Abstract
Fermentation-derived foods (FDFs) such as cultured animal cells, edible microbial biomass, and recombinant food proteins have recently seen a notable surge in both investment and public interest. FDFs have been promoted as a more ethical, resilient, and environmentally sustainable food alternative to animal-derived foods such as meat, eggs, and dairy. However, some of the manufacturing process choices and sociopolitical assumptions made by actors in this space could risk undermining the goals of the technology as stated above. This article highlights five aspects of FDFs that should be carefully considered if they are to assist human society successfully transition to a more ethical, sustainable, and resilient food production system: (1) sustainable nitrogen fixation and management, (2) transitioning away from sugar feedstocks, (3) realistic expectations of consumer adoption rate, (4) careful consideration of the role of intellectual property, and (5) greater emphasis on food products that do not require cold or frozen storage.
The idea of culturing cells and tissues rather than whole organisms for food has attracted significant attention and capital investment in just the past few years. 1 For readers less familiar with this area of food technology, this category of food products broadly falls into three types: cultivated animal cells (muscle and fat), 2 edible microbial biomass (bulk cellular protein and/or lipid), 3 and recombinant animal food proteins (e.g., casein, β-lactoglobulin, and ovalbumin). 4
Cultivated animal cells (Fig. 1A) are primarily employed to create meat imitations, which typically involves taking a biopsy from a live animal and coaxing the constituent muscle or fat cells to proliferate into tissues reminiscent of “real” muscle and fat with the help of physiological growth factors.2,5 Some companies have circumvented the need for growth factors by generating so-called immortalized cells that no longer require conventional growth factors for proliferation. 6
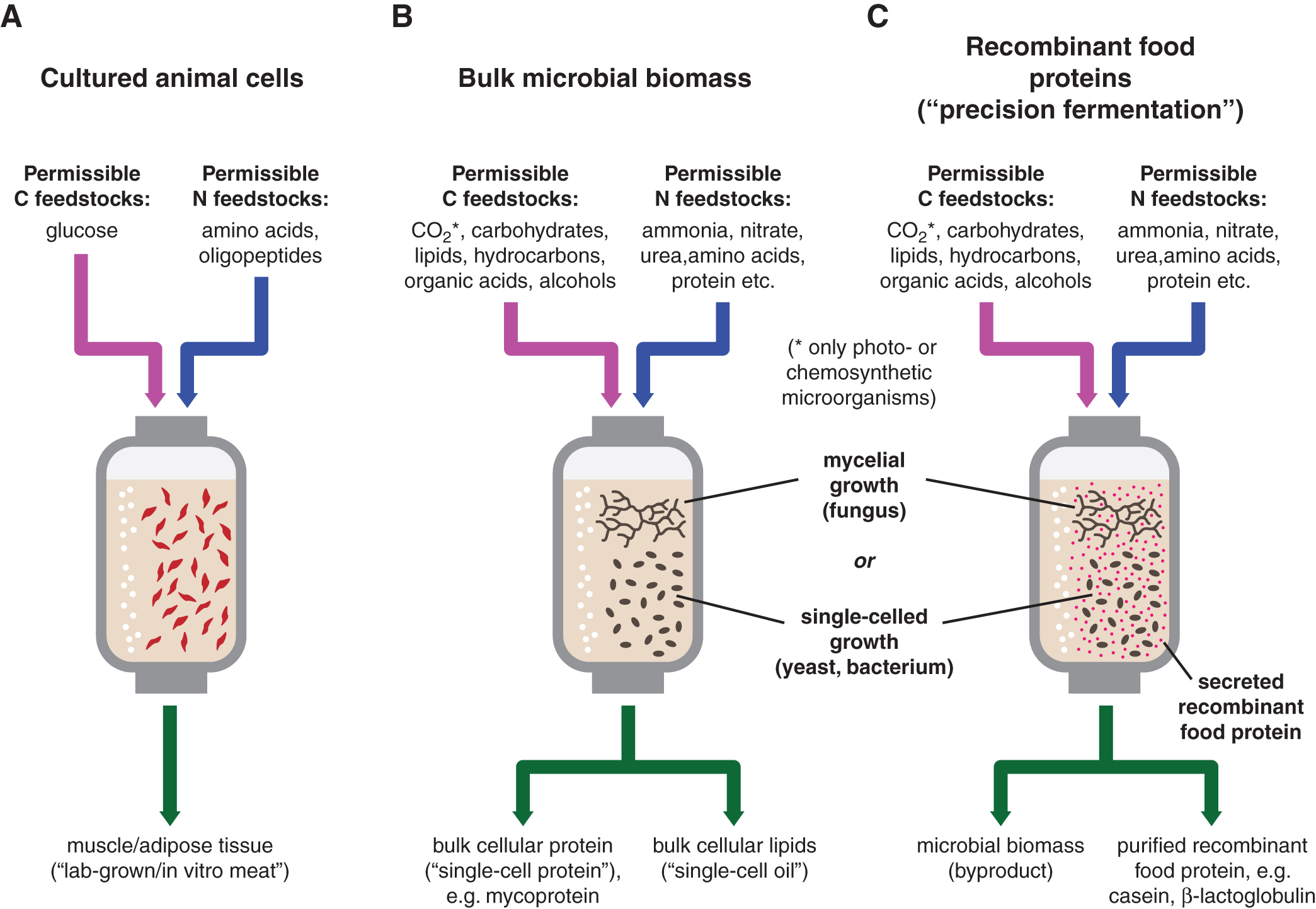
Production of edible microbial biomass (Fig. 1B) involves large-scale cultivation of certain microorganisms such as yeasts, fungal mycelium, microscopic algae, and certain nonpathogenic bacteria in nutrient-rich broth. 3 The microbial cells are then harvested, cracked open, and the desired cell fraction (protein or lipids) is extracted and processed into food products. This category of foods includes products that are already commercially available such as Marmite® and mycoprotein.7,8
The production of recombinant food proteins (Fig. 1C) involves inserting the gene for a particular food protein (e.g., milk casein or β-lactoglobulin from cow or egg ovalbumin from chicken) into the genome of a microorganism such as yeast or a mycelial fungus. These genetically modified microorganisms are then cultivated in nutrient-rich broth and secrete the desired food protein, which can then be extracted from the surrounding culture liquid.3,4 Recombinant protein technology using genetically engineered microorganisms has existed for >40 years but has until now been used to produce certain peptide hormones (e.g., insulin), vaccine antigens, industrial enzymes, and certain food additives (e.g., soy leghemoglobin in plant-based meat imitations and fish ice-structuring protein in low-fat ice cream).
As all three types of foodstuffs are all produced with the help of bioreactors known as fermenters, they will collectively be referred to fermentation-derived foods (FDFs) in this article. Some readers may be familiar with buzzwords such as “cellular agriculture” and “precision fermentation,” which are also commonly used to refer to these technologies. However, since both cellular agriculture and precision fermentation also encompass nonfood applications within biotechnology, these terms will not be used in this text to avoid unnecessary confusion.
FDFs should not be confused with fermented foods such as beer, wine, yoghurt, olives, kimchi, and tempeh (Table 1). In the case of fermented foods, a pre-existing food substrate such as milk or cabbage is inoculated with microorganisms that subsequently metabolize a portion of that substrate (typically soluble sugars) while releasing metabolic byproducts such as ethanol or lactic acid. During this type of fermentation process, the food substrate is modified with respect to taste, smell, texture, and/or visual appearance, but the bulk of the final product typically derives from the initial substrate. FDFs on the other hand completely metabolize their growth substrate to produce edible biomass either in the form of whole cells (animal or microbial) or secreted food molecules such as protein or fats.
Distinguishing fermented foods from fermentation-derived foods
In principle, FDFs are well positioned to address many of the problems of our current global food production system. The use of bioreactors for food production—although capital intensive—ensures greater control of nutrient inputs and potential metabolic waste products. This in turn can minimize nutrient leakages and associated downstream effects such as nitrous oxide (N2O) emissions and eutrophication.
The controlled environment inside the bioreactor means not only that food production can be situated in geographical locations unsuited to conventional agriculture (deserts, polar regions, and underground spaces) but is also expected to be more resilient to climate change. A cruel irony is that the high capital costs associated with the construction of industrial-scale bioreactors currently put them out of reach for many developing economies—exactly where they would be of greatest use.
A number of recently published anticipatory life cycle assessments claim that FDFs can be produced in ways that result in lower greenhouse gas (GHG) emissions than many forms of conventional food production.7,9,10 Therefore, rapid deployment of this technology would in theory ensure that food can still be produced in a predictable way despite climate change and perhaps play an important part in stalling and even reversing climate change.
The most obvious and immediate practical hurdle toward achieving this proposed goal is the enormous fermentation infrastructure that would have to be built to scale up FDF production capacity to a level comparable with the animal-derived foods they have been designed to replace. This in itself is a colossal challenge: 337 million tons of meat, 887 million tons of milk, and 87 million tons of hen eggs were produced in 2020. 11 For some process concepts—in particular cultured animal cells and recombinant food proteins—this will require lowering production costs to be competitive with conventional meat, eggs, and dairy.12,13
However, this article will assume not only that the challenges of cost-efficiency and scaling up can somehow be surmounted but also that the significant amounts of electricity that would be required to power this fermentation infrastructure could be provided using only zero emission sources of electricity generation. This article instead wishes to focus on specific aspects of FDF production that the author feels have been neglected in the larger discussion regarding the role of FDFs in the global food system of the future. Just because a technology has the potential to usher in a more ethical, sustainable, resilient, and equitable global food production system, care must be taken to avoid poor choices in how FDFs are produced—especially at an early stage of process development, which could risk undermining their promised benefits.
A parallel could be drawn with the production of corn (maize) ethanol as biofuel in the United States. The stated aim of fuel ethanol was in part to decrease the environmental impact of land transportation. Yet this practice has been plagued by controversy.14,15 A recent study found that the environmental impacts of current practices of growing corn for bioethanol were not much better than fossil fuels due in part to increased GHG emissions from fertilizer manufacture and applications as well as increased land-use change. 16 Policy makers and investors must, therefore, proceed with caution to avoid creating incentive structures that end up impeding rather than accelerating a transition to a more ethical, sustainable, resilient, and equitable food system.
In this context it should be noted that all FDF products and processes are not created equal. Some types of FDFs—for example, cultured animal cells—are more constrained in their process choices, as will be discussed in greater detail. In the remaining text, five key issues have been highlighted that should be taken into concern both when making investments and policies regarding this technology.
Sustainable Nitrogen Fixation Needs to Be a Greater Priority
Nitrogen is essential to all life and is a constituent of most biological molecules including all proteins and nucleic acids as well as many carbohydrates and lipids. The Earth's atmosphere consists of 78% nitrogen gas (N2) by volume and is the original source of all nitrogen found in living organisms. The chemical stability of the N2 molecule makes the extraction and conversion of nitrogen from the atmosphere into a form that can be widely assimilated by organisms a nontrivial obstacle.
Biological nitrogen fixation of N2 into ammonia (NH3) is carried out by bacteria and possibly some archaea. The oxygen sensitivity of the biological nitrogen fixation process restricts it either to oxygen-poor environments or to specialized cell types or tissues that can exclude molecular oxygen, such as is found within the root nodules of legumes, which are colonized by symbiotic nitrogen-fixing bacteria.
In the early 20th century, German chemists Fritz Haber and Karl Bosch succeeded in developing a commercially viable process for direct chemical synthesis of NH3 from nitrogen gas, which ended humanity's absolute dependence on biological nitrogen fixation. The Haber–Bosch process has been instrumental in increasing agricultural yields worldwide and it is commonly thought that 50% of current global food production is only made possible through industrial nitrogen fixation. 17 However, industrial NH3 synthesis is not without controversy. The process requires high temperatures and pressures, which today are overwhelmingly powered by combustion of fossil fuels.
In addition, fossil methane provides a source of hydrogen for NH3 synthesis. The other major environmental concern—which is common to all nitrogen fertilizers—is the leakage of nitrogen from the food system into the surrounding environment. This can lead to eutrophication of rivers, lakes, and oceanic waters. In addition, NH3 can be metabolized into the highly potent GHG N2O by microorganisms in the environment.
With respect to nitrogen inputs, FDFs offer both benefits and challenges. The use of bioreactors for cell cultivation allows for precise control of nutrient inputs, thus potentially minimizing nutrient leakage and subsequent downstream effects such as eutrophication and N2O emissions. However, cultivation of microbial or animal cells for food applications generally requires refined forms of nitrogen substrates such as NH3, nitrate, urea, amino acids or protein hydrolysates from soy. Crucially, animal manure cannot be used as a source of nitrogen for FDFs, which may limit options for sustainable manure management and nitrogen recycling in a future scenario where FDFs have achieved greater market share.
Another important factor to consider is that although plants and most microorganisms can use NH3, nitrate, and urea directly as sources of metabolic nitrogen, cultured animal cells are restricted to single amino acids or short peptides as nitrogen sources (Fig. 1A). Unlike cultured animal cells, whole animals can be supplied with low-grade protein from nitrogen-fixing legumes such as alfalfa and soybean. It is conceivable that some of the amino acids requirements of cultured animal cells can be supplied through hydrolyzed legume protein. However, amino acid proportions will need to be adjusted through supplementation of individual amino acids to maintain optimal growth.
These supplementary amino acids overwhelmingly derive from microbial fermentations, 18 which depend on inorganic nitrogen sources such as NH3. Taken together, this raises the possibility that FDF products designed to replace animal-derived food products will increase the dependence on industrial nitrogen fixation over biological nitrogen fixation. A transition toward fossil-free “green” NH3 synthesis must, therefore, become a more urgent priority. 19
FDFs Need to Wean Themselves Off Sugar Feedstocks
One key potential of microbially derived FDFs—edible microbial biomass and recombinant food proteins produced using transgenic microorganisms—is that they have the ability to convert an inedible or nutrient-poor substrate to a nutrient-dense foodstuff. 3 Such substrates can include agricultural and forestry carbohydrate side streams but also hydrocarbons, alcohols, and organic acids (Fig. 1B, C).3,20 This is highly significant since many such feedstocks (methanol, ethanol, and acetic acid) can be synthesized directly from carbon dioxide. 3
The implications of using carbon feedstocks that do not originate from photosynthesis for the cultivation of edible microbial food products (“carbon capture, conversion, and cultivation”) are profound. Decoupling food production from factors such as light availability, soil quality, and favorable climate conditions for crop cultivation means that food (in the form of microbial FDFs) can be produced essentially anywhere on the planet. 3 The high productivity of bioreactors 21 also means that the geographical area that must be dedicated to food production can be orders of magnitude smaller than conventional crops.22,23
It is perhaps surprising then that the majority of mycoprotein and recombinant food protein producers have settled on sugar as carbon feedstock (Table 2), presumably due to its good product yields and comparatively low price at present.
12
Unlike microorganisms, cultured animal cells have an absolute requirement for the monosaccharide
Selected companies within the fermentation-derived food space
“Fungus” here denotes fungal mycelium (e.g., Fusarium and Trichoderma species) rather than yeast.
Information derived from either company website or from reference. 63 Question mark (“?”) indicates that the feedstock was inferred from basic biological principles but the company declined to respond to requests for confirmation.
In case FDFs gain significant consumer acceptance and market share, a growing dependance on sugar feedstocks may become a concern. First, the cultivation of sugar-yielding crops such as maize, sugarcane, and sugar beet does still result in a measurable environmental footprint.16,26 Second, there is the question of how much future competition there will be from other sugar-dependent fermentation products such as fuel ethanol and organic compounds derived from microbial cultivation including enzymes, amino acids, and bioactive secondary metabolites.
Added on top of that is the possibility that global per capita consumer demand for FDFs could actually surpass current per capita levels of consumption for conventional meat, eggs, and dairy. Taken together, it is conceivable that future demand for sugar will be significantly higher than at present.
Third, future sugar feedstock availability is a point of vulnerability for sudden shocks to the global food system such as crop diseases, extreme weather events, and larger social conflicts. 27 In addition, a recent report from the Stockholm Environment Institute predicted that global maize and sugarcane yields may decrease by 27% and 58%, respectively. 28 It would, therefore, seem prudent from food security perspective to begin transitioning to nonsugar feedstocks as soon as possible for those producer organisms capable of assimilating nonagricultural organic compounds.
This category includes the filamentous fungi used in mycoprotein production as well as yeasts and filamentous fungi used for expression of recombinant food proteins.
3
This is not an option for cultivated animal cells since they are absolutely dependent on
To gauge any concern within the FDF space regarding future carbon feedstock supplies, the author reached out to a number of companies in the FDF sector, of which two responded. Perfect Day, which currently produces recombinant dairy proteins, responded that crop residues could represent a more sustainable source of feedstock should the need arise. OnegoBio, which produces recombinant ovalbumin, noted that their chosen production organism—the wood-degrading fungus Trichoderma reesei (which is also used by Perfect Day)—has the ability to utilize carbohydrates derived from indigestible plant matter such as straw and wood residues.
Although technically feasible, the author is currently unaware of any published data regarding process yields for recombinant food proteins produced using such feedstocks. However, there is some precedent for the use of inedible carbohydrate feedstocks for the production of mycoprotein. The Finnish company eniferBio has revived the so-called Pekilo-process, which employs wood-derived carbohydrates as feedstock for the cultivation of the fungus Paecilomyces varioti for use as both food and animal feed. 29 A recent pilot study of the fungus used in Quorn®-brand products (Fusarium venenatum) found that using enzymatically hydrolyzed rice straw as feedstock could be feasible provided that the enzymatic hydrolysis of the substrate was sufficiently thorough. 30
Do Not Assume That Consumer Acceptance and Adoption of FDFs Will Be Rapid or Comprehensive in the Near Term
Reducing the impact of animal-derived foods—primarily meat, eggs, and dairy—is by far the greatest challenge in trying to achieve a sustainable global food system. The dependence of animal-derived foods on plant-based inputs (feed crops and pasture) introduces inefficiencies into the global food system as well as detrimental side effects such as destruction of natural habitat, eutrophication, and emissions of the potent GHGs methane and N2O. 31 Conceptually, the simplest solution to this problem would be a societal shift toward plant-dominated diets. However, the global trend is currently the opposite with an uninterrupted increase of per capita consumption of meat.31,32
FDFs have been promoted as a solution to the unsustainable environmental footprint of conventional meat, eggs, and dairy. A prevailing narrative within the FDF space is that the replacement of animal-derived foods is more or less inevitable once the gap in global fermentation capacity has been filled. 33 In other words, the transition to FDFs is framed strictly as a supply-side challenge. However, this narrative completely dismisses the intricacies underlying the demand side of such a food system transformation and simply assumes a rapid change in consumer behavior that would be unparalleled in human history. The central argument is essentially that if food products can be developed at low enough costs that perfectly mimic conventional meat, eggs, and dairy, rapid consumer adoption will be more or less guaranteed.
However, it is important to consider that the challenges facing the global food production system today are in large part a sociological and political problem rather than a technological problem. The assertion that a transition to FDFs will be both rapid and comprehensive completely ignores the cultural aspects of food choice. Individual food choice is driven not simply by cost and sensory experience but also by other intangible factors. One good example of the cultural complexities of consuming animal foods is the trade in so-called bushmeat that caters for urban populations in Africa 34 and China. 35
In both instances, meat from wild animals in perceived as healthier than farmed animals,34,35 while largely disregarding detrimental effects to endangered species of animals as well as the risk of zoonotic infections. As such, the choice of wild meat is not motivated by low cost or sensory experience but because of what it is.
For many developing nations, livestock are not just an important source of protein and micronutrients but can also provide transport, perform heavy agricultural work, and boost societal status, especially for women. 31 In geographical locations where the soil or climate is not suited to growing crops, grazing livestock is often crucial for localized food production. Another complicating factor is that societies that are initially more reliant on plant protein eventually tend to transition to diets higher in animal protein once per capita wealth increases.31,32 However, lower income consumers in developing countries who are poised to make a dietary transition from plant-derived protein to animal-derived protein may actually present an opportunity for a technological intervention to break this trend.
Since this particular group lacks any long-standing personal tradition of consuming animal-derived food products, they may be the most promising target for adoption of FDF imitations of conventional meat, eggs, and dairy rather than consumers in developed countries with more established traditions of consuming animal-derived protein.
In the case of developed nations, it is perhaps informative to look specifically at the United Kingdom due its pioneering role in developing and commercializing microbial FDFs such as yeast extract products 8 (Marmite) and mycoprotein meat imitations. 7 Although Marmite is predominantly used as a sandwich spread, mycoprotein was specifically developed to serve as a major source of dietary protein. 7 In 1985—the year that mycoprotein was first commercialized—British per capita meat consumption was 71 kg per person per year. 36 By 2019, British per capita meat consumption had actually increased somewhat to 79 kg per person per year. 36
A recent survey of England, Wales, and Northern Ireland conducted by the British Food Standards Agency found that 53% of respondents had tried meat imitation products (including mycoprotein) and 32% respondents claimed that they consumed meat imitation products with some regularity. 37 (The same survey found that only 28% of respondents expressed willingness to try cultured meat while 59% of respondents expressed hesitancy or completely unwillingness to try cultured meat.) 37 From these data it would seem clear that moderate consumer adoption of meat imitation products was insufficient to decrease consumption levels of conventional meat.
How realistic is it then to expect that British consumer adoption of meat imitation products (mycoprotein, plant protein, or cultured meat) will increase further in the coming decades to a point where they actually start to significantly suppress consumption of conventional meat? (It should be noted that plant-based meat imitations are currently struggling to gain greater consumer adoption beyond vegans and vegetarians in the United States.) 38
More heavy-handed approaches such as simply trying to legislate away animal-derived foods in favor of FDFs are fraught with political risk. In the case of dairy, many countries have traditions of cheesemaking and other fermented dairy products (yoghurt, kefir, oggtt, shubat, airag, etc.) that go back centuries or even millennia.39,40 In the United States, which is one of the biggest per capita consumers of meat, 31 meat consumption is closely correlated with conservative political ideology41,42 and any perceived threats to food choice (real or imagined) are likely to be exploited for political ends. 43
Since rapid and comprehensive consumer acceptance and adoption of FDFs are clearly far from certain, FDFs should, therefore, be thought of as merely one possible tool for decreasing the environmental impacts of global food production and consumption. The urgency44,45 of stalling and reversing climate change will require a multipronged approach, which must include a “plan B” for animal-derived foods. If one assumes that animal-derived foods will not be replaced within the nearest decades, then more effort must be invested toward decreasing the current environmental footprint of farming animals for food. Such a strategy is not without controversy as it could be argued that any mitigation efforts that do not aim to eliminate animal-derived foods would in fact prolong their presence within the global food system.
For monogastric animals such as swine, poultry, and farmed fish, feed is the predominant factor contributing to their inherent environmental costs. Hence, if conventional animal feeds such as soy and fish meal can be replaced with more sustainable alternatives, this provides one avenue for mitigating environmental impact. Fermentation technology does provide a possible solution in the form of microbial feeds,3,46 for both protein and fats. Just as with edible microbial biomass for direct human consumption, the same product can be used as animal feed with the same benefits, that is, high nutrient utilization efficiency and no absolute requirement for arable land, thanks to the ability to use nonagricultural feedstocks.
Microbial feeds already have a proven track record in terms of product quality and scalability that dates back to the 1970s and 1980s.21,47 Although microbial feeds have not displaced conventional feeds as of yet, they do provide one potential policy opening to mitigate the environmental footprint of animal-derived food consumption that completely bypasses the consumer. This would probably require financial incentives to promote the use of microbial feeds over conventional alternatives.
There are additional technological interventions that can decrease the environmental impacts of animal-derived foods that do not involve fermentation technology. Three key sources of GHG emissions that originate from farming animals for food include methane emissions from enteric fermentation in ruminants, methane emissions from animal manure, and carbon dioxide emissions caused by land-use change as natural habitats are converted either into pasture or cropland for animal feed. 31
Various approaches to mitigate ruminant methane emissions are currently being investigated, which include breeding cattle with lower methane emission profiles, 48 supplementing cattle feed with chemical methanogenesis inhibitors 49 and catalytic conversion of methane in farm environments into carbon dioxide, 50 which has lower warming potential than methane.
Uncontrolled release of methane from manure can be avoided in part through anaerobic digestion where naturally occurring microorganisms degrade the organic content of the manure to produce biogas, which is a mixture of carbon dioxide and methane. 51 The captured methane can then be used as fuel, for electricity generation or as a feedstock—either for chemical synthesis or the cultivation of specialized methane-consuming bacteria that can then be processed into animal feed. 52
Promote Greater Use of Open-Source Technology for FDF Production
Intellectual property (IP) protection is an inescapable fact of food technology innovation. Novel foods that have never been produced at scale before—cultivated meat, chemosynthetic bacteria, and recombinant animal proteins—can in most cases only secure private investment if their technology platform is covered by patents. 53 Nevertheless, the ongoing patenting of cell lines, microbial strains, bioreactor design, downstream processing, and so on will fuel both perceived and real concerns of corporate control over global food production.54,55
At the moment, several FDF startups are partnering with global corporations in the food production and processing sector such Cargill, ADM, Nestlé, Unilever, and Mars.56–58 It is not inconceivable that many of the current startups in the FDF space will inevitably be acquired by some of those same corporations. 59 Such a scenario will have real implications for global food sovereignty. When compared with patented genetically modified (GM) crops that came to dominate the markets in the countries where they were permitted, it is important to stress that these crops did not eliminate their non-GM competition in those countries.
This was in part due to GM crops sharing the same mechanical infrastructure for processing and distribution as their non-GM counterparts. As such, GM crops were not a threat to the infrastructure for non-GM crops. This would probably not be the case for fermentation-derived replacements for conventional dairy, eggs, and meat.
Although the author is skeptical about the potential of cultured meat and most recombinant food proteins to displace their conventional counterparts in the near term, a feasible argument can be made for the displacement of conventional milk and egg protein ingredients in processed foods by recombinant alternatives. 33 Should such a scenario come to pass, a substantial part of the animal protein ingredients industry would likely come under the complete control of just a few corporate actors. Smaller animal-dependent producers would probably struggle to compete, especially if much of the old industrial infrastructure for processing and distribution of conventional eggs and dairy becomes eliminated as a result of shrinking market share.
Needless to say, IP protection in connection with food production is a sensitive topic that needs to be navigated carefully as it may feed a narrative of corporate control of the global food system, 5 which may or may not be a realistic concern. However, it can breed suspicion—especially in developing nations. 60 Investing in local ownership in FDF production could be one way that both local food sovereignty and trust in the technology can be strengthened. This should extend beyond simply licensing more recently developed technology platforms but also include investment in producing FDF using open-source technology.
At the moment, this is only possible for edible microbial biomass, which can offer a variety of microbial strains, bioreactor designs, and process descriptions from the 1970s and 1980s 47 whose patents have now expired. There is, therefore, no need to “reinvent the wheel” to enable immediate production of edible microbial biomass. In addition, there are plenty of past process concepts with hard data on successful upscaling.21,29,61 And should there be hesitancy among local consumers to adopt these types of novel food product, the processes could easily be reconfigured for animal feed production instead as outlined in the previous section.
Prioritize the Development of Nonperishable and Affordable FDFs for Consumers in Developing Nations
One aspect of FDFs that is often overlooked is that many FDF products currently under development for direct human consumption will require cold or frozen storage—especially products intended to mimic meat or dairy. Storage conditions for FDFs are especially important since many parts of the developing world do not yet have access to affordable refrigeration and/or reliable supplies of electricity to ensure stable cold chains. 62 Under such circumstances, consumers would be better served by FDFs formulated as nonperishable dry goods such as protein flours, powdered stock mixes for soups and stews, protein-fortified noodles, protein-fortified biscuits, and so on. Such products could also be fortified with important micronutrients such as vitamins and minerals to help alleviate malnutrition.
Cold or frozen storage will probably be unavoidable for cultured animal cells as their corresponding FDF product—overwhelmingly cultured meat—is a fully hydrated and enzymatically active “whole-cell” product that is expected to spoil at much the same rate as conventional meat. The same constraints do not necessarily exist for microbial FDFs such as bulk cellular protein or lipids as well as recombinant food proteins. These FDF ingredients can be formulated as dehydrated powders or oils and packaged under oxygen-free aseptic conditions.
In response to a query from the author, Perfect Day confirmed that several products made with their recombinant protein ingredients (including protein powders, bakery mix, egg replacer, and chocolate confection) do not require cold storage or transport—although some products, such as aseptic dairy imitation beverages, would require refrigeration after opening.
Greater emphasis on sustainable, nutritious, and above all affordable FDF products intended for the developing world would help dispel any potential narrative that FDFs are mainly aimed at wealthier consumers in the developed world. As part of such an initiative, the manufacture of such products should preferably also be localized to the developing world whenever possible so that local populations can benefit not only from access to FDFs but also well-paying jobs associated with their production.
In addition, FDF processes employing both renewable NH3 synthesis and nonagricultural feedstocks derived from captured CO2 rather than imported feedstocks could significantly strengthen both local food security and food sovereignty. However, such a scenario would require substantial outside investment not only to construct the necessary fermentation infrastructure but also to ensure a reliable supply of electricity.
Concluding Remarks
It is the author's hope that the issues highlighted above make it clear that FDFs are not a guaranteed solution to the challenges currently facing our global food production system. Careful consideration must be given to how each technology—cultured animal cells, microbial biomass, or recombinant food proteins—is implemented in terms of both process configuration and sociopolitical context. The temptation to rush a flawed process design should be tempered by the possibility that once such a process becomes integrated within supply chains and financial incentive structures, it may no longer be a trivial task to reconfigure it for a more sustainable, resilient, and equitable outcome.
Footnotes
Acknowledgments
The author thanks representatives for OnegoBio and Perfect Day for responding to technical queries regarding the issues discussed in this article.
Disclaimer
The opinions expressed in this article are solely those of the author.
Author Disclosure Statement
The author currently serves as an unpaid advisor to the company Arkeon. Arkeon was not involved in authoring or editing the article text in any way.
Funding Information
No funding was received for this article.
