Abstract
Purpose:
We report the first multicenter, prospective, randomized noninferiority controlled trial of steerable ureteroscopic renal evacuation (SURE) for nephrolithiasis treatment.
Materials and Methods:
Candidates for laser lithotripsy ≥18 years with ≥1 renal stone ≥7 mm and 7–20 mm stone burden were randomized 1:1 SURE vs ureteroscopy (URS). SURE was performed using the CVAC Aspiration System, a novel steerable irrigation–aspiration catheter. The primary efficacy end point was noninferiority in stone-free rate (SFR) based on zero residual fragments (RFs) 30 days postprocedure on noncontrast computed tomography. Secondary end points were superiority tests, stone clearance (percent stone volume reduction), residual stone volume (RSV), SFR (no RF >2 mm), and SFR (no RF >4 mm) 30 days postprocedure.
Results:
Of the subjects, 123 were randomized and 101 qualified for efficacy analysis (SURE 46 vs URS 55). Despite randomization, baseline URS stone volume was higher (SURE 485.0 ± 432.5 mm3 vs URS 713.3 ± 558.5 mm3). The primary noninferiority end point was achieved (SURE SFR 48% vs URS SFR 49%, p = 0.027, −1.3% [90% confidence interval; −18%, 15%]). Stone clearance was significantly higher for SURE vs URS subjects (SURE 96.9 ± 5.6% vs URS 92.9 ± 11.6%, p = 0.036); RSV was significantly lower (SURE 14.3 ± 3 0.9 mm3 vs URS 70.2 ± 144.9 mm3, p = 0.012). SURE stone clearance and RSV were independent of baseline stone volume, but for URS degraded with increasing baseline stone volume. Safety was comparable between groups.
Conclusions:
SFR for SURE subjects was noninferior compared with URS subjects; stone clearance and RSV were significantly better with SURE, independent of baseline stone volume.
Introduction
Ureteroscopy (URS) is one of the most common urolithiasis treatments and has seen technology advances, including digital endoscopes and high-power lasers. 1,2 Even so, stone-free rates (SFRs) as defined by computed tomography (CT) remain 55%–73% 3,4 and can differ by stone burden, type, and location. 1,5 –8 Residual fragments (RFs) can increase the risk for stone recurrence and future stone events. 9 –15
Steerable ureteroscopic renal evacuation (SURE) using the CVAC Aspiration System, a novel irrigation and aspiration catheter, has recently been introduced. A single-center study showed that SURE removed significantly greater stone volume and achieved significantly higher stone clearance (percent stone volume reduction) vs basketing, as measured by CT on postoperative day 1. 16 A multicenter study of large stone burden patients (mean 29.3 mm) showed that SURE is safe and effective for this population, including high-risk patients. 17 In these studies, SURE significantly reduced stone volume and achieved high stone clearance independent of baseline stone volume, in contrast to previous studies of conventional URS which found that as baseline stone size increases, SFR decreases, and the number of operations needed increases. 6 –8,18
As a next step, the ASPIRE (ASPiration to Improve Renal Calculi Removal Effectiveness) prospective, randomized, controlled multicenter study was conducted. The aim was to evaluate the safety and effectiveness of SURE vs URS at 30 days through 2 years. In both groups, the goal of treatment was to achieve a “surgically stone-free” outcome (defined as no visible fragments or dust at the end of the procedure). The hypothesis was that SFR would be greater than or noninferior to that of URS with SURE and that SURE would achieve higher stone clearance and lower residual stone volume (RSV) vs URS.
Materials and Methods
Study protocol and objectives
ASPIRE complied with the Good Clinical Practice Guidelines, the International Organization for Standardization 14155, and the Declaration of Helsinki 2013 (NCT04519294; clinicaltrials.gov). Each investigator had the protocol approved by a central or local institutional review board. Written informed consent was obtained for study participation, each subject understanding the risks/benefits.
Eligible subjects were ≥18 years old, had a total renal stone burden of 7–20 mm, had ≥1 renal stone ≥7 mm, were candidates for URS with laser lithotripsy, and could comply with all study-related follow-up. CT measurements using the longest dimension from the axial, coronal, or sagittal view of each stone were used to calculate stone size and total stone burden (sum of the longest dimensions of each stone). Baseline CT scans were taken as much as 90 days before the index procedure. Subjects with body mass index (BMI) ≥45, significant comorbidities, bladder, ureteral, or kidney abnormalities, inability to accommodate a 12/14F ureteral access sheath (UAS), pregnancy, or unable to meet the protocol requirements were excluded.
Subjects underwent noncontrast CT (NCCT) imaging using 1.25 mm slices at 30 days postprocedure. A blinded analyst at an independent central laboratory used MIMICS semi-automated segmentation software (Materialise NV, Belgium) to measure and record stone volume at baseline and 30 days to evaluate stone clearance and RSV. For added rigor, the CT images were also read by a blinded independent endourologist. Safety was assessed and adjudicated by an independent medical monitor and the principal investigator, both blinded to randomization. Adverse events (AEs) were graded using the Clavien–Dindo system and assessed whether device or procedure related. Stone events were defined as subject-reported, documented stone passage and/or any recurrent RF-related symptom(s) requiring clinic or ED visit, hospitalization, or additional interventions. Event rates between groups were compared.
Effectiveness was compared between groups. The primary effectiveness end point was noninferiority in SFR, defined as the percentage of subjects with zero RF at 30 days postprocedure observed on 1.25 mm slice NCCT by a blinded central reviewer. Secondary end points were stone clearance, RSV, and modified SFR defined as no RF >2 mm and no RF >4 mm at 30 days.
Statistical methods
Subjects were randomized 1:1 SURE to URS using permuted block sizes 2 and 4, stratified by study center. Computer-generated randomization schedules were administered using sequentially numbered sealed envelopes opened by site personnel during the index procedure. Randomization was administered intraoperatively after effective 12/14F UAS placement and retrograde pyelogram to delineate the collecting system. Any ureteral stones were treated prior to randomization.
PASS 2021 was used to estimate the sample size (80% power) for a noninferiority test for the difference between two proportions (20% noninferiority margin; one-sided Z-test, α = 0.05). We chose 20% based on published URS studies with comparable SFR (55%−73%), 3,4 although absence of multicenter randomized controlled trials with 30-day follow-up and inconsistent SFR reporting made it challenging to find comparable studies. Assuming 25% URS SFR, an estimated 58 subjects per group would be needed. For secondary end points, if the noninferiority test passed, a superiority test was conducted (two-sided α = 0.05) per protocol. Categorical variables were tested using chi-squared tests; continuous variables were tested using two-sample t-tests. The same primary and secondary end point analyses were repeated excluding >2 cm stone burden subjects (since their inclusion represented protocol deviations) and in lower pole stone subjects. Stone clearance and RSV linear regressions on baseline stone volume showed evidence of treatment group and baseline stone volume interaction, so these analyses were stratified by treatment group. Safety was assessed based on the intent to treat (ITT) population (all subjects in whom treatment was initiated), which includes the roll-in/training subjects (SURE, n = 6 and URS, n = 3). Efficacy was analyzed based on the modified ITT (mITT) population (all subjects treated without major protocol deviations preventing 30-day outcomes assessment).
Study procedures
After effective UAS placement and randomization, subjects underwent SURE or URS. In both groups, subjects initially underwent laser lithotripsy, and for consistency, each site used its ureteroscope of choice (disposable, reusable, 6.9–9.9F) and standard laser for both groups, including thulium fiber laser (3 sites) and high-power holmium Yttrium Aluminum Garnet (YAG) (8 sites). Laser settings and a dusting or fragmenting approach were based on investigator’s preferences. In both groups, the surgeon was instructed to attempt a surgically stone-free outcome; accordingly, basketing was allowed in both groups.
The SURE procedure was performed using the CVAC Aspiration System (Calyxo, Inc., Pleasanton, CA), a steerable ureteroscopic irrigation and aspiration catheter designed to remove fragments ≤2.5 mm (Fig. 1). The 11.9F outer diameter catheter has an outer irrigation lumen for continuous irrigation and a 7.5F inner aspiration lumen. The large aspiration lumen facilitates continuous passive efflux of irrigant and stone debris, preventing intrarenal pressure buildup during use of the device. The steering control dial enables the surgeon to deflect the catheter tip into all parts of the collecting system under fluoroscopy. The vacuum port connects to standard operating room suction (150–200 mm Hg), and aspiration is activated by covering the vacuum controller opening. Irrigation is applied using a 10-cc syringe system and quickly removed by both passive and active aspiration through the aspiration lumen.
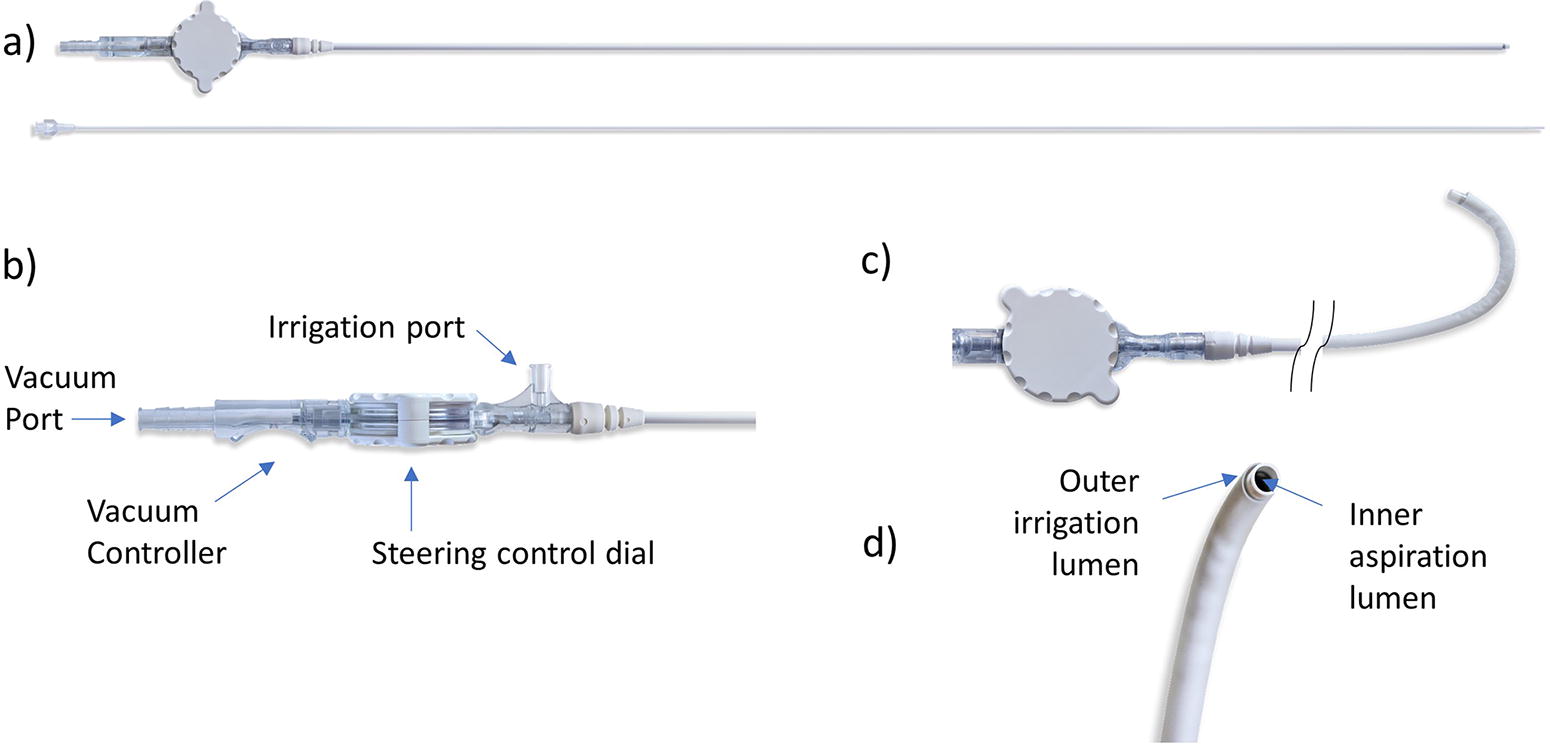
CVAC© Aspiration System.
During SURE (Fig. 2), the surgeon utilized laser lithotripsy to ablate the stones until fragments were ≤2 mm. After ureteroscope removal, CVAC was introduced over a 0.035” or 0.038” guidewire through the UAS under fluoroscopy. Once CVAC was placed in the target location, the introducer and guidewire were removed. CVAC was manipulated under fluoroscopy throughout the collecting system. In each calyx, fragments, including “dust,” were removed using simultaneous irrigation/aspiration. After CVAC removal, the ureteroscope was reintroduced to assess for RF. If necessary, CVAC and/or a basket was reintroduced and treatment continued until the subject was surgically stone free.
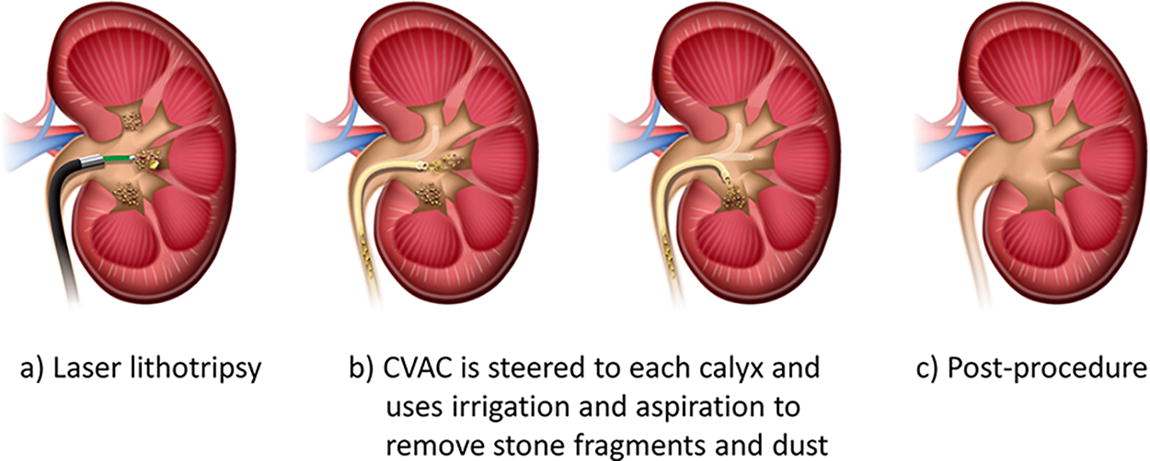
Schematic of the steerable ureteroscopic renal evacuation (SURE) procedure using the CVAC© Aspiration System.
In the control group, URS was performed based on the investigator’s standard of care. Surgeons chose a dusting vs fragmenting approach and used basket extraction as per their clinical judgment to render the subject surgically stone free.
In both groups, a retrograde pyelogram was performed to assess for kidney/ureter injury postprocedure. A stent was placed for ≥3 days, total duration as per surgeon discretion. Alpha-adrenergic blockers, analgesics, and/or anticholinergics were prescribed as per investigator standard of care.
Any protocol changes initiated by investigators were reviewed and approved by the principal investigator. Since CVAC had been previously used for large stone burdens, investigators requested this deviation when they felt that they could achieve a surgically stone-free result with either treatment. As such, 7 subjects with stone burden >2 cm were admitted into the study, randomized 6:1 URS to SURE (by chance).
Results
From June 2021 to February 2023, 123 subjects were randomized across 11 U.S. institutions. A total of 101 subjects qualified for the efficacy analysis in the mITT population (SURE n = 46 and URS n = 55, Fig. 3). Baseline demographics and clinical characteristics were similar between groups, except for stone burden; despite randomization, the baseline stone burden and volume were lower in the SURE vs URS groups (Table 1). Stone composition was also comparable between SURE and URS subjects (Table 1).
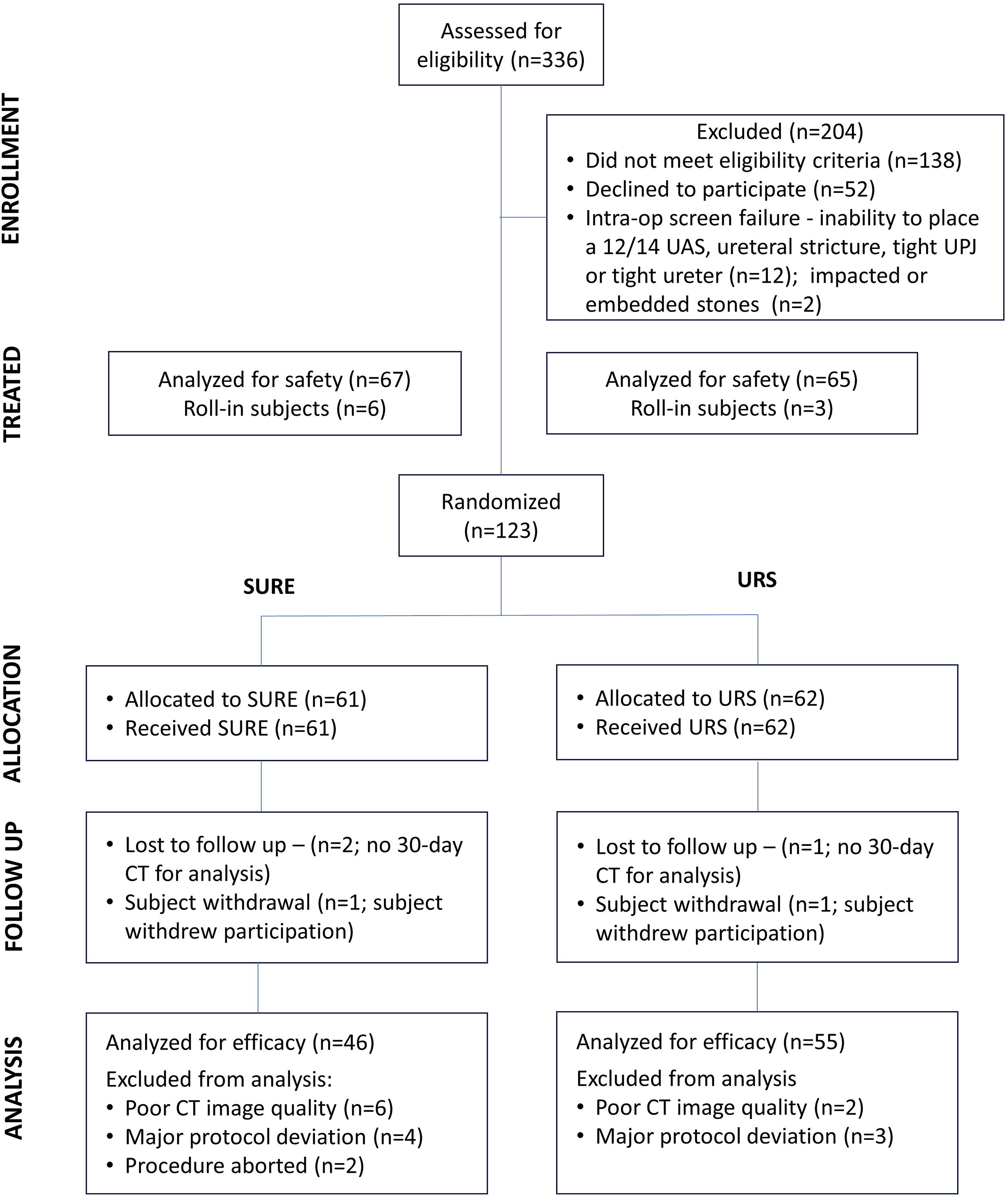
CONSORT (Consolidated Standards of Reporting Trials) flow diagram (CT = computed tomography; SURE = steerable ureteroscopic renal evacuation; UAS = ureteral access sheath; URS = ureteroscopy).
Baseline Demographics and Clinical Characteristics
Subjects with stone composition analysis.
HU, Hounsfield units; SD = standard deviation; SURE = steerable ureteroscopic renal evacuation; URS = ureteroscopy; BMI = body mass index.
Procedure time, laser time, and laser power were similar between groups (Table 2). Mean fluoroscopy time was greater for SURE vs URS, and the mean number of basket passes was lower for SURE vs URS.
Procedure and Perioperative Parameters
kJ = kilojoules; YAG = Yttrium Aluminum Garnet.
All end points were met (Table 3). Secondary end points of SFR (no RF >2 mm or no RF >4 mm) showed no significant difference between groups. SURE demonstrated significant superiority in stone clearance and RSV vs URS at 30 days. Subgroup analyses excluding subjects with stone burden >2 cm showed findings consistent with the entire cohort (Table 4). RSV (mm3) in the lower pole was significantly lower after SURE vs URS (SURE n = 17, 25.8 ± 39.1 vs URS n = 22, 97.4 ± 147.3, p = 0.04). SFR by stone-free status grade is shown in Table 5.
Stone Removal Outcomes
Chi-squared test for noninferiority.
Two-sample t-test for superiority.
Chi-squared test for superiority.
RF = residual fragment; RSV = residual stone volume; SFR = stone-free rate.
Subgroup Analysis Excluding Subjects with Stone Burden >2 cm
Chi-squared test for noninferiority.
Two-sample t-test for superiority.
RF = residual fragment; RSV = residual stone volume; SFR = stone-free rate.
Stone-free rates based on noncontrast CT scans with 1.25 mm slices.
Chi-squared test for noninferiority.
Chi-squared test for superiority.
RF = residual fragment.
With SURE, there was no evidence that stone clearance was dependent on baseline stone burden (Pearson’s correlation coefficient = 0.02, p = 0.89; Fig. 4). In contrast, with URS, there was a significant negative correlation between stone clearance and baseline stone volume (correlation coefficient = −0.30, p = 0.025; Fig. 4).
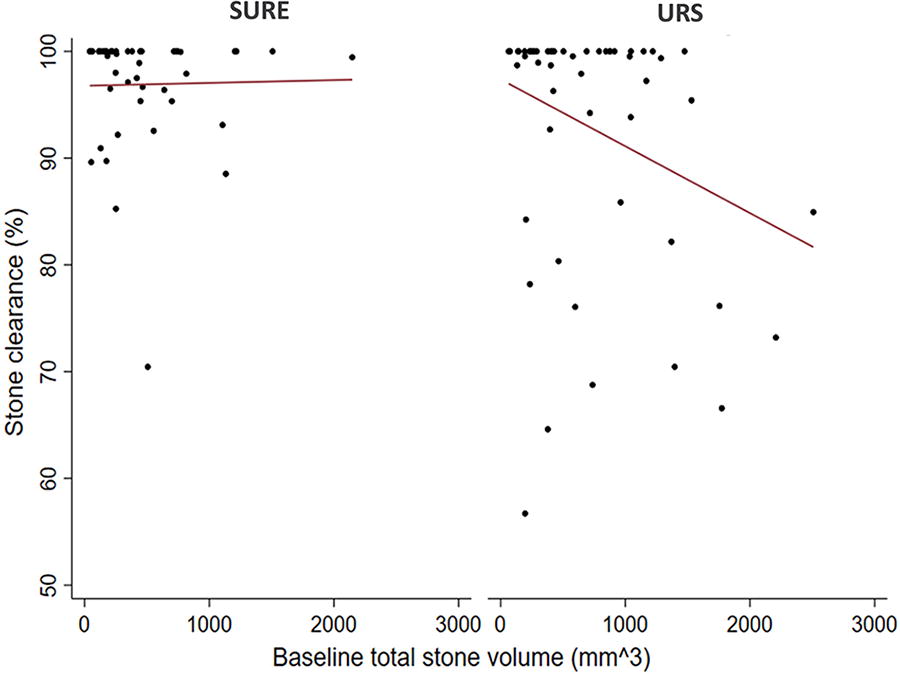
Regression analysis of stone clearance vs baseline total stone volume.
Regression analysis also indicated that with SURE, there was no significant evidence of a relationship between RSV and baseline total stone volume (correlation coefficient = 0.24, p = 0.11), whereas with URS, there was a significant, positive correlation between RSV and baseline stone volume (correlation coefficient = 0.68, p < 0.001; Fig. 5).
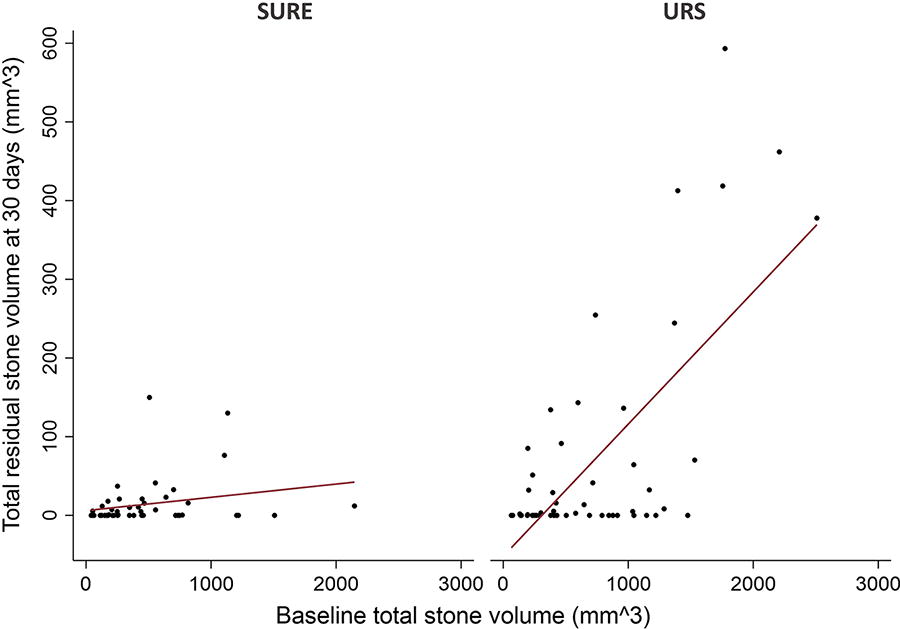
Regression analysis of residual stone volume (RSV) vs baseline total stone volume.
There were no significant differences in safety between groups (Table 6). Most AEs were mild, occurred within the first 7 postoperative days, and resolved spontaneously. There were no serious device-related AEs in either group. There were 3 Clavien–Dindo Grade I SURE-related AEs; all involved a tear/perforation to the tissue. Two had bleeding that obscured visualization and the procedures stopped. Since CVAC was manipulated without direction visualization, it is unknown whether suction and/or device manipulation caused the bleeding. All events resolved without intervention. In the URS group, there was one Clavien–Dindo Grade I device-related AE in which the laser caused tissue injury and bleeding that resolved before the end of the procedure.
Adverse Events at 30 Days
Fisher’s exact test.
Three subjects per group experienced stone events. Two SURE subjects had documented stone passage; one underwent stent placement. One URS subject passed a stone fragment; one had flank pain requiring an ED visit; and one was scheduled for retreatment for RF. No SURE subjects required retreatment within 30 days.
Discussion
SURE using the CVAC Aspiration System achieves high stone clearance with minimal RSV independent of baseline stone volume. A recent study of SURE in large stone burden patients also reported high effectiveness independent of stone burden. 17 ASPIRE also demonstrates what was previously reported in the literature that URS efficacy declines with increasing baseline stone volume. 6 –8,18 To our knowledge, SURE is the first ureteroscopic procedure to demonstrate an efficacy and reliability profile independent of baseline stone parameters.
A strict, standardized CT imaging protocol enabled rigorous outcomes assessment and demonstrated the limitations of the SFR metric. The mean SFR with zero RF was <50% in both groups, in line with the 55% SFR in a study of aggressive basket extraction. 3 The seemingly low SFR is likely due to the increased sensitivity of 1.25 mm CT slices and reflects a more accurate assessment vs other studies where 3–5mm slices are typically used. The fact that SFR improved as the criteria for “stone-free” changed from zero fragments to no RF >2 mm to no RF >4 mm highlights the problematic variability of the SFR metric. These distinctions in SFR definition have clinical implications, as RFs >4 mm have been associated with increased rates of stone events and RFs >2 mm are more likely to grow than smaller fragments. 11,12
Due to the variability of how SFR is measured and defined and its bimodal nature, it may be that continuous variables such as stone clearance and RSV are more clinically relevant and useful for comparing treatments. Both RSV and RF size pertain to how much stone is left. Studies have demonstrated that the largest RF is a predictor for stone events 12 and that larger RFs are more likely to grow vs RF. 11 With longer follow-up, we anticipate understanding whether similar observations can be made between RSV and stone events, including retreatment.
There has been recent discussion in the urologic community of possible elevated intrarenal pressure during URS contributing to increased sepsis. 19,20 The sepsis rates for SURE (1.5%, 1/67) and URS (3.1%, 2/65) compared favorably to the 5.0% average published rate for URS. 21
Several limitations must be acknowledged. The baseline imbalance in stone size between the SURE and URS groups, despite randomization, introduces a confounding factor. The subgroup analysis excluding patients with stone burden >2 cm addresses this concern, and the findings similar to those in the entire cohort are reassuring (Table 4). In addition, the regression analyses (Figs. 4 and 5) showed no relationship between baseline stone volume and RSV or stone clearance for SURE.
Each site used its standard laser, which represents real-world practice but introduces variability. To control for different laser types and dust quality, the same laser type and settings were used in both groups. Future studies could be enhanced by limiting the laser to one type. Subjects were censored when major deviations impacted accurate assessment of the primary and secondary efficacy end points. It is not known whether these unavailable data would have affected the results.
The benefits of increased stone clearance with SURE need to be balanced with potential drawbacks as follows: increased fluoroscopy time, the need for a 12/14F UAS, and device costs. Although fluoroscopy time for SURE was significantly higher than for URS—potentially increasing radiation exposure to the patient—there was wide variation reflecting differing investigator techniques. A next-generation CVAC system that integrates direct vision, laser lithotripsy, irrigation/aspiration, and stone collection through a single device expected to improve safety and effectiveness outcomes has been developed and will be the subject of future studies. The benefit–drawback balance for SURE vs URS will also be further understood with longer term follow-up.
Conclusions
In this largest and most rigorous study of SURE compared with URS, SFR was noninferior and stone clearance and RSV were significantly better with SURE, independent of baseline stone volume. These results suggest that SURE has a role in the treatment of nephrolithiasis; longer follow-up will better inform the clinical and health economic effect of improved stone clearance, including the need for retreatment. Additional outcome metrics beyond SFR such as stone clearance and RSV are needed to better assess procedural outcomes.
Footnotes
Acknowledgments
The authors would like to thank the following collaborators for their support in investigation: Brian Mazzarella, MD; Urology Austin; Austin, TX. Kalpesh Patel, MD; Arizona Urology Specialist; Tucson, AZ Gregg Eure, MD; Urology of Virginia; Virginia Beach, VA Patrick Springhart, MD; Prisma Health; Greenville SC Eric Katz, MD; Johns Hopkins University School of Medicine; Baltimore, MD Kevin Wymer, MD; Mayo Clinic; Rochester, MN. The authors would also like to thank Sharon Lam Wang for her assistance in preparation of this article and Miranda Kunz of Technomics Research for the statistical support.
Authors’ Contributions
Conceptualization: B.R.M. and G.M.P. (both supporting), investigation: B.R.M. (lead); T.J.M., B.J., J.P., J.S.W., G.M.P., L.J., I.S., K.S., D.C., K.C., R.B., R.M., C.M., N.N., M.W., and M.L. (all equal), supervision: B.R.M. and T.C. (both equal), and writing—review and editing: B.R.M., B.J., J.S.W., G.M.P., K.S., and T.C. (all equal).
Author Disclosure Statement
J.S.W. serves as a consultant to Applaud Medical, Calyxo Inc., and Lilac Therapeutics and is on the editorial board of Urology Times, G.M.P. serves as a consultant to Calyxo, Inc., I.S. serves as a consultant to Ambu and Storz, N.N. serves as a consultant to Endotheia, and T.C. serves as a consultant to Calyxo Inc., Boston Scientific Company, Becton Dickinson and Company, Cook Medical, and Uroviu. The remaining authors have nothing to disclose.
Funding Information
This study was sponsored by Calyxo, Inc.
