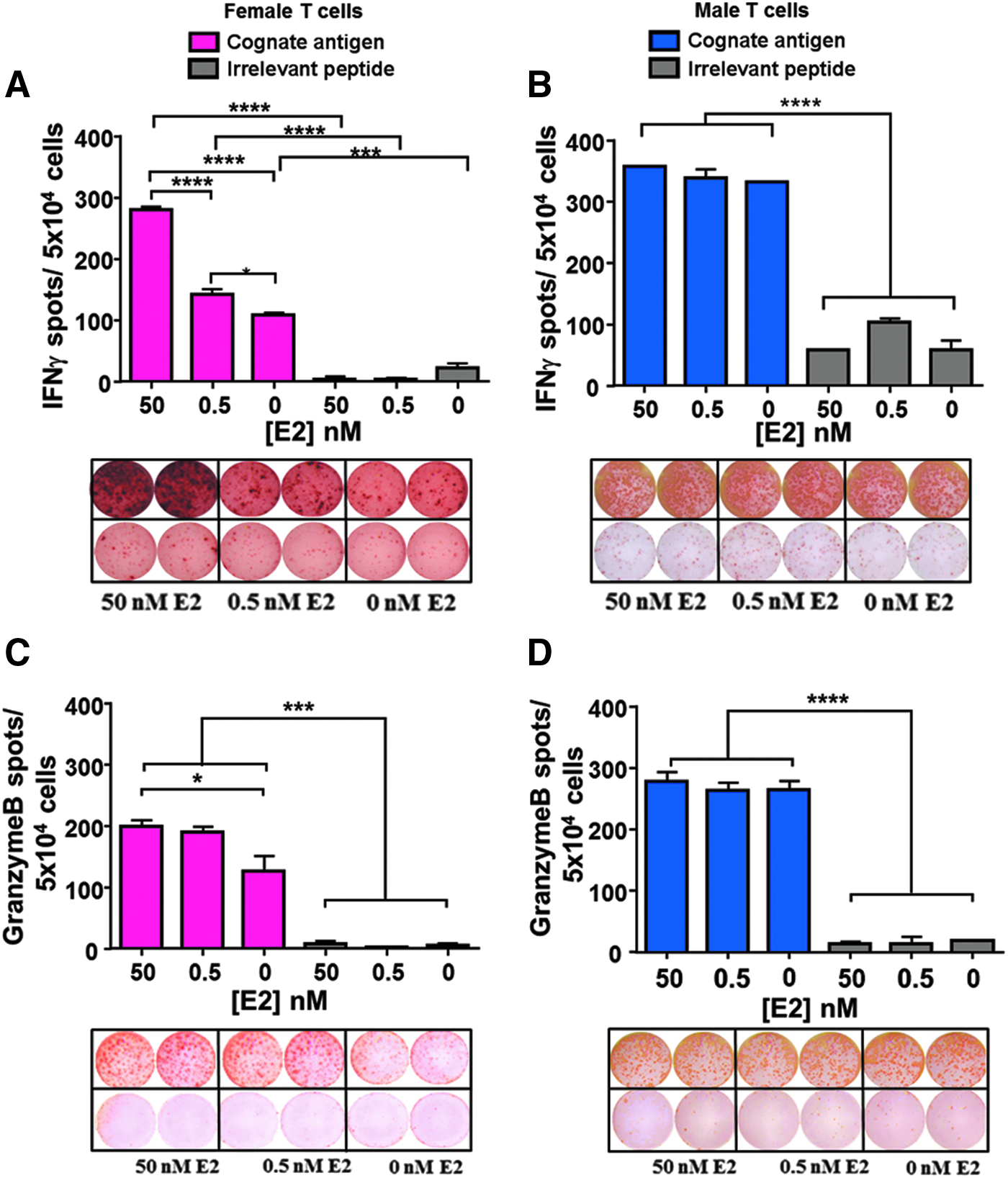
IFNγ and granzyme B secretion is significantly increased in response to E2 treatment in T cells from female but not male donors. T cells stimulated with 0, 0.5, or 50 nM E2 and their cognate Ag or an irrelevant peptide secreted increased IFNγ and granzyme B in a dose-dependent manner in T cells from female donors
T helper cytokines TNFα and IL-4 are increased in response to estrogen stimulation in both females and males
Given the differential impact on effector function, we next tested whether estrogen differentially impacted cytokines important for helper T cell function including IL-4, TNFα, IL-2, IL-6, and IL-17a. Ag-specific T cells from female and male donors had increased IL-4 and TNFα production as measured by ELISA upon treatment with increased hormone dosage (Fig. 3). The increase in TNFα production was similar at physiological doses but much greater in female T cells compared to male T cells at superphysiological levels of E2 (Fig. 3B). The increase in IL-4 was equivalent for both sexes (Fig. 3A). For each of the other cytokines such as IL-6, IL-2, and IL-17, there was no significance between male and female or in changes in secretion on E2 treatment (data not shown).
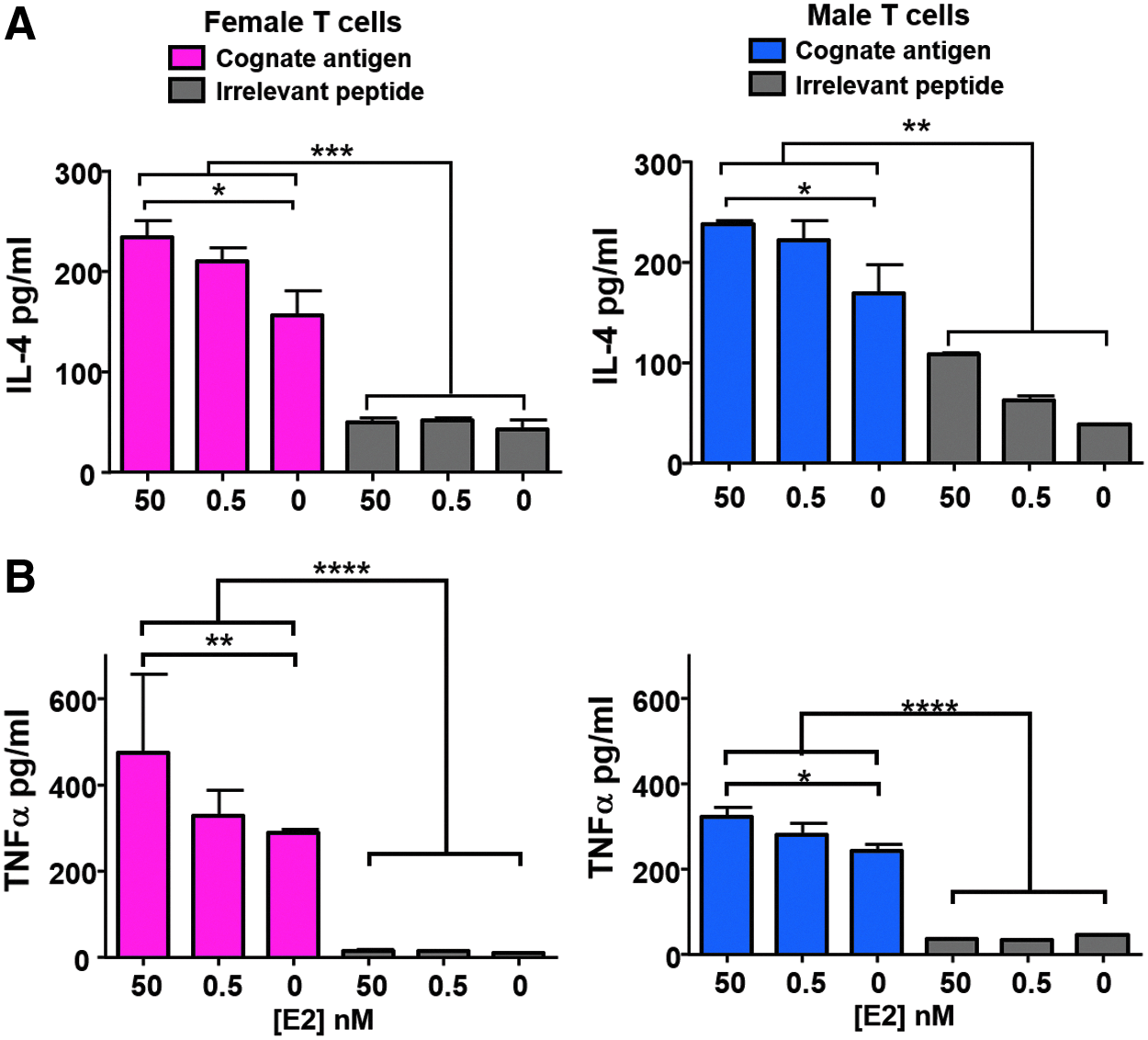
TNFα and IL-4 are significantly increased in response to E2 treatment in females and males. Ag-specific T cells from female and male donors were treated with E2 for 2 h before stimulation with their cognate Ag or an irrelevant peptide. After a 20-h coculture, the amount of
Estrogen stimulation differentially modifies overall Ag-specific T cell function in males compared to females
To obtain a clearer understanding of why helper T cell functions are more significantly affected by E2 treatment, we next tested the polyfunctionality of the Ag-specific T cells in response to E2 treatment. We hypothesized that the combination of cytokines produced simultaneously to either IL-4 or TNFα is important for influencing the level and proficiency of the effector molecules as well as the sustainability of those functions in response to tumor Ag. To test this, T cells were stimulated with their cognate Ag, which was presented by autologous DCs. T cells were then stained with 11 different antibodies for extracellular markers and intracellular cytokines, as well as the lytic marker CD107a, in the presence of protein transport inhibitors. After running flow cytometric analysis, T cells were first gated on CD3 and CD34 followed by either CD8 or CD4 to ensure data collection on the specific transduced T cell population and to generate results for CD4 and CD8 T cells separately. The cytokines assessed included IFNγ, TNFα, IL-2, IL-4, IL-17, and IL-22. The lytic marker CD107a was assessed on the surface as an indicator of granzyme B release. 20
Polyfunctionality profiles of CD4 and CD8 T cells were generated for four separate male and four separate female donors. The results vary slightly from donor to donor as would be expected among human donors not genetically identical or previously exposed to the same environments or past infections. However, the pattern of cytokine changes is remarkably consistent across the donors of each sex. Due to the amount of possible combinations of cytokines, and CD107a expression, the analysis for these data is very complex and difficult to analyze. However, it is immediately apparent on observation that T cell polyfunctionality differs dramatically between T cells from male and female donors, and further differentiates on E2 treatment at the different doses in both the CD8 and CD4 populations (Figs. 4–6).
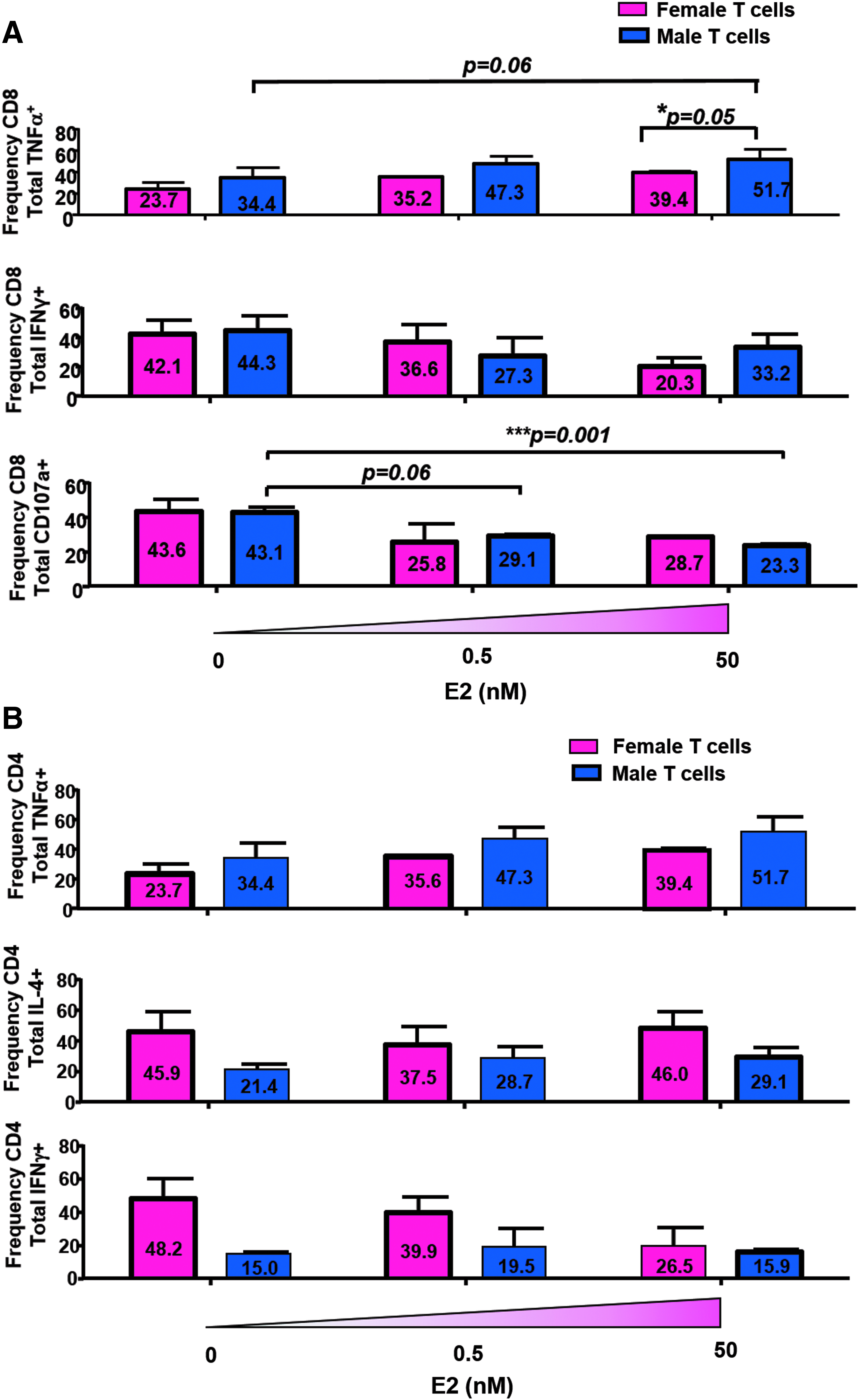
Estrogen impacts the frequency of CD8+ but not CD4+ T cells producing TNFα and having lytic function. Transduced T cells from male and female donors were stimulated with E2 and DC loaded with cognate Ag or an irrelevant peptide before intracellular staining for a panel of cytokines: IL-2, IL-4, TNFα, IFNγ, IL-22, IL-17a, and the lytic marker CD107A. Overall frequency of each cytokine was determined followed by an analysis for cytokines coexpressed in CD34+/CD3+/CD8+ T cells. The bar graphs represent the total frequency of
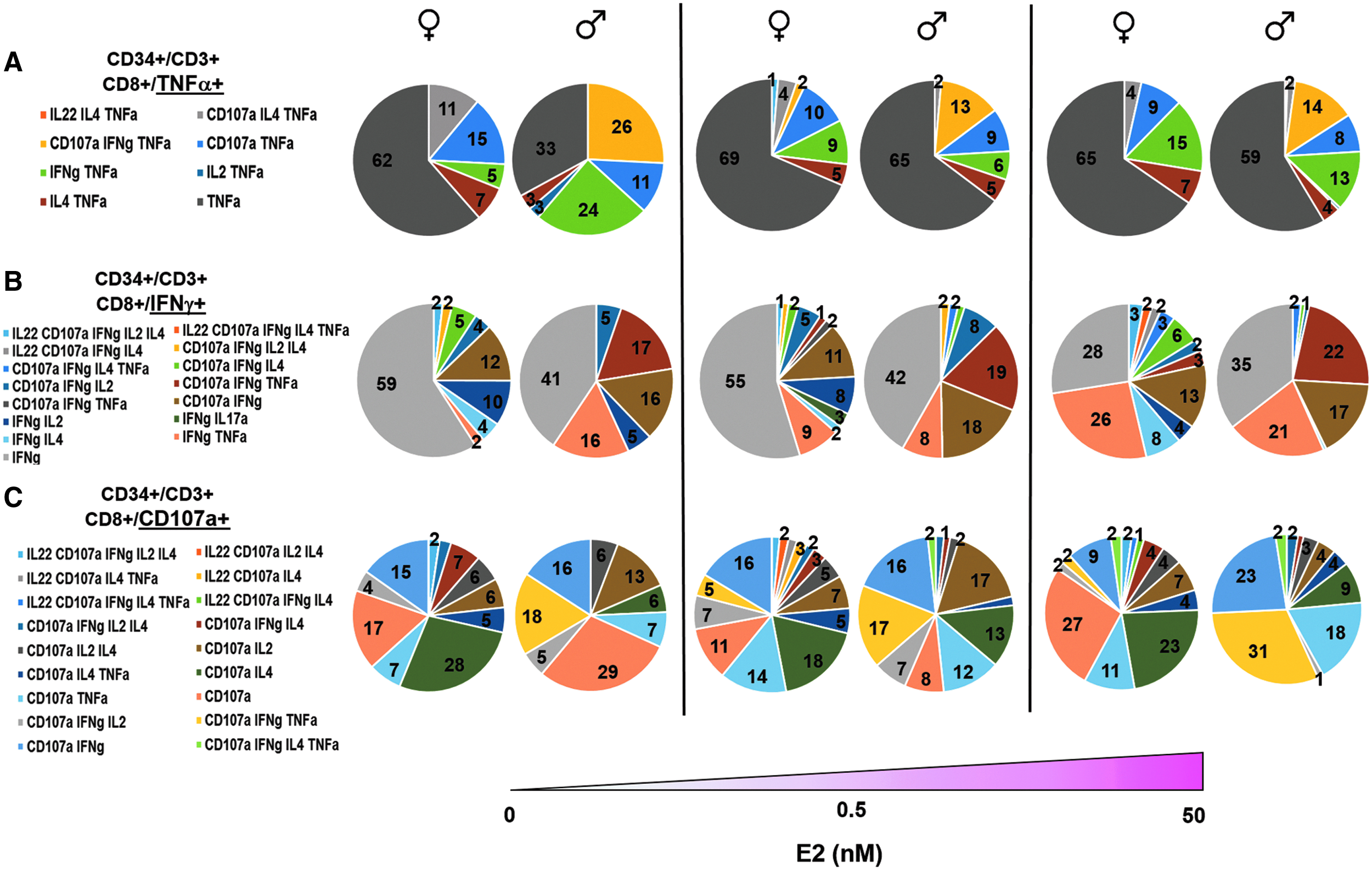
CD8+ T cell polyfunctional ability changes depending on sex and E2 treatment. Flow cytometry data were analyzed with Pestle and Spice software, and observed frequencies of cytokines produced in combination were graphed in Excel using parts of a whole analysis. The pie charts represent the different combinations of cytokines produced simultaneously within the total percentage of CD8+ T cells that produce
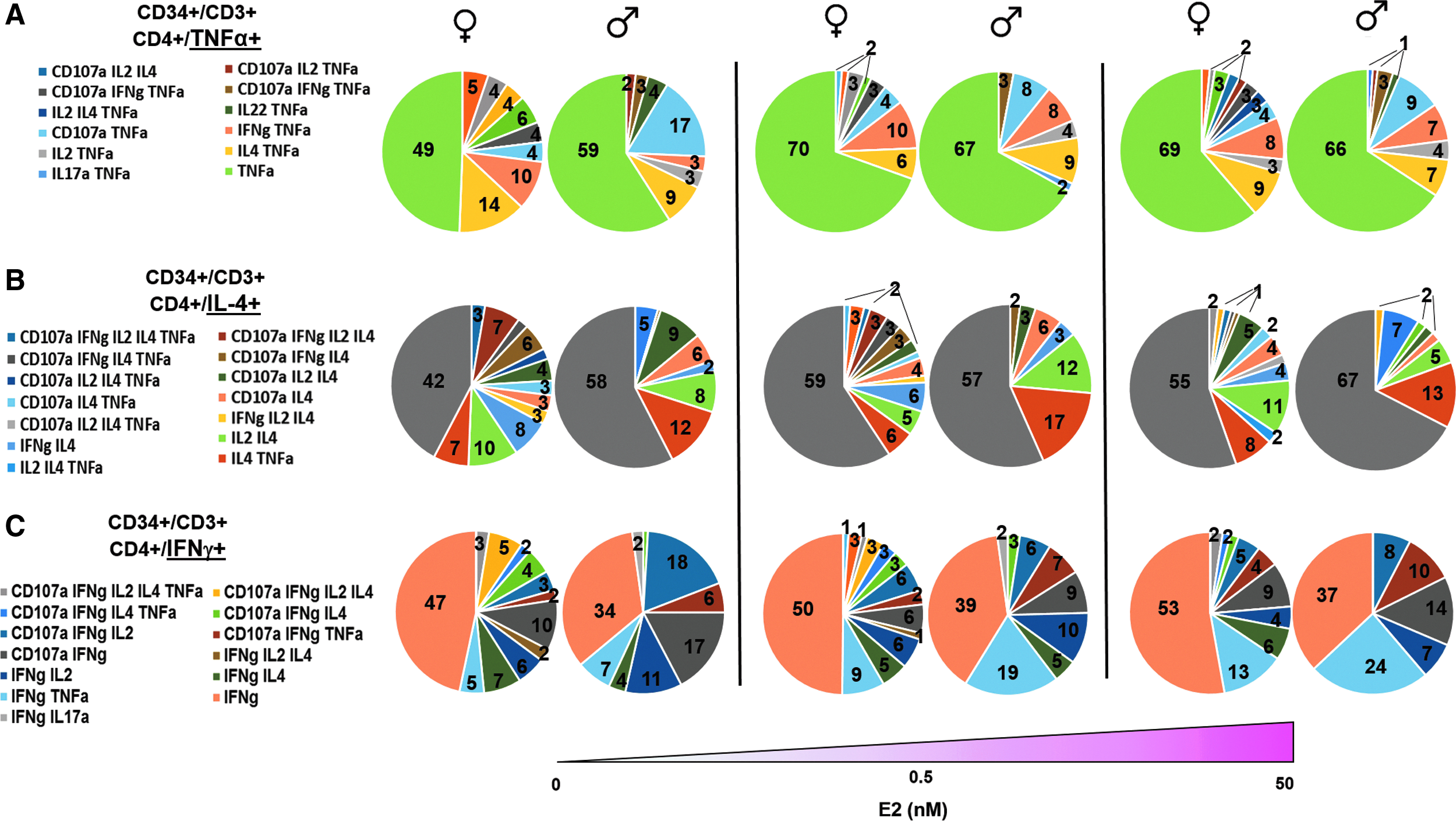
CD4+ T cell polyfunctional ability changes depending on sex and E2 treatment. The same analysis described in Figure 5 was performed, but in this case, overall frequency of each cytokine was determined followed by an analysis for cytokines coexpressed in CD34+/CD3+/CD4+ T cells. Similar to Figure 5, the pie charts represent the combination of cytokines produced simultaneously with
To best assess these results, gates were first set for CD34 and CD3. From there, the CD8 and CD4 populations were selected. First analyzing the CD8+ T cells, a gate was set to first encompass all the T cells producing TNFα (Fig. 4A). The total percentage of cells producing TNFα increases in both the male and female T cells, which is consistent with the previously mentioned finding of increased TNFα production by both groups in response to E2 during Ag stimulation (Fig. 4A). Taken together, there is quantitatively more TNFα as demonstrated by the ELISA (Fig. 3B) and there is also a higher frequency of T cells producing TNFα as found by the flow cytometry analysis (Fig. 4A). The total percentage of cells producing IFNγ decreases in both females and males as E2 doses increase from 0 to 0.5 nM. It further decreases in females from 0.5 to 50 nM E2. The only exception is in males in which the total percentage of IFNγ producing cells increases again at 50 nM from 27.3% to 33.2%. These data are particularly interesting considering that the female T cells were first observed to increase the secretion of IFNγ on E2 treatment (Fig. 2). Therefore, although there is more IFNγ, there are fewer T cells producing it as the E2 dose increases (Fig. 4A). In assessment of lytic function, it was found that the total percentage of CD107a expressing cells decreased on E2 treatment equivalently in T cells from both male and female donors from 43% to around 25% (Fig. 4A).
We next analyzed the cytokine expression patterns of the CD4+ T cells in a similar manner (Fig. 3B). The total percentage of CD4+ T cells producing TNFα increased as E2 dose increased in T cells from both male and female donors (Fig. 4B), consistent with the observed increase in quantity of TNFα measured in the ELISAs (Fig. 4B). The total percentage of CD4+ T cells producing IL-4 is more than two times higher in female T cells (45.9%) compared to male T cells (21.4%) in the untreated group (Fig. 4B). CD4+ T cells producing IL-4 slightly decrease in female T cells at 0.5 nM E2, but it rebounds at 50 nM E2.
To better understand the overall function, we use Pestle and Spice software to examine the combinations of these cytokines and lytic factors produced and expressed simultaneously for CD8+ T cells (Fig. 5). In the untreated group, 62% of the female and 33% of the male CD8 Ag-specific T cells did not produce any of the other cytokines tested in combination with TNFα (Fig. 5A). Interestingly, on treatment with E2, the percentage of TNFα+ alone did not change in the female donors; however, the T cells from the male donors did shift to produce equivalent levels of TNFα in the absence of other cytokines (Fig. 5A). This effect seems to come as a result of decreasing simultaneous production of IFNγ and expression of CD107a (yellow portion of pie chart) at the higher E2 concentrations (Fig. 5A). Since the E2-mediated increase of TNFα seemed to effect the frequencies of CD8+ T cells expressing IFNγ and CD107a, we next set up the gates for CD8+IFNγ+ (Fig. 5B) and CD8+CD107a+ (Fig. 5C) T cells, and analyzed the combination of cytokines produced simultaneously with either IFNγ or CD107a, respectively.
In males, the cells producing only IFNγ in combination with no other cytokine or CD107a remain constant across E2 treatments. The other major combinations observed are IFNγ simultaneously expressed with TNFα or CD107a, but in general, the male IFNγ+ T cells are not as highly polyfunctional as the female T cells. In females, the cells producing only IFNγ in combination with no other cytokine or CD107a decrease as the E2 dose increases, but the polyfunctional cells producing IFNγ in combination with other markers increase across E2 treatments. Specifically, we observe a big increase in the female T cells producing both IFNγ and TNFα at superphysiological concentrations of E2 (50 nM) similar to the male cells.
In females, the CD107a+-only cells decrease at 0.5 nM but increase again at 50 nM. The cells positive for CD107a in combination with other cytokines remained relatively constant across E2 treatments with an increase in TNFα and a concomitant decrease in IL-4 expression (Fig. 5C). In males, the cells producing CD107a in combination with no other marker decrease dramatically in response to E2 doses from 29% at 0 nM to 0% at 50 nM. However, an increase in the polyfunctionality of the male T cells was observed specifically in the ones producing both IFNγ and TNFα in combination with CD107a expression.
After studying the effect of E2 on the polyfunctionality of male and female CD8+ T cells, similar analysis was performed on CD4+ T cells. In the untreated group, 49% of female and 59% of male T cells did not produce any other cytokine measured nor did they express CD107a in combination with TNFα. However, the frequencies of T cells producing TNFα increased as E2 dosage increased to 0.5 nM, but remained constant at the 50 nM E2 treatment. In contrast to what was observed in the CD8+ T cells, CD4+ T cells from males had a higher percentage of T cells producing TNFα alone at 0 nM E2 treatment compared to females. Although the exact percentages changed from donor to donor, this trend was consistent from male to female. Overall, T cells from female donors were found to be more polyfunctional across E2 treatments. The range of polyfunctionality remained constant in both sexes (with female more polyfunctional than male) (Fig. 6A).
The cells producing only IL-4 with no other cytokines or expression of CD107a increased at physiological E2 due to a loss of cells that at 0 treatment expressed IL-4 and IL-2 simultaneously. In the male T cells, the total percentage of cells producing IL-4 increases slightly as E2 dose increases to 0.5, and again to 50 nM. The cells producing IL-4 with no other cytokines or CD107a increased at 50 nM E2 with a simultaneous decrease in T cells producing IL-4 and IL-2 together (Fig. 6B). Overall, the percentage of cells producing only IL-4 is similar in both sexes, however, female T cells are more polyfunctional than male T cells at all three E2 dosages. In addition, a greater frequency of cells producing more cytokines and CD107a in combination with IL-4 was detected in female T cells compared to male.
Analysis of CD4+ T cells producing IFNγ, as a helper cytokine to support CD8 effector responses, demonstrates that the total percentage of female CD4+ T cells expressing IFNγ decreases as E2 dose increases. This is interesting given that the total quantity of IFNγ produced is significantly upregulated (Fig. 2). Furthermore, the cells expressing only IFNγ with no other cytokines or CD107a marker increase. The total percentage of CD4+ T cells expressing IFNγ is more than two times lower in male T cells than in the female T cells in the nontreatment group, and slightly increases at 0.5 nM E2 (Fig. 6C). Similar to the data from previous gating, female cells are more highly polyfunctional than male cells at all E2 doses, expressing higher amounts of cytokines or CD107a in combination with IFNγ. Taken together, these results suggest that although T cells from male donors maintained greater effector function, T cells from females were more polyfunctional suggesting memory T cell potential.
ERβ competes with ERα for ligand binding and affects T cell function
To confirm that changes in T cell frequency expressing cytokines, male and female donor T cells were stimulated with E2 at various concentrations along with Ag stimulation in the presence of either an ERα or ERβ inhibitor. T cells were again analyzed for expression of TNFα, IFNγ, and IL-4 by flow cytometry on stimulation. Variability among donors in response to the inhibitors in these experiments was fairly high resulting in larger error bars. However, although not all cytokine changes were statistically significant, it was observed that the overall trend was that a higher frequency of CD8+ T and CD4+ T cells did express IFNγ, TNFα, and IL-4 on blockade of the ERβ during E2 stimulation. These results suggest that ERβ competes with ERα for available E2. On blocking ERβ, more E2 is available to stimulate ERα and induces more T cells to produce cytokines. These results were observed primarily in T cells from female donors (Fig. 7A). When blocking ERα, the observed increase in T cells producing each cytokine was not observed (Fig. 7A, B). Furthermore, it was statistically significant in both male and female donors that when ERα was blocked, expression of IL-4 was dramatically reduced in both CD8+ T cells (Fig. 7A) and CD4+ T cells (Fig. 7B). Taken together, these results suggest that T cells, both CD4 and CD8 from males and females, respond to E2 stimulation predominately through the ERα. Therefore, the differences in function mediated by E2 are downstream of the receptor.
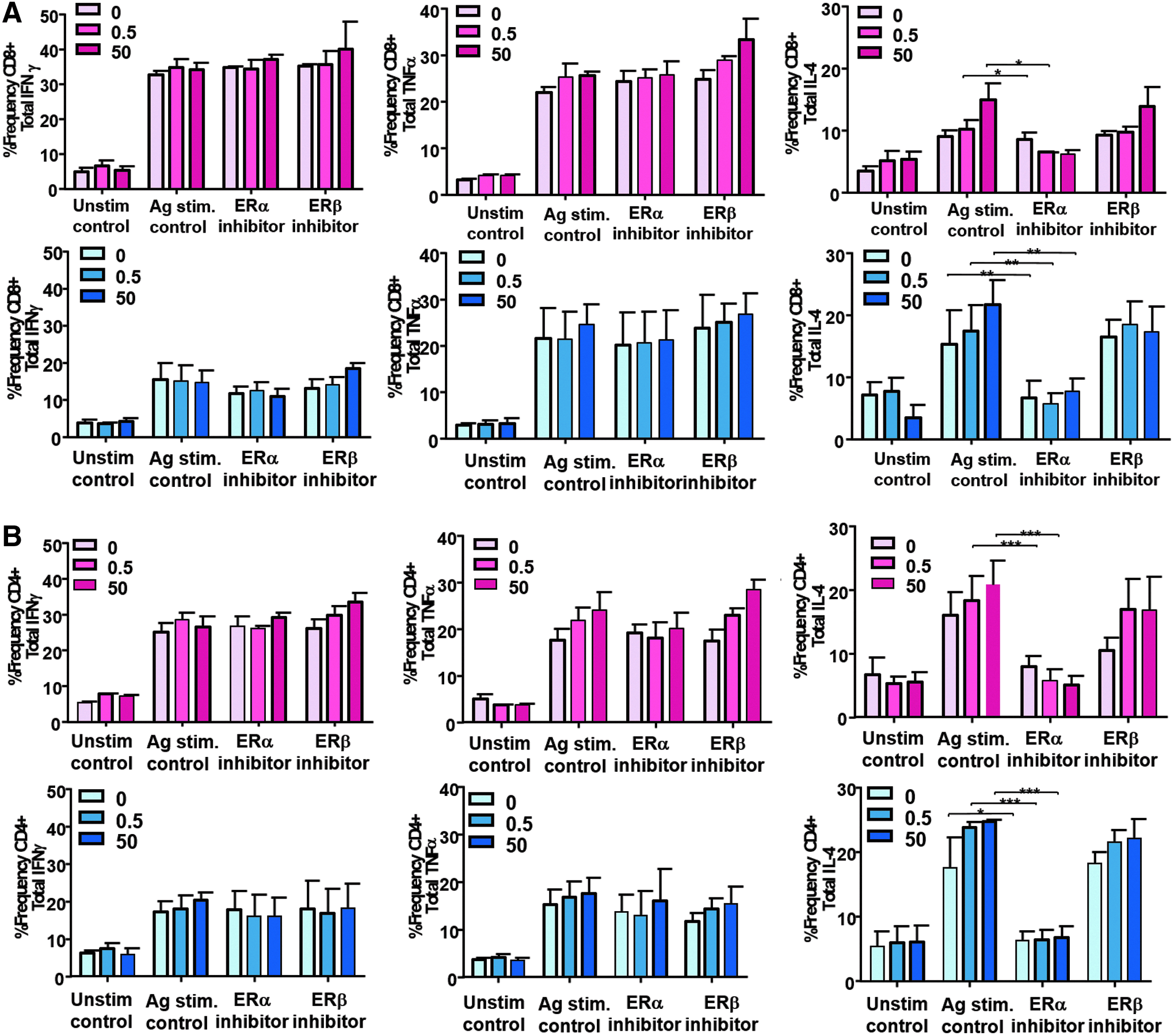
Blocking ERα or ERβ alters T cell functional profiles on Ag stimulation. An inhibitor to ERα (MPP-dihydrochloride) or to ERβ (PHTPP) was added to T cells before Ag stimulation with or without E2 present.
Discussion
In this work, we sought to determine the contribution of gender and ER activation in regulating Ag-specific T cell effector function and helper T cell function. For the first time, we have demonstrated the importance of hormone signaling on the overall polyfunctionality of human Ag-specific T cells. These results may have far reaching importance in terms of boosting immunotherapy against cancer as well as in regulating T cells during autoimmune disease or even as a result of chronic inflammation due to Ag stimulation.
Herein we showed that male and female Ag-specific T cells express equivalent levels of the canonical ERs, ERα and ERβ. Both receptors are known to bind E2 with the same affinity but result in different cell signaling and function. 21 Gender and E2 treatment did not increase protein expression of the receptors (Fig. 1), and so, the differences observed in T cell function must be due to the dominance of signaling of one receptor over the other, or due to additional mechanisms such as differences in dimerization on ligation.
It is notable that E2 can also bind a third receptor, G protein-coupled estrogen receptor 1 (GPER), which mediates rapid nongenomic responses to E2. 22 GPER has been shown to activate multiple signaling pathways, including the MAP kinase signaling cascade activation. 23 The changes in cytokine secretion and polyfunctionality observed in the Ag-specific T cells could be the work of not only the canonical ERs but also be influenced by GPER. This needs to be further studied to better understand how to improve the beneficial signaling cascades in these T cells that will promote stronger antitumor responses, such as the combined secretion IFNγ and TNFα and simultaneous lytic activity as suggested by the expression of CD107a. Furthermore, E2 had a dose-dependent effect on the production of IFNγ and granzyme B in T cells from female but not in male donors, which secreted higher levels of these molecules at 0 nM E2 (Fig. 2A, B).
On the contrary, we showed that E2 had a dose-dependent increase on the secretion of IL-4, and TNFα in both female and male T cells, both show very similar trends (Fig. 3A, B).
There are many studies reporting the effects of E2 and ER signaling on T cell and other immune cell development and maturation, but the effect on their overall function is still controversial. Studies have reported that E2 can act on T cells in both ER-dependent and ER-independent mechanisms. Estrogen can modulate both CD4+ (Th1, Th2, Th17, and Tregs) and CD8+ T cell subsets. Estrogen has been shown to increase IFNγ secretion via ERα binding to the Ifnγ promoter or through upregulation of the Th1 transcription factor T-bet.24–26
The effect of E2 on Th2 subsets and its prototypic cytokine IL-4 has is also controversial and remains unclear. Some studies show either a stimulatory effect 27 or no effect26,28,29 of E2 on IL-4 secretion and GATA-3 expression. High levels of E2 have been shown to skew the T cell differentiation from Th1 to Th2 in human unstimulated PBMCs. 30 The effects of E2 on Th17 cells have been recently reported but they vary depending on the experimental conditions. Estrogen deficiencies correlate with increase on Th17 cells and IL-17a secretion, 31 but they also correlate with increased IL-17a production and signaling in postmenopausal women. 32 Estrogen has also been shown to elevate expansion of Tregs and upregulate expression of FoxP3, PD-1, and CTLA-4 via ERα signaling.33–35 Each of these previous studies has focused on endogenous T cells in the body; this is the first study to demonstrate the effect of E2 on genetically modified and stimulated Ag-specific T cells that would be used for adoptive T cell transfer immunotherapy against cancer.
There is only one study in which the effect of sex on checkpoint blockade inhibitors was reported. Blockade of the immune inhibitory molecule PD-L1 resulted in better antitumor immune responses in female mice than in male mice due to reduced regulatory T cell numbers. It is important to determine the effect that estrogen will have on adoptively transferred T cells in the patient body and in the tumor microenvironment (TME). Tumors and metastases can have dysregulated and fluctuating amounts of estrogen that will affect immune responses differently in males and females.36–38 Our data demonstrated that by increasing E2 during Ag stimulation in females, the result is even more polyfunctional T cells. This may be important for increasing helper T cell responses, effector T cell responses, and importantly, the development of memory T cells with antitumor function. Previous studies have reported that polyfunctionality is critical for memory T cell formation. The more polyfunctional the better the effector memory survival. 39 It is still not clear what the effect of elevated concentrations of E2, found in the TME, had on the modification of T cell function and survival. However, in this study, we have reported the effect of not only physiological concentrations but also superphysiological concentrations of E2 in both male and female Ag-specific T cells used for immunotherapy.
Our data suggest that T cells given as adoptive transfer therapy could continue to be modulated by superphysiological concentrations of hormone in the TME. We have described the effect of different E2 concentrations on single cytokine production and polyfunctionality of both male and female T cells. Importantly, we have shown that the ERα is important in E2 stimulation of T cells regardless of whether they are from male or female donors. Inhibition of the ERβ promoted the increases in cytokine production on E2 treatment. Therefore, ERβ is a competitor of ERα in T cells. The differences in T cell function driven by E2 between male and female donors as demonstrated by our results are downstream of the receptors. Therefore, future studies are required to determine whether coactivators or corepressors are triggered on ER engagement, impacting T cell function, or whether an entirely different signaling pathway is triggered.
Footnotes
Acknowledgments
The research presented in this study was made possible by funding from the American Cancer Society, Research Scholar Grant award number RSG-16-242-01-LIB, and Loyola University Chicago Research Funding Council, Bridge Funding. We thank Keith E. Watkins for assistance in image preparation. We thank Dr. Pablo Saenz-Lopez for assistance and guidance with polyfunctionality analysis of human T cells.
Author Disclosure Statement
No competing financial interests exist.
