Abstract
Introduction:
To describe the medium-term outcome of robotic augmentation ileocystoplasty by posterior and anterior approaches in the management of contracted low-capacity bladder from cystitis.
Materials and Methods:
Data on consecutive cases of robotic augmentation ileocystoplasty between 2011 and 2021 were prospectively collected and reviewed in our center. Retzius-sparing posterior approach was performed by anastomosis of an M-configuration small bowel plate to the posteriorly located U-shaped cystostomy. Conventional anterior augmentation ileocystoplasty was performed by anastomosis of an S-configuration small bowel plate to the anterior cystostomy. The procedure was as per conventional robotic pelvic surgery setting with the da Vinci Surgical System (Intuitive Surgical, Sunnyvale, CA, USA). Perioperative data and postoperative complications were assessed. Preoperative and postoperative functional outcomes were compared.
Results:
Six patients underwent posterior augmentation cystoplasty and 20 patients underwent anterior augmentation cystoplasty with a mean follow-up of 51 ± 26 months. Mean operative time was 274 ± 113 minutes and 267 ± 102 minutes, respectively (p = 0.889), and mean hospital stay was 9.0 ± 9.6 days and 8.3 ± 6.1 days, respectively (p = 0.831). Additional procedures include eight ureteral reimplantations and one ileal interposition. Mean functional bladder capacity (FBC) at 2 years of the two approaches were similar, which were 350.2 ± 126.3 mL (posterior approach) and 310.2 ± 151.4 mL (anterior approach) (p = 0.5936). Overall, FBC before the operation and at 24 months was 68.9 ± 48.2 mL and 318.5 ± 144.8 mL (p = <0.0001), respectively. Analgesic requirement was decreased after the operation (preoperative 19 patients, postoperative 4 patients; p < 0.0001).
Conclusions:
Posterior approach of robotic augmentation ileocystoplasty provided an alternate option to expand the bladder capacity for patients with a small contracted bladder. It could provide a similar symptom relief and functional improvement as the anterior approach. The clinical trial number is CRE-2011.454.
Introduction
Augmentation cystoplasty has been used as a surgical treatment for low capacity, poorly compliant, or refractory overactive bladder. 1 It is a valid option in neurogenic and non-neurogenic bladder dysfunction when medical treatments and endoscopic treatments have been exhausted. 2 Furthermore, augmentation cystoplasty has a substantial role in managing pathologies like interstitial cystitis and ketamine-associated cystitis. 3,4 Robot-assisted laparoscopic augmentation ileocystoplasty with total intracorporeal reconstruction has been reported in the literature as case reports or small case series with relatively short-term follow-up. Furthermore, although the robotic system provides technical agility and access to almost every corner of the pelvis, the basic surgical approach to augmentation cystoplasty has not been modified in response to these additional advantages coming from this technological advancement. Retzius-sparing posterior approach augmentation cystoplasty has the potential advantage of improved functional outcome and preserving the suprapubic access to the bladder. In the study, we reported the medium-term outcome of posterior approach robotic augmentation ileocystoplasty and compared its effects with the conventional anterior approach.
Materials and Methods
Study population and preoperative preparation
The current study used a prospectively maintained database approved by the institutional review board (IRB approval reference number CRE-2011.454) and adhered to the principles outlined in the Declaration of Helsinki, including patients of cystitis treated with augmentation cystoplasty. After excluding open and laparoscopic procedures, consecutive cases of robotic augmentation cystoplasty were reviewed and analyzed. The diagnosis of cystitis was confirmed with clinical history and relevant endoscopic findings. Before the operation, functional bladder capacity (FBC) of the patients was assessed by urodynamic study or repeated uroflowmetry if the refractory pelvic or perineal pain from cystitis precluded patients from catheterization and a formal urodynamic study. Creatinine clearance (CrCl) was measured by 24-hour urine creatinine and serum creatinine. FBC of <200 mL and CrCl >40 mL/min/1.73 m2 were selected for consideration of augmentation cystoplasty. Ultrasound was performed in all patients to screen for hydronephrosis. Contrast CT urogram, Tc-99m DTPA scan, micturating cystogram, antegrade pyelogram, or retrograde pyelogram would be performed according to clinical indication and to determine the site of ureteral strictures if present. All patients were counseled to ascertain that they were sufficiently motivated to learn and comply with clean intermittent self-catheterization (CISC) if it was eventually required. Patients were started on clear fluid diet 1 day before procedure. A penicillin-class antibiotic and metronidazole were given on induction of surgery as prophylaxis or according to previous urine culture sensitivity.
Surgical technique
Conventional anterior augmentation cystoplasty technique was adopted in the initial cases of the cohort. Since 2020, posterior augmentation cystoplasty technique has been adopted because of its convenience in simultaneous bilateral ureteral reimplantation, as well as its feasibility to preserve the Retzius space. For both techniques, the patient was placed in 25° Trendelenburg split-leg position. Port placement was as per conventional robotic radical cystectomy setting with the da Vinci Surgical System (Intuitive Surgical, Sunnyvale, CA, USA) (Fig. 1).
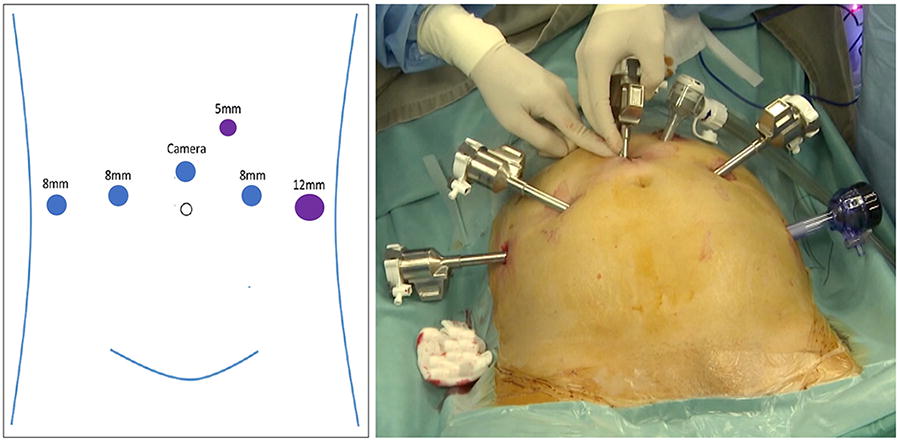
Port placement for robotic augmentation ileocystoplasty. Blue circles represent robotic trocars. Purple circles represent assistant ports.
Anterior augmentation cystoplasty
Urinary bladder was dissected away from the anterior abdominal wall, and the urachus was taken down. Perivesical spaces and anterior bladder neck were exposed for visualization of the full breadth and length of the bladder. Cruciate incision was made on the bladder anteriorly making use of the full extent of the available bladder dimensions to avoid any possible waiting phenomenon after augmentation reconstruction. Next, a 25- to 30-cm segment of ileum 15 cm proximal to the ileocecal junction was isolated with laparoscopic stapler. Small bowel continuity was resumed using a functional end-to-end anastomosis. The isolated ileal segment was positioned in an “S-configuration,” and the limb closest to the bladder was anchored behind the cystostomy edge before detubularization (Fig. 2). This anchoring suture helped keep the S-shaped ileal segment in position despite the gravitational pull from the patient’s Trendelenburg position. Once the ileal segment had been detubularized, the edges of the small bowel wall were joined together to form a plate, which was then flipped over to cover the cystostomy opening.
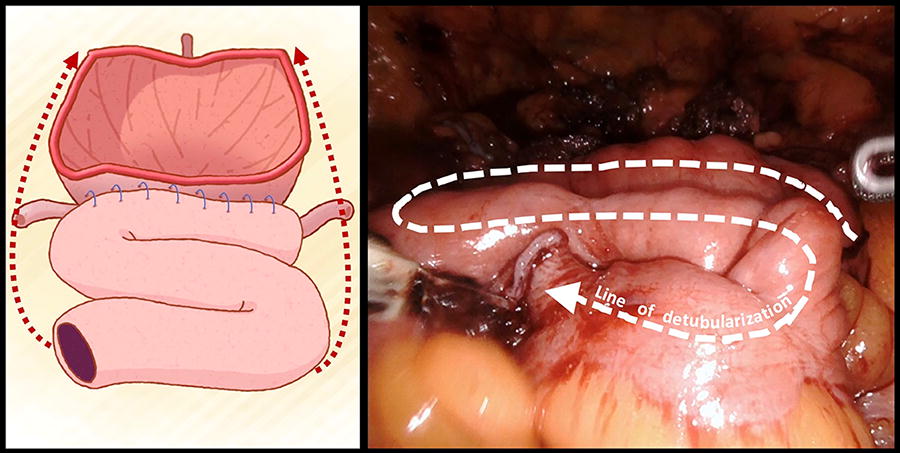
Anterior augmentation cystoplasty with S-configuration of small bowel plate. Cystostomy is made on the anterior surface of the bladder. Blue line between the posterior wall of bladder and small bowel limb represents the anchoring suture. Red dotted line represents the expected direction of folding of the small bowel plate. White dotted line represents the expected line of small bowel detubularization.
Posterior augmentation cystoplasty
Unlike the anterior approach, the Retzius space was preserved in the posterior approach (Supplementary Video S1). Without the need for bladder mobilization, a U-shaped cystostomy incision was created with the apex approaching the trigone. Attention was paid not to injure the ureteral orifices. Once the cystostomy had been completed, the U-shaped bladder flap was flipped over and anchored to anterior abdominal wall, maximizing the bladder wall circumference for subsequent bowel anastomosis (Fig. 3). A 25- to 30-cm segment of ileum was prepared similar to the anterior approach. A longer segment of small bowel could be isolated if the additional reconstructive procedure was contemplated, for example, ileal interposition. The small bowel segment was configured to an “M” shape in the posterior approach (Fig. 4). The two ends of the M-shaped segment could act as two chimneys with variable length, tailoring to the need of ureteral anastomosis if concomitant ureteral stricture is present. After detubularization, the small bowel plate was pulled forward and anastomosed to the cystostomy. This maneuver created minimal additional tension to the ileal segment mesentery because there was no change in the orientation of the small bowel plate.
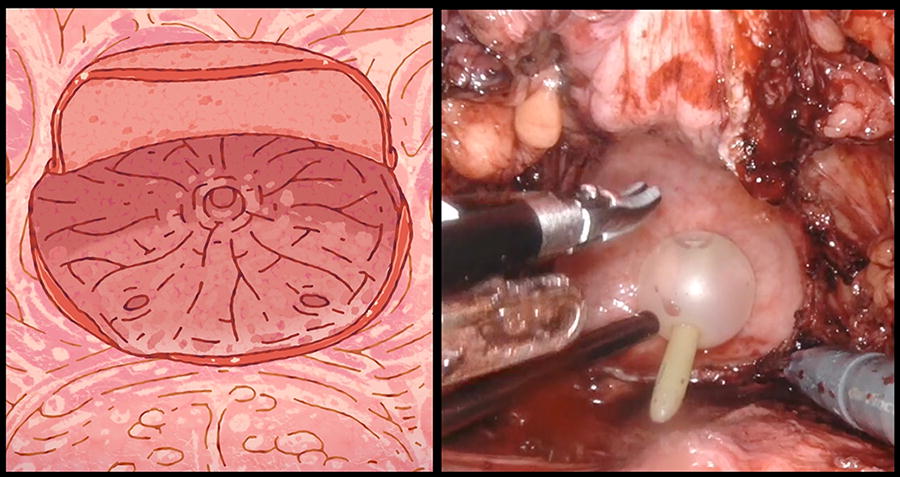
Posterior augmentation cystoplasty with an U-shaped cystostomy flap being flipped toward the anterior bladder wall without disturbance of the Retzius space.
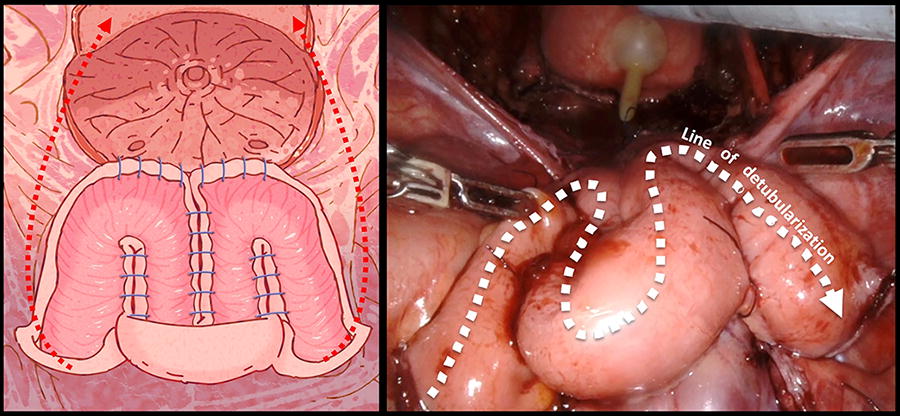
Posterior augmentation cystoplasty with M-configuration of small bowel plate. An U-shaped cystostomy flap is fixed to the anterior bladder wall. Blue line represents the suture for the small bowel plate and the bladder anastomosis. Red dotted line represents the expected direction of apposition between the small bowel plate and the bladder. White dotted line represents the expected line of small bowel detubularization.
Postoperative treatment and follow-up
Suprapubic catheter was not inserted toward the end of the procedure in either approach. Pelvic drain was taken off when the output was less than 100 mL. Foley catheter was taken off on postoperative Day 14 unless there was a clinical indication. Routine cystogram was not performed unless there was suspicion of urinary leakage. Complications were graded using the Clavien-Dindo classification. Patient was followed up every 3 months in the first year and every 4 months in the second year. Standardized pro forma to document patients’ urinary symptoms was used during each follow-up. Follow-up interval after second year depended on clinical situation. Blood gas for pH and base excess, renal function test, and uroflowmetry were performed at every visit with interval FBC assessment. Ultrasound of the urinary system was performed to screen for upper tract abnormalities.
Statistical analyses
Descriptive statistics included the frequency of categorical variables. The mean and standard deviation were reported for continuous data. Differences in categorical data distributions were assessed using the chi-square test. Differences in means between the two groups were assessed using t test. A p-value of <0.05 was considered statistically significant. All statistical analyses were performed using SPSS version 23.0 (Armonk, NY: IBM Corp.).
Results
Six patients underwent posterior robotic augmentation cystoplasty, and 20 patients underwent anterior augmentation cystoplasty from 2011 to 2021. The patient demographics and clinical parameters are shown in Table 1. The mean follow-up time for the posterior approach and anterior approach was 43.8 ± 1.5 months and 88.2 ± 27.4 months, respectively (p < 0.01). All patients had clinical features of cystitis with a FBC less than 200 mL before operation. Most of the patients in the cohort had a small contracted bladder secondary to ketamine-associated uropathy. The mean duration of ketamine abuse was 112 ± 37 months, and the mean duration of abstinence before procedure was 42 ± 24 months. In the cohort, 12 (46.2%) patients had treatment with intravesical sodium hyaluronate instillation before operation because of intractable pelvic or perineal pain from cystitis. One (3.8%) patient had hydrodistention 16 months before his robotic augmentation. Two (7.7%) patients were found to have concomitant bilateral ureteral strictures, and 2 (7.7%) other patients had concomitant unilateral ureteral stricture as a complication of ketamine-associated uropathy. Two (7.7%) patients were found to have bilateral vesicoureteral reflux (VUR).
Patient Characteristics
FBC = functional bladder capacity; NSAID = nonsteroidal anti-inflammatory drug; SD = standard deviation.
Perioperative details are listed in Table 2. The mean operative time for posterior approach robotic augmentation cystoplasty was 274 ± 114 minutes, and the mean catheter time was 17.2 ± 5.2 days. No significant difference was observed compared with the anterior approach group. For patients with concomitant upper tract pathology, additional procedures were performed. Bilateral ureteral reimplantation was performed in 2 (7.7%) patients with bilateral VUR and 2 (7.7%) patients with distal ureteral stricture. Two (7.7%) patients with unilateral distal ureteral stricture also had ureteral reimplantation. One (3.8%) patient had right distal ureteral stricture and left proximal ureteral stricture. Same session augmentation cystoplasty with right ureteral reimplantation and left ileal interposition was performed. In the postoperative 3-month period, overall 10 (38.5%) patients had Clavien-Dindo Grade I complication. The three complications in the posterior approach group included two patients developing fever before removal of Foley catheter who responded to antibiotics treatment and one patient having readmission for blocked Foley catheter. The rest of the complications happened in the anterior approach group, which included 6 (30.0%) patients developing fever before removal of Foley catheter who responded to antibiotics treatment, 1 (5.0%) patient developed diarrhea which was managed conservatively, and 1 (5.0%) patient was found to have urinary leakage, which was managed by keeping the Foley catheter for 19 days after the operation. One (5.0%) patient was found to have intestinal obstruction at the ileoileal anastomosis on postoperative Day 9 and subsequently laparotomy and limited right hemicolectomy were performed as the distal ileal stump was found not healthy enough for primary anastomosis. The patient was discharged on postoperative Day 31.
Perioperative Outcomes
Comparison between posterior approach and anterior approach.
CD = Clavien-Dindo.
Outcomes of both posterior augmentation cystoplasty and conventional anterior augmentation cystoplasty are shown in Table 3. Posterior approach achieved an increase in mean FBC by 622%. Both posterior and anterior approaches were found to have significantly improved FBC with similar perioperative parameters. No statistically significant difference was found in the postoperative mean FBC between these two approaches (310.2 ± 151.4 mL vs 350.2 ± 126.3 mL, p = 0.5936). At 1-year follow-up, 1 (16.7%) patient required CISC in the posterior approach group and 2 (10.0%) patients for the anterior approach group to drain the augmented bladder. At 2-year follow-up, 2 (33.3%) patients and 3 (15.0%) patients required such measure, respectively. Urinary symptoms assessment per pro forma protocol during follow-up did not reveal any incontinence after either approach of augmentation cystoplasty.
Long-Term Functional Outcome
The functional outcome of the whole cohort after the operation at 6, 12, and 24 months was charted (Fig. 5). The mean FBC before the operation was 68.9 ± 48.2 mL. Comparing the mean FBC against the last value, the mean FBC at 6, 12, and 24 months was 280.0 ± 109.0 mL (p < 0.0001), 322.9 ± 157.1 mL (p = 0.0599), and 318.5 ± 144.8 mL (p = 0.4809), respectively. Two (7.7%) patients were on anticholinergic, and 1 (3.8%) patient was on beta-3 agonist at 2-year follow-up. Four (15.4%) patients had a mildly deranged serum creatinine level before surgery. After surgery, this derangement persisted at 1 year and 2 years. An addition of 1 (3.8%) patient developed a mildly deranged renal function at 1 year and 2 years after surgery. No patients developed metabolic acidosis upon follow-up at 2 years, and none required sodium bicarbonate. At the latest follow-up, the mean FBC of the cohort was 344.6 ± 171.8 mL. The number of patients with deranged renal function and the number of patients on CISC were the same as at 2-year follow-up. Overall, one (3.8%) patient was started on sodium bicarbonate because of metabolic acidosis at postoperative 3 years.
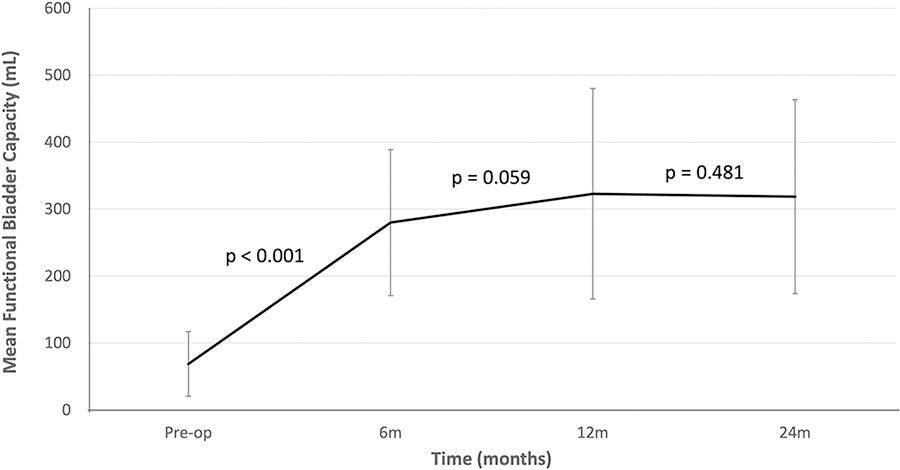
Mean functional bladder capacity before and after robotic augmentation ileocystoplasty. p-Value signifies the difference between the adjacent values.
Discussion
Augmentation ileocystoplasty was popularized by Couvelaire who adopted this technique to treat patients with a small contracted bladder. 5 A small contracted bladder could be resulted from interstitial cystitis, radiotherapy, chemotherapy cystitis, ketamine-associated uropathy, or tuberculosis. 6 –8 Taking reference from the treatment outcome of augmentation cystoplasty in interstitial cystitis, more significant symptomatic relief was observed in patients with a smaller bladder capacity. 9 In our current series, the mean bladder capacity was 68.9 ± 48.2 mL, and all patients’ capacity was <200 mL. This could account for the significant decrease in analgesic demand after augmentation cystoplasty in the current study.
Gundeti and colleagues reported the first case of robotic augmentation ileocystoplasty in 2008 in a 10-year-old girl with a neurogenic bladder, which had a capacity of 220 mL. 10 The same center later reported that comparing with its open counterpart, robotic augmentation cystoplasty had a similar functional outcome but a longer operative time. 11 Since then, a number of reports were in the literature addressing the outcome of robotic augmentation ileocystoplasty, but all were in the format of case reports or small case series (Table 4). Thus far, our current study presented the largest series of robotic augmentation ileocystoplasty available in the literature, as well as the series in adult with the longest follow-up duration. Furthermore, although most of the augmentation ileocystoplasty series in the literature were on patients with neurogenic bladder, our cohort was one of the few series that investigated the outcome in a population with contracted bladder secondary to an inflammatory process. This also explained the relatively smaller preoperative bladder capacity in our series compared with other reports in the literature. Results from the current study demonstrated the efficacy of robotic augmentation ileocystoplasty in this specific clinical situation.
Results of Robotic Augmentation Ileocystoplasty in the Literature
BC = bladder capacity; CISC = clean intermittent self-catheterization; N/A = not available.
Since the first introduction of robotic surgery in radical prostatectomy in 2000, robotic platform has been extended to different aspects of urologic surgery. 24 Taking advantage of the immersive 3-dimensional visualization of the pelvis and free range of movement provided by the robotic surgical system, our current study has introduced a new approach for augmenting the bladder capacity with a posterior U-shaped cystostomy flap. Such approach provides a few potential advantages over the conventional anterior approach, which requires complete mobilization of the bladder from the anterior abdominal wall. Taking from the experience of Retzius-sparing robotic-assisted radical prostatectomy, the preservation of Retzius space allows the anterior bladder to be fixed in the high position, which acts as a fulcrum during abdominal pressure. This would translate to a compression force on the membranous urethra and a high-resting urethral closure pressure, resulting in better continence control. 25 The preservation of continence mechanism would be relevant for patients of augmentation cystoplasty especially when some of the patients have significant symptoms of urgency or detrusor overactivity. Furthermore, the posterior approach in robotic augmentation cystoplasty allows later an easier access to extraperitoneal space if it is needed in the future. Such access can give convenience to simple procedures such as suprapubic catheter insertion or complex procedures such as extraperitoneal suprapubic radical prostatectomy if indicated later in life. Last but not least, the posterior approach gives possibility to the use of an M-shaped ileal segment for reconstruction. Although from the laparoscopic vision perspective the ileal segment is shaped in “M,” in fact it resembles the “W-configuration” Hautmann neobladder. 26 The Hautmann W-configuration of ileum gives a large-volume reservoir. 27 Furthermore, its bilateral chimneys make it easy to reach the distal ureteral stumps with minimal tension and ureteral mobilization in cases of concomitant ureteral strictures necessitating additional ileo-ureteral anastomosis. This decreases the risk of anastomotic strictures. 28 In addition, such configuration of ileum together with the posterior U-shaped cystotomy flap means that there is no need to fold the posterior wall of the small bowel patch to the bladder neck (see Supplementary Video S1). This avoids the extra tension in the small bowel mesentery from such maneuver. 29
In our current study, one patient had deterioration in renal function 2 years after augmentation cystoplasty. Patients with CrCl more than 40 mL/min/1.73 m2 had a lower risk of further renal impairment. 30 In our cohort, all patients had CrCl more than 40 mL/min/1.73 m2. This serves as a guide for our patient selection. For patients with a poorer CrCl, alternative gut segment can be considered for augmentation. In our center, robotic augmentation gastrocystoplasty was offered to patients with poor renal function as an option. 31 This alternative approach is another example of the effort in extending the application of robotic platform and minimally invasive surgery in reconstructive surgery.
Our study has several limitations. Bowel disturbance can be a potential long-term troublesome complaint after augmentation cystoplasty. This aspect was not addressed in our current study. Patient-reported outcome could provide another perspective on the evaluation of surgical success. It can be included in the follow-up assessment in future studies. The postoperative length of hospital stay in this current study was similar to open augmentation cystoplasty series. 11 The implementation of enhanced recovery after surgery pathways was not a routine in our cohort. Its impact on surgical outcome would require further assessment.
Conclusions
Medium-term outcome of robotic augmentation ileocystoplasty was demonstrated to be satisfactory with respect to bladder capacity improvement and symptomatic relief. In contrast to the conventional anterior approach, the posterior approach manages to preserve the Retzius space. Its combination of an U-shaped cystostomy flap and an M-shaped ileal segment achieved a similar outcome to the anterior approach. A larger number of patients are warranted to validate the observation.
Footnotes
Authors’ Contributions
C.H.Y.: Study conception and design, data collection, analysis and interpretation of results, and draft article preparation. M.H.M.T.: Data collection. Y.L.H.: Data collection. P.T.L.: Data collection. Y.H.T.: Data collection. E.S.Y.C.: Study conception and design and data collection. C.K.C.: Data collection. K.L.L.: Data collection. W.H.C.C.: Data collection. J.Y.C.T.: Data collection. P.K.F.C.: Data collection. C.F.N.: Study conception and design and data collection. All authors reviewed the results and approved the final version of the article.
Author Disclosure Statement
All authors have no conflict of interest to disclose.
Funding Information
No funding has been received for the study.
Supplementary Material
Supplementary Video S1
Abbreviations Used
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
