Abstract
Background:
Large cutaneous defects of the face have traditionally been reconstructed with cervicofacial rotation flaps.
Objective:
To measure advancing distance and assess vascular reliability of superficial musculoaponeurotic system (SMAS) island flaps for reconstruction of large cutaneous facial defects.
Methods:
The study design was a retrospective case series of all patients who had undergone reconstruction of facial defects 3 cm or greater with fasciocutaneous SMAS island flaps from 2009 to 2023. Flap advancing distance and vascular compromise were analyzed.
Results:
The average patient age at the time of surgery was 69 (standard deviation [SD] 15) years with more males (n = 33, 92%) than females (n = 3, 8%). Of 36 patients, 18 had posterior defects, 16 had anterior defects, and 2 had defects straddling both regions. Fifteen cases had a full fasciocutaneous SMAS island flap, and 21 underwent the partial island modification. Flap ischemia occurred in three cases (8%). The mean flap advancing distance was 3.8 cm (SD 1.1). Of the 5 smokers, none had ischemia of the flap. The median follow-up time was 2 years (range 12–3,170 days).
Conclusions:
Fasciocutaneous SMAS island and partial island flaps are reliable options for the reconstruction of large cheek defects (3 cm or greater). Anterior pedicle blood supply is more reliable for defects anterior to the line from the lateral brow to the angle of the mandible, whereas the posterior pedicle is a better choice for defects posterior to that line.
Introduction
Reconstruction of large cheek defects with subcutaneous cervicofacial flaps was described as early as 1917 by Esser. 1 Juri and Juri in 1979 described the classical subcutaneous cervicofacial flap and reviewed its history. 2 Cook et al. in 1991 presented a series of these flaps with various modifications. They experienced 1 case of flap ischemia out of 14 cases, and at least one other case of previously irradiated flap failure. 3 Liu et al. in 2011 described a cervicofacial and cervicothoracic flap case series of 21 patients with a 9.5% full-thickness necrosis rate. 4
The subfascial modification of this flap was touched on by Crow and Crow in 1976 who used just a subplatysmal neck dissection paired with subcutaneous cheek dissection for large cheek defect reconstruction. 5 The first description of the deep plane (sub-superficial musculoaponeurotic system [SMAS]) cervicofacial flap for large cheek defects was published by Kroll et al. in 1994. 6 The first case series of the deep plane cervicofacial flap was described by Tan and MacKinnon in 2006. Of the 18 patients in that case series, one marginal flap necrosis occurred in irradiated skin. 7
All of the cervicofacial flaps for large cheek defects were based on the rotation advancement principles. Our case series introduces the full island and partial island modifications of the deep plane cervicofacial flap. This sub-SMAS fasciocutaneous island flap serves as a versatile technique for large defect reconstruction.
The nomenclature of the island flap dates back to Esser, who described it a century ago. 8 He describes two general types of island flaps: those that are adjacent to the defect and those with intervening skin between the flap and the defect. We evaluated the island flap with its leading edge sharing the defect perimeter.
Barron and Emmett published an excellent review of the subcutaneous pedicle flaps (island flaps) 60 years ago. 9 Island flaps have also been described as myocutaneous flaps,10,11 myocutaneous sliding flaps, 12 axial flaps, 13 myocutaneous island pedicle flaps, 14 and V-Y island flaps.15–17 These should not be confused with the V-Y advancement flaps without deep subfascial undermining and fascial release of the pedicle.18,19 Partial island flap modification was introduced by us in 2024 and has a similar surface topography as the hatchet rotation flap described 50 years before.20,21
Fasciocutaneous SMAS island flaps derive their perfusion from the blood supply of the cheek and SMAS.22–24 We set out to review the reliability of SMAS fasciocutaneous island flaps as compared with traditional cervicofacial rotation flaps in patients with large cheek skin cancer defects (3 cm or greater) as measured by flap viability and reach.
Methods
Study design
Institutional review board review and exemption were obtained from St. Joseph Health Center for Clinical Research. A retrospective chart review and photo analysis were performed of patients who underwent the fasciocutaneous SMAS island flap for facial defect reconstruction from 2009 to 2023 by the senior author. A photo database was used to search for patients tagged with “fasciocutaneous cheek flap,” “partial,” “temple,” “ear,” and “cheek.”
Of the initial 53 cases identified, 36 cases met our inclusion criteria. Patients selected for analysis had the reconstructed defect measure 3 cm or greater in one dimension. Cases were limited to the anatomical cheek area anterior to the ear, posterior to the nasolabial fold, superior to the mandibular line, and inferior to the temporal line. Defect locations were categorized as anterior, posterior, or straddling both regions. The dividing line between anterior and posterior was drawn from the lateral brow to the angle of the mandible. Hair-bearing temple and lower cutaneous eyelid defects were excluded. The flap advancing distance was defined from the flap leading edge to the most distal defect edge covered by the flap following the vector of advancement.
Surgical technique
Incisions of the SMAS island flap are extended from the medial and lateral aspects of the cutaneous defect inferiorly. The curves of the relaxed skin tension lines are followed for aesthetic scar placement. For medial cheek defects, a postero-inferior extension of incisions can also be used. Subcutaneous dissection is then performed either anteriorly or posteriorly depending on the choice of pedicle. If an anterior subcutaneous dissection is performed, an anterior or medial pedicle will be used. In that case, a full-thickness incision is made on the posterior edge of the flap to the sub-SMAS and subplatysmal plane (Fig. 1). Then, the sub-SMAS dissection is carried anteriorly under the island flap and the pedicle. In this deep plane, the parotid fascia and zygomaticofacial ligaments are released from the pedicle, providing mobility. Familiarity with this plane is critical for the avoidance of nerve injury. Additional mobility is achieved when the broad myofascial pedicle is cut inferiorly to release the flap and allow superior movement. Posterior (lateral) pedicle flap dissection is reversed from the above description of the anterior (medial) pedicle flap. Most of the flaps had blood supply based either on an anterior pedicle or on a posterior pedicle. A minority of cheek flaps had inferior-based pedicles where the fascial release was performed both medially and laterally.
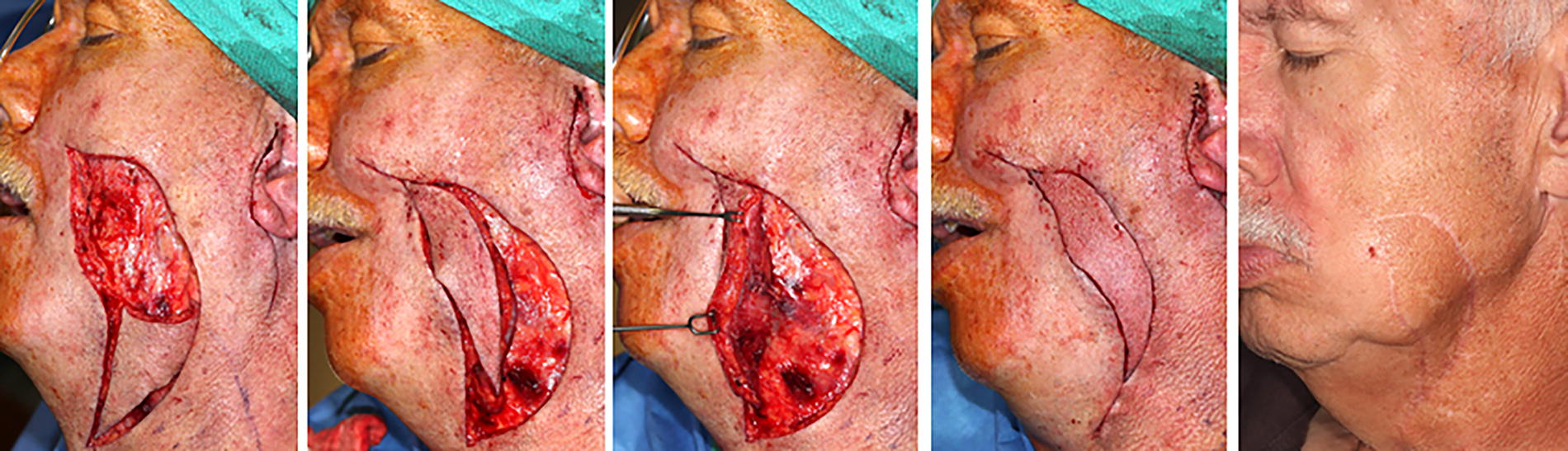
Anterior cheek defect reconstructed with a fasciocutaneous SMAS/platysma island flap based on anterior blood supply with a 5.5 cm advancing distance. The skin is elevated anteriorly off the medial pedicle. The flap is elevated in sub-SMAS/subplatysmal plane from the posterior incision medially under the pedicle. The platysma is backcut inferiorly to allow for superior movement of the myofasciocutaneous flap. The final result is 6 months post completion of adjuvant radiation therapy. SMAS, superficial musculoaponeurotic system.
Partial island modification leaves a cutaneous pedicle that is <50% of the length of the island (Figs. 2, 3, and 4). The dissection and release remain mostly the same. The difference is that the skin bridge is not entered in the subcutaneous plane.
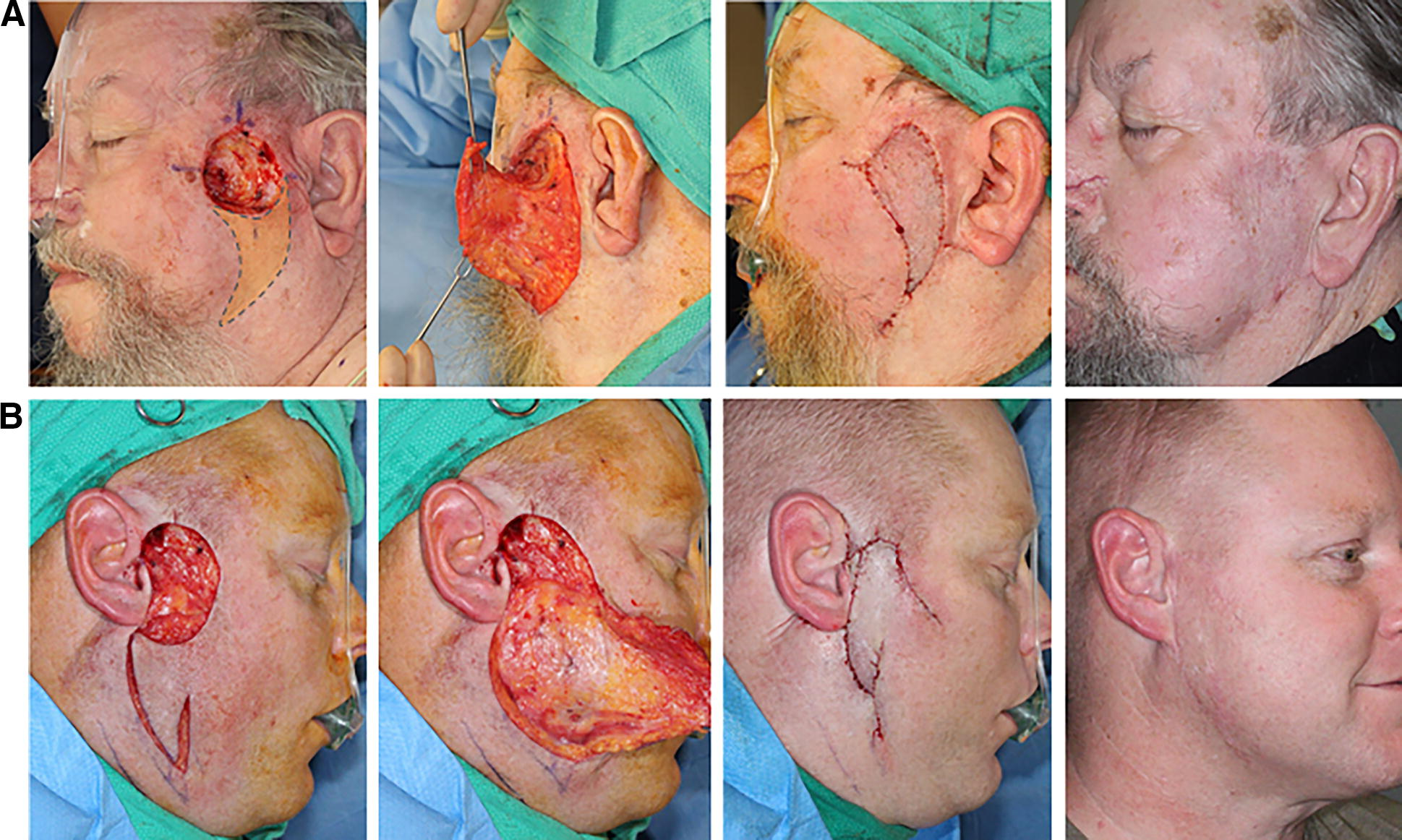
Posterior cheek defects reconstructed with anterior (medial) pedicle-based fasciocutaneous SMAS island flaps.
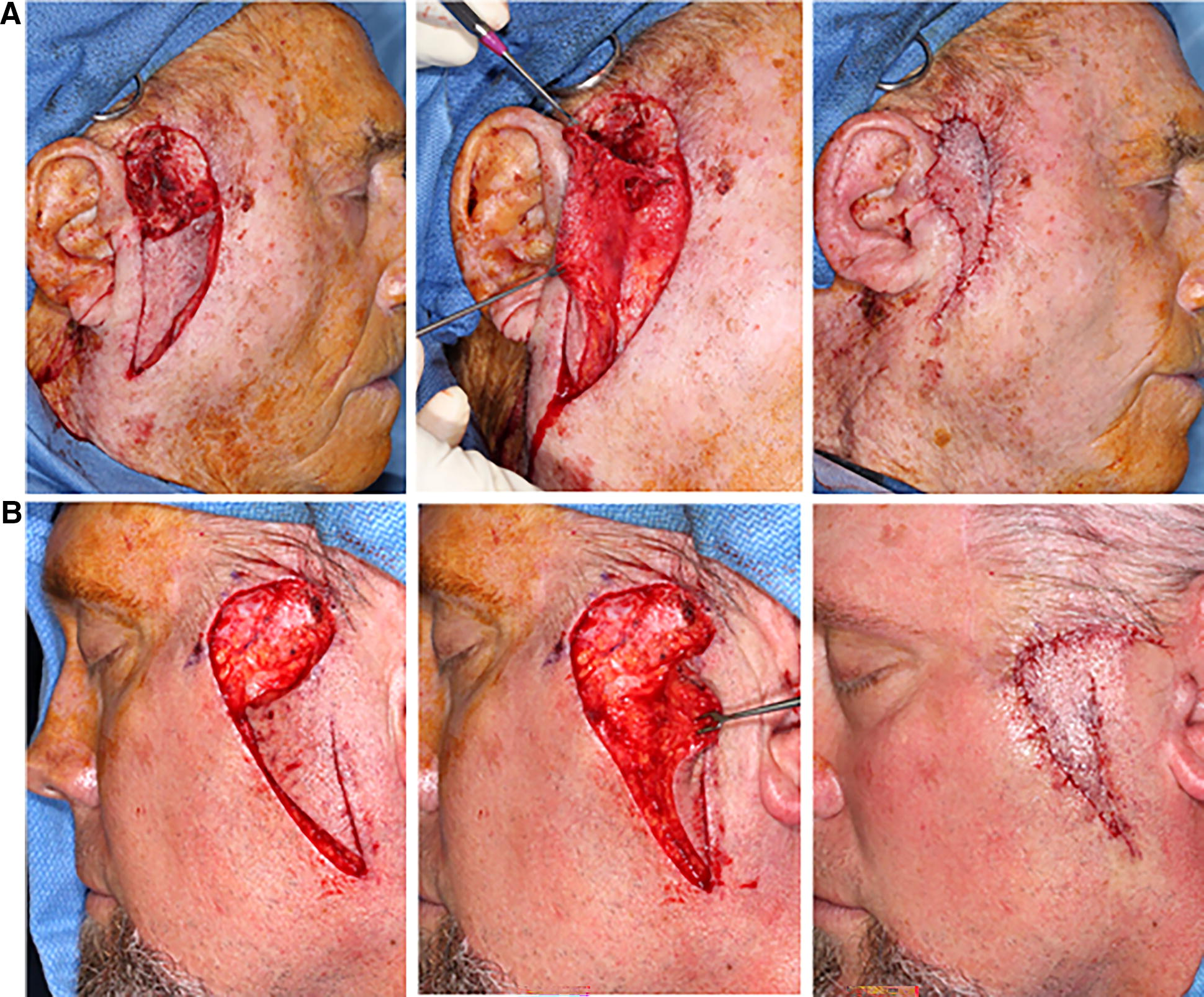
Posterior cheek defects reconstructed with posterior (lateral) pedicle-based fasciocutaneous SMAS island flaps.
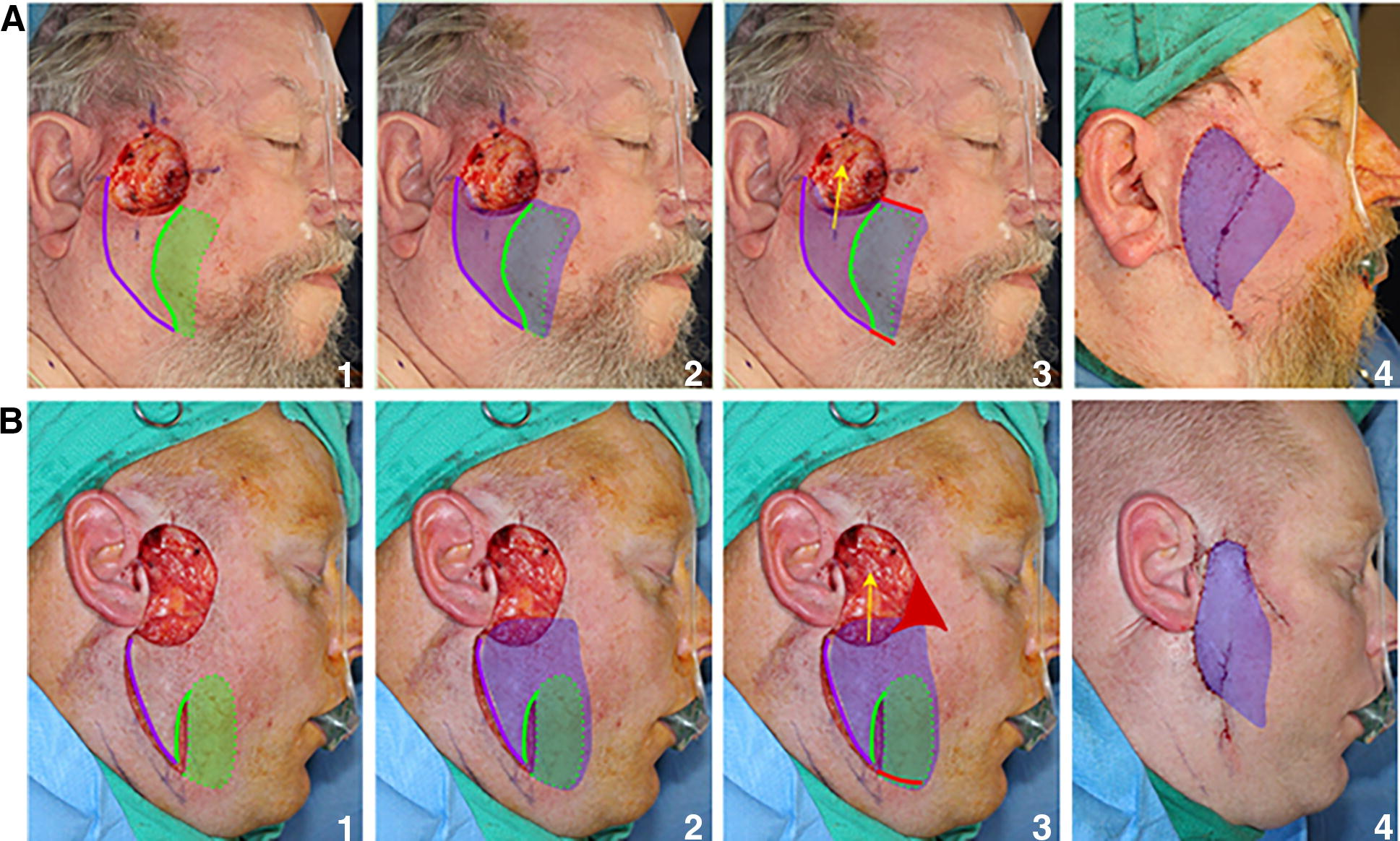
Surgical technique illustration of fasciocutaneous SMAS island flaps.
Results
Over the last 14 years, 36 patients’ defects met inclusion criteria and were reconstructed with full or partial fasciocutaneous SMAS island flaps. Patient demographics and characteristics are summarized in Table 1. Mostly male patients (n = 33/36, 92%) with an average age of 69 (standard deviation [SD] 15) years were studied. Five of the 36 patients (14%) were smokers. The indications for surgery were defects resulting from skin cancer excisions. The median follow-up time was 2 years (range 12–3,170 days).
Patient demographics for fasciocutaneous SMAS island flap
This table demonstrates patient demographic data and indications for surgery.
SMAS - superficial musculoaponeurotic system.
The mean defect size was 4.2 cm (SD 1.0 cm). For classification purposes, a single-number defect size was calculated by averaging the height and width of the defect. The distribution of reconstructions based on defect size included 4 cases (11%) of 2.9–3.0 cm, 16 cases (44%) of 3.1–4.0 cm, 9 cases (25%) of 4.1–5.0 cm, 5 cases (14%) of 5.1–6.0 cm, and 2 cases (6%) of 6.1–7.0 cm.
There were 18 cases (50%) of posterior defects located in the preauricular cheek (11 cases), cheek and temple (3 cases), and temple (4 cases). There were 16 anterior defects (44%) with the majority located in the central cheek. Two cases (6%) were large defects straddling both of these regions.
Out of the 36 cases, 15 defects (42%) were reconstructed with a full island flap, and 21 defects (58%) were reconstructed with a partial island flap. The flap advances superiorly in most cases and anterosuperiorly in cases of medial defects. The advancing distances are represented by the histogram in Figure 5 and range from 2.5 to 6.8 cm, with a mean advancing distance of 3.8 cm (SD 1.1 cm) and a median of 3.5 cm. The average advancing distance for full islands was 4.2 cm (range of 3.0–6.5 cm) and for partial islands was 3.5 cm (range 2.5–6.8 cm).
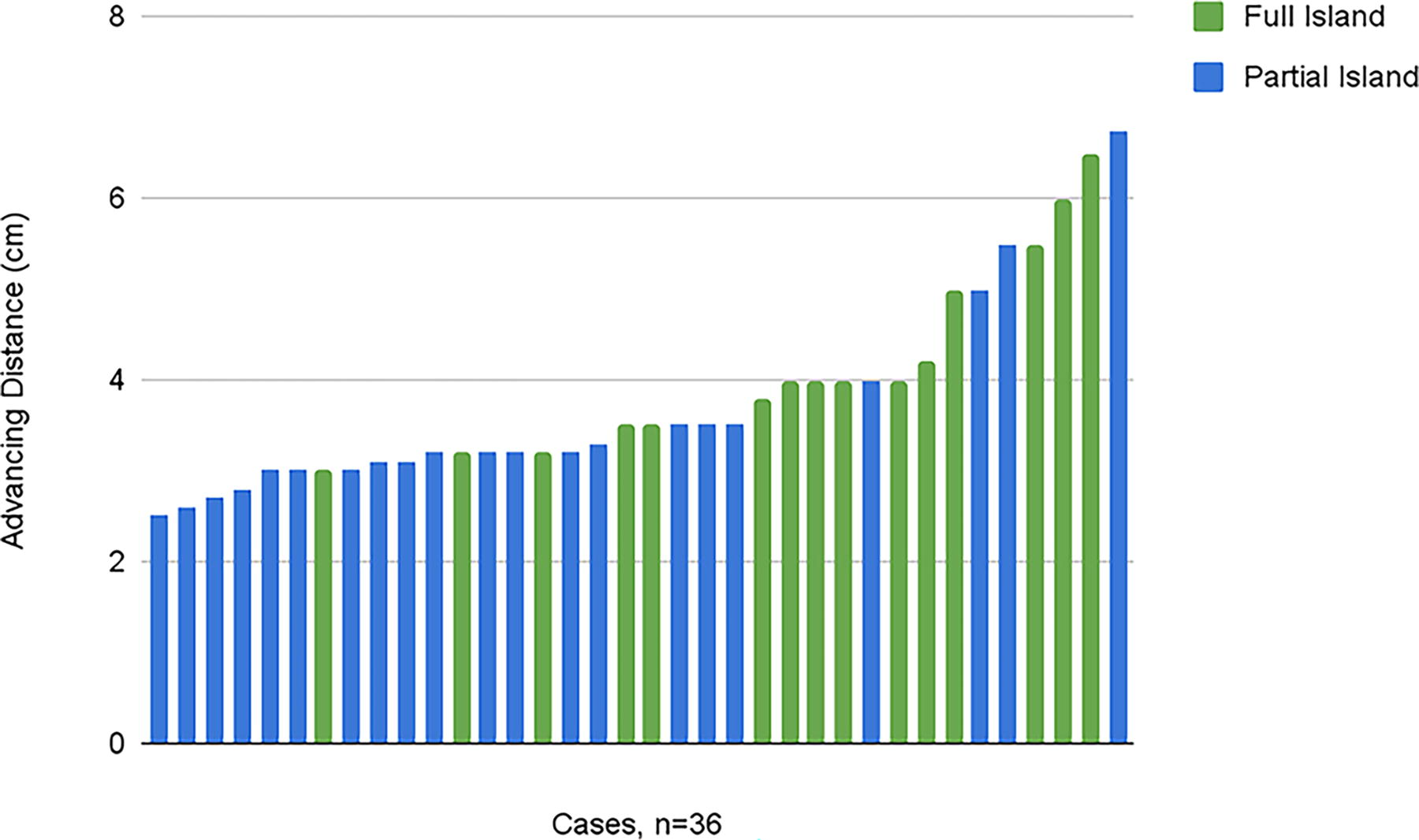
Histogram of advancing distances in 36 cases of the fasciocutaneous SMAS island flap.
The fascial pedicle blood supply was based either anteriorly, posteriorly, or inferiorly. Anterior defects were reconstructed with 7 (19%) anteriorly based flaps, 5 (14%) posteriorly based flaps, and 4 (11%) inferiorly based flaps. Of the defects located in the posterior region, 5 (14%) were reconstructed with anteriorly based flaps, 8 (22%) with posteriorly based flaps, and 5 (14%) with inferiorly based flaps. Both defects straddling both regions (6%) were reconstructed with flaps utilizing an anteriorly based blood supply.
Additional reconstructive techniques, including other island flaps and full-thickness skin grafts, were used for initial defect closure in 10 cases (28%). Distal tip ischemia occurred in three cases (8%) of posterior defects. All three defects were reconstructed with full fasciocutaneous SMAS island flaps. No ischemia was found in reconstruction of anterior defects. No distal tip ischemia was seen with partial fasciocutaneous SMAS island flaps. Of the five smokers, none had ischemia of the flap.
One small hematoma required incision and drainage. No Frey’s syndrome complication was observed. No facial paresis was caused by flap elevation alone. Two patients experienced temporary facial branch paresis due to oncologic resection with extended nerve dissection. We experienced no lower lid ectropion or retraction. We do not utilize this flap for lower eyelid defects due to the preference for a different flap technique.
Discussion
The principal finding of the study is that the fasciocutaneous SMAS island flap is able to advance an average of 3.8 cm, covering large cheek defects. The reliability of the flap was limited by partial ischemia in only 3 of 36 patients studied. The pattern of ischemia revealed limits of the blood supply. Partial island modification avoided ischemia in all cases.
Vascular supply was described by Esser in his 1917 paper. The cheek island flap has a deep pedicle supplied by the external maxillary artery (facial artery). 8 Barron and Emmett in 1965 posited that the viability of these flaps does not depend on the presence of the main feeding artery in the base of the pedicle. Instead, a “meshwork of intimate small vessels” is sufficient. 9
To help decide the flap pedicle choice, we view the vascular supply of the face as based on three vertical axes: facial artery anteriorly, transverse facial artery centrally, and external carotid artery posteriorly. Our fasciocutaneous SMAS island flaps were based on three types of fascial pedicles: anterior (n = 14), posterior (n = 13), and inferior (n = 9). Anterior pedicles derived their blood supply from the facial artery, while posterior and inferior pedicles from the external carotid artery. Few cases relied on the anterior pedicle supplied by the transverse facial artery perforators. This was due to the flap design requiring the release of the zygomaticofacial ligament with the perforator of the transverse facial artery contained within. This deep plane release is necessary for flap mobility.
Two cases of ischemia were due to the failure of perfusion to the posterior preauricular cheek from the facial artery anterior axis via the SMAS. Whetzel and Stevenson's description of SMAS (1977) as an avascular layer through which vessels pass to the subdermis in a way predicts that ischemia. 22 Schaverien et al. (2009) contradicts Whetzel and Stevenson by describing SMAS as containing a continuous anastomotic arterial network while describing a superficial venous system within the subdermal plexus. They also describe the importance of the transverse facial artery in the perfusion of the lateral face. 23 The central axis of the transverse facial artery within a medial pedicle is a reliable source for posterior defects as demonstrated by our case in Figure 2A. The medial pedicle supplied by the anterior axis of the facial artery is an unreliable source for posterior defects.
One of the ischemic cases involved a posterior cheek defect reconstructed with an inferiorly based island pedicle. Although deep perforators of the external carotid artery supply the dermis, the superior-inferior axis does not support an axial blood supply and is thus unreliable.
The solution to these vascular limitations is to use the posterior axis (external carotid artery) with a lateral pedicle for posterior defects as defined by the line from the lateral brow to the angle of the mandible. For anterior defects, the medial pedicle supplied by the facial artery is the obvious choice. Ultimately, the partial island modification, where no cases of ischemia occurred, is an excellent choice for vascular predictability.
Both anteriorly and posteriorly based fascia flaps could be modified as a partial island reconstruction (Figs. 2B and 3B). In some cases, the partial island modification affords better scar placement than a full island. The downside of the partial island flap is the greater tension of closure. Our advancing distance data indicates that - the full island average advancing distance was 4.2 cm, whereas the partial island advancing distance was 3.5 cm.
The precursor of the partial island flap is a hatchet flap that was first described in 1977 by Emmett. The only aspect that they have in common is the surface incisions. The hatchet flap deep blood supply is a “central sit-on” pedicle. 21 Although there is a deep vertical release below the skin incisions on each side of the “central sit-on” pedicle, there is no deep fascial release under the flap pedicle. In essence, this is a modification of the V-Y advancement. Thus, a hatchet flap is a “partial” V-Y advancement. The partial island flap differs from the hatchet flap by the deep subfascial undermining and release similar to the deep release of the island flap (Fig. 4). We had previously described a similar island flap based on TPF (temporoparietal fascia) vascular pedicle. 20
We experienced no significant hematomas likely due to the subfascial dissection plane of the fasciocutaneous flaps. Whereas anticoagulants should be avoided with subcutaneous cervicofacial flaps, continued anticoagulant use is not a contraindication for fasciocutaneous SMAS island flaps. Lack of Frey’s syndrome demonstrated the protective effect of the fasciocutaneous flap on the parotid defect as compared with a subcutaneous flap. 24
Although this study defines the fasciocutaneous SMAS island flap, the limited number of subjects with each defect location (anterior or posterior) paired with their flap subtype (full island or partial island) leaves room for further research. Another limitation of the study is the inclusion of one case with limited postoperative assessment, which impairs assessment of delayed flap ischemia of one partial island flap with posteriorly based pedicle. Fasciocutaneous SMAS island flaps are versatile and reliable for the reconstruction of large facial defects. The limitation of SMAS fascia vascularity should dictate flap design and occasionally require a partial island modification of the flap.
Footnotes
Disclaimer
All authors had full access to the design and conduct of the study; collection, management, analysis, and interpretation of data; preparation, review, or approval of the article; and decision to submit the article for publication. All authors had full access to all the data in the study and took responsibility for the integrity of the data and the accuracy of the data analysis.
Disclosure Statement
No competing financial interests exist.
Funding Information
Grant funding was provided by the Skin Cancer and Reconstructive Surgery Foundation.
