Abstract
While gamma irradiation is commonly used as a standard for sterilization of single-use systems (SUS) in the biotech industry, an alternative sterilization mode using X-ray irradiation has been introduced. The integrity, safety, and performance of X-ray-treated SUS must be confirmed prior to implementation. This investigation employs gamma- and X-ray-treated SUS, comprised of filters, tubing, and bags, to sterilize media at filter loadings of 4, 20, and 100 L/m2 to evaluate the impact of potential extractables and leachables (E&L) on viable cell density and viability of Chinese hamster ovary cells. Statistical results confirm equivalent cell growth using media sterilized by X-ray and gamma-irradiated SUS, even at the worst-case filter loading. As the industry evolves toward acceptance of X-ray sterilization, this approach supports X-ray as a viable irradiation option and, further, provides a standard for future evaluation of the impact of E&L from SUS treatment on cell performance.
Introduction
Sterilization by gamma irradiation is a standard process for medical devices, pharmaceuticals, and the food industry. Many worldwide industrial sites offer gamma irradiation as a means of sterilization. In addition, over the last decade, new irradiation sources such as X-rays or electron-beams are becoming widely used to meet existing and future sterilization demand. Alternatives to gamma irradiation are driven in part by long lead times, limited element production, site location limitations, and radioactive element control/disposal security concerns, as well as by potential cost reduction opportunities. 1 Regulatory bodies, such as the FDA and EMA, require evidence that sterilization processes are effective and do not compromise product safety. 2 It is crucial to also confirm that sterilization by radiation generates an SAL (sterility assurance level) of ≤10−6, i.e., the probability of not more than 1 non-sterile item in 1 × 106 sterilized items. 3 This ensures that medical devices or pharmaceutical products are sterile and safe for use. 2 Using a common measure of radiation dose (kGy) and following established standards for validating irradiation sterilization, X-ray has been proven to deliver equivalent sterilization capabilities to those offered by gamma irradiation. 4
Gamma irradiation and X-ray sterilization can modify the physical and chemical properties of materials. 4 Therefore, assessing the potential impacts of X-ray irradiation on the chemical structure of the material of construction of single-use systems (SUS) material of construction helps to ensure that the materials remain safe and functional after sterilization and continue to meet regulatory requirements. These evaluations of product performance include any changes in mechanical properties, chemical stability, and overall functionality, such as microbial retention evaluation for filters. 1 Evaluating these changes is not the focus of this work and was covered by SUS manufacturer studies.
The transition from gamma-ray to X-ray necessitates a thorough risk evaluation to confirm that the new sterilization method does not compromise the integrity and performance of SUS. 5 The primary interest and necessity for performing the risk evaluation of X-ray irradiation lie in ensuring that X-ray irradiation is equivalent to gamma irradiation for the sterilization of SUS. This equivalency is crucial for maintaining the safety and quality of SUS that are extensively used in medical and pharmaceutical applications for supporting interactions with health authorities. The approach to risk evaluation involves a comprehensive multi-step X-ray implementation cycle, including the review of existing data related to X-ray and gamma irradiation treatments, including in particular extractables and leachables (E&L) profiles. 5 Several studies have already concluded that equivalent profiles of radical species and extractables are observed after gamma and X-ray irradiation, which aligns with the fact that gamma rays and X-rays interact with matter through the same mechanisms.5–11 Both types of radiation primarily lose energy via the Compton effect, where photons collide with orbital electrons, resulting in scattered photons and ejected electrons. 12 Further, generation of new chemical species undetectable by analytical tools is unlikely. However, hypothetically unexpected chemical species could empirically impact cell culture performance, while bioassays may provide complimentary value because of their sensitivity to potentially toxic compounds.
For example, the necessity of using dedicated bioassays in the context of biopharmaceutical manufacturing to assess the compatibility of SUS materials in contact with cell culture previously was recognized after a cytotoxic leachable from SUS was identified as being detrimental to cell culture growth. 13 Specifically, a study identified bis(2,4-di-tert-butylphenyl)phosphate (bDtBPP), a degradation product from a material of construction antioxidant additive that originated from SUS bioprocess containers becoming more cytotoxic after exposure to ionizing radiation. The study examined the detrimental effects of bDtBPP on cell growth, particularly its impact on mitochondrial membrane potential, which is crucial for cell viability and energy production. 13 Upon further investigation, it has been shown that common biocompatibility assays used to assess material compatibility of medical devices in animal studies are not sensitive enough to cover the specific requirements in biomanufacturing. For this reason, the ASTM guideline on Cell Culture Growth Assessment of SUS Material was developed and published in 2019. 14 The guideline focuses on recommendations with respect to designing an in-vitro cell growth study to evaluate the compatibility of SUS material in biopharmaceutical manufacturing processes. Although SUS used in upstream processes are usually not considered a medical device, biocompatibility testing of the latter allows the early identification of potentially adverse effects, streamlining the development process of new SU materials and systems. The present study proposes a methodology designed to isolate and enhance any signal arising from gamma versus X-ray irradiation of SUS containing sterile filters, tubing, and bags concerning cell compatibility.
The experimental workflow for this study followed that described in the ASTM Standard Guide for Cell Culture Growth Assessment of Single-Use Material (designation E3231-19). 14 Briefly, SUS was sterilized by either gamma or X-ray irradiation. Media filtered through and stored in SUS sterilized by either gamma or X-ray irradiation under various process conditions was used for cell cultivation of Chinese Hamster Ovary (CHO) cells. CHO cells, specifically a CHO DG44 cell line, were chosen due to their widespread industrial use and reasonable sensitivity to potential cytotoxic leachables.15,16 Both positive and negative controls were used to check the sensitivity and reproducibility of each test. Each test condition was tested in triplicate.
The study aims to demonstrate the equivalency of gamma and X-ray irradiation for SUS indirectly used in the culture of cells and provide a standardized approach for testing cell performance based on SUS components’ sensitivity to irradiation. Furthermore, the test assembly can be provided to any company to repeat the testing in-house with their media and cell lines.
Worst-Case Considerations
The study was designed to encompass many worst-case situations to identify any potential impact for a range of applications. First, SU filter assemblies were tested as quickly as possible after irradiation, with a target of less than 15 days. The short time allows for better observation of potential free radicals created by the irradiation process, which will decrease over time and so reduce any potential negative impact on the cell culture growth. The SU filter assemblies were irradiated at a dose range of 45–55 kGy, which is higher than the typical dose range of 25–45 kGy used for routine SU sterilization. The test involved a full assembly, including the filter, tubing, and bags. A well-established test system, using a cell line with reasonable sensitivity based on published data, was employed. 15
The experimental set also considered worst-case scenarios from an E&L perspective. A long extraction time of 23 h, instead of a flow-through process, was utilized. Extraction occurred at an elevated temperature, higher than typical filtration temperatures. The initial flush of the experimental filter was included in the study sample (bag #3) to represent the highest possible leachable level. The low 4 L/m2 ratio condition, corresponding to 5 liters passed through a 10-inch (0.8 m2) filter, was included to represent a worst-case surface area-to-volume ratio compared to typical loading volumes during routine manufacturing. Three dilution steps were used to cover a broad application range, from large-scale down to small-scale, and to allow for the evaluation of dilution-response trends. A 1:20 split ratio was employed, meaning only 5% v/v from the pre-culture was used, eliminating the need for dilutions by inoculation. Additionally, the study started with a low cell density, resulting in a high ratio of potential leachables per cell, which increased sensitivity. The cultivation time was extended to 6 days instead of the usual 3 days, with the first 5 days being the most suitable for cell growth. An additional day was added to observe potential latent effects of the different sterilization treatments.
Results
Cell growth
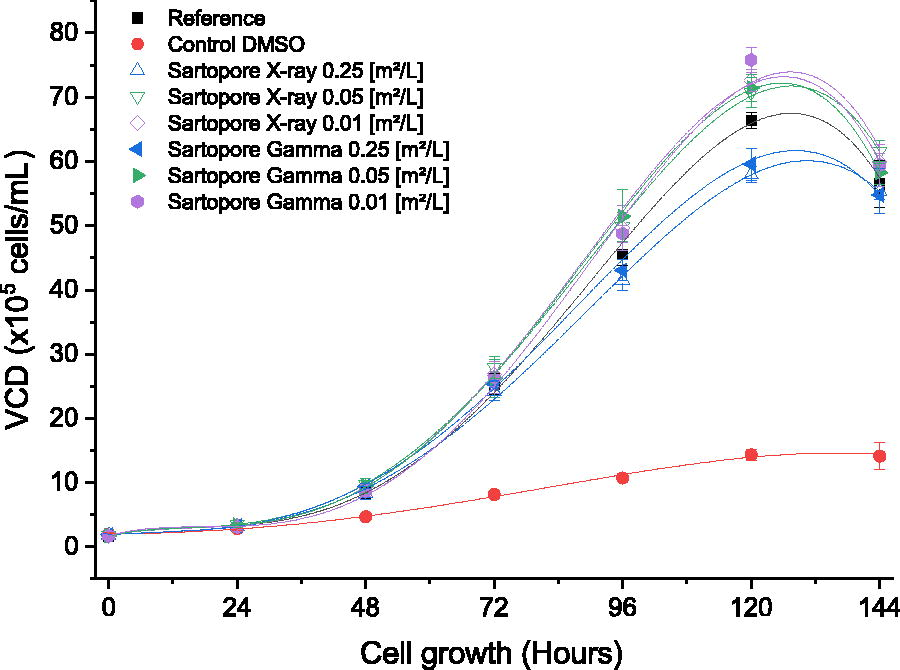
The overall viable cell density (VCD) curve with respect to time shows that all conditions, except for the DMSO control, followed the general trend observed for the untreated reference (Fig. 1, black line). The VCD curves showed the typical trend of a batch cultivation, consisting of a lag phase followed by exponential growth, a stationary phase, and a death phase due to nutrient depletion. The cell growth of the reference resulted in a VCD of approximately 25.3 × 105 cells/mL at 72 h and a VCD of approximately 56.5 × 105 cells/mL at 144 h. The 2% DMSO positive control led to reduced cell growth, indicating the cell culture was sensitive as expected. The filter membrane surface area to volume (S/V) ratios of 0.05 m2/L (Fig. 1, green triangles) and 0.01 m2/L (Fig. 1, purple diamonds) did not impact cell growth in a negative way, irrespective of the radiation technology applied (gamma or X-ray). The lowest dilution, representing the highest S/V ratio of 0.25 m2/L, had a small impact on cell growth after both gamma irradiation and X-ray irradiation (Fig. 1, blue triangles), which confirmed inhibition was detected by the method under the worst-case scenario for small-scale low-loading applications.
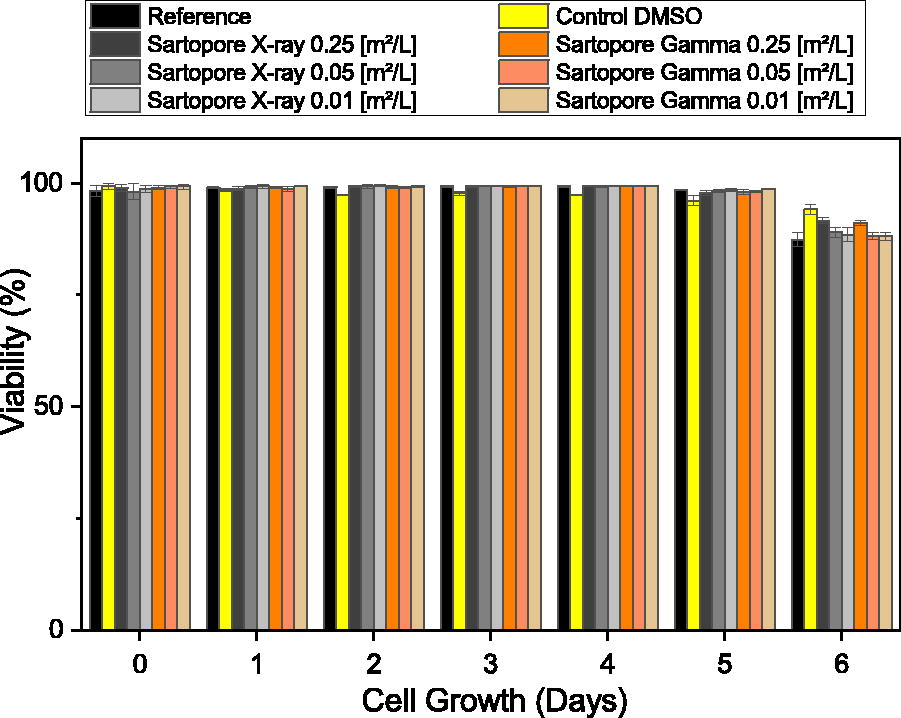
The cell viability (Fig. 2) was not affected by either irradiation technology and was above 86% for all tested surface-area-to-volume ratios. Interestingly, the lowest viability of 87% (average from three replicates) was found for the untreated reference at day 6, considered the death phase at the end of a batch cultivation. However, on day 6, all test conditions showed a lower viability compared to the first day of the cultivation. Therefore, no impact from the two irradiation technologies on cell viability of the CHO DG44 cell line was observed.
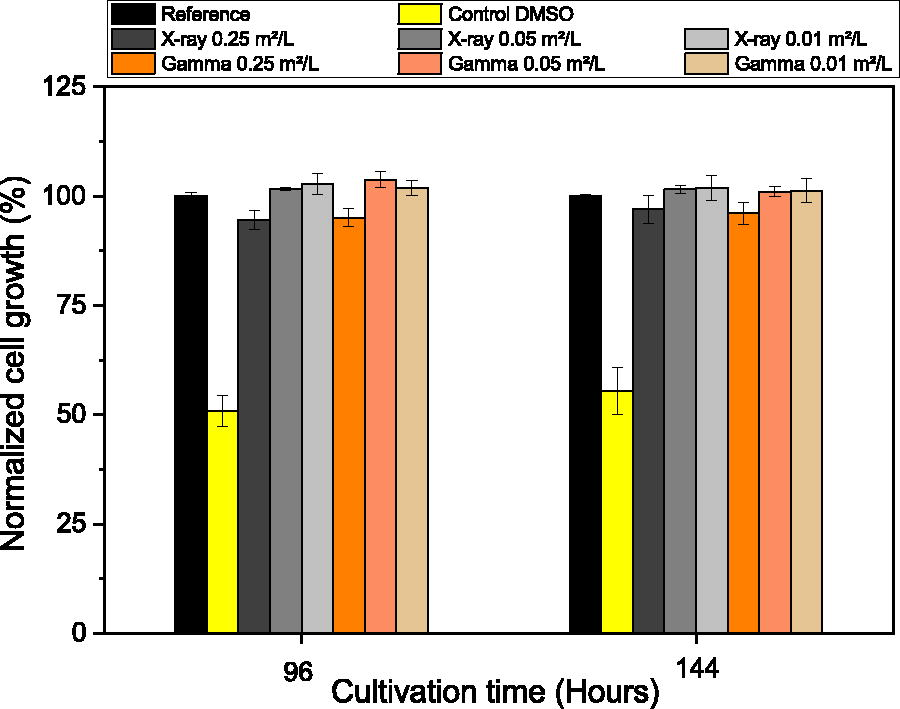
The cell growth at 96 and 144 h of cultivation was normalized to the cell growth of the reference condition. The results (Fig. 3) showed that the DMSO control resulted in around 50% normalized cell growth, establishing that the assay were as sensitive as expected to a known standard cytotoxic substance. 16 The SU assembly extracts resulted in a normalized cell growth above 90%, which demonstrated the absence of cell growth inhibition. When comparing gamma to X-ray-irradiated materials, the trends were highly similar and matched the no-irradiation reference within 6%. The 0.25 m2/L S/V ratios consistently had slightly lower normalized cell growth under both treatments compared to the 0.01 and 0.05 m2/L S/V ratios.
Thus, the filter material and irradiation technology revealed minimal effect on cell growth. An in-depth statistical analysis was applied to the whole data set to further quantify the degree of impact.
Statistical evaluation
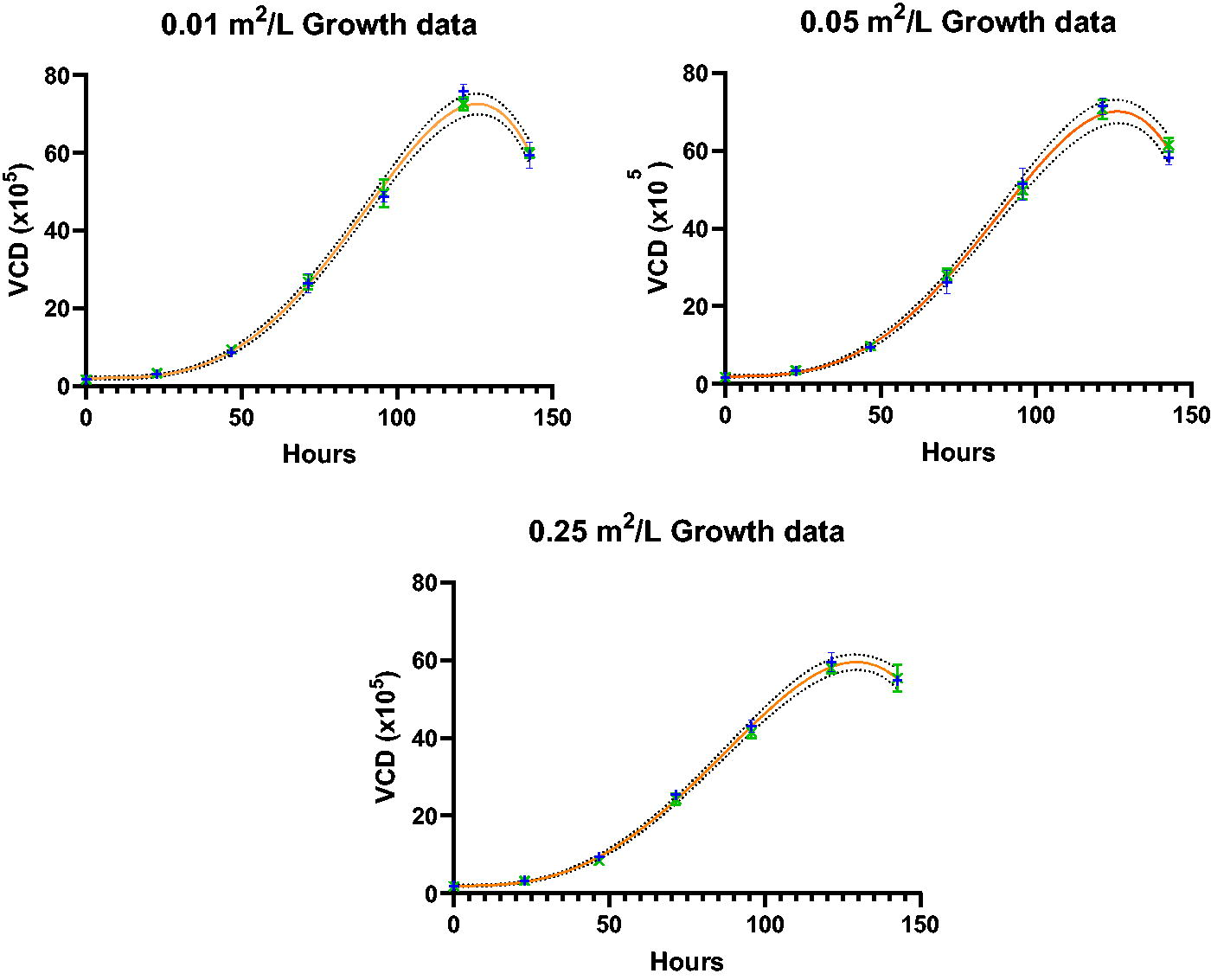
Statistical analysis was performed per the Data Analysis section. Graphs of the compared paired data sets were prepared with two-sided 99% confidence interval bands for the fit models calculated to bracket the likely location of the true growth profile curves, as shown in Figure 4. As expected, the resulting 99% confidence intervals increased with the Y-axis response magnitude and contained most of the replicate means. For each set of three replicates, the ±1 standard deviation intervals for the replicate means showed the replicate variation was well controlled. Therefore, the graphical and statistical analysis confirmed that for each paired data set, one model adequately described the profile. As shown in the Inset Table, for all three data sets with S/V ratios of 0.25, 0.1, and 0.05 m2/L, the gamma and X-ray data sets fit single models with >95% probability. Therefore, the X-ray irradiation method had no impact on the cell culture growth profiles for S/V ratios up to 0.25 m2/L when compared to using gamma irradiation.
Discussion
This work demonstrated a technical approach to empirically assessing differential impacts on cell culture growth from gamma- and X-ray irradiation. The approach used a CHO DG44 cell line, which had been used previously to assess SUS for cytotoxic residuals after Gamma-irradiation. Assessment conditions were designed to achieve high filter membrane surface-area-per-volume ratios that were conservative compared to typical biomanufacturing process applications. For this testing approach, it is noted that the incubated filters were not flushed prior to exposure to media to assess all extractables in the cell culture. The above conditions were highly worst-case to provide high confidence that the approach is predictive of success for real-world applications.
Based on ISO 10993-5 guidelines, a cell growth inhibition >30% is regarded to be cytotoxic. 17 The study methods were designed to support detection of a difference in comparative cell growth profiles of ±10% relative to the untreated reference control. This sensitivity was based on experience and considered natural variations caused by manual handling steps (i.e., pipetting) and cell counting. The range for cell growth inhibition caused by the 2% DMSO control was 40%—65% of the normalized cell growth of the reference.
The irradiation methods were assessed using triplicate assemblies under each challenge condition. Growth profiles were measured daily, enabling statistical analysis to determine the potential cytotoxic impact from gamma- and X-ray irradiation of the SU assemblies. Critically, no inhibition was observed on cell culture growth by gamma irradiation versus X-ray irradiation. When considering the averaged cell growth profiles, the differences between gamma and X-ray irradiation were smaller than 10%; therefore, the results are suitable for robustly concluding the absence of differential cytotoxicity.
At the highest surface-area-to-volume ratio of 0.25 m2/L, there was a minor effect on cell culture growth when compared to other loadings, but critically, the impact was independent of the choice of irradiation method. This minor effect at high surface-to-volume ratios is not expected to affect normal manufacturing use conditions, where filters are typically loaded to achieve ratios ≤0.05 m2/L (or ≥ 20 L/m2). Also, this minor effect demonstrates the sensitivity of the measurement approach used in the presented method relative to the untreated reference control.
The conservative, worst-case conditions tested in this work provide ≥10-fold safety margins for CHO DG44 cell lines relative to typical filter loadings in the 200 L/m2 range or 0.005 m2/L. It is possible that some cell lines may be more sensitive to differential irradiation impacts. For instance, NS0 cell lines in certain applications may be more sensitive to cytotoxic substances, such as bDtBPP, and the use of safety margins relative to CHO cell results could be a prudent approach. Therefore, the ≥10-fold safety margin for the CHO cell line minimizes the risk that cell-line-specific sensitivities could be realized when the filters and assemblies are used for real manufacturing applications.
In addition to the qualification of the primary filter elements for X-ray irradiation, this study also confirmed prior vendor work showing the tubing and bags are also compatible with cell culture use after X-ray irradiation.
Conclusion
Use of X-ray sterilization of SUS is an alternative to the industry standard gamma-irradiation. To supplement gamma-irradiation with X-ray irradiation capabilities, an empirical evaluation is valuable to demonstrate the integrity, safety, and performance of X-ray irradiation for SUS. This study focuses on the performance aspect, specifically any potential cytotoxic impacts from gamma- and X-ray-irradiated SUS filters for CHO cell culture.
Three filter surface area to volume ratio loadings of 0.25 m2/L, 0.05 m2/L, and 0.01 m2/L (4 L/m2, 20 L/m2, and 100 L/m2, respectively) showed no significant differential impacts from gamma versus X-ray irradiation on CHO DG44 cell culture performance. Specifically, all gamma and X-ray-irradiated SUS yielded cell growth at 96 h and 144 h within ±6% of the untreated reference control condition. Further, the complete cell growth profiles were statistically analyzed and compared using a Beta-plus-Offset model, which showed a single trend fit to each pair of gamma and X-ray irradiation conditions. These results demonstrate that CHO DG44 cell growth performance is not impacted by the use of media filtered through either gamma- or X-ray-irradiated Sartopore 2 XLM capsule filters under the worst-case conditions tested.
This work supports more generally the gamma and X-ray irradiation compatibility of the single-use materials of construction tested, including polyethersulfone (PES) as a material of construction for cell culture media filter membranes; polypropylene and polyethylene terephthalate as materials of construction for filter capsule construction; thermoplastic elastomer tubing; and polyethylene bag contact layer materials.
This study also serves as a practical approach to explore the differential impacts of gamma- and X-ray irradiation on cell growth in single-use systems.
Materials and Methods
Materials
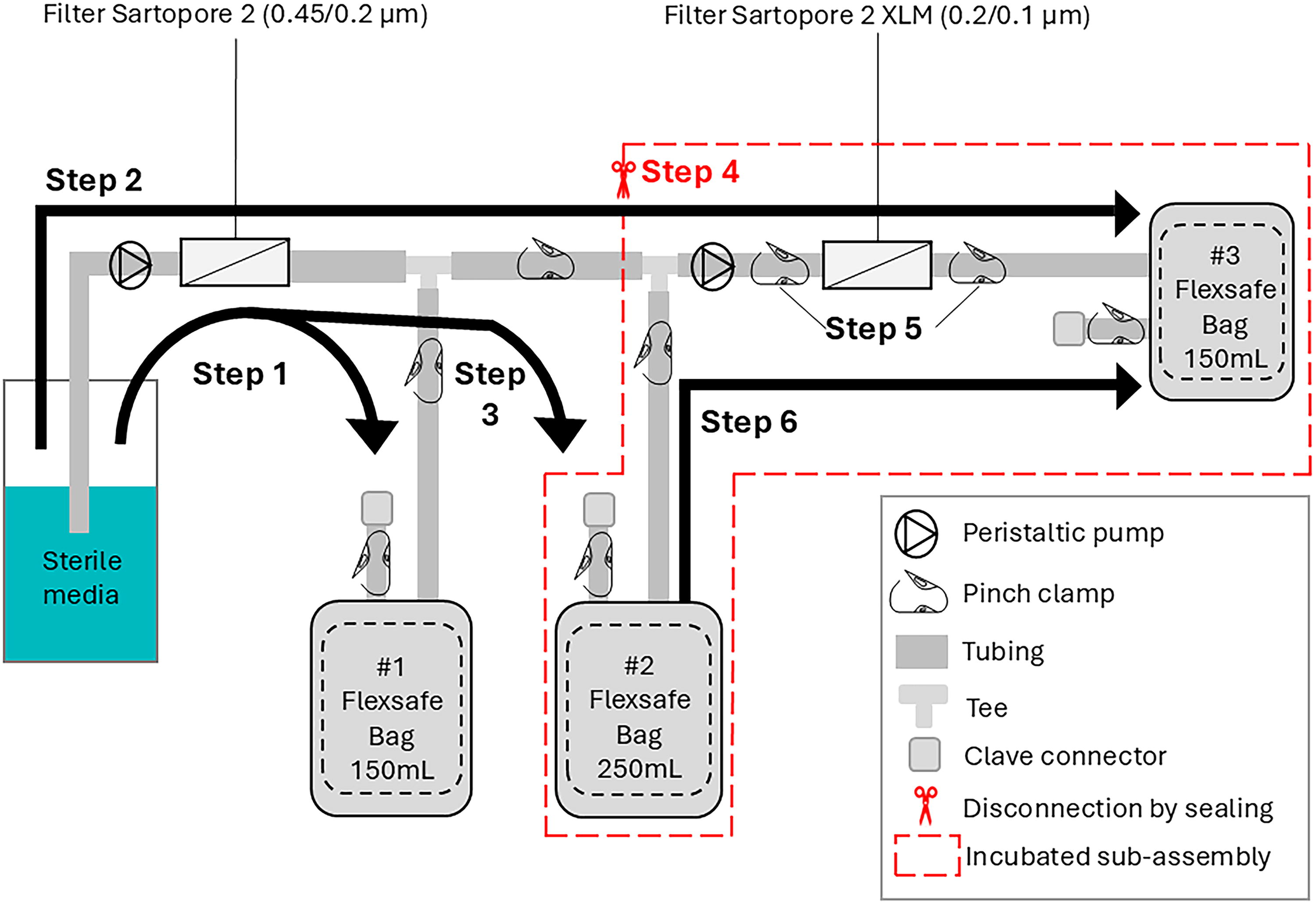
The schematic overview of the filter assembly is shown in Figure 5. The SUS assembly is composed of two PES sterilizing-grade filters (Sartopore® 2 0.45/0.2 µm 150 cm2 part number 5441307H4G-OO used to sterilize the media prior to incubation; Sartopore® 2 XLM 0.2/0.1 µm 210 cm2 5441358M4G-OO used to incubate the media before the cell culture study); in capsules made of polypropylene with polyethylene terephthalate support fleece, and three storage bags (Flexsafe® S80 polyethylene film used respectively as waste bags, intermediate storage for incubation and final container for the treated media); tubing (Tuflux® TPE thermopolymer elastomer used for connection and disconnection); fittings (polypropylene); and needle-free connectors (acrylic, polybutylene terephthalate, polypropylene, silicone). These components are typical of SU filter assemblies. The schematic overview of the filter assembly is shown in Figure 5. Six filter assemblies from the same manufacturing lot were used for the study.
Three SU filter assemblies were gamma-irradiated at a dose of 52.5 kGy and three SU filter assemblies were X-ray irradiated at a dose of 47.4 to 48.2 kGy. This difference is related to the ability to control the dose during the irradiation process and was not considered significant compared to the typical doses for SU assemblies of 25 to 45 kGy. The time between irradiation and extraction was 14 days for gamma irradiation and 17 days for X-ray irradiation; again, this difference was not considered significant.
The test SUS were stored prior to use at an average room temperature of 20 ± 5°C.
Stock and adaptation media (SAM) was sterilized by filtration through an autoclaved (not irradiated) Sartopore® 2 0.45/0.2 µm filter (5441307H4–OO–B), SAM media was stored at 2°C to 8°C for less than 2 weeks, which was well within the established expiry. SAM media was warmed to 37 ± 1°C just prior use.
The study included PC Erlenmeyer flasks (125 mL, Corning), Stock and Adaptation Medium (4Cell® SmartCHO SAM, Sartorius, Cat No. CQP3FB1147), Dimethyl sulfoxide (DMSO, Sigma Aldrich, Cat No. D2438-50ML), and CHO DG44 cells expressing human IgG1 (Sartorius Cellca, as described by Budde & Maier [2020]). 15
The instruments used in the study included a CO2 incubator for extraction and cell growth (Certomat® CT plus, Sartorius), a laminar flow hood (HeraSafe KS-18), a cell counter (Cedex HiRes, Roche Diagnostics), and a Pipetus® by Hirschmann.
Test specimen
The SU filter assemblies were filled with cell culture media according to the following procedure, illustrated in Figure 5. In Step 1, the Sartopore 2 sterile filter (0.45/0.2 µm) was pre-flushed with approximately 100 mL of media into the waste bag #1. This flush aligned with the recommended minimal flushing volume in order to minimize leachables from this filter, which was not part of the extraction study. 18 In Step 2, both waste bag #1 and intermediate bag #2 were clamped off, and 30 mL of media (approximate filter void volume) was pumped through both the Sartopore 2 and the Sartopore 2 XLM filters (0.2/0.1 µm) into the final bag #3, ensuring the Sartopore 2 XLM filter was filled. Minimal material was collected in bag #3. In Step 3, the Sartopore 2 XLM filter was clamped off and bag #2 was unclamped; then 200 mL of cultivation media was filtered through the Sartopore 2 filter into bag #2. In Step 4, a sub-assembly consisting of bag #2, Sartopore 2 XLM filter, and bag #3 was disconnected from the rest of the assembly by sealing and cutting, as indicated by the scissor icon. The tubing lines and bags included in step 4 had media contact surface areas of 270 and 730 cm2, respectively. In Step 5, while bag #2 and bag #3 remained attached, the sub-assembly was incubated at 36.8°C for 23 h while shaking at 120 rpm with a 50 mm orbital diameter. In Step 6, after incubation, 50 mL of media was pumped from bag #2 through the Sartopore 2 XLM filter to bag #3. This produced the starting exposed media that was further diluted as described below. This procedure produced 83 mL of filter-exposed media that was further diluted as described below.
As shown in Table 1, three dilutions at 4, 20, and 100 L/m2 were tested, corresponding to the filter surface area to media exposure volume (S/V) of 0.25, 0.05, and 0.01 m2/L, respectively. This range of filter loading covered the full range of worse-case uses for laboratory-to-commercial scale applications. Typical loading volumes in commercial manufacturing for filtration of chemically defined media range from 300 to 2000 L/m2. Loading volumes could be lower, in the range of 20 L/m2 for low volume applications such as media for seed trains. The lowest dilution of 0.25 m2/L (or 4 L/m2) was therefore chosen as a worst-case scenario of the smallest possible loading volume on small-scale filters. The other dilutions were chosen as 5x increments of that initial dilution.
Overview of sample extracts and further dilution of extracts.
Triplicate test conditions were used where “GI-1 to 3” correspond to gamma irradiation and “XI-1 to 3” to X-ray irradiation. Each condition had a volume of 34 mL, and further dilutions were made using media sterile filtered by autoclaved sterile filters to achieve the target volume per filter area. For instance, a 1:5 dilution involved adding 6.8 mL of extract to 27.2 mL of cell culture medium to achieve a target volume of 20 L/m2. Similarly, a 1:25 dilution involved adding 1.36 mL of extract to 32.64 mL of cell culture medium to achieve a target volume of 100 L/m2.
Untreated reference and inhibition controls (2% DMSO) were also included in the study. For the untreated reference, three sterile extraction glass vessels were filled with sterile cultivation medium only; no test sample was added. These vessels were then incubated next to the filter units in the same incubator so that the media in the extracts and the reference were treated similarly.
Cell growth
A cell line vial was thawed/centrifuged, and the recovered pellet was resuspended in 50 mL media for a first passage. About 10% of this first passage was inoculated in 150 mL of media for a second passage before inoculation into treated media. This ensured full recovery from the thawing process. The cells were cultivated for six days, considering the day of inoculation as time 0. The cultivation conditions were as follows: the temperature was maintained at 36.8°C, the shaking speed was set to 120 rpm with a 50 mm orbital diameter, the humidity was kept at 80%, and the CO2 concentration was 7.5%. The initial cell density was 0.2 × 106 cells/mL, and the working volume was 35 mL (see Table 2).
The Bigger picture: Implication for biopharmaceutical applications
The growing need to augment continuity of SUS manufacturing through the establishment of X-ray sterilization options is driven by constraints on gamma radiation sources. The stringency of the evaluation provided in this study demonstrates cell culture equivalency for a representative SUS within a class of materials and supports the acceptance of gamma and X-ray sterilization as interchangeable technologies. It is anticipated that the results can plausibly be used to assess risk of treatment for that class and for similar applications, thereby minimizing or avoiding routine cell culture for the risk assessment of X-ray-treated SUS for individual applications. When cell culture studies are deemed prudent, it is valuable to employ a proven metric for assessing impact. In this context, the current work provides an example of a well-controlled, practical cell culture evaluation method that contributes to the safe transition from gamma to X-ray sterilization of SUS. Ultimately, the work presented here contributes to enhancing supply continuity for SUS needed to deliver healthcare materials and reliably manufacture pharmaceuticals.
Cultivation conditions.
VCD was assessed daily by manual sampling of the cell cultures. Cell counting was performed using the Cedex HiRes automated cell counting device. The viability of each cell culture was also assessed using the trypan blue exclusion method. Cell cultures were sampled each day at the same time ±2 h. The normalized growth rate was calculated for each culture individually, as shown in the Results section. Afterwards, the mean of three replicates was assessed, and subsequently, the cell growth rate was normalized to the untreated reference that had not contacted irradiated SU assembly material.
Data analysis
Statistical analysis was performed to evaluate the impact of X-ray irradiation compared to gamma-irradiation of SU filter assemblies. Complete statistical analysis details are available in the Supplementary Data Package Prism file. Briefly, for each analysis, two data sets were paired based on the S/V ratio dilution conditions. Each data set had seven triplicate time points. Non-linear regression was used as it provided increased statistical power to detect potential growth profile differences. Calculations were performed using GraphPad Prism (V 10.1.2.324). The probabilities that the paired profiles were best fit using one versus two non-linear regression models were computed using a BGD model, which followed the Beta Growth then Decay model described by Yin et al., with an added offset to reflect the initial cell culture seed density:
19
Where:
X: Time in hours
Y: Viable Cell Density (VCD)
Offset : Initial VCD
Ym : Peak VCD
Te : Time of VCD peak
Tm : Time of VCD profile inflection point
The model was simultaneously fit to paired data sets using least-squares minimization weighted by (1/[YK]), with K = 1.00 or 1.35. Model adequacies were confirmed after successful tests for acceptable residual normality (alpha = 0.01), weighting (p > 0.01), skewedness (<0.5), 99% confidence interval computability, and adjusted R2 (weighted) (≥0.990). As shown in Table 3, sum-of squares F tests and Akaike’s Information Criterion tests were used to assess whether a single model fit to each paired data set from gamma and X-ray irradiation test assemblies.
Data Set Analysis Parameters used in Figure 4.
Footnotes
Authors’ Contributions
D.B.: Methodology (lead); conceptualization (contributing); writing—review and editing (equal). L.R.: Investigation (lead). C.H.: Methodology (contributing); writing—review and editing (contributing). C.R.: Methodology (contributing); visualization (lead); writing—review and editing (equal). D.N.: Conceptualization (lead); formal analysis (lead); writing—review and editing (equal). B.C.: Project administration. K.H.: Formal analysis (contributing); writing—review and editing (equal). S.C.: Formal analysis (contributing); writing—review and editing (contributing). M.K.: Investigation (contributing); formal analysis (contributing), writing—review and editing (equal). V.P.: Writing—review and editing (contributing). S.D.: Conceptualization (lead); writing—original draft (lead), visualization (contributing); writing—review and editing (equal).
Author Disclosure Statement
The authors of this article have affiliation with organizations with a commercial interest in use of the subject matter discussed in this article. D.B., L.R., C.H., C.R., and S.D. are affiliated with Sartorius, while D.N., B.C., K.H., S.C., M.K., and V.P. are affiliated with AstraZeneca. The authors have no financial conflicts of interest to disclose.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
