Abstract
Introduction:
The goal of this study is to report the updated 3-year safety and efficacy outcomes of the intervention arm of the ROBUST III randomized controlled trial, comparing the Optilume® drug-coated balloon (DCB) with standard endoscopic management of recurrent male anterior urethral stricture.
Methods:
Eligible patients included adult men with recurrent anterior urethral stricture ≤3 cm in length and ≤12 Fr in diameter, International Prostate Symptom Score (IPSS) ≥11, and peak flow rate (Qmax) <15 mL/s. Patients were randomized to treatment with the Optilume DCB or standard-of-care endoscopic management. Primary endpoints evaluated over the 3-year follow-up period included freedom from reintervention and alterations in IPSS, Qmax, and postvoid residual (PVR). The primary safety endpoint was freedom from serious procedure- or device-related adverse events (AEs).
Results:
Descriptive statistics of the intervention cohort have been published previously. The treatment arm maintained a high percentage of patients free from repeat intervention (71%), nearly equal to the 2-year results and three times higher than that observed in the control group at the 1-year mark. Clinically significant subgroups, including those with history of repeat endoscopic dilations (≥5 prior dilations) and longer stricture lengths (≥2 cm), did not demonstrate any significant differences in observed metrics, including IPSS score, Qmax, or PVR. In the crossover cohort of patients who experienced treatment failure with standard endoscopic management and opted for DCB treatment, the Kaplan–Meier curve for freedom from reintervention closely resembles that of the original DCB cohort. Treatment-related AEs were rare and generally self-limited (hematuria, dysuria, and urinary tract infection).
Conclusion:
The Optilume DCB continues to achieve significant improvements in symptoms and reintervention rates through 3 years posttreatment and represents a viable endoscopic alternative with durable results and a low-risk safety profile for the management of recurrent anterior urethral strictures ≤3 cm in length.
Introduction
The Optilume® drug-coated balloon (DCB) (Urotronic Inc./Laborie Medical Technologies, Plymouth, MN, USA) utilizes a dual mechanism of action for treating urethral stricture disease (USD). It combines mechanical urethral dilation with targeted, circumferential delivery of paclitaxel, a chemotherapy agent with antifibrotic and antiproliferative properties. Previous phase I/II trials have demonstrated success, as defined by improvement in the International Prostate Symptom Score (IPSS) questionnaire, an 8-question screening tool used to assess and track obstructive urinary symptoms. Results from the ROBUST I trial, which has now completed its 5-year duration, showed that 63% of patients treated with Optilume experienced an IPSS score improvement of ≥30% without retreatment, and freedom from repeat intervention was maintained through 5 years at 71.7%. Additionally, no serious adverse events (SAE) were reported, and there was no impact on erectile function. 1 –4
ROBUST III is a randomized controlled trial comparing the DCB with standard endoscopic management. The control group was followed for 12 months, whereas the intervention group will be followed for 5 years. The 12- and 24-month results have demonstrated a durable response to therapy. At 24 months, 61% of patients experienced an IPSS improvement of at least 30% without the need for repeat intervention. Additionally, the freedom from repeat intervention rate was 77.8% in the treatment group at 24 months compared with 23.6% in the control group at 12 months. 5,6 We now report the updated 3-year safety and efficacy outcomes of the DCB cohort of the ROBUST III trial.
Materials and Methods
ROBUST III is a prospective, multicenter, single-blind, randomized controlled study designed to evaluate the safety and efficacy of the Optilume DCB for treating recurrent male anterior USD. The study was conducted at 22 sites in the United States and Canada (NCT03499964) following the guidelines of the Declaration of Helsinki. Protocol approval was obtained from the institutional review boards at each participating site, and written informed consent was acquired from all participants before their involvement. The study was overseen by an independent clinical events committee and data monitoring committee for adjudicating the seriousness and relatedness of all potential device- and/or procedure-related events and evaluating the overall study outcomes, respectively.
Study design and participants
Adult males meeting the following criteria were considered eligible: anterior urethral strictures ≤12 Fr and ≤3 cm in length, a history of ≥2 prior endoscopic treatments, an IPSS of ≥11, and a peak urinary flow rate (Qmax) of <15 mL per second. Exclusion criteria included prior urethroplasty, hypospadias repair, lichen sclerosus, or unresolved confounding etiologies such as bladder neck contracture, neurogenic bladder, or benign prostatic hyperplasia. All participants provided written informed consent.
Participants were randomly assigned in a 2:1 ratio to receive treatment with the Optilume DCB or undergo standard endoscopic management. Follow-up assessments were conducted postprocedure at Foley removal (2–5 days), 30 days, 3 months, 6 months, and yearly thereafter. Randomized participants remained unaware of their treatment group assignment until the 6-month mark, after which the study entered the open-label phase. For the DCB group, annual follow-ups are ongoing up to 5 years. The standard-of-care (SOC) group completed the required study follow-up up to 1 year, with no further follow-up required beyond this point. However, after the 6-month mark, any patients in the SOC group who failed treatment were allowed to cross over to the DCB and were subsequently followed.
Procedure
Prior to treatment with the Optilume DCB, predilation of the stricture was performed using an uncoated balloon or direct visualization internal urethrotomy (DVIU). The main requirement for predilation was to achieve a minimum caliber of 20 Fr prior to use of the DCB; however, the method of dilation was site-dependent and not standardized. To prevent multiple doses of paclitaxel, no patients underwent primary dilation with the DCB if the stricture was not sufficiently dilated on first attempt. Optilume DCBs were available in diameters ranging from 18 to 36 Fr and lengths of 3 and 5 cm. Balloon size selection was based on the diameter of the lumen and stricture length to ensure a 0.5 to 1 cm overlap of normal tissue on both ends of the stricture. The balloon was then inflated to its rated burst pressure for a minimum of 5 minutes, followed by the insertion of a 12–14 Fr Foley catheter.
Summary of endpoints
The efficacy and safety analyses encompass all participants who were randomized to the DCB group. Anatomical success was defined as achieving a urethral lumen of 14 Fr or larger through calibration or cystoscopy at the 6-month mark, as previously reported. Primary endpoints evaluated over the 3-year follow-up period included freedom from reintervention and alterations in IPSS, Qmax, and postvoid residual (PVR). The primary safety endpoint measured freedom from SAEs related to the procedure or device.
Statistics
Descriptive statistics were used to summarize results. Participants initially treated with Optilume who required alternative therapy were considered treatment failures and discontinued from the study. To accurately reflect those participants who received alternative therapy in the efficacy data (IPSS, Qmax, and PVR), timepoints after study exit because of treatment failure are imputed as failures (i.e., the worst observed value is assigned for visits after the repeat intervention) and included in the denominator. Comparisons to baseline were evaluated with a paired t-test, whereas comparison between subgroups was evaluated with an unpaired t-test.
The required sample size was based on the randomized portion of the study. There were no additional sample size requirements associated with long-term follow-up. Significance was evaluated at the 0.05 level with no adjustments for multiplicity. Statistical analyses were performed using SAS 9.4 (SAS Institute, Cary, NC, USA).
Results
A total of 127 patients were randomized into the treatment arm (n = 79) and control arm (n = 48). The average age of men undergoing treatment with the DCB was 58.7 years old (range: 25–87), with a history of 3.2 prior endoscopic treatments (ranging from 2 to 10) at the time of enrollment. Notably, around 15% of men (12/79) in the treatment arm had undergone ≥5 dilations. The majority of strictures were located in the bulbar urethra (89.9%, 71/79) with an average length of 1.6 cm. The etiology of the strictures, previously documented, was comparable between the control and DCB groups, with idiopathic strictures being the most prevalent, followed by iatrogenic and traumatic causes. Following the 6-month visit, 75 participants entered the open-label phase. At the 3-year follow-up, 20 patients experienced treatment failures, and 2 participants had non-study-related deaths—one from intestinal infarction and another from lung cancer (Fig. 1).
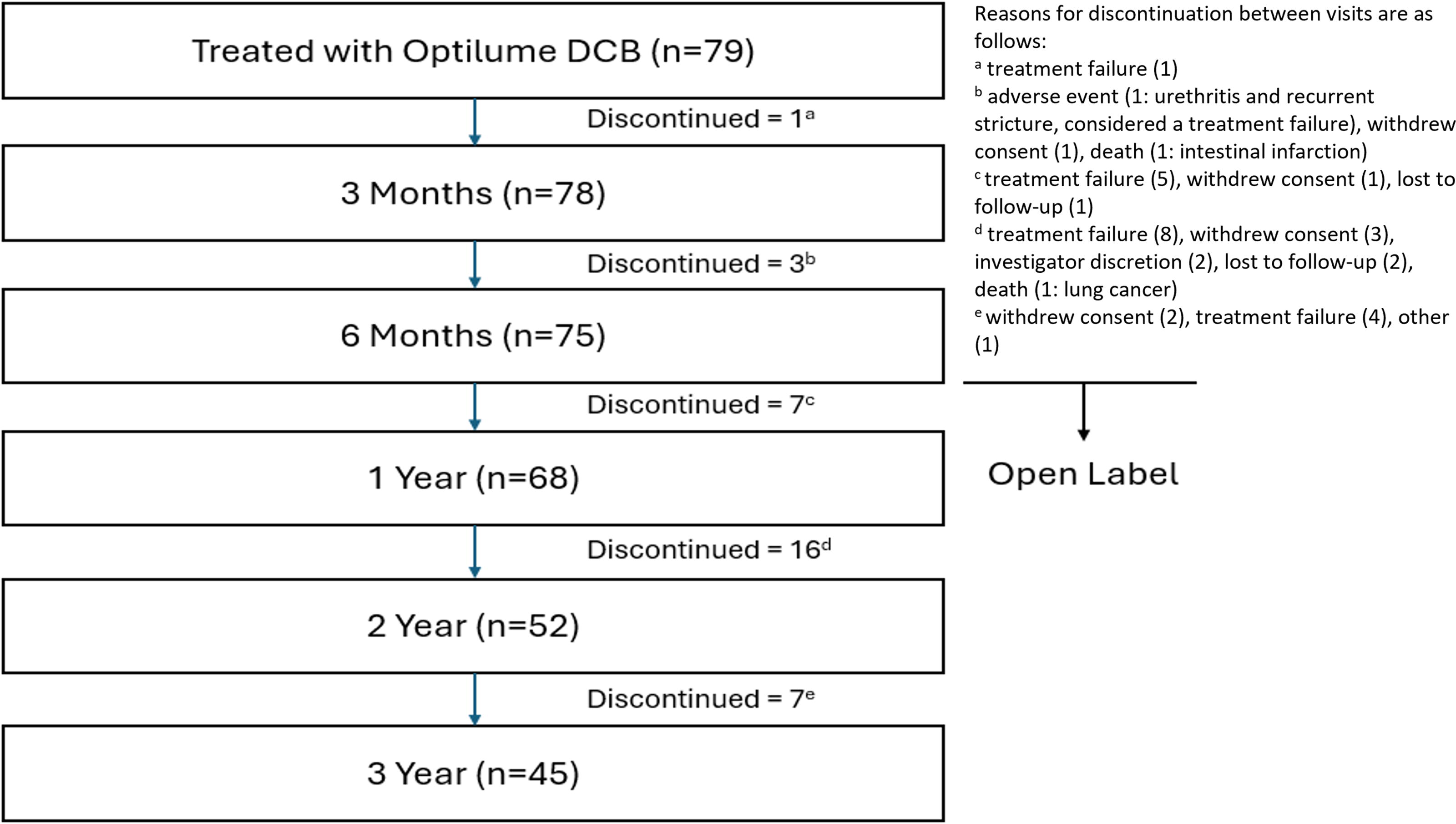
Participant timeline.
Efficacy
The average IPSS improved from 22.0 at baseline to 9.0 at 1 year, 10.1 at 2 years, and 11.6 at 3 years, showing a steady annual increase at a relatively stable rate. Comparatively, the average Qmax improved from 7.6 mL at baseline to 15.5 mL/s at 1 year, 12.6 mL/s at 2 years, and 10.6 mL/s at 3 years (Table 1). At 3 years, 48% (27/56) of participants in the treatment arm experienced an IPSS improvement of at least 30% without repeat intervention. The Kaplan–Meier estimate for freedom from repeat intervention was significantly greater in the DCB group at 3 years (71.9%) compared with the control group at 1 year (23.6%), yielding a 50% difference between groups (Fig. 2). Of the 48 control group participants, 32 experienced stricture recurrence and opted to cross over into the DCB treatment arm. This subgroup’s outcome data can be found in Table 2.
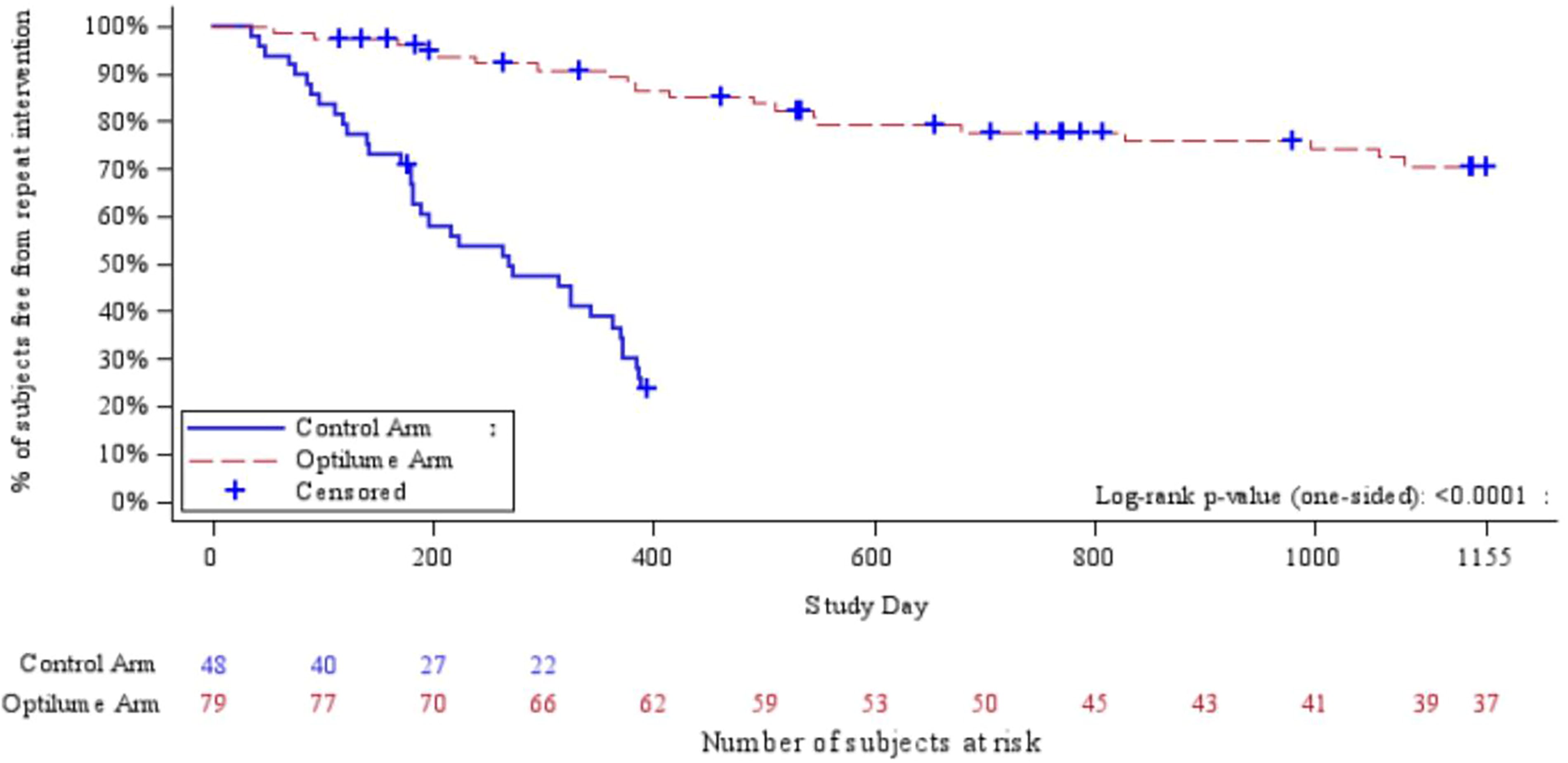
Kaplan–Meier curve—freedom from reintervention (Optilume® vs control) at 3-year follow-up.
Summary of Outcome Measures Through 3 Years for the Optilume® DCB Group
DCB = drug-coated balloon; IPSS = International Prostate Symptom Score; SD = standard deviation.
Summary of Outcome Measures Through 3 Years for Crossover Cohort
Safety
The AEs most commonly associated with treatment included postprocedural hematuria (11.4%), dysuria (7.6%), and urinary tract infection (6.3%). SAEs attributed to treatment were rare, with one case of aspiration/aspiration pneumonia and one case of urinary tract infection occurring in each treatment arm. No late-onset treatment-related SAEs or AEs were observed throughout the study.
Discussion
USD affects approximately 0.6% of males over their lifetimes, presenting significant challenges in both diagnosis and management. 7 Historically, treatments such as DVIU and mechanical dilation have been widely employed, albeit with the notable shortcoming that recurrence rates after these procedures remain unacceptably high. 8,9 A recent review of patients diagnosed with USD between 2008 and 2016 shows a shift in treatment trends, with a decline in repeat endoscopic procedures and an increase in urethroplasty for managing the condition. 10
The 3-year findings from the ROBUST III trial demonstrate that the Optilume DCB is a viable endoscopic alternative for the management of recurrent anterior urethral strictures ≤3 cm in length, offering durable results and a low-risk safety profile. This trial meticulously evaluated multiple success metrics to assess its efficacy. Notably, when comparing the 1-year outcomes of the control arm with the 3-year outcomes in the DCB arm, Optilume continues to outperform standard endoscopic management in multiple metrics, including freedom from retreatment and improvements in IPSS, Qmax, and PVR.
Recurrence vs retreatment
Given the susceptibility of urethral stricture tissue to rescarring from stretch trauma and mucosal tearing, even with paclitaxel administration, it is plausible that some stricture recurrence may occur over time following dilation. 11 This can be observed in the steady decline in Qmax over the course of 3 years (7.6 mL/s at baseline, compared with 15.5 mL/s at 1 year and 10.6 mL/s at 3 years). The average IPSS has also demonstrated an upward trend, though at a much slower rate. The average IPSS increased from 7.4 at 3 months posttreatment to 10.1 at 2 years and 11.6 at 3 years. Similarly, between the 2- and 3-year follow-ups, the percent of participants in the treatment arm with an IPSS improvement of at least 30% declined from 61.3% to 48.2%. In comparison with the baseline average IPSS of 22, this demonstrates a more sustained improvement in comparison with the average Qmax values in the same patients.
However, despite these trends, 71.9% of patients remain free from repeat intervention at 3 years, a rate consistent with the 2-year results and three times higher than that observed in the control group at 1 year. The sustained symptom improvement appears sufficient to deter patients from opting for additional procedures at the 3-year mark, even in the context of decreasing Qmax values and increasing PVRs. These are encouraging statistics to utilize when counseling patients seeking a minimally invasive yet durable option, particularly those who are not ideal candidates for urethroplasty or who prefer to avoid more invasive procedures while still achieving meaningful and durable symptom relief. Looking ahead, 4- and 5-year follow-up data will be critical in determining whether more patients ultimately require retreatment as symptoms or flow rate continue to evolve over time.
Clinically significant subgroups
Certain subgroups in this study are more prone to stricture recurrence after traditional endoscopic management. These high-risk groups include a history of repeat endoscopic dilations, longer strictures (≥2 cm), or previous pelvic radiation. 12,13 Within the DCB treatment arm, 15.1% (12/79) of patients had a history of ≥5 prior dilations and 46.1% (36/78) had a stricture length ≥2 cm. Despite these risk factors, these high-risk subgroups demonstrated sustained improvements in IPSS and Qmax over a 3-year period (Table 3). Patients with <5 prior dilations showed an average IPSS improvement from 22.4 at baseline to 15.0 at 3 years, whereas those with ≥5 prior dilations had a baseline IPSS of 20, improving to 16.6 over the same period. Similarly, patients with strictures <2 cm had an average IPSS reduction from 21.2 at baseline to 14.3 at 3 years, whereas those with strictures ≥2 cm had an improvement from 23.0 to 18.0. No statistically significant differences in outcomes were noted between these subgroups, indicating that Optilume maintains its efficacy even in patients traditionally considered at higher risk for recurrence.
Summary of Outcome Measures by Subgroup for the Optilume DCB Group
This study included a limited number of irradiated patients (6/48 in the control arm and 9/79 in the DCB arm), making it challenging to generalize the statistical findings to the broader population of patients who have received radiation. There was no significant difference in IPSS or Qmax (mL/s) between the nonirradiated and irradiated groups in the DCB arm at 3 years. Similarly, there was no significant difference in these values between the control irradiated group and the DCB irradiated group. Patients with USD and history of radiation can be a challenging population to treat, often requiring additional endoscopic treatments for stricture recurrence. 14 Therefore, it is important to consider patient-centered outcomes and to understand that the definition of success may be different. For example, for certain patients within this cohort, treatment failure at 1 year requiring retreatment may be considered an improvement over a repeat dilation every 2–3 months.
Crossover
In analyzing the pre/postcrossover Kaplan–Meier data, the focus shifts to observing how the curve closely resembles the randomized curve, albeit with a distinct approach. Here, subjects serve as their own control, allowing for a unique exploration of treatment efficacy within the same individuals over time. A total of 32 patients initially assigned to the control arm, who had undergone standard endoscopic management, required retreatment and subsequently received urethral dilation with the Optilume DCB. Figure 3 illustrates the Kaplan–Meier curve of these patients. Notably, all men who transitioned from the control arm to the DCB arm had experienced treatment failure within 1 year of the study; however, following DCB treatment, more than 75% of these same patients remained free from repeat intervention at 1 year posttreatment. This contrast highlights the durability of the DCB therapy when compared with standard endoscopic management in this subset of patients.
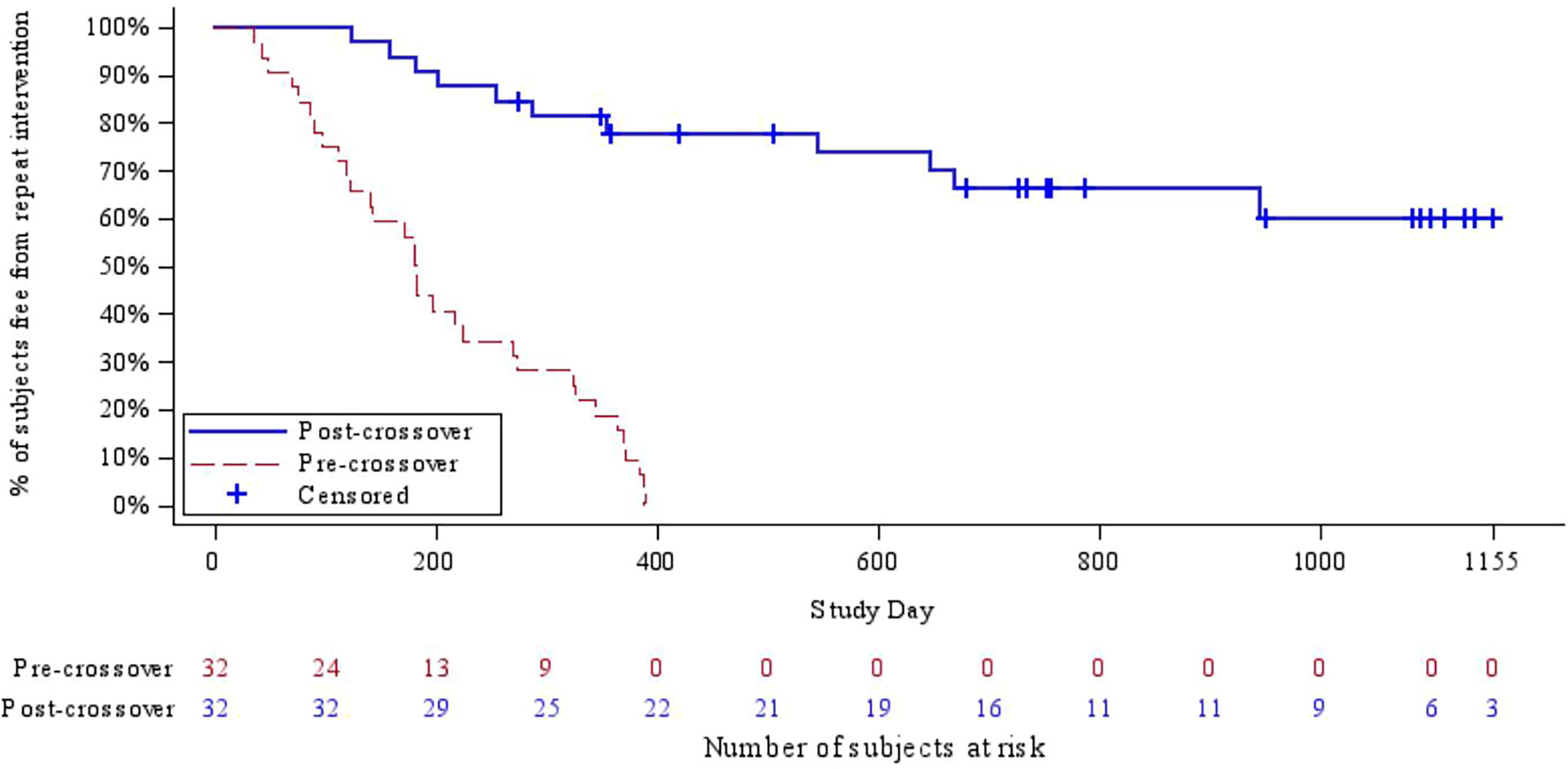
Kaplan–Meier curve—freedom from reintervention pre- and postcrossover at 3-year follow-up.
One limitation of the crossover cohort is the potential for selection bias, as patients in the control group who experienced recurrent symptoms may have been more interested in pursuing retreatment upon learning they had not been randomized to the DCB arm. Since unblinding occurred at the 6-month mark, the knowledge of available treatment options could have influenced their decision to seek treatment with the DCB. If symptoms recurred after crossover treatment with the DCB, they may not have been as eager for a second treatment with the DCB.
Future directions
Future research should explore the potential of Optilume as a first-line treatment option for USD, given its ease of use and similarities in technique to traditional balloon dilation. The patients in this study had an average of 3.2 prior endoscopic procedures, representing a failure of traditional management prior to treatment with the DCB. Additionally, although the DCB involves a higher initial cost compared with standard endoscopic management, its potential for reducing the frequency of repeat interventions suggests the possibility of significant long-term savings, which warrants further economic evaluation. Future studies could also focus on specific populations, such as those with radiation-induced urethral strictures, penile urethral strictures, and ureteral strictures, to assess the broader applicability of this treatment.
Limitations
The limitations of this trial have been previously described. Briefly, these include the single-blinding study design in which the surgeons were not blinded to the type of treatment administered, a lack of standardization in the method of predilation prior to use of the DCB, and the unblinding of the control arm patients at 6 months. In addition, not all patients initially enrolled in the DCB arm reached the completion of the 3-year endpoint. Among these patients, some withdrew their consent, whereas others were lost to follow-up or passed away because of unrelated causes. Future multi-institutional studies in the following years will serve to validate these findings, along with the long-term follow-up data from this cohort, which is intended to extend up to 5 years.
Conclusion
The Optilume DCB demonstrates sustained efficacy in the treatment of recurrent anterior urethral strictures, achieving significant and durable improvements in symptom relief and freedom from reintervention through 3 years posttreatment. Objective voiding measures such as Qmax do show a steady decline, though still improved from baseline. These findings support its role as a viable and minimally invasive alternative to traditional endoscopic management, offering a promising long-term solution for patients with recurrent urethral strictures. Further research will help refine patient selection and establish its potential as a first-line therapy for USD.
Footnotes
Authors’ Contributions
P.S.: Analysis, manuscript drafting and preparation; R.V. and J.D.: Investigation, analysis, manuscript preparation; S.P.E.: Conceptualization, methodology, investigation, manuscript preparation.
Author Disclosure Statement
No competing financial interests exist.
Dr. Virasoro, Dr. DeLong, and Dr. Elliott serve as consultants for Urotronic. The remaining authors report no competing personal or financial interests related to this work.
Funding Information
Urotronic, Inc., is the sole funder of this study. The authors had access to all the primary data and drafted all parts of the article. The sponsor had no role in the final approval of the article or the decision to publish.
