Abstract
Campylobacter is a globally important pathogen with well-studied risk factors, but the burden of risk factors has not been quantified. We quantified the cost of illness attributable to specific domestic risk factors for C. jejuni and C. coli in Australia. We used data from a 2018–2019 case–control study to estimate odds ratios and attributable fractions for risk factors. We used data on national incidence, hospitalization, and premature mortality to quantify burden. We then applied costs related to healthcare utilization, pain and suffering, premature mortality, and lost productivity to each risk factor. In Australia, C. jejuni caused 83.0% of campylobacteriosis infections and chicken consumption resulted in the highest attributable fraction (30.0%), costing approximately US$110 million annually. The excess burden of campylobacteriosis associated with the use of proton-pump inhibitors (PPIs) was US$45 million, with almost half these costs due to disease in adults over 65 years of age. Contact with young dogs (US$30 million) and chicken feces (US$10 million) also contributed to costs and burden. Campylobacteriosis is a significant cost to Australia, particularly because of lost productivity. Effective cross-sectoral interventions to improve chicken meat safety and reduce inappropriate use of PPIs might have substantial economic and human benefits.
Introduction
The World Health Organization (WHO) estimates that Campylobacter causes 166 million infections globally each year, making it a leading cause of bacterial gastroenteritis worldwide (Kirk et al., 2015). Among industrialized countries, Australia has one of the highest rate of campylobacteriosis at approximately 145 reported cases per 100,000 persons (Australian Government Department of Health and Aged Care, 2024). The incidence has increased in Australia over the last decade, leading government to develop a national foodborne illness reduction strategy focusing on Campylobacter and Salmonella (Australian Government Department of Health and Aged Care, 2024; Food Regulation Standing Committee, 2018). Despite this, national notification rates of Campylobacter have continued to increase unabated.
The main risk factors for campylobacteriosis identified from case–control studies and outbreaks are overseas travel, stomach acid suppression medications (e.g., antacids and proton-pump inhibitors), contact with young pets, untreated drinking water, and poultry consumption (Fravalo et al., 2021). Other countries such as neighboring New Zealand implemented successful interventions following identification that poultry consumption was the predominant risk factor (Sears et al., 2011).
We conducted CampySource—a comprehensive study into the epidemiology of Campylobacter infections in Australia that incorporated epidemiological, modeling, and genomic approaches to understanding sources of domestic infections (Cribb et al., 2022). In a cost of illness study, we estimated campylobacteriosis transmitted via contaminated food cost Australia US$253 million annually, with the majority of costs coming from lost productivity and pain and suffering (Glass et al., 2023). In this study, we synthesize data to characterize costs attributable to C. jejuni and C. coli due to specific risk factors.
Methods
Study population and pathogen identification
We used data from an Australian case–control study (2018–2019) to identify risk factors for campylobacteriosis. The methods are published elsewhere (Cribb et al., 2022), but in brief, we recruited 571 case–patients who were culture-positive for Campylobacter spp. in the Australian Capital Territory (ACT), Hunter New England region of New South Wales (NSW), and Queensland (Qld). We frequency-matched cases to 586 controls by sex, age group, and location. We excluded cases and controls if they traveled outside of Australia during, or interstate for the entirety of, the 2 weeks before illness or if a household member was positive for Campylobacter in the 4 weeks before illness or interview. We interviewed cases and controls using telephone-administered questionnaires covering clinical illness, demographics, medication use, food history including dining venues and types of consumed foods and kitchen practices, and exposures to animals including pets. We sequenced Campylobacter isolates on an Illumina® sequencing platform (Illumina, San Diego, CA) and classified isolates to the species level by analyzing read sets with Kraken and the PlusPf database (Wood and Salzberg, 2014). We characterized 508 case isolates as C. jejuni (n = 422), C. coli (n = 84), or C. lari (n = 2). We were unable to revive 63 Campylobacter isolates in culture, so we could not identify the species.
Burden estimates
We estimated the burden of disease (cases, hospitalizations, and deaths) for initial diarrheal illness and three sequelae: reactive arthritis, irritable bowel syndrome (IBS), and Guillain–Barré syndrome (GBS) in line with our previous work (Glass et al., 2023). To estimate the total number of cases, we adjusted national notifications for campylobacteriosis in 2019 using multipliers for domestic acquisition and underreporting. We produced separate estimates for three age groups (<5 years old, 5–64 years old, and ≥65 years old) for all transmission pathways. We estimated the number of campylobacteriosis cases that would lead to sequelae, hospitalization, or death by multiplying the number of cases by age- and outcome-specific multipliers derived from previous work (Supplementary Tables S1, S4, S5, S8). This previous work estimated the number of deaths and hospitalizations from the Australian Bureau of Statistics Causes of Death 2001–10 and the Australian Institute of Health and Welfare principal diagnosis data cubes for 2018–19 and 2019–20 (Australian Bureau of Statistics, Causes of Death [data available on request]) (Australian Institute of Health and Welfare, 2024). We assumed that all cases of GBS were hospitalized. We estimated the number of case visits to primary health care (all outcomes) or an emergency department (initial illness only) based on data from the National Gastroenteritis Survey II (NGSII) (Supplementary Tables S1, S2, S3, S4). We assumed no ongoing illness from campylobacteriosis (i.e., all gastrointestinal illness resolved) but determined the proportion of sequelae cases experiencing ongoing illness from the literature (Supplementary Tables S2, S3, S4). A health outcome tree visualizes the possible pathways for campylobacteriosis healthcare use and related sequelae (Supplementary Fig. S1).
Estimating attributable costs of illness
Cost of illness estimates included premature mortality, direct healthcare costs (including healthcare visits, tests, and medications; Supplementary Tables S5, S6, S7, S8, S9, S10), lost productivity, and the cost of pain and suffering. We provide all model data and assumptions in the supplementary materials and our previous costing work (Glass et al., 2023). We applied the Office of Best Practice value for a statistical life of AU$4.9 million (US$3.4 million) (Australian Government Department of Prime Minister and Cabinet Office of Best Practice Regulation, 2019). We sourced direct costs for medications, costs, tests, and some procedures from the Pharmaceutical Benefits Schedule and the Medicare Benefits Scheme—two parts of the Australian national medical insurance scheme. We sourced public and private hospitalization costs from the Australian Refined Diagnosis-Related Groups published by the Australian Department of Health (Australian Government Department of Health, 2016). We assumed that test and medication costs were included in hospitalization costs for GBS.
We estimated lost productivity for cases and their carers using a human capital approach with data from the NGSII, length of hospital stays from separation statistics, workforce participation data, and additional literature (Supplementary Table S11). To cost pain and suffering, we incorporated willingness-to-pay (WTP) values from an Australian discrete choice experiment. We used daily WTP values for primary illness and yearly values for sequelae (Supplementary Tables S12, S13). We costed ongoing pain and suffering for a proportion of sequel cases (Supplementary Table S13). Our cost estimates associated with ongoing disease burden were conservative and calculated only to the first year of illness for sequelae due to insufficient case follow-up in supporting studies.
Pathogen fractions, adjusted odds ratios, and attributable fractions
We estimated the fractions of national campylobacteriosis due to C. jejuni and C. coli from the numbers of isolates in our study with known species. We estimated adjusted odds ratios (aORs) for risk factors using logistic regression in a multivariable generalized logistic regression model. The model controlled for age, sex, location, and season, with other risk factors included in the final model selected using backwards stepwise selection based on p value < 0.05, variance inflation factors, Cramér’s V, and expert judgement. The strength of association between risk factors may vary by age. We assessed model fit using Akaike information criterion with two-level (<65 years and ≥65 years) and three-level age group adjustments, with and without interaction terms. The three-level age group models performed best, showing little difference between models with and without interaction terms. Therefore, we calculated the aOR for risk factors for the entire population. For our final estimates, we used a Bayesian framework through the “rstanarm” package (version 2.21.4) with default uninformative priors to generate 100,000 draws from the posterior distribution for aOR estimates (Gabry et al., 2023). Our aORs differ slightly from previous work, which fitted the same model in a frequentist paradigm.
We used population attributable fractions (AF) to quantify the proportion of campylobacteriosis due to certain risk factors and apply this fraction to burden and cost measurements. We assumed causality in that removing an exposure from the population would reduce disease incidence (Bruzzi et al., 1985). To calculate the AF for each risk factor by age group, we multiplied the aOR by the proportion of all cases exposed to each factor in each of three age groups, estimated from the self-reported exposure in the cases in our study. Estimation for the proportion of cases exposed was in a Bayesian paradigm, using the binomial model and the uninformative Jeffrey’s prior (i.e., Beta[0.5,0.5]). While there is some evidence linking proton-pump inhibitor (PPI) use to higher odds of hospitalization from gastroenteritis (Chen et al., 2016) and higher odds of developing IBS following initial illness (Esan et al., 2020), we did not include these assumptions in our calculations. We assumed that probability of hospitalization, death, and postinfectious sequel illnesses depended on age of the case, but not the risk factors that led to the initial illness. To reflect uncertainty in the final burden and cost estimates, we chose to report a 90% credible interval (90% CrI) for estimates in a Bayesian framework or a 90% uncertainty interval (90% UI) if combining multiple sources of uncertainty.
Results
We estimated that there were 349,000 (90% UI 221,000–554,000) cases of campylobacteriosis in Australia, with 83.0% (90% UI 80.0–86.0%) of cases due to C. jejuni and 17.0% (90% UI 14.0–19.0%) due to C. coli. This equated to 290,000 (90% UI 183,000–461,000) C. jejuni and 57,900 (90% UI 35,800–92,900) C. coli cases after accounting for underreporting and domestic acquisition (Table 1). Among five primary campylobacteriosis risk factors, consumption of cooked chicken in the 7 days before illness had the highest AF across all age groups. This ranged from 30.0% (90% CrI 3.0%–49.0%) in adults aged over 65 years to 35.0% (90% CrI 3.0%–56.0%) in children aged under 5 years (Fig. 1). Compared with consumption of cooked chicken, consumption of undercooked chicken had a much larger aOR (102 vs. 1.67) but was much less frequently reported, so the overall attribution proportion was much smaller (Table 2; Fig. 1). For other risk factors, we observed a variation between age groups, with PPI use greatest in adults aged over 65 years and contact with young dogs or chicken feces greatest in children aged under 5 years (Fig. 1).
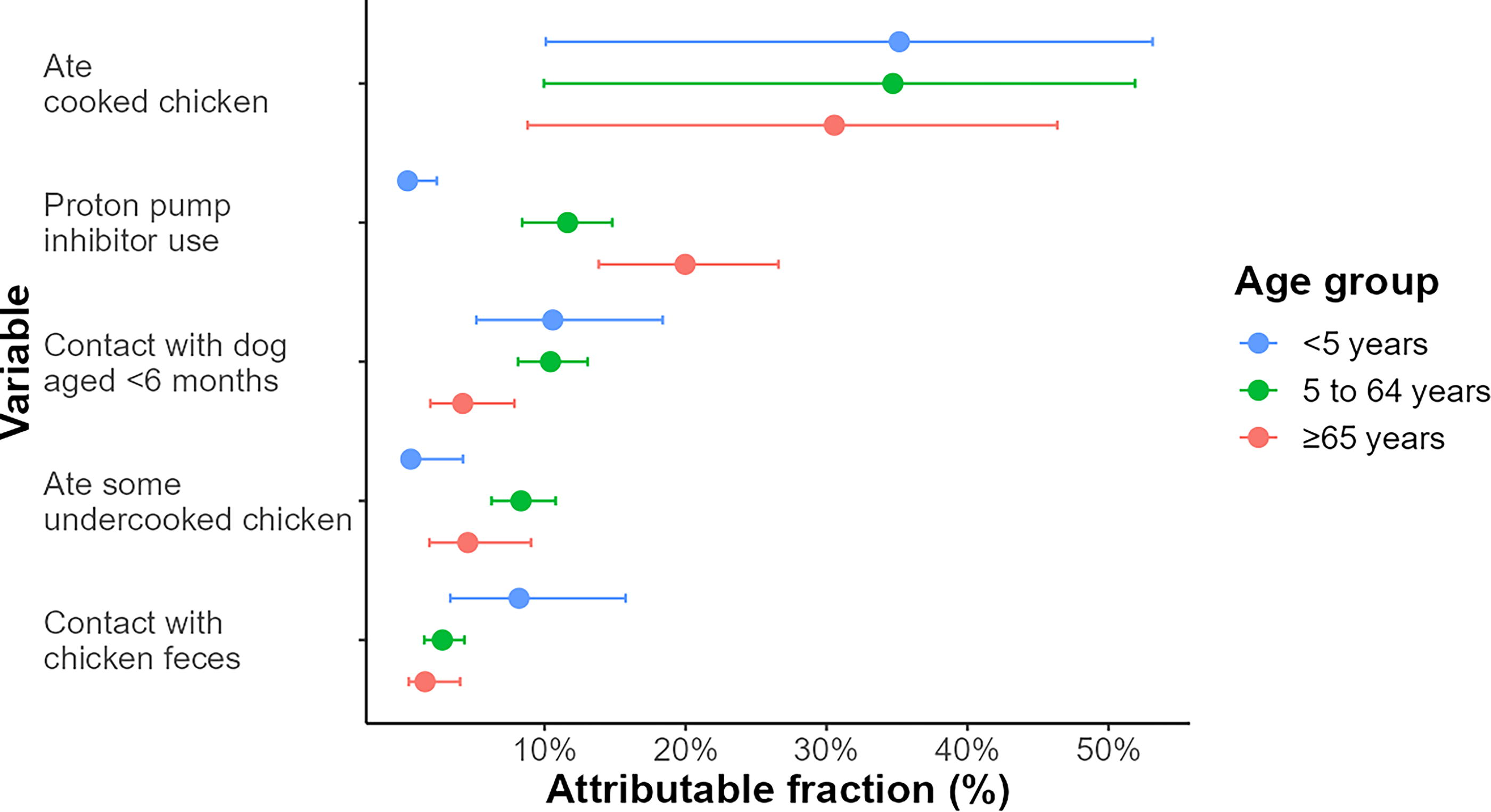
Population attributable fraction of five main campylobacteriosis risk factors by age group in Australia circa 2019. Point estimates for each age group are surrounded by error bars that represent the 90% credible interval.
Attribution of Campylobacteriosis Cases in Australia Circa 2019 by Campylobacter Species with 90% Credible Intervals or Uncertainty Intervals
Number of Exposed and Unexposed Cases and Controls by Age Group and Adjusted Odds Ratios with 90% Credible Intervals for Five Main Campylobacteriosis Risk Factors in Australia Circa 2019
We attributed 114,000 cases (90% UI 31,800–220,000) of campylobacteriosis to eating cooked chicken and 24,200 cases (90% UI 14,200–40,800) to knowingly eating chicken that appeared undercooked. We estimated that cooked chicken consumption resulted in 2,400 hospitalizations (90% UI 668–4,110) and one death (90% UI 0–3) and that undercooked chicken consumption resulted in 487 (90% UI 308–748) hospitalizations and less than one death (90% UI 0–1). Cases, hospitalizations, and deaths attributed to sequelae are in Supplementary Tables S14 and S15. When including all cost components for initial illness and sequelae, this amounted to an estimated US$111 million for transmission from cooked chicken and US$23.1 million from undercooked chicken infection (Fig. 2; Supplementary Tables S16 and S17).
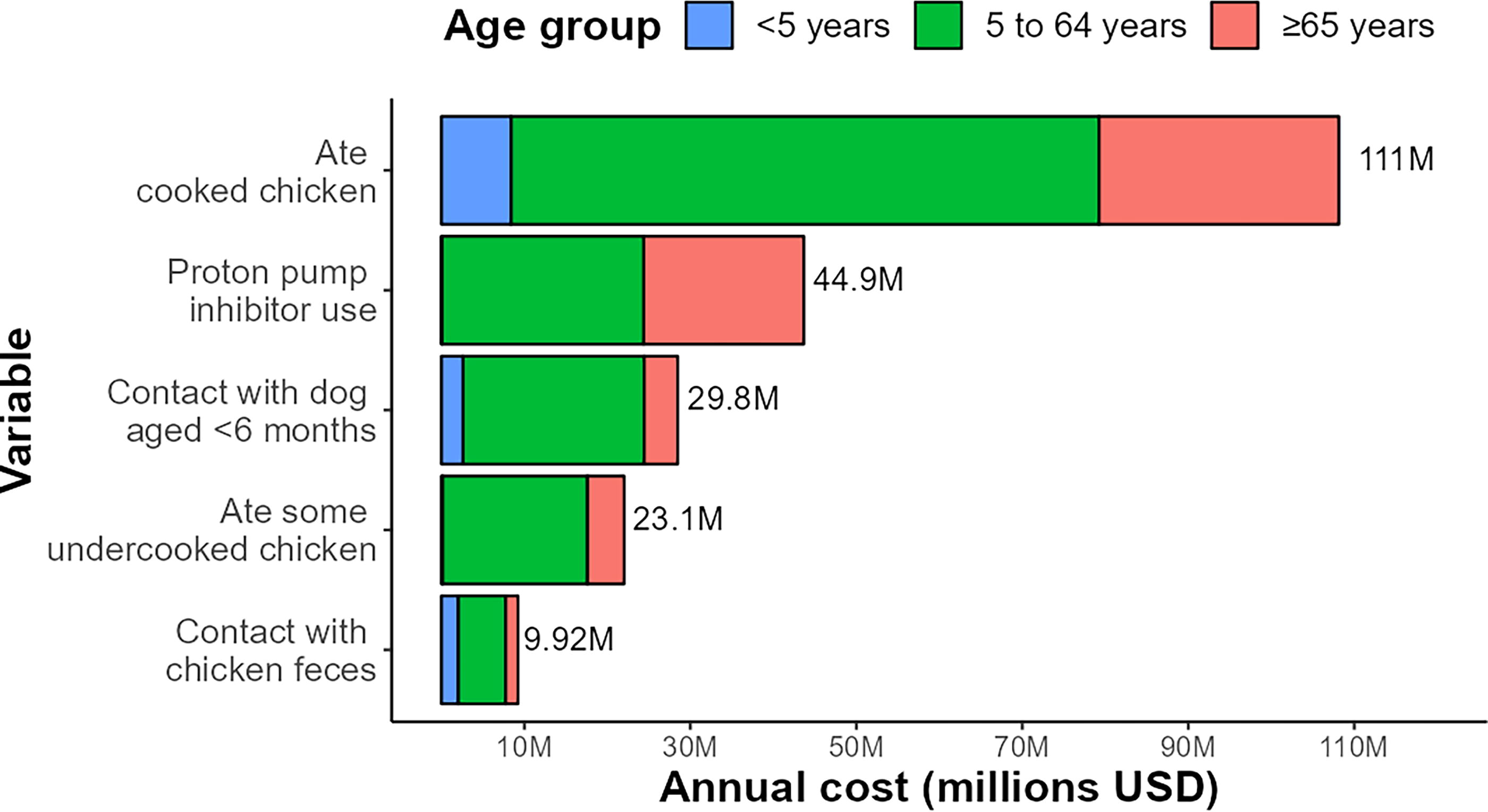
Annual cost attributable to five main campylobacteriosis risk factors in Australia circa 2019. Total cost per risk factor is provided in the figure. Costs are in US dollars.
While not a source of Campylobacter spp., PPI use increases the risk of gastrointestinal infections. We did not have power to include interactions as to how PPI use affects these individual pathways, so we instead present values for PPI use as a standalone risk factor. We attributed 42,500 (90% UI 24,500–71,100) excess cases of campylobacteriosis to PPI use in the 4 weeks before illness, along with 1,070 (90% UI 686–1,590) hospitalizations and one (90% UI 0–1) death (Supplementary Table S18). We estimated that the excess societal cost of campylobacteriosis and sequelae due to PPI use was US$44.9 million (Fig. 2; Supplementary Table S19).
We attributed 32,500 (90% UI 19,300–54,000) campylobacteriosis cases to contact with dogs aged less than 6 months and estimated 595 (90% UI 389–884) hospitalizations and less than one death (90% UI 0–1; Supplementary Table S20). This equated to US$29.8 million annually (Fig. 2; Supplementary Table S21). We attributed 10,800 (90% UI 5,300–20,000) campylobacteriosis cases to contact with chicken feces in the 7 days before illness and 192 (90% UI 99–329) hospitalizations and no deaths (90% UI 0–0; Supplementary Table S22). This equated to over US$9.92 million annually (Fig. 2; Supplementary Table S23).
Discussion
We demonstrated the significant impact and costs of campylobacteriosis from key risk factors in Australia, and particularly that associated with chicken consumption. Previous studies have attributed campylobacteriosis cases to risk factors (Cribb et al., 2022; Stafford et al., 2008). Other economic studies have quantified the total costs of campylobacteriosis and related sequelae for all illness and illness attributed to foodborne sources, estimated at US$2.18 billion in the United States circa 2018 (U.S. Department of Agriculture [USDA], Economic Research Service [ERS], 2021) and £50 million in the United Kingdom circa 2008–9 (Tam and O’Brien, 2016). Furthermore, a study in the United States estimated basic costs attributed with unspecified poultry consumption at US$1.46 billion circa 2019 (Scharff, 2020). This current study is the first to estimate costs of infection risk factors at the country level from primary data sources for both foodborne and nonfoodborne exposures. Attributable costs and disease burden metrics can provide insight into the relative importance of risk factors from an economic perspective and can highlight priority areas for public health resources and interventions in a more targeted way than disease-wide estimates, as seen with a WHO noncommunicable disease attributable burden study (World Health Organization, 2018).
Campylobacteriosis linked to eating contaminated chicken meat, even when reported as cooked, incurred costs of approximately US$111 million, which accounts for almost half of the estimated total costs of foodborne campylobacteriosis of US$253 million (AU$365 million) (Glass et al., 2023). We found that ∼30% of campylobacteriosis cases were attributed to eating cooked chicken meat, greater than a previous Australian study at 21.2% conducted in 2001–2002 but similar to a Dutch study at 28% from 2002 to 2003 (Mughini Gras et al., 2012; Stafford et al., 2007). As cooking meat kills Campylobacter spp., attribution to eating cooked chicken is likely from people unknowingly eating undercooked chicken or from poor handling of raw chicken and cross-contamination during preparation (Stafford et al., 2007). Recent mathematical modeling studies have attributed high proportions of cases to poultry sources at 84% of cases in New Zealand and 80% of cases in Australia (Lake et al., 2021; McLure et al., 2023). These results highlight the importance of broiler meat production on campylobacteriosis cases. Costs associated with chicken consumption can be used to evaluate the cost-effectiveness of interventions. For example, a study in New Zealand assessed the cost-effectiveness of interventions within the poultry meat supply chain, comparing disability-adjusted life years (DALYs) with actual costs of different interventions (Lake et al., 2013). Similarly, a previous Dutch study evaluated interventions in the poultry supply chain and compared intervention costs with both DALYs and healthcare costs, estimated at €4.2 million circa 2005 (US$6.28 million circa 2019) (Havelaar, 2005). Increased chicken meat production has resulted in a high burden of campylobacteriosis, which emphasizes the importance of the poultry industry and food safety authorities working to identify interventions to reduce Campylobacter infections (Skarp et al., 2016).
Proton-pump inhibitors are one of the most commonly prescribed medications used to treat stomach acid-related disorders and are available without a prescription in Australia (Chen et al., 2016, Jackson et al., 2016). Excess campylobacteriosis attributable to PPI use represented the second-highest costing risk factor, with nearly half of these costs from illness in adults over 65 years. PPI use increases the risk of acquiring illness from Campylobacter and other foodborne pathogen-contaminated foods by altering the gut biome and increasing the pH of stomach acid (Jackson et al., 2016). In addition to enteric infections, long-term PPI use has been associated with adverse effects, particularly in older adults, including bone fractures, kidney disease, and vitamin B12 deficiency (Maes et al., 2017). There is evidence that PPI use is associated with an increased risk in hospitalization from campylobacteriosis (Chen et al., 2016) and an increased risk of developing IBS following campylobacteriosis (Esan et al., 2020). Our estimates do not account for these increased risks and therefore are conservative.
Our study has some limitations. Our analysis relied on self-reported exposure to risk factors from cases and controls to calculate aORs and AFs, which may be influenced by recall and reporting biases. We minimized this bias by conducting interviews within 2 weeks of illness. Cases may be more likely to remember contact with known risk factors for illness than controls, and this may result in differential misclassification resulting in an overestimation of the actual impact of these factors. We validated the questionnaire before interviews to minimize misclassification. We tested PPI use as an interaction term with age and other significant risk factors in initial models but were unable to include these estimates due to insufficient statistical power. Lastly, the risk factors we considered had different levels of exposure between different age groups, and though we could estimate exposure by age group, the study was inadequately powered to estimate these age-specific odds ratios for each risk factor (Unicomb et al., 2008).
Quantifying the burden and costs associated with specific risk factors for campylobacteriosis translates risk and burden measurements into meaningful and actionable insights that can inform the prioritization of interventions. These costs can be compared with the costs of implementing measures to reduce Campylobacter prevalence in risk factors. Priority should be given to direct pathways from contaminated chicken exposure to human illness, emphasizing targeted interventions (e.g., on-farm biosecurity measures, primary processing changes, and consumer education) that require collaboration between primary producers, retail, government, and researchers. Recognizing the impact of risk modifiers such as prolonged PPI use, and the importance of less common infection pathways like maintaining proper hygiene after contact with animals and contaminated animal products, is paramount for a comprehensive understanding of the overall burden of campylobacteriosis. Our estimates of costs attributable to risk factors should serve as the basis for evaluating interventions and programs aimed at reducing campylobacteriosis.
Footnotes
Acknowledgments
The authors thank the extended CampySource Project team, reference panel, and additional contributors to the study. The CampySource Project team comprises three working groups and a reference panel. The working groups focus on food and animal sampling, epidemiology and modeling, and genomics. The reference panel includes expert representatives from government and industry. The study includes the following partner organizations: the Australian National University, Massey University, University of Melbourne, Queensland Health, Queensland Health Forensic and Scientific Services, New South Wales Food Authority, New South Wales Health, Hunter New England Health, Victorian Department of Health and Human Services, Food Standards Australia New Zealand, Commonwealth Department of Health and AgriFutures Australia–Chicken Meat Program. CampySource also collaborates with the following organizations: ACT Health, Sullivan Nicolaides Pathology, University of Queensland, Primary Industries and Regions South Australia, Department of Health and Human Services Tasmania, Meat and Livestock Australia, and New Zealand Ministry for Primary Industries. The CampySource Project Team consists of: Nigel P French, Massey University, New Zealand; Mary Valcanis, The University of Melbourne; Dieter Bulach, The University of Melbourne; Emily Fearnley, South Australian Department for Health and Wellbeing; Russell Stafford, Queensland Health; Amy Jennison, Queensland Health; Trudy Graham, Queensland Health; Keira Glasgow, Health Protection NSW; Kirsty Hope, Health Protection NSW; Themy Saputra, NSW Food Authority; Craig Shadbolt, NSW Food Authority; Arie Havelaar, The University of Florida, USA; Joy Gregory, Department of Health and Human Services, Victoria; James Flint, Hunter New England Health; Simon Firestone, The University of Melbourne; James Conlan, Food Standards Australia New Zealand; Ben Daughtry, Food Standards Australia New Zealand; James J Smith, Queensland Health; Heather Haines, Department of Health and Human Services, Victoria; Sally Symes, Department of Health and Human Services, Victoria; Barbara Butow, Food Standards Australia New Zealand; Liana Varrone, The University of Queensland; Linda Selvey, The University of Queensland; Tim Sloan-Gardner, ACT Health; Deborah Denehy, ACT Health; Radomir Krsteski, ACT Health; Natasha Waters, ACT Health; Kim Lilly, Hunter New England Health; Julie Collins, Hunter New England Health; Tony Merritt, Hunter New England Health; Rod Givney, Hunter New England Health; Joanne Barfield, Hunter New England Health; Ben Howden, The University of Melbourne; Kylie Hewson, AgriFutures Australia–Chicken Meat Program; Dani Cribb, The Australian National University; Rhiannon Wallace, The Australian National University; Angus McLure, The Australian National University; Ben Polkinghorne, The Australian National University; Cameron Moffatt, The Australian National University; Martyn Kirk, The Australian National University; and Kathryn Glass, The Australian National University. The authors acknowledge the contributions of Siobhan Bourke towards previous cost estimates work and Food Standards Australia New Zealand staff and reference panel members from University of Western Australia, University of Tasmania, Australian Government Department of Health, Nutrition Policy Section, NSW Department of Primary Industries, United States Department of Agriculture, and UK Food Standards Agency on the costing model and associated report. This work was completed while ELL was employed at the ANU.
Authors’ Contributions
Conceptualization: K.G., M.D.K., and A.T.M.; Data curation: D.M.C. and A.T.M.; Formal analysis: D.M.C. and A.T.M.; Funding acquisition: K.G. and M.D.K.; Investigation: D.M.C. and A.T.M.; Methodology: D.M.C., K.G., E.L.L., M.D.K., and A.T.M.; Project administration: D.M.C.; Software: A.T.M.; Supervision: K.G., R.L.W., M.D.K., and A.T.M.; Validation: K.G., E.L.L., R.J.S., and A.T.M.; Visualization: D.M.C. and A.T.M.; Writing—original draft: D.M.C., K.G., M.D.K., and A.T.M.; Writing—review and editing: All authors.
Ethical Approval
The CampySource Project was approved by the Australian National University Human Research Ethics Committee (Reference No. 2016/426), ACT Health Human Research Ethics Committee (Reference No. ETH.8.17.168), Qld Health Human Research Ethics Committee (Reference No. RD007108), HNE Human Research Ethics Committee (Reference No. 17/08/16/4.03), and the University of Melbourne Office of Research Ethics and Integrity (Reference No. 1750366.1). Human ethics approval was obtained for patients with campylobacteriosis in the ACT, NSW, and Qld as part of the CampySource case–control study. Informed consent was obtained from all subjects and/or their legal guardian. All notified data and cost components were from publicly available data and did not require ethics clearance.
Disclosure Statement
The authors declare no competing interests for this work.
Funding Information
This work was supported by multiple funding sources: an Australian Government Research Training Program scholarship to D.C.; a National Health and Medical Research Council fellowship [grant number GNT1145997] to M.K.; National Health and Medical Research Council [grant number GNT1116294], AgriFutures, Australian Government Department of Health, Food Standards Australia New Zealand, New South Wales Food Authority, Queensland Health, and ACT Health for the CampySource Project; the Australian Government Department of Health and Aged Care through a tender for Food Standards Australia New Zealand [grant number 2020-21/03] for previous costing work.
Supplementary Material
Supplementary Figure S1
Supplementary Table S1
Supplementary Table S2
Supplementary Table S3
Supplementary Table S4
Supplementary Table S5
Supplementary Table S6
Supplementary Table S7
Supplementary Table S8
Supplementary Table S9
Supplementary Table S10
Supplementary Table S11
Supplementary Table S12
Supplementary Table S13
Supplementary Table S14
Supplementary Table S15
Supplementary Table S16
Supplementary Table S17
Supplementary Table S18
Supplementary Table S19
Supplementary Table S20
Supplementary Table S21
Supplementary Table S22
Supplementary Table S23
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
