Abstract
In recent years, vegetables gain consumer attraction due to their reputation of being healthy in combination with low energy density. However, since fresh produce is often eaten raw, it may also be a source for foodborne illness. The presence of antibiotic-resistant bacteria might pose a particular risk to the consumer. Therefore, this review aims to present the current state of knowledge concerning the exposure of humans to antibiotic-resistant bacteria via food of plant origin for quantitative risk assessment purposes. The review provides a critical overview of available information on hazard identification and characterization, exposure assessment, and risk prevention with special respect to potential sources of contamination and infection chains. Several comprehensive studies are accessible regarding major antimicrobial-resistant foodborne pathogens (e.g., Salmonella spp., Listeria spp., Bacillus cereus, Campylobacter spp., Escherichia coli) and other bacteria (e.g., further Enterobacteriaceae, Pseudomonas spp., Gram-positive cocci). These studies revealed vegetables to be a potential—although rare—vector for extended-spectrum beta-lactamase-producing Enterobacteriaceae, mcr1-positive E. coli, colistin- and carbapenem-resistant Pseudomonas aeruginosa, linezolid-resistant enterococci and staphylococci, and vancomycin-resistant enterococci. Even if this provides first clues for assessing the risk related to vegetable-borne antimicrobial-resistant bacteria, the literature research reveals important knowledge gaps affecting almost every part of risk assessment and management. Especially, the need for (comparable) quantitative data as well as data on possible contamination sources other than irrigation water, organic fertilizer, and soil becomes obvious. Most crucially, dose–response studies would be needed to convert a theoretical “risk” (e.g., related to antimicrobial-resistant commensals and opportunistic pathogens) into a quantitative risk estimate.
Introduction
V
However, vegetables are also implicated in foodborne outbreaks due to various—most often unknown—sources of contamination, the most recent familiar case in Europe being the enterohemorrhagic Escherichia coli (EHEC) outbreak of 2011, with 4321 reported cases, of which at least 50 had been fatal (Robert-Koch-Institute, Germany, cited from Buchholz et al., 2011). Besides other unusual features, the outbreak strain was characterized as an extended-spectrum beta-lactamase (ESBL) producer due to the presence of bla CTX-M15 genes (Mellmann et al., 2011). The press reported extensively on antimicrobial resistance (AMR), but not on the fact that this resistance was meaningless for therapy, due to contraindication of antibiosis in EHEC infections (see Hazard Identification and Characterization section).
According to the FAO definition, quantitative risk assessment is based on four steps: hazard identification, hazard characterization, exposure assessment, and risk characterization (
There have been well-recognized approaches for risk assessment related to general microbial hazards imposed by vegetable consumption (Hamilton et al., 2006; Franz et al., 2010; Danyluk and Schaffner, 2011; Pang et al., 2017). However, no specific quantitative risk assessment has been published for hazards related to antimicrobial-resistant bacteria (ARB) in vegetables, although AMR is seen as an obligate “risk amplifier” in public awareness. Steps to assess specific risks due to AMR would involve all steps of quantitative risk assessment. This starts from hazard identification (i.e., is a known adverse health effect aggravated by AMR features of the causative agent? Does an adverse health effect arise from spread of antibiotic resistance features in commensals, which otherwise would not be considered as microbial hazards?) but also includes hazard characterization as well as exposure assessment.
This literature survey aims to initiate a discussion on quantitative risk assessment related to the spread of antibiotic resistance in the vegetable food chain. For this purpose, we tried to provide an overview over current screening data starting from 2007. To allow risk prevention, sources of contamination were highlighted as well. Finally, important knowledge gaps were identified, which should be addressed in the future to facilitate quantitative risk assessment within a reasonable term.
Literature Search and Exclusion Criteria
Our PubMed search combined the keywords “antimicrobial resistance” or “antibiotic resistance” with “vegetables” or “fresh produce” in May 2018. We aimed to provide a systematic overview on recent screening data for antibiotic-resistant bacteria isolated from vegetables, so we covered the period from January 2007 to April 2018 and found 169 studies, after excluding double hits. The search result comprised 93 studies that reported screening data for antibiotic resistance in vegetable-borne isolates (Tables 1 –7). We excluded 22 obvious mishits (one study dealing with the inhibition of bladder cancer cell proliferation by mustard oil, for example) and 13 studies that did not investigate vegetable-borne isolates (instead, these studies reported on, e.g., reptiles). Eight studies were excluded since they did not attribute resistant isolates to their source (these studies presented resistance rates from different sources, e.g., meat and vegetables, as one common value). We excluded opinion reports, reviews (unless they comprised relevant screening data), and publications that described modeling or method implementation or whole genome sequencing data of single isolates (in total 14 studies). We also excluded three studies in a language other than English (Polish, Ukrainian, and Chinese). One study that would not have met the exclusion criteria was excluded afterward since it presented mainly resistance data for Escherichia coli and vancomycin, a substance that is not effective in Enterobacteriaceae. All other studies presenting intrinsic resistance data (e.g., erythromycin in Salmonella spp.) were kept since significant results for acquired resistance were presented as well.
Studies on Major Antimicrobial-Resistant Foodborne Pathogens (Salmonella enterica subsp. enterica, Listeria spp., Bacillus cereus, Campylobacter spp.) in Vegetables
Font normal: resistance tested but not found; underlined: resistance in one single isolate; bold face: resistance in two or more isolates. Resistance defined as in the original study.
In L. ivanovii and L. welshimeri.
Studies on Antimicrobial-Resistant Escherichia coli in Vegetables
Font normal: resistance tested but not found; underlined: resistance in one single isolate; bold face: resistance in two or more isolates. Resistance defined as in the original study.
STEC, Shiga-toxin producing E. coli.
Studies on Antimicrobial-Resistant Pseudomonads in Vegetables
Font normal: resistance tested but not found; underlined: resistance in one single isolate; bold face: resistance in two or more isolates. Resistance defined as in the original study.
All 13 experimental studies returned by the keywords were included in the text (e.g., experimental application of manure or biosolids to soil), as were two studies that reported metagenomic screening data.
Besides our systematic approach, we used targeted searches with individual keywords to add details (e.g., on vegetable consumption).
Hazard Identification and Characterization
A recent outbreak due to vegetables was related to a multiresistant strain of EHEC O104:H4, which produced ESBL enzymes (Buchholz et al., 2011). Did this fact add adverse effects to the outbreak? In general, antibiotics are not the first-line agents for treatment of diarrhea (Guerrant et al., 2001). On the contrary, for EHEC, as compiled by Goldwater and Bettelheim (2012), antibiotics—and especially beta-lactams—are even contraindicated due to the fact that cell-wall-impaired dead bacteria release more toxin (Tarr et al., 2005; Smith et al., 2012). Even subinhibitory concentrations are thought to increase toxin production and/or toxin release (Grif et al., 1998). Thus, in the absence of treatment indication, AMR could not lead to treatment failure. However, for other bacterial infections such as systemic listeriosis, antibiotic therapy is the treatment of choice (Safdar and Armstrong, 2003), and clinical resistance against first-line antibiotics is likely to impose additional health hazards. Centers for Disease Control and Prevention report recent cases of listeriosis in the United States due to vegetable consumption, which led to several deaths (
Providing comprehensive hazard identification of all foodborne pathogens associated with vegetables goes beyond the scope of this review. Thus, three more general situations will be exemplarily illustrated and connected to AMR: (1) primary foodborne infectious disease due to obligatory or opportunistic pathogens, (2) foodborne microbial intoxication, (3) foodborne colonization, maybe followed by opportunistic disease after a considerable time-shift (Fig. 1a–c).
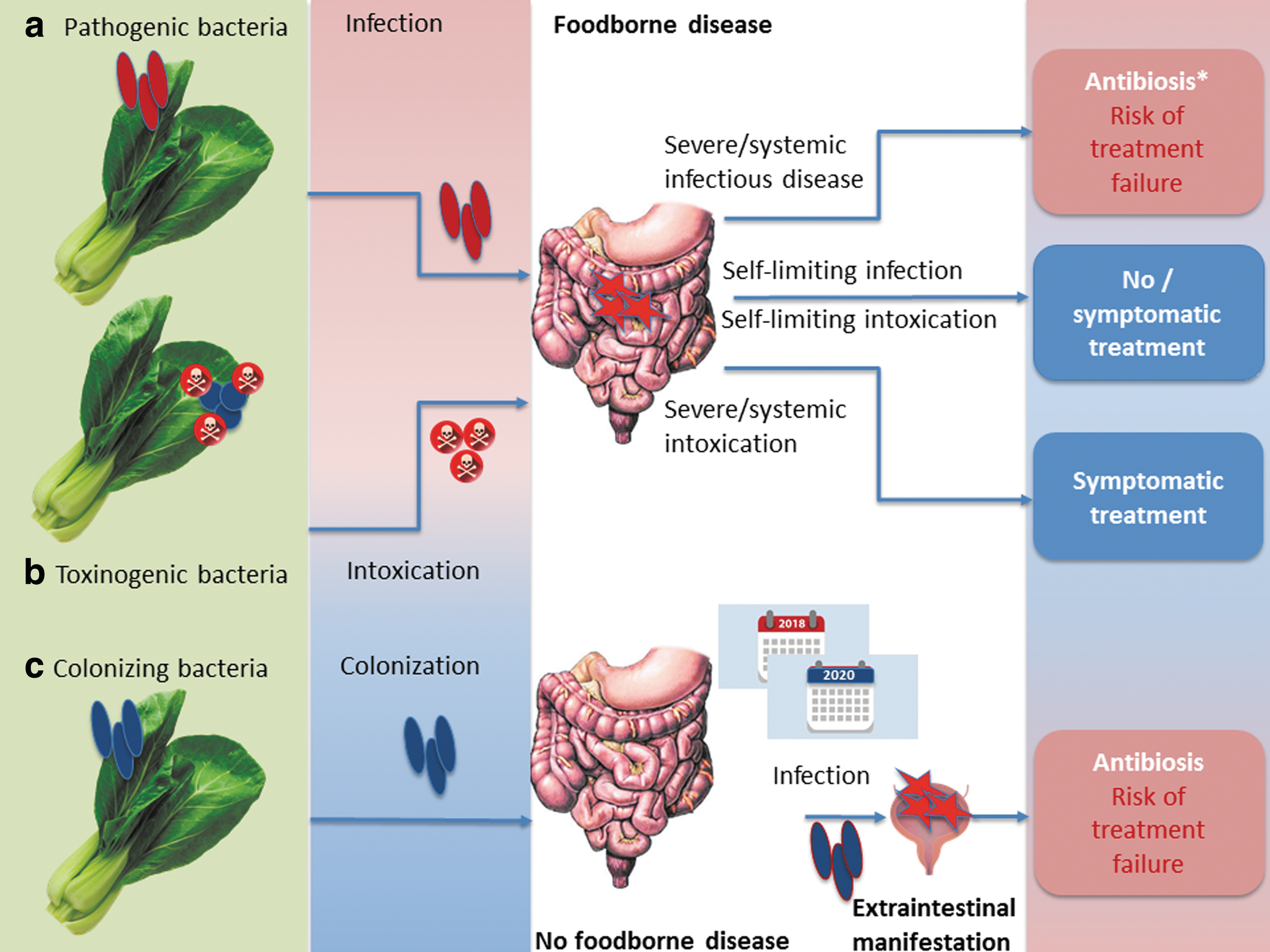
Impact of antimicrobial resistance in different situations following vegetable-borne
Primary foodborne infectious disease is seen after infection with pathogenic serovars of, for example, Salmonella enterica subsp. enterica, Campylobacter spp., Listeria monocytogenes, Bacillus cereus, and E. coli (e.g., EHEC, Shiga-toxin producing E. coli [STEC]) (Butler et al., 2015). In case of systemic infection, as already mentioned, antimicrobial treatment is often indicated (except for EHEC), and AMR to clinically relevant antimicrobial drugs will lower the number of therapeutic options (Fig. 1a). In contrast, foodborne Staphylococcus aureus exclusively causes food poisoning due to the presence of preformed heat-stable toxins independent of viable bacteria (Kadariya et al., 2014). Thus, this kind of food poisoning is self-limiting, and antibiotic treatment is never indicated (Fig. 1b). However, S. aureus might cause opportunistic infections of wounds like cuts (van de Sande-Bruinsma et al., 2015), which might occur during food preparation and might call for topical treatment. Only a limited number of B. cereus lineages (Ehling-Schulz et al., 2005) are able to cause mere food poisoning due to the presence of cereulide (see Rosenquist et al., 2005, for example). Instead, in most cases, B. cereus needs to infect the organism to cause gastrointestinal disease (due to synthesis of Nhe and other enterotoxins in the intestine; EFSA, 2016). This is similar to the situation seen in Clostridium perfringens (Uzal et al., 2014). Antibiotic treatment of B. cereus infections is recommended in severe disease, whereas it is unreasonable in self-limited food poisoning (Spiliopoulou et al., 2014). C. perfringens might contribute to antibiotic-associated diarrhea (Borriello et al., 1984; Kim et al., 2017b); treatment of choice is discontinued primary antibiosis; only severe cases might be treated with glycopeptides or metronidazole (Bergogne-Bérézin, 2000).
A particular situation is found for organisms that are able to colonize the human gastrointestinal tract (Fig. 1c), such as E. coli or enterococci. These bacteria are useful indicators for AMR (Franklin et al., 2001; Schwaiger et al., 2011b) and were included in a recent EFSA recommendation to provide harmonized European monitoring of AMR (EFSA, 2012), as authorized by Directive 2003/99/EC. In addition, E. coli might produce ESBL enzymes, and this ESBL production (or other resistance phenotypes) might complicate the treatment of opportunistic infections. However, it is impossible to relate such colonization to a particular foodborne source, such as it is done for foodborne outbreaks: the onset of opportunistic infectious disease (e.g., urinary tract infection due to ESBL E. coli) might sporadically happen years after ingestion of the contaminated food. Thus, according to Depoorter et al. (2012), the human health risk posed by a given exposure to foodborne ESBL E. coli cannot be estimated yet.
In general, the situation described in Figure 1a–c applies for all foods, not only vegetables. However, the risk to encounter infection or colonization is elevated in vegetables, due to a high share of raw consumption.
Studies on Other Antimicrobial-Resistant Gram Negatives in Vegetables
Font normal: resistance tested but not found; underlined: resistance in one single isolate; bold face: resistance in two or more isolates. Resistance defined as in the original study.
R > 8 mg/L.
Studies on Antimicrobial-Resistant Gram-Positive Cocci in Vegetables
Font normal: resistance tested, but not found; underlined: resistance in one single isolate; bold face: resistance in two or more isolates. Resistance defined as in the original study.
E. faecalis; b E. casseliflavus, E. faecalis; c E. casseliflavus.
Comparative Prevalence (%) of Antimicrobial-Resistant Escherichia coli in Meat and Vegetables (NonSelective Approach; Calculated from Percent Positive Samples and Percent Resistant Isolates)
Different superscripts in a row: values differ significantly in a chi-squared test (Fisher's exact test, if expected values are below 5 per cell).
Opportunistic pathogens (and apathogenic commensals) might further serve as a vehicle for the transfer of antimicrobial resistance genes (ARGs) to pathogens. This might happen in the intestine, but also before consumption as observed on lettuce with a resistance gene of clinical relevance in humans (bla SHV18, Jung and Matthews, 2016). DNA is partly degraded by heat, so raw consumed food is also more likely to pass high concentrations of ARGs into the human intestine.
In terms of hazard identification, additional hazard due to AMR is identified in systemic foodborne infection and in opportunistic colonization, but not in self-limiting diarrhea and intoxication.
In terms of hazard characterization, dose–response relationships are the most critical point for quantitative risk assessment. In case of obligate pathogens that would be antibiotically treated (Fig. 1a), the dose–response relationship is that of foodborne disease, so data are available for many pathogens. In contrast, dose–response data are completely missing for stable intestinal colonization or opportunistic extraintestinal disease. For colonization, hints are provided from studies using oronasal infection to colonize animals: Schoeni and Doyle (1994) isolated E. coli O157:H7 from cecal tissue of single chickens 3 months after experimental inoculation with as low as 2.6 × 101 colony-forming unit (cfu).
Exposure Assessment
Produce and human consumption of vegetables
Community members of the EU produced vegetables (including melons and strawberries) on a total area of 224,126,000 ha in 2016 (
Prevalence of ARB in vegetables
A PubMed search combining the keywords “antimicrobial resistance” or “antibiotic resistance” with “vegetables” or “fresh produce” covered the period between January 2007 and April 2018. After applying the exclusion criteria as described above, 93 hits were condensed in Tables 1 –7.
The studies included bacteria of more than 20 different genera (Tables 1 –7), mainly E. coli, S. enterica subsp. enterica, Listeria spp., enterococci, pseudomonads, and B. cereus. To provide basic information from the studies, resistances are marked as present or absent in Tables 1 –5 (irrespective of whether they are acquired or intrinsic). Additionally, prevalences are included in Tables 6 and 7. In general, comparisons between studies should be avoided since different choices of breakpoints introduce significant bias when comparing resistance data according to the European EUCAST organization with the U.S. Clinical and Laboratory Standards Institute (CLSI). Moreover, many studies lack the information of whether they used appropriate reference strains in antimicrobial susceptibility testing.
Prevalence of Vegetable-Borne Bacteria with Particularly Relevant Antimicrobial Resistance (Examples)
R > 8; CLSI breakpoint: R ≥ 8, for colistin recently adjusted to R ≥ 4.
?, unknown number; CLSI, Clinical and Laboratory Standards Institute; ESBL, extended-spectrum beta-lactamase; MSSA, methicillin-sensitive Staphylococcus aureus.
For Salmonella, studies are hard to compare due to a high diversity in serotypes. A huge number of vegetables (more than 100,000 samples) were investigated in the United States from 2002 to 2012, resulting in isolation of 152 Salmonella strains, of which as few as 10 had detectable AMRs (Reddy et al., 2016; Table 1). However, CLSI warned that Salmonella might be falsely reported as susceptible to several antibiotics (CLSI M100-S22E, table 2A in the CLSI document) due to differences between in vitro and in vivo susceptibility. Yoke-Kqueen et al. (2008) reported 56.7% of 134 isolates with a multiple antimicrobial resistance (MAR) index of more than 0.2. However, erythromycin was included in the MAR. Corrected for this fact, 44.3% of isolates from Malaysian vegetables were resistant to two or more antibiotics (mainly tetracycline+streptomycin—substances that are not the first- or second-line drugs in human therapy of salmonellosis). Relevant multiresistance (>5 resistances, including third-generation cephalosporins) was found in Salmonella Albany, Salmonella Brunei, and Salmonella Kralingen (Yoke-Kqueen et al., 2008).
All identified studies dealing with AMR in vegetable-borne B. cereus (n isolates = 39–110, Table 1) referred to Korean fermented or traditional food (Lee et al., 2012; Kim et al., 2015; Yim et al., 2015), thus statements are restrained to a limited variety of food and regions. In addition, resistance was assessed only by means of disk diffusion, and the source and validity of breakpoints is unclear, since these studies did not refer to CLSI or other acknowledged standards, but to the secondary literature. Apart from substances to which B. cereus should be considered as intrinsically resistant (e.g., ampicillin, oxacillin, penicillin, cefepime, rifampin), resistance rates were moderate, with complete susceptibility to gentamicin, imipenem, ciprofloxacin, tetracycline, and vancomycin in one study (Kim et al., 2015). However, resistance to vancomycin—a first-line therapeutic in (extraintestinal) B. cereus infections—was reported in 1/87 isolates from fermented soybeans (Yim et al., 2015) and 5/39 isolates from Sunsik (Lee et al., 2012), a ready-to-eat food made from grains, fruits, and vegetables.
Significant data for L. monocytogenes (n = 144, Table 1) from fresh and frozen vegetables were reported from a Polish study using E-test with CLSI-breakpoints for listeria and staphylococci (Korsak et al., 2012): only one vegetable-borne isolate had detectable AMR at all (to tetracyclines). In Turkish ready-to-eat salad, listerial AMR was most pronounced to erythromycin and cephalothin (Gurler et al., 2015). However, cephalosporin resistance is considered as an intrinsic feature of L. monocytogenes (Collins et al., 2012), and erythromycin is not considered as a treatment of choice (Morvan et al., 2010).
Campylobacter jejuni (n = 33, Table 1) from Malaysian “salad style vegetables” had moderate resistance rates except for intrinsic resistances and erythromycin resistance (60.6%; Khalid et al., 2015). The latter fact might be remarkable since macrolides have for long been used as a therapy of choice (Blaser et al., 1979; Engberg et al., 2001). However, the study is generally limited by the fact that breakpoints were outdated (taken from a document from 2003, when the CLSI was still named NCCLS). In any case, fluoroquinolones—a first-line therapy of today—were highly effective in this study.
Resistance data for E. coli (60–239 isolates, Table 2) differed significantly between studies; this might be related to the very different locations (South Africa, India, United States, South America, Europe, Lebanon, Iran), the different nature of samples, and the different choice of antibiotics and breakpoints.
The majority of studies reported pronounced resistance to streptomycin and tetracyclines—a finding of limited significance, given the fact that clinical breakpoints cut within natural populations. However, studies also reported significant resistance to amoxicillin combined with clavulanic acid (>15%) (e.g., Araújo et al., 2017) or resistance to carbapenems (Shakerian et al., 2016) and third- or fourth-generation cephalosporin as well as presence of bla
TEM1-genes (Araújo et al., 2017). Remarkably, Gómez-Aldapa et al. (2016) reported 100% resistance to amikacin and colistin in diarrheagenic E. coli pathotypes from Mexican cactus salads. The study referred to CLSI; however, CLSI does not provide a breakpoint for colistin in E. coli. In addition, colistin resistance was assessed by disk diffusion, a practice that is discouraged nowadays (
For pathogenic E. coli, one study assessed the prevalence of plasmid-borne colistin resistance genes mcr1/mcr2 in STEC E. coli from the United States; not a single positive isolate was found in 1000 strains from different sources, including about 230 isolates from vegetables (Mavrici et al., 2017).
Due to their environmental sources, pseudomonads are frequently found in vegetables. Pseudomonads are not considered as major foodborne pathogens; however, six studies were identified, which reported resistance data of up to 401 Pseudomonas isolates from, mostly Pseudomonas aeruginosa (Table 3). Using microdilution and DIN58940-breakpoints where available, one study identified significantly higher resistance rates to aminoglycosides in Pseudomonas isolates from fruit vegetables compared with root vegetables or salads (Schwaiger et al., 2011a). In this study, resistance also included antibiotics used in clinics, such as gentamicin, tobramycin, amikacin, ciprofloxacin, colistin, piperacillin, ceftazidime, imipenem, or meropenem; however, such resistances were rare (Schwaiger et al., 2011a). Using CLSI breakpoints and disk diffusion, most of these resistances were also observed in Jamaican vegetable isolates, apart from resistance to carbapenems (Allydice-Francis and Brown, 2012).
Cronobacter spp. acts as a foodborne pathogen in immunocompromised patients (Healy et al., 2010) and will then be antibiotically treated, for example, with ampicillin plus gentamicin or chloramphenicol (Lai, 2001). However, vegetable isolates were characterized by a high rate of pansusceptible strains and lacked resistance against clinically relevant antimicrobials (Vojkovska et al., 2016; Table 4). For Enterobacter spp. (n = 264), several AMRs were significantly lowered at retail, compared with farm level, pointing toward a need for sampling close to consumption for particular questions of risk assessment (Schwaiger et al., 2011a; Table 4). Once more, resistance depended on the vegetable group: resistance to colistin was significantly more frequent in Enterobacter cloacae from fruit vegetables than from roots (Schwaiger et al., 2011a).
None of the studies on Gram-positive cocci (Table 5) reported staphylococci to be resistant to vancomycin, a critically important antibiotic especially for infection caused by methicillin-resistant S. aureus (MRSA). Korean leafy vegetables were partly contaminated with multiresistant MRSA strains (Hong et al., 2015). One methicillin-susceptible isolate was resistant to linezolid, a first-line drug for the treatment of MRSA-infected wounds (Gurusamy et al., 2013). Single linezolid-resistant strains of Enterococcus faecalis (1/100) and Enterococcus faecium were isolated from German vegetables (Schwaiger et al., 2011a). One linezolid-resistant E. faecium was detected in Canadian vegetables as well (Allen et al., 2013). Detectable resistance in E. faecalis isolates (n = 20) from Portuguese ready-to-eat salad was restrained to tetracyclines and erythromycin due to the presence of tet(M), tet(L), and erm(B) (Campos et al., 2013). From Tunisia, Ben Said et al. (2016) reported additional resistance to high level concentrations of aminoglycosides and chloramphenicol. By multilocus sequence typing, Leavis et al. (2006) identified vegetable-borne ARB of a so-called high-risk enterococcal clonal complex. Importantly, vancomycin–teicoplanin-resistant enterococci were isolated from fresh produce or its environment in Korea (Kim et al., 2017a).
Human exposure to vegetable-borne ARB
Assessing the exposure of humans to vegetable-borne ARB is not the same as assessing the prevalence of AMR in vegetable-borne bacteria: Considerable exposure to ARB might arise in a situation when bacteria are rarely found, but frequently resistant, or vice versa in the situation when AMR is moderate, but the prevalence of bacterial contamination is very high. For Salmonella or Listeria spp., both prevalence of bacteria and prevalence of AMR within vegetable-borne bacteria are low (Reddy et al., 2016). For other bacteria such as pseudomonads, prevalence of bacteria is high, but the prevalence of clinically significant resistance is low.
Vice versa, the prevalence of E. coli or E. faecalis is much lower in vegetables than in food of animal origin, but the prevalence of AMR is remarkably high for some antibiotics and, although rare, resistances to critically important isolates are present as well. For E. coli, we had the chance to comparatively calculate the prevalence of ARB (prevalence of bacteria × prevalence of resistance) in pork, poultry, and vegetables from two studies (Schwaiger et al., 2011a, 2012 and unpublished data) using identical methods; pork and poultry were included for the sake of comparison. The studies resembled each other in identical (nonselective) bacterial isolation, identical susceptibility testing (microdilution, DIN 58940-81), and identical source of breakpoints. Results are shown in Table 6. In total, the prevalence of E. coli was lowest in vegetables (3.4% compared with 75% in chicken and 25% in pork). However, for several antibiotics, the prevalence of resistant E. coli was quite comparable between pork and vegetables. This was because a much lower prevalence of E. coli in vegetables was accompanied by a high prevalence of resistance: up to 55% of the vegetable-borne E. coli were resistant to beta-lactams.
For quantitative risk assessment, it would be crucial to know not just the prevalence but also the concentration of ARB in vegetables since risk characterization has to consider dose-related data. However, this information is more than rare. Some of the studies listed in Tables 1 –7 quantified bacteria (e.g., Hassan et al., 2011; Campos et al., 2013; Holvoet et al., 2013; Araújo et al., 2017; Du Plessis et al., 2017) but did not specifically refer to ARB. To assess these data, selective quantification for resistant bacteria would be needed, which is performed even rarer. Relevant data are provided by Ruimy et al. (2010), who reported densities of resistant bacteria as high as 104 cfu/g of product for Gram negatives grown in the presence of tetracycline, chloramphenicol, and nalidixic acid. Yang et al. (2016) quantified antibiotic-resistant endophytic bacteria in experimentally grown pak choi and found concentrations of up to 9 × 102 per g.
Risk Characterization
Risk characterization integrates the informations from hazard identification, hazard characterization, and exposure assessment into a quantitative estimate of risk (probability of adverse outcome or number of cases). Hazard identification related to resistant bacteria has to account for the fact whether this resistance is of therapeutic interest. This review revealed that clinically relevant antibiotics are underrepresented in current studies so that many pathogen-AMR combinations listed above do not imply any therapeutic consequence (hazard). However, ESBL production in Enterobacteriaceae or linezolid resistance in enterococci and staphylococci is surely of clinical interest.
Risk characterization would also include hazard characterization, which means dose–response relationships. Infectious doses vary in the individual host and situation; for many environmental bacteria, such infectious doses have not been defined at all. In addition, the amount of opportunistic pathogens or commensals, which is necessary to produce stable colonization in the human intestine, is unknown for most bacteria, and dose–response studies are absent. For foodborne outbreaks in general, case numbers are also used as a substitute in hazard characterization if dose–response data are missing. However, no specific case numbers are available for infectious disease due to vegetable-borne ARB.
In the absence of known colonization doses or known frequencies of colonization events, at least qualitative data might help to further characterize the risk. Thus, attempts were made to identify genetically related antimicrobially resistant organisms in food and human intestines or human clinical samples in a distinct region and period (e.g., Abriouel et al., 2008; Hannah et al., 2009; Burgos et al., 2014), but without showing significant association. Several studies tried to relate the presence of ARB in human feces to consumption behavior and found that vegetarians are at higher risk to carry ARB (Elder et al., 1993; Sannes et al., 2008). However, Sannes et al. (2008) also showed that this might be linked to a confounder related to the lifestyle of vegetarians: traveling abroad.
Exposure assessment identified vegetables as a relevant vehicle in terms of a high consumption frequency and in terms of frequent bacterial contamination in a ready-to-eat state. However, the exposure to clinically relevant pathogens or to commensals conferring clinically relevant resistance is low, compared with other food like meat.
Ultimately, taking together the individual steps of risk assessment, we have to state that the absence of dose–response data and the absence of published estimates on treatment failure due to AMR in foodborne infections do not allow quantitative risk characterization for vegetable-borne ARB yet.
Risk Prevention: Sources of Contamination
While sprout seeds from Egypt were considered as the most probable outbreak source for the multinational EHEC outbreak in 2011, the primary source of sprout contamination remained unknown. This is seen in most outbreaks (not only) related to vegetables, thus indirect clues are important for assessing infection chains. In rare cases, molecular typing of bacterial isolates from possible sources and contaminated vegetables is available to strengthen these indirect clues.
Irrigation water as a source of ARB
Worldwide, the FAO indicates a need for irrigation water as high as 1500 km3/year; of this irrigation water, 7.38% are used for vegetables and roots (
Several studies found that irrigation water and vegetables shared the same clones, as confirmed by repetitive-element polymerase chain reaction (rep-PCR: BOX-, REP-, and ERIC-PCR) (Jongman and Korsten, 2016; Araújo et al., 2017). Njage and Buys (2015) identified irrigation water as a possible pathway of transmission for ESBL E. coli.
Soil and organic fertilizer as sources of ARB
Vegetables produced in or close to soil—such as carrots and leaf vegetables—are at special risk for contamination with soil-borne bacteria, either belonging to natural soil microbiota or introduced into soil by manure fertilization. While the application of manure in ready-to-eat crops is discouraged, for example, by the U.K. Food Standards Agency (
Several studies focused on the prevalence of ARB or AMR genes in vegetables at harvest and the soil from which these vegetables were harvested (Marti et al., 2013, 2014; Wang et al., 2015; He et al., 2016; Lau et al., 2017; Tien et al., 2017); these studies found temporary—if any—impact of manuring compared with natural soil, which has a natural base level of ARG due to the presence of antimicrobial-producing microorganisms (Nesme and Simonet, 2015). Ruimy et al. (2010) showed that resistance scores were significantly higher in vegetables grown in or close to soil compared with fruits and vegetables grown above the soil. Rahube et al. (2014, 2016) reported that human biosolids are a relevant—but also temporary—source of ARG on vegetables at harvest, and Duan et al. (2017) found that ARG contents in soil and lettuce decreased after experimental application of biochar. Zhu et al. (2017) reported eightfold higher absolute copy numbers or ARG in manure-fertilized Chinese organic lettuce compared with conventionally produced lettuce; this difference was mainly due to different microbial communities in both types of samples. One recent study searched for ARGs in the viral DNA fraction (bacteriophages) from vegetables and soil and found that concentrations were highest for lettuce and soil, pointing toward soil as a relevant source of ARGs (Larrañaga et al., 2018).
Antibiotic residues in manure or soil might be more critical than is presence of manure-borne ARB, due to higher persistence. Antibiotic residues were able to select antimicrobially resistant endophytic bacteria, which are not removed by washing or peeling (Zhang et al., 2017; Esteban-Cuesta et al., 2018).
Direct contamination by humans
To our knowledge, systematic studies on human contamination of vegetables with ARB during production or processing are missing, as is systematic source tracking by molecular microbiological methods.
In the 2011 EHEC outbreak, it was assumed that, in a regional suboutbreak, vegetable dishes were contaminated by an infected caterer (
Besides the mentioned routes of contamination, other plausible ways of contamination include a broad variety of sources, either for the primary products (e.g., Salmonella contamination by reptiles and amphibians, Gorski et al., 2013) or during transport, processing, and distribution. Macrorestriction plus pulsed-field gel electrophoresis suggested that two different kinds of vegetables from two different countries were contaminated with a clonal E. coli strain due to packaging and distribution at the same factory. The role of processing is further illustrated by cut salads, which are generally considered as a risk food (
Outlook: Transfer of ARB to Humans by Food of Plant Origin—Current Lacks and Future Needs for Risk Assessment
A considerable share of studies used nonvalidated methods, reported intrinsic resistance, dealt with nonpathogenic bacteria, or provided data on AMRs/ARGs of low clinical relevance. Such limitations should be proactively addressed, if present. Commensal bacteria might be useful indicator organisms and serve as donors for AMR genes in the gut; in addition, the label pathogenic/nonpathogenic is conferred more carefully than in former since pathogenesis is more and more treated as a nonstatic interaction between strain-specific virulence and susceptibility of individual hosts. However, this fact gives even more emphasis to a need for prudent study design, overcoming the actual strategy of “count and collect” in favor of more functional approaches leading to dose–response data.
Important lacks of knowledge affect almost every part of risk assessment and management, starting from identifying sources of contamination and ending up to the black box of intestinal and infectious processes long after consumption. With regard to sources of contamination, search for plant-derived ARB should include processing and distribution steps since contamination close to consumption is more likely to result in human exposure, and human contamination might be more likely to introduce strains with AMR against human therapeutics. Further research should thus focus on clinically relevant antibiotics, should provide (or reject) evidence of transmission at clonal level, and should clarify and quantify the involvement of commensals in ARG transfer to pathogens as well as in extraintestinal opportunistic infections after transmission by food. Future studies addressing these gaps would be more than welcome.
Footnotes
Disclosure Statement
No competing financial interests exist.
