Abstract
In biotechnology, the term ‘cell-free’ describes a wide range of established and emerging processes and technologies. However, broad application of the term ‘cell-free’ leads to confusion and ambiguity in the field, especially when applied to biomanufacturing. When engaging with investors, government agencies, and industry partners, we have found that the association of ‘cell-free’ with numerous technologies spanning diverse applications creates unnecessary confusion. As a result, we believe the lack of clarity complicates communication, impacts technology adoption, and creates challenges in securing funding and regulatory recognition. Specifically, in this opinion, we focus on terminology used to describe enzyme-driven biocatalytic approaches for manufacturing small molecules, often referred to as ‘cell-free biomanufacturing.’ This ‘cell-free’ approach harnesses biocatalytic pathways, in the absence of living cells, to synthesize small molecules used as pharmaceuticals, nutraceuticals, flavors, and fragrances, but also specific commodity chemicals and fuels. To clarify terminology, we introduce the new terms ‘exozymes’ and ‘exozyme systems’ as unambiguous descriptors for ‘cell-free biomanufacturing.’
Introduction
The use of the term ‘cell-free’ has a long-standing history that has inspired and contributed to the rise of biotechnology. Importantly, ‘cell-free’ has served as a crucial concept and method for studying and manipulating biochemical processes outside the confines of living cells.1–4 Today, the most well-known and successful applications of ‘cell-free’ technologies include next-generation sequencing-based diagnostics utilizing cell-free DNA/RNA (cfDNA/RNA),5,6 cell-free protein synthesis (CFPS),7–9 and cell-free biomanufacturing of small molecules2,10–14 among others. While all these technologies are ‘cell-free,’ their operational mechanisms, processes, and final products differ significantly (Fig. 1). In this opinion, we will focus primarily on biocatalysis and its role in ‘cell-free biomanufacturing’ for the production of small molecules.
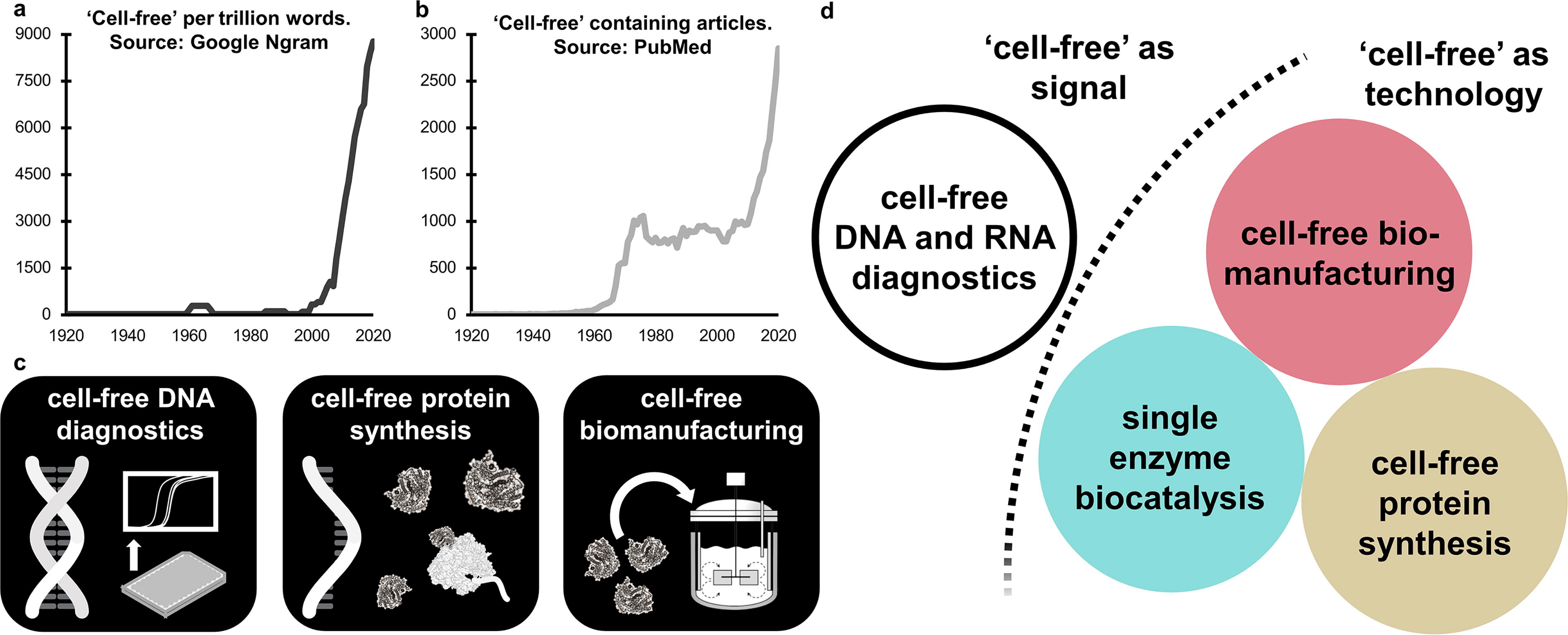
Historically, Eduard Buchner’s groundbreaking experiment with yeast extracts in 1897 demonstrated that ethanol fermentation could occur without living yeast cells,3,15 alerting scientists and bioengineers to the possibilities and usefulness of conducting biochemical reactions in vitro, ex vivo, or ‘cell-free’.2,7,8,16–18 While in vitro, ex vivo, and ‘cell-free’ carry nuanced distinctions—in vitro highlights in a test tube, ex vivo emphasizes outside a living organism, and ‘cell-free’ focuses on extracted molecular components—all refer to biochemical reactions outside living organisms and, in the cases described here, align with the concept of biocatalysis. Early work by Buchner and others studying the forces responsible for the activity of ‘ferments’ laid the foundation for what would become the diverse field of ‘enzymology’ focused on the study of ‘enzymes’, a term derived from Greek meaning ‘in leaven’ or ‘in yeast’.3,15,19,20 As the field of enzymology advanced, the ability to study isolated biochemical reactions within a controlled environment enabled scientists to better understand, manipulate, and regulate cellular metabolism as well as elucidate the genetic code with unparalleled precision.21–23
Modern ‘cell-free’ technologies, particularly those applied to synthetic biology and metabolic engineering, offer numerous distinct advantages, including a controllable, open platform where individual components can be added or removed at will.2,24–28 For example, high-throughput screening platforms using cell-free protein synthesis have been developed to rapidly optimize enzyme variants29,30 for industrial use. Additionally, cell-free systems for protein synthesis allow for the integration of unnatural amino acids, expanding the scope of protein engineering. In biomanufacturing, enzyme cascades are being employed to produce complex small molecules such as pharmaceuticals, mitigating toxicity and feedback inhibition that typically effect cell-based production systems and eliminating the multitude of metabolic pathways and processes toward which a living organism naturally channels carbon flux.2,10,13 Ongoing advancements in cell-free systems continue to enhance accessibility and efficiency while reducing costs and development timelines.
So, what is actually meant when we say ‘cell-free’?
As described above, expansion of ‘cell-free’ methods, driven by many different use cases, has caused the term ‘cell-free’ to gain immense popularity over the last 15 years. While the growth of ‘cell-free’ as a method and term is supported by both Google Ngram data (Fig. 1a) and PubMed records (Fig. 1b) ambiguity surrounding the terminology makes it challenging to distinguish between different ‘cell-free’ technologies and their ultimate application. Interestingly, comparing Ngram data, it appears that the growth rate of term usage for ‘cell-free’ from 2000 to 2020 was higher than that of ‘metabolic engineering’, ‘biocatalyst’, ‘enzyme cascade’, ‘biocatalytic cascade’, or any combination of enzyme engineering (e.g., rational design, directed evolution, etc.). Historically, limited usage of the term ‘cell-free’ started in the 1960s, primarily in reference to biochemical studies focused on the characterization of individual enzymes.23,31 The most prominent early ‘cell-free’ application was the Matthaei-Nirenberg experiment, which led to the discovery of the first codon of the genetic code.22,32 Since the early 2000s, however, the term ‘cell-free’ has seen a dramatic surge in the general language and scientific literature (Fig. 1a and b). This increase was likely driven by the development of cfDNA diagnostics based on next-generation sequencing (Fig. 1a) and the introduction of novel methods for cell-free protein synthesis (Fig. 1b). Yet despite increased popularity, the term ‘cell-free’ has become broad and ambiguous, lacking clarity and specificity and diluting its meaning.
Multiple fields now use ‘cell-free’ to describe a wide range of disparate applications and underlying technologies, each with distinct and often unrelated operational principles, objectives, and challenges 33 (Fig. 1d). To provide clarity, we briefly summarize how the term ‘cell-free’ is used across major biotechnological disciplines with industrial and commercial importance, highlighting overlaps in technologies, and challenges associated with the terminology (Fig. 1c).
First, the use of cell-free DNA (cfDNA) and RNA (cfRNA) has transformed diagnostics, enabling noninvasive genetic analysis for prenatal screening, cancer detection, and disease monitoring.5,25 By isolating and sequencing nucleic acids from bodily fluids, these ‘cell-free’ approaches allow the detection of genetic mutations, epigenetic modifications, and disease biomarkers, revolutionizing early diagnosis and personalized medicine.5,24,34–37 These advances have not only expanded the diagnostic landscape but also contributed to the widespread adoption of ‘cell-free’ as a term in clinical applications.
Next, in synthetic biology and protein production, ‘cell-free’ has been used primarily in relation to cell-free protein synthesis (CFPS), which is a critical tool for rapid in vitro transcription–translation (TX-TL) and is used to facilitate enzyme engineering, functional protein studies, and the development of vaccines and biologics.7–9,29,30,38–40 By bypassing the constraints of living cells, CFPS enables the expression of complex or toxic proteins, allows for the incorporation of nonnatural amino acids, and can accelerate screening and optimization processes.41–46 While CFPS is often used as a prototyping platform for metabolic engineering and enzyme design,9,47–51 it has also found applications in the scalable production of therapeutic proteins.52,53
Finally, ‘cell-free’ biomanufacturing refers to technologies that apply enzyme-driven processes to synthesize small-molecule chemicals, pharmaceuticals, and sustainable bio-based commodities.2,13,18,33,54–57 Cell-free biomanufacturing leverages the use of multi-enzyme cascades outside of living cells, which allows for the construction of new-to-nature biosynthetic pathways, improved reaction control, expanded substrate compatibility, and elimination of barriers associated with cellular toxicity and metabolism.2,13,18,54,58
Other technologies (e.g., in immunotherapy, regenerative medicine, or biocomputing59–63 ) that are essentially ‘cell-free’ also contribute to the broader conversation, making clear communication even more difficult. As a result, we argue that there is a need to clarify terminology and precision for the term ‘cell-free’ but especially as it applies to ‘cell-free’ biomanufacturing as described below.
The need for clarity and precision in ‘cell-free’ terminology
The term ‘cell-free’ has become increasingly ambiguous, describing a wide range of technologies across different applications (see above). This lack of clarity complicates communication, obscures the unique advantages of specific systems, and creates challenges when engaging with collaborators, investors, funding agencies, regulatory bodies, and industry partners. Through our own experiences in fundraising, grant reviews, and industry discussions, we have encountered significant confusion. For example, when introducing ‘cell-free’ as a method for manufacturing small molecules, the common refrain is, “So, you use lysates?” or “Is there one enzyme that can do that?” or “I hear that cell-free protein synthesis (or in vitro transcription/translation) is growing in popularity!” Clearly, many stakeholders either have a narrow, preexisting definition of ‘cell-free’ or struggle to understand its diverse applications, let alone quickly navigate to the specific ‘cell-free’ use case related to small-molecule biomanufacturing. While ‘cell-free’ was once a straightforward descriptor, the rapid expansion of these technologies now demands a more precise and structured approach to ensure effective communication and continued innovation in this emerging bio-industrial sector.
Through this opinion, we aim to reconcile the language ambiguity surrounding ‘cell-free’ by i) distinguishing distinct fields within ‘cell-free,’ such as cell-free biomanufacturing, cell-free DNA/RNA diagnostics, and cell-free protein synthesis, in order to streamline communication, foster collaborations, and avoid misunderstandings that could hinder the growth of this emerging bio-industrial sector; and ii) proposing a standardized language specific to cell-free biomanufacturing to resolve discrepancies in the description of multi-step biocatalytic processes, creating a clear pathway for understanding, development, and commercialization.
Exozymes: A new terminology for ‘cell-free’ biocatalysis
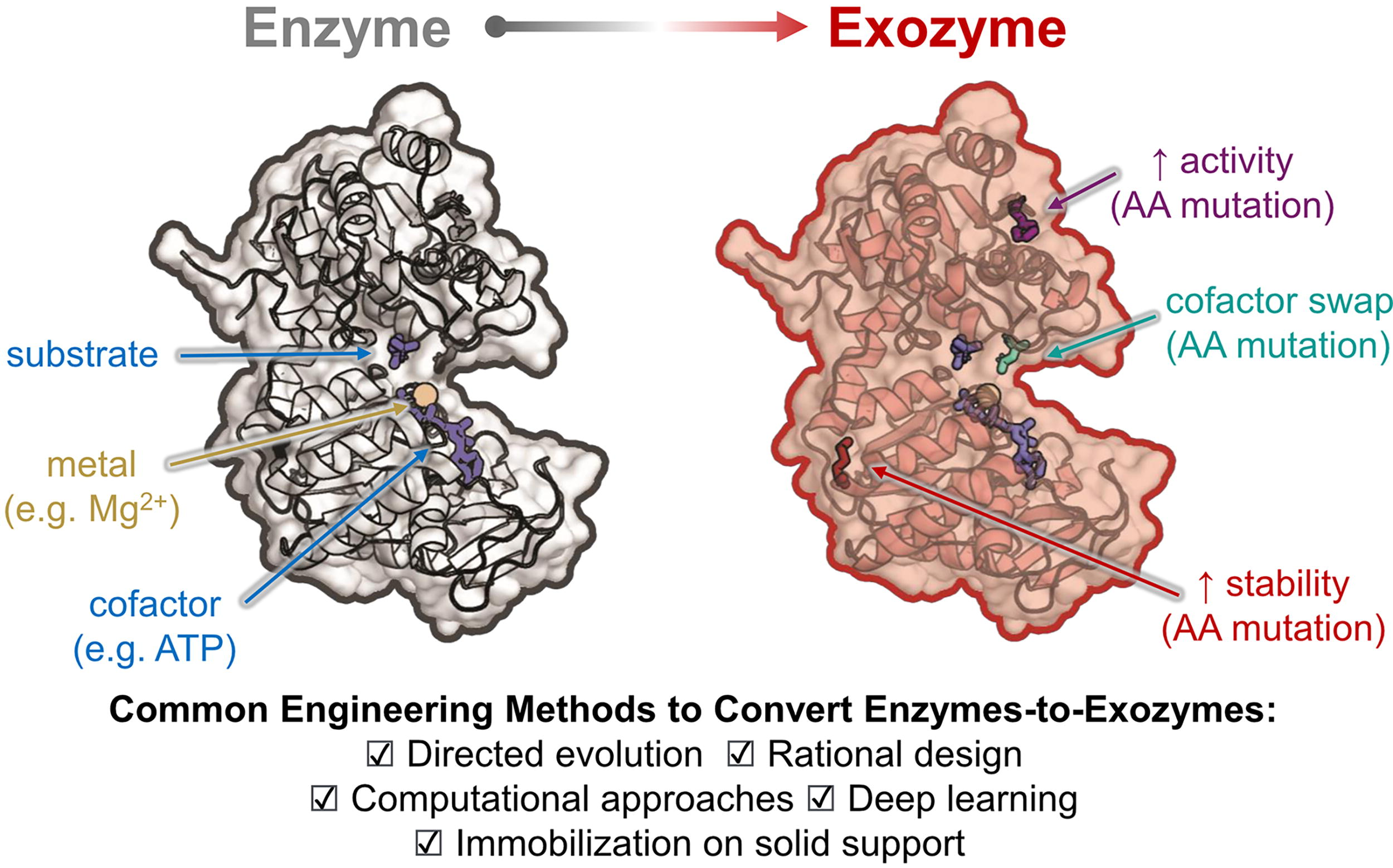
We define exozymes (Box 1) as enzymes that are primarily used ex vivo in bio-industrial applications (Fig. 2). Specifically, exozymes perform biocatalytic reactions independent of any living cell and may be naturally selected or are engineered to exhibit exceptional stability, resilience, and activity under industrially relevant conditions that would typically not be suitable for intracellular enzymes. One of the primary reasons for the growing relevance of exozymes in industrial applications is the increasing ability to obtain enzymes that maintain high turnover rates, even in harsh environments such as elevated temperatures, variable pH levels, or the presence of organic solvents.2,54,64–68 Additionally, exozymes can be tailored for specific purposes by modifying their amino acid sequences to exhibit improved properties using methods such as genome mining, structure-based rational design, directed evolution, and computer- or AI-aided next-generation enzyme engineering approaches. Alternatively, exozymes can be generated by attaching an enzyme to solid supports through tagging with specific epitopes or linker sequences to improve their stability, longevity, and recyclability (Fig. 3).69–74 The concept of exozymes therefore expands upon traditional enzymology by distinguishing enzymes with unique properties that either retain functionality for use outside of the cellular context or are specifically designed for practical ex vivo activity with the primary goal of manufacturing small molecules. The term exozymes also shifts focus from traditional intracellular enzymes, which primarily are used to sustain cellular life, to biocatalysts that function in vitro, ex vivo, or in a ‘cell-free’ environment (Fig. 2 & Fig. 3). Further distinguishing their use, exozymes are not restricted to the same biological constraints as intracellular enzymes, allowing more diverse and challenging industrial processes. Exozymes can either be based on enzymes naturally secreted from the cell (i.e., secreted enzymes)75–77 or can be normally intracellular enzymes that are inherently stable or engineered to remain stable and functional after being isolated from the intracellular environment. In either case, the primary function of an exozyme is to catalyze the transformation of a small-molecule substrate into a product outside the context of a cell with the goal of being utilized for industrial biomanufacturing.
Box 1. A New Language for Cell-Free Biomanufacturing
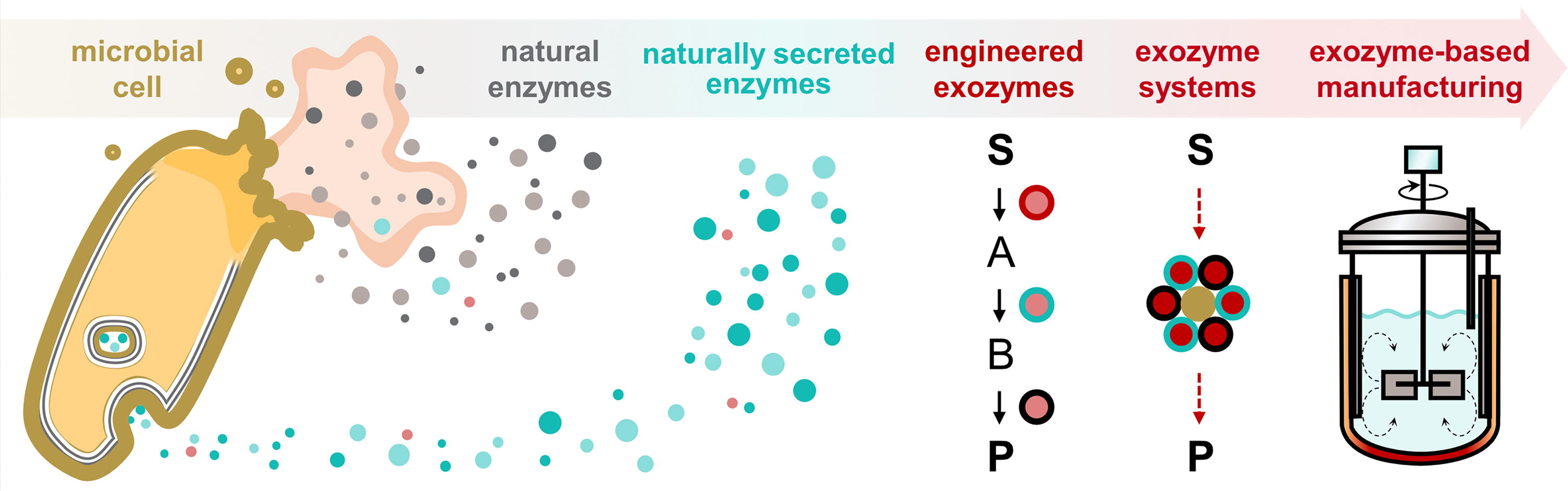
A prime example of an exozyme in industrial use is xylose isomerase (XI). 78 XI is central to the large-scale production of high-fructose corn syrup (HFCS), a common sweetener in processed foods and beverages. 79 XI catalyzes the conversion of glucose to fructose, which is a crucial reaction step for HFCS production. Initially, the native form of the XI enzyme presented challenges in terms of stability and operational efficiency, limiting its (re)use in industrial processes.78,79 However, through immobilization—a process that physically binds the enzyme to a solid support—the stability and operational lifespan of XI were significantly enhanced, 80 transforming it from a regular enzyme to an exozyme. As an exozyme, immobilized XI can withstand the rigors of a continuous flow system, maintaining functionality over extended periods of time (up to 200 days) and multiple cycles of use. 68 In this case, the enzyme-to-exozyme transformation enabled cost-effective production by reducing the need for frequent enzyme replenishment, thereby facilitating the economic feasibility of HFCS production on an industrial scale. Similar to XI, many other exozymes are used outside of cells in the food and beverage industry to enhance production and quality. For example, amylases break down starch into sugars for brewing and baking, pectinases clarify fruit juices, and lactase removes lactose from dairy products.
A class of naturally secreted enzymes that fulfill the definition of exozymes are fungal laccases. Laccases are multicopper enzymes that catalyze one-electron oxidations.81–83 Secreted fungal laccases play key physiological roles in breaking down lignin and other complex organic molecules in the fungus’ environment to mobilize nutrients for cell survival. 81 For instance, fungal laccases enable lignin degradation to provide access to cellulose used to generate energy via central metabolism in vivo. 84 Hence, laccases are ecologically vital in nutrient cycling by returning carbon to the soil. 81 The broad substrate specificity of laccases, their ability to use oxygen as an electron acceptor, and their stability across diverse pH levels make laccases well-suited for flexible industrial processes and classified as exozymes.82,83,85–87 Consequently, companies like Novonesis (formerly Novozymes), Amano Enzyme, IFF (after merging with DuPont’s Nutrition & Biosciences), AB Enzymes, and BASF have commercialized laccases for various applications, including wastewater treatment, pulp bio-bleaching, delignification, textile treatment, and food processing.82,85,87
While most naturally occurring secreted enzymes perform reactions without the need for cofactors beyond inorganic ions, allowing them to catalyze basic biochemical reactions outside of cells, the lack of cofactor utilization also limits the types of biotransformations they can perform. In contrast, enzymes selected for their exozyme properties or that are engineered to be exozymes can integrate cofactor-dependent reactions to perform a more diverse array of biochemical reactions and unlock applications far beyond what can be achieved with traditional secreted enzymes. The ability to utilize various cofactors becomes exceptionally beneficial when combining exozymes into exozyme systems (described in “Exozyme systems for cell-free biomanufacturing”). However, it is important to note that not all intracellular enzymes used ex vivo qualify as exozymes. For example, cytochrome P450s, employed in drug metabolism studies and chemical synthesis, rely on complex electron transfer partners. Similarly, iron-sulfur (Fe-S) cluster-containing enzymes, such as nitrogenase used for ammonia production, and enzymes dependent on intricate cofactors like methionine synthase (requiring cobalamin) or thymidylate synthase (dependent on tetrahydrofolate), face limitations due to their complex cofactor requirements and fragile catalytic cycles. These enzymes, while valuable for specific applications, lack the inherent autonomy and robustness that define true exozymes. Indeed, P450s and Fe-S containing enzymes would require additional engineering to be considered exozymes. The ability of exozymes to function autonomously in varied environments, combined with advances in enzyme engineering and immobilization techniques, makes them indispensable for the future of modern bioindustrial manufacturing. Furthermore, by engineering exozymes to be functionally compatible with other exozymes and assembling them into coordinated cell-free cascades that maintain cofactor homeostasis, complex ‘exozyme systems’ can be developed (see section “Exozyme systems for cell-free biomanufacturing”).11,13,54,56,88–90 These ‘exozyme systems’ are specifically designed to carry out multiple sequential reactions, converting low-cost, simple substrates into complex, high-value molecules through a series of linked biocatalytic steps. As a result, exozymes and exozyme systems are reshaping industrial biocatalysis by offering robust, efficient, and scalable solutions across industries. We argue that adopting the standalone terms ‘exozyme’ and ‘exozyme system’ when referring to cell-free biomanufacturing will help provide focus and standardize language, while also supporting the creation of a clear framework for understanding, development, external investment, and commercialization in the cell-free space.
Exozyme systems for cell-free biomanufacturing
As introduced in the section above, exozyme systems (Box 1) are purposely designed biochemical pathways composed of optimized exozymes that are selected for—or engineered to—function in industrial applications ex vivo. Exozyme systems leverage multiple exozymes working in concert, strategically integrated with essential components, such as cofactors, chemical reagents, and environmental controls, to enable continuous, efficient, and cost-effective production of commodity chemicals, biofuels, nutraceuticals, pharmaceuticals, and other bio-based products (Fig. 2). A key advantage of exozyme systems is their ability to enable cost-effectiveness and scalability in ways traditional fermentation cannot match. This scalability is due to the properties of exozymes (Box 1 & Fig. 3), which can be amplified by advancements in biotechnology, including enzyme engineering, high-throughput screening, and AI-driven protein design. Unlike fermentation, which faces cellular constraints, exozyme systems bypass metabolic bottlenecks, allowing for direct optimization of catalytic efficiency, stability, and specificity. Moreover, exozyme systems are inherently green catalysts, contributing to sustainability by reducing the need for living cells and minimizing waste. Sustainability can be directly linked to exozyme robustness and is a crucial feature that enhances the industrial viability of exozymes and exozyme systems. Unlike whole-cell systems, which require tightly controlled conditions, exozyme systems operate in optimized environments that maximize reaction efficiency and durability, further distinguishing them from enzyme-based or cell-free approaches of the past. This is why we believe that exozyme systems directly tie to current market trends (e.g., the abovementioned sustainability, resilience, cost-effectiveness, and scalability) that are increasingly important, especially in the pharmaceutical and nutraceutical sectors.
Ideally, exozyme systems are also autoregulatory, which means that intermediate compounds and energy (e.g., cofactors ATP or NADH) are produced and consumed in tightly controlled steps, allowing the system to achieve high overall yields and conversion efficiencies that can also help overcome thermodynamic bottlenecks by pulling the unfavorable equilibrium reactions in the desired direction. Or in other words, exozyme systems are autoregulated for efficient, targeted production while cells will always autoregulate for maximum growth. Additionally, exozyme systems often address a common limitation in enzymatic reactions: the need for cofactor regeneration. Many biochemical reactions depend on cofactors, such as ATP, NAD(P)+/NAD(P)H, FAD, CoA, or S-adenosyl methionine (SAM), to provide a thermodynamic driving force. Recycling these cofactors is essential to ensure the continued function of biochemical cascades and reduce costs. Many older biocatalytic systems often faced high operational costs and reduced efficiency because they required the continuous addition of cofactors or the addition of sacrificial co-substrates. In modern examples of engineered exozyme systems however, cofactor regeneration can be directly incorporated into the pathway design, enabling the recycling and reuse of cofactors. The ability to regenerate cofactors significantly enhances sustainability and economic viability in large-scale biomanufacturing, making exozyme systems a promising choice for industrial processes where cofactor availability would otherwise be a limiting factor.
We believe exozyme systems hold the potential to deliver on synthetic biology’s promises of sustainable and efficient biomanufacturing by providing a stable, adaptable platform that can be engineered for a wide range of bioproducts. By operating in controlled ex vivo environments, exozyme systems provide increased control over reaction conditions, optimizing efficiency and scalability to levels beyond those achievable in vivo. The ex vivo nature and modular design of exozyme systems also offer additional levels of resilience and reliability, positioning exozyme systems as a compelling answer to many of the longstanding issues in biomanufacturing such as low product yields, the inability to produce cytotoxic products (e.g., some fuels, polymer building blocks, flavors, fragrances, nutraceuticals, and pharmaceuticals), and scalability. Demonstrating the versatility and promise as a biomanufacturing solution, exozyme systems of varying complexity have been applied to produce a wide range of chemicals (see Table 1). With the following three examples, we illustrate how exozyme systems have been scaled, outperform highly developed fermentations, or are revolutionizing drug manufacturing (Fig. 4).
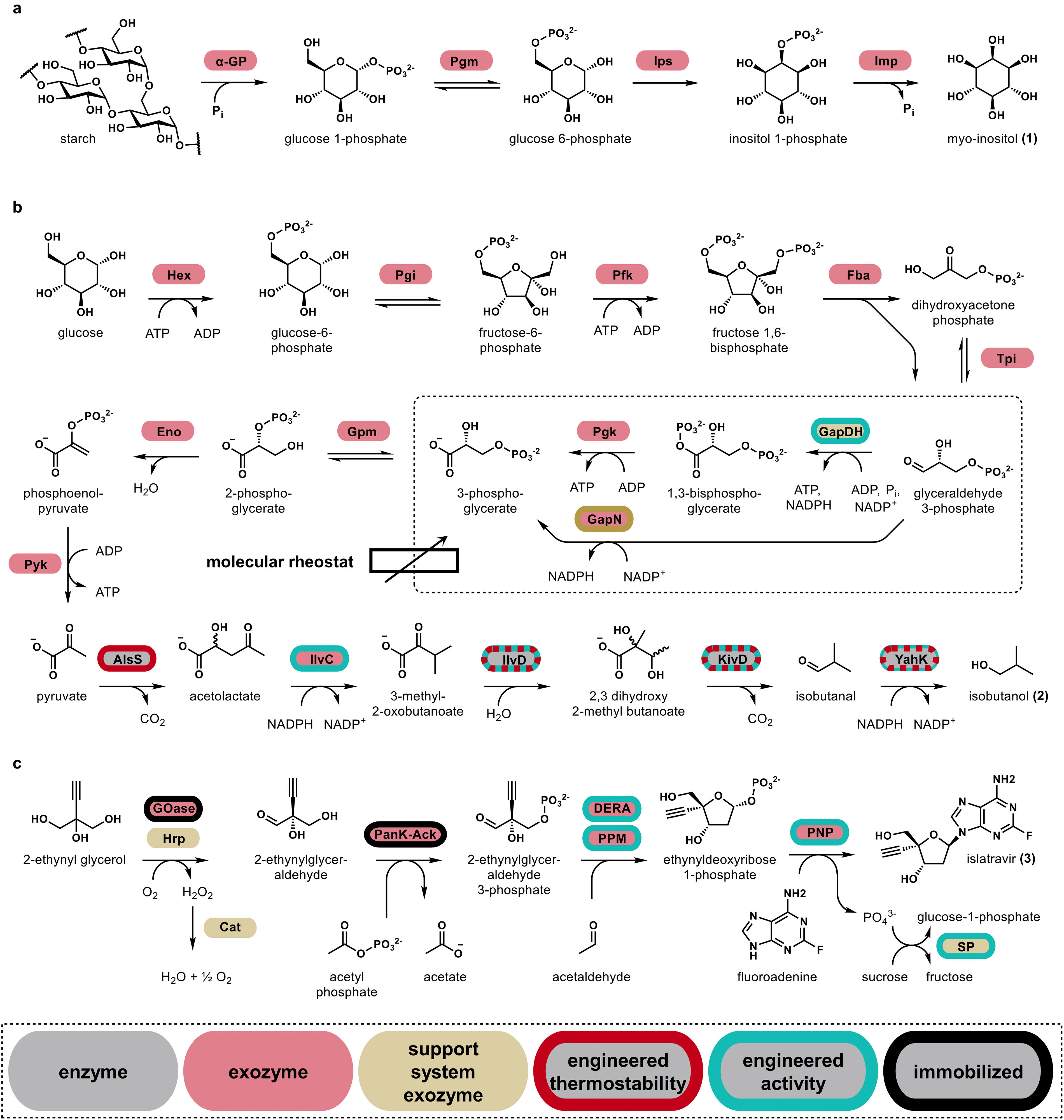
Examples of different exozyme systems
There are different reaction systems that can be associated with exozymes: An
Example I — commodity chemicals
A prominent example of an exozyme system that has been impressively scaled is the 20,000 L scale production of myo-inositol, a commodity chemical used in the drug, cosmetic, and food industry. 13 The myo-inositol exozyme system uses a highly efficient ex vivo process to convert starch directly into myo-inositol without ATP or NAD+ supplementation (Fig. 4a). The pathway comprises exozymes from hyperthermophilic microorganisms, such as Thermotoga maritima and Thermococcus kodakarensis, enabling operation at elevated temperatures suitable for industrial applications. In this case, normally intracellular enzymes were selected for properties like thermostability that allow them to function as an exozyme system outside of a cell. By implementing the five-step exozyme system for biochemical transformation of maltodextrin to myo-inositol, the system achieved a high product yield of 98.9% and reached a titer of 95 g/L over 48 h 13 demonstrating the potential of exozyme platforms to efficiently transform common feedstocks into commodity product at an industrially relevant scale.
Example II —biofuels
Exozyme systems may also be transformative for biofuel production.2,58 The production of isobutanol—a biofuel and commodity building block chemical with promising properties as a petroleum alternative—is a good case study for the transformative potential of exozyme systems. 54 Fermentative microbial systems for isobutanol production struggle to meet cost and yield metrics for commercial viability. Although moderate titers exceeding 100 g/L have been reported, reaching these higher titers requires an expensive and energy-intensive in situ separation process to maintain isobutanol concentrations in the fermentation broth below 2%, which is critical for cell viability.106,124 The isobutanol exozyme system is a key innovation (Fig. 4b) that overcomes toxicity and process limitations that historically have led to low yields and productivity in cell-based processes. For full implementation, the isobutanol exozyme system relied on the development and application of several exozymes with enhanced stability, derived either from hyperthermophilic organisms or engineered for thermo- and solvent tolerance from less optimal enzyme variants.54,88 The engineered robustness of the exozymes in the isobutanol system allows the exozyme system to function continuously for many days even in the presence of high isobutanol concentrations and an organic overlay, which is not compatible with most in vivo applications. The organic phase serves a dual purpose: It not only facilitates the removal of isobutanol as it is produced but also helps shift the equilibrium of the reaction toward product formation. 54 Thus, the overlay essentially acts as a thermodynamic sink, preventing reactions from stalling and allowing the biochemical transformations to progress at high rates. Importantly, the ability to maintain an isobutanol concentration at 4% or higher in the aqueous phase significantly improves extraction efficiency and can reduce the energy costs of separation in future processes. 54 The isobutanol pathway also illustrates another key aspect of exozyme systems: cofactor recycling and regulation. In particular, the isobutanol exozyme system efficiently recycles NAD(P)H and implements a so-called ATP rheostat to autoregulate ATP levels. 88 As a result, the exozyme system provides its own biochemical energy during production, allowing for long-term, continuous production of isobutanol without the addition of costly, sacrificial substrates. 54 The exozyme system with an organic phase overlay improves both titers and yields, achieving a maximum productivity of 4 g/L/h and an effective titer of 275 g/L with a 95% yield54—values that exceed even the best-performing ethanol fermentation processes and also approach the economic viability required for isobutanol to compete with petroleum-based fuels. 125
Example III—pharmaceuticals
One of the most remarkable examples of an exozyme system in pharmaceutical manufacturing is the production of islatravir, an experimental HIV drug developed by Merck Sharp & Dohme (MSD) and Codexis. 126 The islatravir example underscores the power of enzyme engineering coupled with immobilization strategies to enhance stability and scalability for successful implementation of an exozyme system.64,73 The production of islatravir involves nine distinct exozymes (Fig. 4c) in a one-pot synthesis. Five of the enzymes were engineered, and three were immobilized to optimize stability, specificity, and catalytic activity, turning them into exozymes (using the nomenclature proposed here) and allowing the cascade to function seamlessly in the ex vivo process. 126 Converting the enzyme cascade into an exozyme system enabled MSD and Codexis to effectively couple reaction steps, eliminating the need for intermediate product purifications and facilitating a streamlined process suited for industrial-scale production of islatravir. 126 The islatravir exozyme system is currently supporting clinical trials.127,128 Innovative exozyme systems to produce islatravir, molnupiravir, and other small-molecule therapeutics may reflect a broader shift in the pharmaceutical industry, from solely organic synthesis or synthetic biology-based microbial fermentation systems, to integrated approaches that combine medicinal and organic chemistry with bio-based exozyme systems.129–131
In summary, the above examples underscore the versatility and transformative potential of exozyme systems across various markets—from commodity chemicals and biofuels to pharmaceuticals. A non-exhaustive list in Table 1 highlights additional examples of exozyme systems, exozyme system prototypes, and ‘classical’ multi-enzyme biocatalytic systems that have been previously developed, establishing a strong foundation and rationale for advancing the field. We argue that by combining advanced natural and engineered exozymes with innovative process design, exozyme systems are the logical successors to traditional fermentation-based SynBio and chemical processes to address challenges in a wide range of applications. As technologies to develop and improve exozymes continue to evolve, driven in part by advanced computational methods and improved high-throughput enzyme characterization and engineering capabilities, exozymes and exozyme systems are well-positioned to drive the transition to novel or more sustainable bio-based production processes, underscoring the transformative role that exozymes could play in advancing a more sustainable global bioeconomy.
Conclusion
By clarifying and redefining terminology around ‘cell-free’ (Box 1), with a specific focus on ‘cell-free’ biomanufacturing, we aimed through this opinion to reduce confusion, foster meaningful collaboration, attract investment, and inspire innovation, ultimately making a positive and lasting impact on industry-redefining technologies. As the biomanufacturing field grows to encompass additional technologies and capabilities like the development and use of exozyme systems, it shifts focus away from a myopic view of fermentation as the only industrial solution. By redefining ‘cell-free biomanufacturing’ as ‘exozymes’ and ‘exozyme systems,’ it will, in our opinion, streamline communication and establish a structured framework for this rapidly growing bio-industrial field (Figs. 1d and 2). By leveraging exozymes optimized for stability and activity to be utilized in ex vivo conditions and assembling them into exozyme systems, bioengineers can effectively overcome traditional challenges such as cellular toxicity, metabolic burden, and process inefficiency. The versatility, robustness, and ability to integrate cofactor regeneration into exozyme cascades further highlight the role of exozyme systems as a transformative innovation for building a new bioeconomy, to reduce dependency on fossil resources and advance green chemistry principles. Therefore, modifying terminology to focus on exozymes and exozyme systems that provide scalable and efficient pathways for producing bio-based chemicals ensures that the field is aligned and consistent when referencing technologies, developments, and manufacturing. We believe the use of exozymes and exozyme systems will allow scientists, entrepreneurs, and investors, to immediately know that the technology being described references a ‘cell-free’ process for the production of small molecules, further streamlining conversation and development. As global efforts to establish and scale bio-based manufacturing accelerate, we firmly believe that a common language will ensure that progress can be tracked and evaluated without confusion.
Outlook
For the first time in history, bioengineers now have access to the tools necessary to design and create strains, enzymes, exozymes, and exozyme systems with unprecedented precision and scalability. Additionally, advances in artificial intelligence (AI) and machine learning (ML) are unlocking new possibilities that were previously unimaginable. With AI tools, bioengineers can quickly predict protein structures with exceptional accuracy, enabling the creation of highly optimized exozymes that function efficiently outside of cells for a wide range of industrial applications. The use of AI and ML for enzyme engineering is no longer speculative; AI and ML are ‘catalysts’ that are accelerating the discovery and design of novel biocatalysts. Hence, with AI’s ability to analyze vast datasets, predict enzyme behavior, and suggest beneficial mutations, it is now possible to design exozymes with optimized stability, specificity, and catalytic efficiency, tailored for even the most challenging industrial processes. The convergence of AI with cutting-edge molecular biology has unlocked this new frontier of possibilities for producing bio-based chemicals, pharmaceuticals, nutraceuticals, and even fuels, in ways that were once thought impossible or limited to a few selected examples. As the field continues to evolve, the integration of AI and machine learning into enzyme engineering will only accelerate, fueling innovation that will reshape industries and drive the next wave of AI-driven biomanufacturing revolution.
In a broader sense, we assert that this biotechnical AI-catalyzed revolution will push global efforts to improve sustainability by reducing reliance on petroleum while addressing climate challenges through innovation. However, to make these ambitious goals a reality, strategic investments in R&D, workforce development, and international collaborations remain essential, further highlighting the need for a common lexicon. As exozymes and exozyme systems continue to mature, integration of exozyme systems into key sectors, such as sustainable aviation fuel, biomaterial building blocks, nutraceutical production, and biopharmaceuticals, will solidify their role in the global bioeconomy. With nations worldwide converging on biomanufacturing as a solution to environmental and economic challenges, the future of biobased manufacturing is not just promising—it is inevitable.
Footnotes
Acknowledgment
The authors would like to thank Emily Roberts, Berge Hagopian, Chris Rabot, and Miyoshi Haruta for their valuable input during the writing process.
Authors’ Contributions
A.B.: Visualization—figures, writing—original draft, writing—review and editing. B.V.: Writing—original draft, writing—review and editing. J.M.B.: Visualization—figures, writing—original draft, writing—review and editing. P.H.O.: Writing—review and editing. E.V.: Writing—review and editing. S.A.S.: Writing—original draft, writing—review and editing. M.H.: Conceptualization, writing—original draft, writing—review and editing. L.G.: Conceptualization, writing—original draft, writing—review and editing. J.U.B.: Writing—original draft, writing—review and editing. Z.Z.S.: Writing—original draft, writing—review and editing. J.L.: Writing—review and editing. B.B.: Writing—review and editing. Y.J.B.: Writing—original draft, writing—review and editing. T.P.K.: Conceptualization, visualization—figures, writing—original draft, writing—review and editing. All authors have read and approved the article.
Disclaimer
The opinions in this perspective are solely those of the authors. The views expressed in the article do not necessarily represent the views of the DOE or the U.S. Government. The U.S. Government retains and the publisher, by accepting the article for publication, acknowledges that the U.S. Government retains a nonexclusive, paid-up, irrevocable, worldwide license to publish or reproduce the published form of this work, or allow others to do so, for U.S. Government purposes. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author Disclosure Statement
A.B., B.V., J.M.B., P.H.O., E.V., M.H., L.G., and T.P.K. are employees of eXoZymes, Inc., a company that commercializes enzyme and cell-free technologies. J.U.B., T.P.K., and P.H.O. are scientific co-founders of eXoZymes, Inc., and J.U.B. and J.L. serve on its board of directors. The terms exozymes and exozyme systems introduced in this article are intended to provide a clearer framework for the field and are not proprietary to eXoZymes, Inc. These terms are freely available for use by the broader scientific community, and no trademarks have been filed.
Funding Information
Work at eXoZymes Inc. was in part supported by the U.S. Department of Energy’s Office of Energy Efficiency and Renewable Energy (EERE) under the Conversion Research and Development Program award number DE-EE0011113 and received additional support by National Institute of General Medical Sciences of the National Institutes of Health under award number 1R43GM153108-01. This work was authored in part by the National Renewable Energy Laboratory, operated by Alliance for Sustainable Energy, LLC, for the U.S. Department of Energy (DOE) under Contract No. DE-AC36-08GO28308. Funding provided by U.S. Department of Energy Office of Energy Efficiency and Renewable Energy under the Cell Free and Immobilization Technologies initiative.
