Abstract
Purpose:
Urodynamics is used to diagnose lower urinary tract dysfunction; however, conventional urodynamics results often poorly correlate to patient symptoms because of the nonphysiological nature of the test. The GleanTM Urodynamics System is a novel, wireless, catheter-free ambulatory urodynamics system that allows clinicians to study lower urinary tract dysfunction in a more physiological manner. The objective of this study was to evaluate the feasibility, efficacy, and safety of the Glean Urodynamics System for use in clinic.
Materials and Methods:
This prospective, single-arm, multicenter interventional trial enrolled adults with lower urinary tract dysfunction. Participants underwent conventional urodynamics per standard of care followed by ambulatory urodynamics using the intravesical Glean bladder sensor. After the removal of the Glean sensor, participants completed comfort and preference questionnaires. Participants were followed up within 7–14 days to assess the incidence of adverse events.
Results:
Thirty-eight participants were enrolled. Glean sensor insertion was attempted in 33 participants and successful in 32 (97.0%). The median sensor insertion time was 33.62 seconds (range: 12.32–256). The Glean Urodynamics System recorded vesical pressure, and all 32 participants were able to void with the sensor indwelling. Median sensor removal time was 5.71 seconds (range: 0.79–22.15). There were 14 adverse events in 12 participants, of which 7 events (in 7 participants) were attributed to the Glean Urodynamics System. There were no serious adverse events. Overall participant feedback on the Glean Urodynamics System was positive, and clinicians reported it was easy to use.
Conclusions:
The Glean Urodynamics System is a feasible, efficacious, safe, and well-tolerated device that enables ambulatory urodynamics with physiological bladder filling and without catheters impeding lower urinary tract function.
Introduction
Urodynamics (UDS) is used to diagnose lower urinary tract dysfunction (LUTD). Conventional UDS involves inserting catheters to empty and fill the bladder, allowing for the measurement of storage dysfunction and voiding parameters. The physiological relevance of conventional UDS is often called into question because of the rapid, retrograde filling of the bladder and voiding with catheters in place (including the potential inability to void), leading to false or inconclusive diagnoses. 1 –3 It is also uncomfortable and distressing for patients. 4 –7
Ambulatory UDS has been shown to significantly improve diagnostic capability and patient comfort 2,8 –10 ; however, it still requires catheters, which can impact patient comfort and impede voiding. 11,12 A physiologically relevant assessment of bladder function via catheter-free ambulatory UDS could avoid the issues of current UDS and provide an effective tool for accurate diagnostics and improved patient outcomes.
The GleanTM Urodynamics System (Bright Uro Inc., Irvine, CA) is a novel, wireless, catheter-free ambulatory UDS system (Fig. 1), which allows clinicians to assess LUTD through continuous bladder pressure monitoring during natural bladder filling and voiding. The Modern Urodynamics System Efficacy (MUSE) study evaluated the feasibility, efficacy, and safety of collecting ambulatory UDS data with the Glean Urodynamics System in males and females with LUTD.

Glean Urodynamics System.
Materials and Methods
The MUSE study was a prospective, single-arm, open-label, multicenter, interventional feasibility study conducted at five US-based urology clinics. This study was approved by the WCG Institutional Review Board and completed in accordance with the Declaration of Helsinki. The study is registered on clinicaltrials.gov (NCT05959655).
Study participants
Adult patients with LUTD who were recommended or scheduled for conventional UDS were eligible. Patients were excluded if they were pregnant (current or within the past 6 months) or breastfeeding; intended to become pregnant during the study; had an active urinary tract infection (UTI) 13 ; unable to tolerate 18 French catheterization; unable to provide informed consent; and those who, at the clinician’s determination, would not be appropriate for the study.
Glean Urodynamics System
The Glean Urodynamics System is composed of a biocompatible bladder sensor and insertion tool, uroflowmeter, and software applications (Fig. 1). It is designed to quantify pressure and flow characteristics of the lower urinary tract and can be used to perform standard UDS tests (e.g., uroflowmetry, cystometrogram, urethral pressure profile, and micturition studies). The sensor (Fig. 2; 15 French) consists of a coudé tip (18 French), removal string, and pressure-measuring circuit board housed in a flexible, curled, medical-grade silicone tube designed to be delivered into the bladder via the urethra using an insertion tool (20 French). The sensor straightens when it is loaded into the insertion tool and, once deployed in the bladder using the advancing rod, returns to a curled configuration. The sensor measures and then transmits vesical pressure data via Bluetooth once it is withdrawn from the body. The single-use sensor and sex-specific insertion tool are provided in sterile packaging. The Glean uroflowmeter is a Bluetooth-connected scale that measures voided volume and flow rate. The Glean UDS data are viewed using a web-based application that displays the vesical pressure tracing, voids (i.e., flow rate and volume), and annotated events (e.g., Valsalva).

Glean bladder sensor.
Study procedures
Participants provided informed consent prior to being screened for eligibility and enrolled. A detailed medical history and physical examination were completed, including baseline uroflowmetry and postvoid residual (PVR) measurement. A urine sample was collected for urinalysis and urine culture. Those with positive urinalysis and exhibiting symptoms were treated for UTI at the discretion of the clinician following standard of care and, upon resolution, were allowed to continue with the study.
Prior to the UDS examination, antibiotic prophylaxis was given at the discretion of the clinician following the standard of care. All participants first underwent conventional UDS and any clinically relevant evaluations (e.g., fluoroscopy, cystoscopy, electromyography). Once these tests were completed and if the participant’s PVR at the end of conventional UDS was <150 mL, 50–150 mL of solution used during conventional UDS was optionally instilled into the bladder at the investigator’s discretion prior to removing the urethral catheter to support confirmation of sensor placement. All instrumentation was then removed, and the participant was prepared for aseptic insertion of the Glean sensor.
Clinicians trained in UDS performed the Glean UDS procedures. Prior to the insertion of the Glean device, the clinician instilled lubricant with or without lidocaine in the urethra. The lubricated sterile insertion sheath with the sensor loaded was then inserted into the urethra and advanced into the bladder, allowing sensor deployment via the advancing rod. The flow of urine through the sheath confirmed sensor placement in the bladder. The insertion sheath was then removed, and the sensor removal string was secured to the body.
Participants were guided through a series of maneuvers meant to simulate activities of daily living (e.g., Valsalva) and subsequently released for in-clinic ambulatory monitoring until they reported a strong desire to void (SDV). Participants voided into a Glean or Minze (Minze Health, Antwerp, Belgium) uroflowmeter, and a PVR was measured. The Glean sensor was then removed transurethrally by pulling on the removal string.
At the end of the visit, participants completed a questionnaire regarding their experience including preference, Net Promoter Score (NPS; higher scores indicate greater satisfaction), level of agreement with predetermined statements (7-point Likert scale with higher scores indicating stronger agreement), and visual analog scales for comfort (5-point Likert scale with higher scores indicating greater comfort). Participants were contacted by research personnel 7–14 days after the UDS examination to record any adverse events.
Clinicians reviewed the Glean Urodynamics System data using the Glean web application. Clinicians also completed point-of-care surveys on the ease of Glean sensor insertion and removal (5-point Likert scale with higher scores indicating greater ease) as well as a questionnaire regarding their overall experience using the Glean Urodynamics System compared with conventional UDS.
Outcome definitions and statistical analysis
Primary efficacy outcomes were the percentage of insertion and removal attempts deemed successful by the clinician. The primary safety outcome was the percentage of participants experiencing a device-related serious adverse event. Secondary endpoints included participant-reported outcomes (e.g., comfort) and successful collection of data from Glean devices. Descriptive statistics were used to summarize the results. Post hoc analyses included Wilcoxon signed ranked tests with continuity correction, McNemar’s test, and exact two-sided binomial tests to compare outcomes between Glean and conventional UDS. Outcomes were also analyzed by sex.
Results
Thirty-eight participants were enrolled in this study (Fig. 3), and 32 (84.2%) underwent Glean UDS. Six participants did not undergo Glean UDS because of a device deficiency (n = 1), early withdrawal because of health issues not related to study (n = 1), investigator-initiated withdrawal because of the participant canceling visits as a result of health issues not related to study (n = 1), the absence of insurance coverage because of study participation (n = 1), being unable to insert the device (n = 1), and an early withdrawal with no reason reported (n = 1). Three additional participants were lost to follow-up after the UDS visit, and one participant did not wish to proceed with the study during ambulatory UDS. Therefore, 28 (73.7%) of the 38 participants enrolled completed the study.
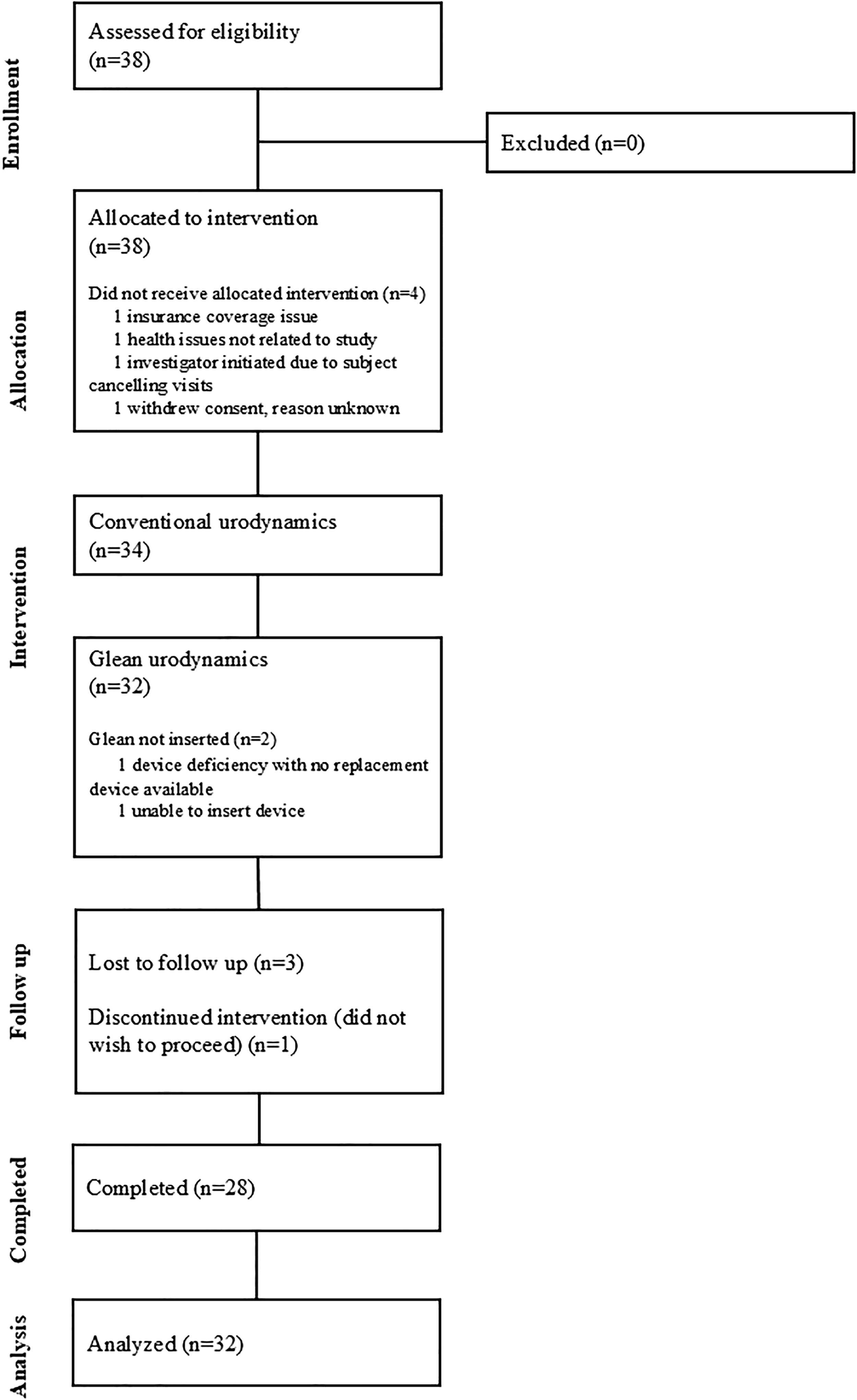
Participant flow diagram.
The baseline characteristics of the participants are found in Table 1. Among those who underwent Glean UDS (n = 32), the median age of the participants was 66.5 years (range: 25–79), approximately half were male (53.1%), and 18.7% were non-White. The most prevalent LUTD conditions were benign prostatic hyperplasia (BPH; among males; 82.4%), overactive bladder (31.3%), and urinary incontinence (59.4%). There were no statistically significant differences, except for age, between those who did and did not experience Glean UDS.
Baseline Characteristics
AI = American Indian; AN = Alaskan Native; BPH = benign prostatic hyperplasia; CKD = chronic kidney disease; MUI = mixed urinary incontinence; NGB = neurogenic bladder; OAB = overactive bladder; PI = Pacific Islander; SUI = stress urinary incontinence; UDS = urodynamics; UUI = urge urinary incontinence.
Efficacy
Of the 34 participants who underwent preparation for Glean UDS, 33 experienced an insertion attempt. One participant was unable to undergo insertion because of a mechanical failure of the device prior to the insertion. Thirty-two (97.0%) of the 33 participants had successful insertions. The median insertion time was 33.62 seconds (range: 12.32–256). Eighty-one percent of sensor insertions were rated as easy or very easy by clinicians (Table 2).
Clinician-Rated Ease of Insertion and Removal of the Glean Sensor
The median Glean ambulatory time was 41.5 minutes (range: 12–96) from the time of confirmed placement to removal. All 32 (100%) participants were able to void with the Glean sensor indwelling in contrast to 27 (84.4%) during conventional UDS (p = 0.0625). There were statistically significant differences in uroflowmetric parameters during Glean UDS compared with conventional UDS (Table 3), but participants reported similar voiding experiences (Fig. 4). The Glean Urodynamics System recorded vesical pressure data during ambulatory monitoring in all 32 participants.
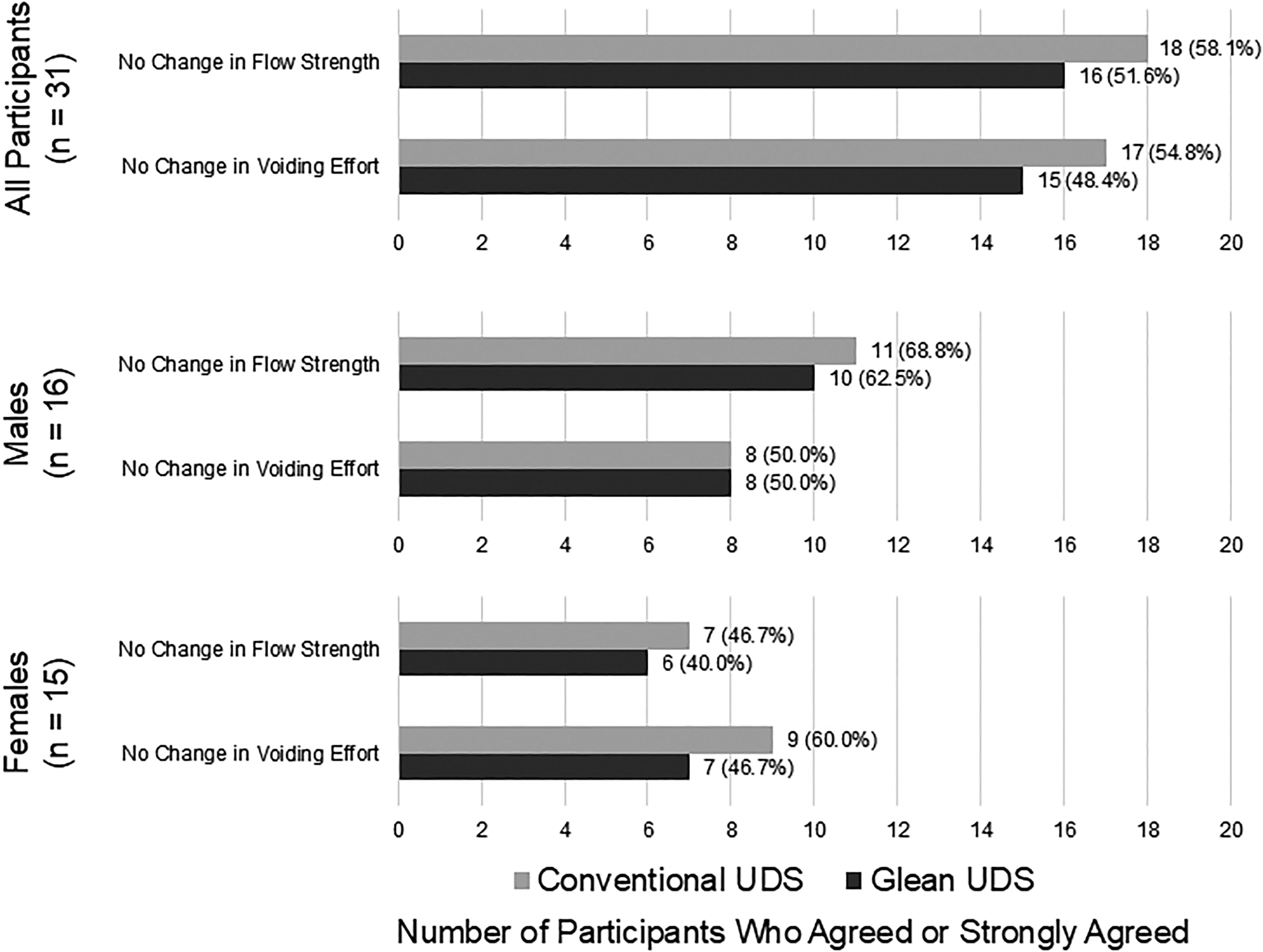
Participant experience during voiding. UDS = urodynamics.
Comparative Evaluation of the Uroflowmetric Parameters
Among participants who had complete uroflowmetric data for both conventional and Glean UDS (n = 22).
Free uroflowmetry immediately prior to conventional UDS examination.
Statistically significantly different from Glean UDS (p < 0.05).
Statistically significantly different from conventional UDS (p < 0.05).
cUDS = conventional urodynamics; IQR = interquartile range; PVR = postvoid residual; Qmax = maximum flow rate.
The removal of the Glean sensor was successful in 100% of those who had a successful insertion. The median removal time was 5.71 seconds (range: 0.79–22.15). Ninety-seven percent of sensor removals were rated as easy or very easy by clinicians (Table 2).
Safety
There were no serious adverse events or deaths. Fourteen adverse events were reported among 12 participants (Table 4): nine gross hematuria events (in nine participants), two dysuria events (in two participants), two genitourinary pain/discomfort events (in two participants), and one asymptomatic bacteriuria event (in one participant). Seven of the adverse events (in seven participants) were attributed to the Glean Urodynamics System including gross hematuria (four events), genitourinary pain/discomfort (two events), and dysuria (one event). All adverse events were rated mild and self-resolved within a few days. The remaining adverse events (Table 4) were attributed to conventional UDS (four events), other instrumentation (one event), or preexisting health conditions (two events).
Adverse Events
Tolerability
Thirty-one (96.9%) participants who experienced Glean UDS completed the survey. Overall, more participants rated Glean UDS as comfortable or extremely comfortable compared with the number of participants who rated conventional UDS as such (41.9% vs 38.7%; Table 5). Fewer participants, however, rated the insertion and removal of the Glean sensor as comfortable or extremely comfortable compared with those who rated the insertion and removal of the conventional UDS catheters as such (Table 5). There were no statistically significant differences in comfort ratings when stratified by sex. More participants also agreed that the filling of their bladder (i.e., physiological vs retrograde) was comfortable (71.0% vs 45.2%, p < 0.05), their symptoms were reproduced (48.4% vs 38.7%, p < 0.05), and they were able to be relaxed (48.4% vs 45.2%, p < 0.05) during Glean UDS than during conventional UDS (Fig. 5).
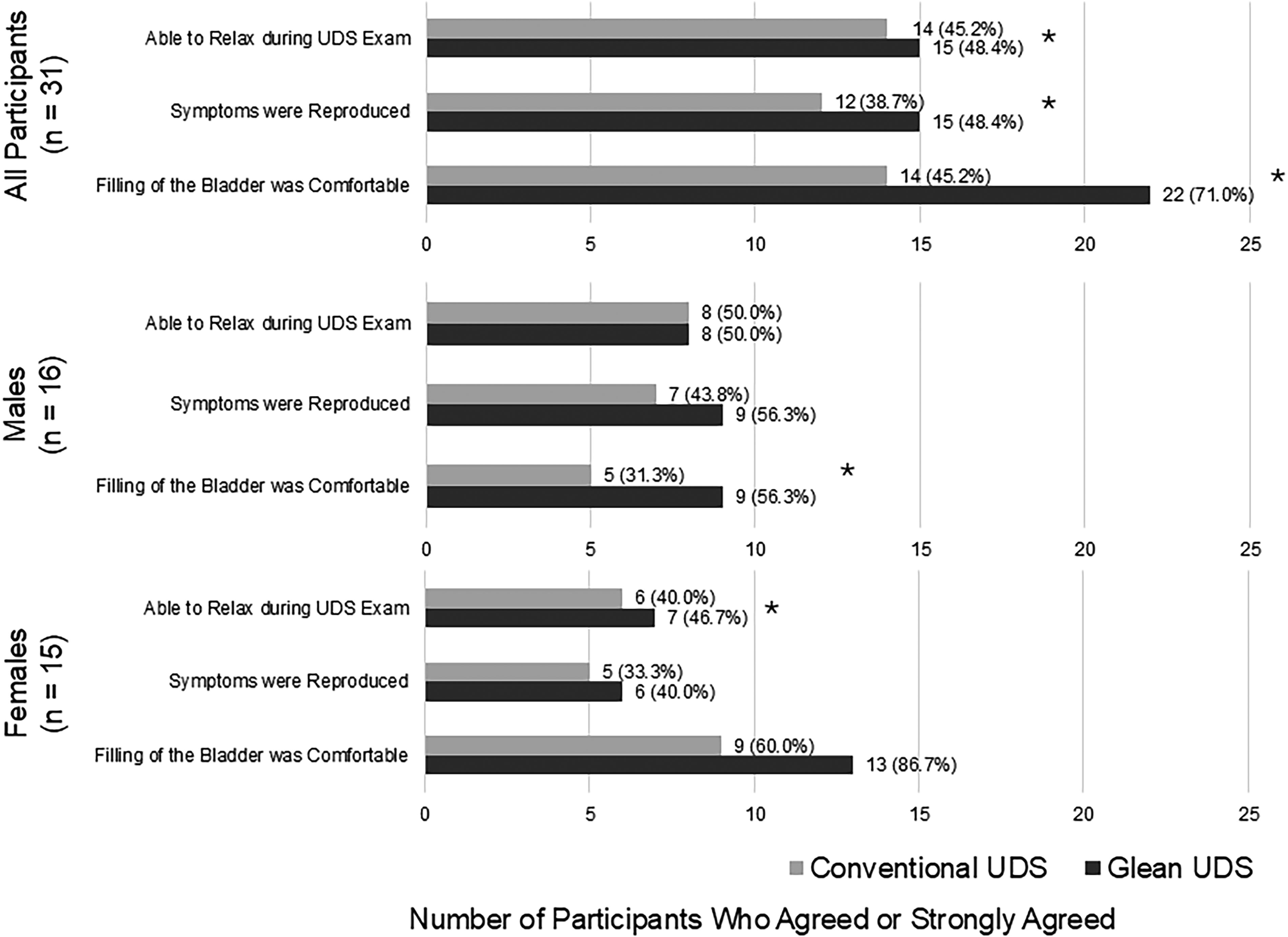
Participant experience during Glean and conventional UDS. *Statistically significant difference between groups (p < 0.05).
Participant-Rated Comfort of the Urodynamics Procedures
cUDS = conventional UDS.
The NPS for Glean UDS was 22.6, whereas the NPS for conventional UDS was −19.4; males rated both examinations negatively (0.0 vs −43.8, respectively), whereas females rated them more highly (46.7 vs 6.7, respectively). Nearly two-thirds (64.5%) of participants who responded to the survey would prefer Glean over conventional UDS if they had to undergo UDS testing again (Fig. 6).
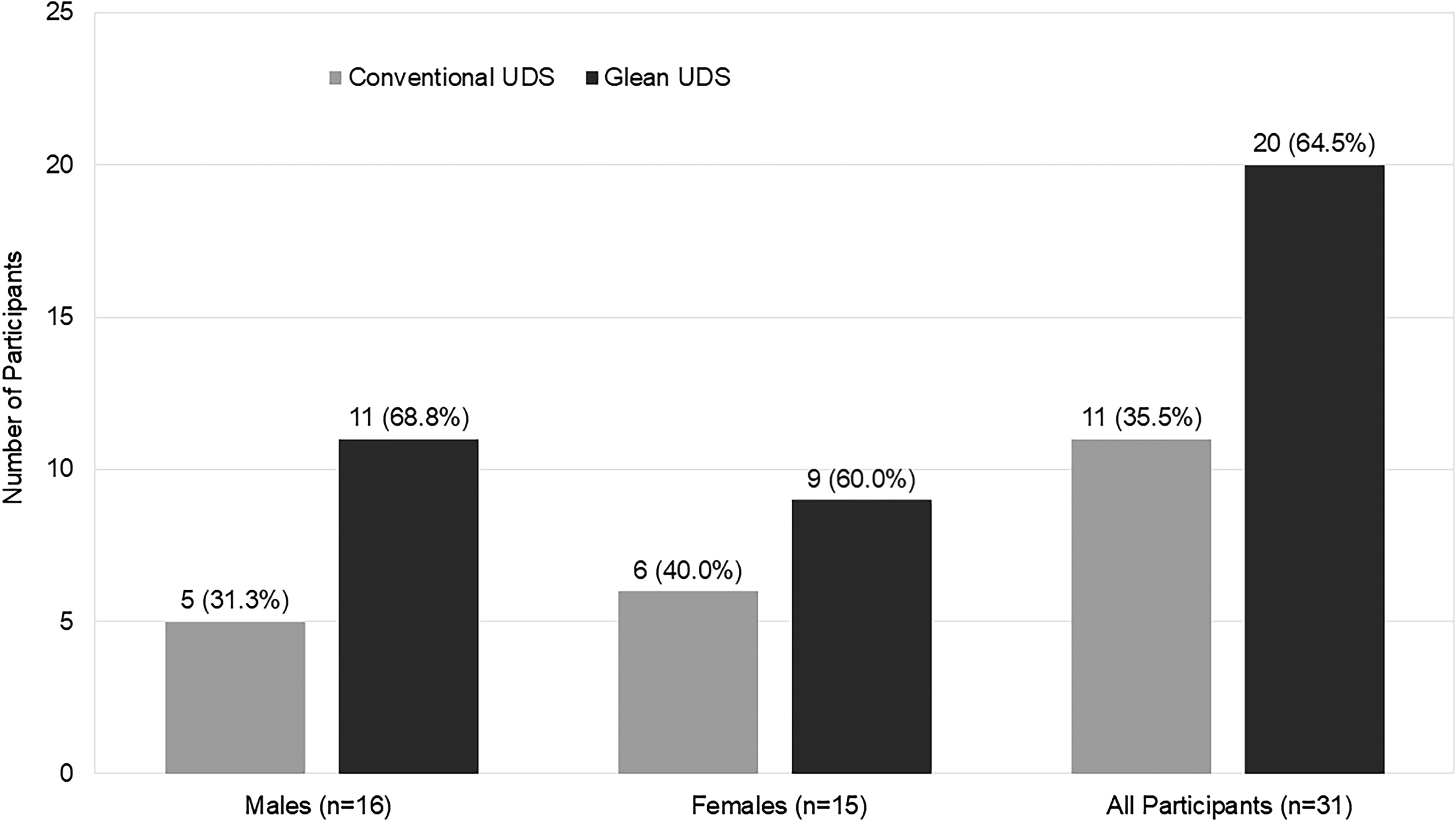
Participant preference.
Discussion
This study reports the initial findings of the Glean Urodynamics System in clinic to evaluate LUTD in male and female patients, showing it provided a clinically feasible, safe, tolerable, and favorable approach for UDS. The Glean Urodynamics System was effectively utilized in the clinic, achieving successful sensor insertion in 97% of participants and successful removal in all. The one failed insertion attempt was in a participant whose subsequent cystoscopy identified bladder neck contracture, which likely prevented the advancement of the Glean device; the placement of the conventional UDS catheters was also challenging in this participant. The Glean sensor may have limited effectiveness in patients with anatomical abnormalities. Otherwise, most sensor insertions and removals were rated as easy or very easy by clinicians furthering its in-clinic feasibility in a population with diverse urological conditions.
The Glean Urodynamics System enabled natural filling and voiding for ambulatory UDS. All participants voided with the sensor indwelling, while five of these participants could not void with conventional UDS catheters in place, improving diagnostic capabilities by ensuring successful voiding. Although the study was not designed to compare Glean and conventional UDS, higher voided volumes and PVRs were observed on conventional UDS likely because of artificially instilled larger volumes during filling compared with physiological filling in Glean. Urethral catheters may also be inhibiting emptying during conventional UDS which has been documented in the literature. 11,12 Similarly, the lower Qmax observed with Glean may be attributed to lower voided volumes, which did not meet standard thresholds for accuracy. 14 Since Glean was the second of two consecutive UDS examinations, participants may have prematurely reported an SDV to shorten their visit or alleviate potential discomfort from the indwelling Glean sensor. When those who voided less than 50 mL during Glean were excluded from analyses of uroflowmetric parameters, Qmax was no longer significantly different from conventional UDS (data not shown). Glean uroflowmetric values may be more reflective of the participant’s natural voiding patterns. Such variances in the urodynamic parameters may show the differences between the natural and artificial approaches to these UDS studies, but more robust data are needed to evaluate physiological bladder storage and voiding.
Few harms were attributed to the Glean Urodynamics System, which aligns with the UroMonitor study that reported no peri-procedural complications in females using a similarly designed sensor. 15 Most adverse events attributed to the Glean Urodynamics System were reported among males (four gross hematuria and two genitourinary pain/discomfort events) where their existing urological condition (i.e., history of BPH) was likely a contributing factor further aggravated by multiple instrumentation. The only adverse event attributed to Glean in females was dysuria (one event). The event rates were similar to those attributed to conventional UDS in this study (four gross hematuria events in four males) and to incidence rates reported in the conventional UDS literature (1.3%–10.3% for gross hematuria, 17.1%–63.3% for dysuria, and 20.7%–54.1% for genitourinary pain/discomfort). 16 –20 Harms of the Glean Urodynamics System may have been overestimated because of the compounding effects of two consecutive UDS procedures. Furthermore, the study evaluated for, but did not observe, other potential harms, such as allergic reactions, removal string entanglement, and accidental removal. Real-world evidence is needed to evaluate Glean’s short- and long-term safety, tolerability, and other potential risks without other UDS procedures. Overall, the Glean Urodynamics System was safe and well tolerated by participants. Participant preference and the NPS showed they would undergo Glean again for UDS testing and recommend it to a friend, which was not the case for conventional UDS.
There are several limitations to this study. This feasibility study was neither designed nor powered for any outcome or any comparisons between conventional and Glean UDS. The study’s small sample size limits validity, generalizability, and applicability. Further, the order of procedures (i.e., conventional UDS occurring before Glean UDS) and the variability of standard-of-care conventional UDS procedures performed at each clinic may have influenced the results. For example, the variable use of single-dose prophylactic antibiotics (used in 55.9% of participants) may have mitigated infection-related adverse events. 14 In addition, the last procedure experienced was the removal of the Glean sensor after which participants were asked to rate their entire UDS experience, and thus recall bias may have influenced participant ratings. Future large-scale randomized trials of the Glean Urodynamics System are needed to further evaluate its diagnostic performance and safety compared with conventional UDS.
Conclusions
The Glean Urodynamics System enables wireless, catheter-free, ambulatory monitoring of bladder pressure that is safe and well tolerated by patients. In addition, it did not appear to interfere with the patient’s void, in contrast to the conventional UDS procedures.
Footnotes
Acknowledgments
The authors thank Rich Cohn, PhD, for statistical support.
Data from this study were previously presented at the following conferences:
Society of Urodynamics, Female Pelvic Medicine, and Urogenital Reconstruction (SUFU) Winter Meeting, Fort Lauderdale, FL, February 20–24, 2024.
Society of Urodynamics, Female Pelvic Medicine, and Urogenital Reconstruction (SUFU) Winter Meeting, Rancho Mirage, CA, February 26–March 1, 2025.
American Urological Association Annual Meeting, Las Vegas, NV, April 26–29, 2025.
Authors’ Contributions
J.K., K.X., T.C.S., T.V., S.V., H.Z., A.A.-H., and N.S.: Investigation and writing—original draft.
Data Availability Statement
Data are not publicly available.
Author Disclosure Statement
K.X. and T.C.S. have financial interests in Bright Uro.
Funding Information
This study received funding from Bright Uro, Inc.
