Abstract
Introduction:
Retrograde intrarenal surgery (RIRS) is associated with complications, many of which are related to the intrarenal pressure (IRP). We aim to describe the design of a novel isoprenaline-eluting guidewire (“IsoWire”) and present the results from the first in vitro release studies and the first animal studies showing its effect on IRP.
Materials and Methods:
The IsoWire comprises a Nitinol core surrounded by a stainless-steel wire wound into a tight coil. The grooves created by this coil provided a reservoir for adding a hydrogel coating into which isoprenaline, a beta-agonist, was loaded. Animal studies were performed using a porcine model. For the control, IRP, heart rate (HR), and mean arterial pressure (MAP) were measured continuously for 6 minutes with a standard guidewire in place. For the experiment, the standard hydrophilic guidewire was removed, the IsoWire was inserted into the renal pelvis, and the same parameters were measured.
Results:
In vitro analysis of the isoprenaline release profile showed that most (63.9 ± 5.9%) of the loaded drug mass was released in the 1st minute, and almost all of the drug was released in the first 4 minutes exponentially. Porcine studies showed a 25.1% reduction in IRP in the IsoWire that released 10 μg in the 1st minute; however, there was a marked increase in HR. The average percentage reduction in IRP was 8.95% and 21.3% in the IsoWire that released 5 and 7.5 μg of isoprenaline, respectively, with no changes in HR or MAP.
Conclusions:
The IsoWire, which releases 5 and 7.5 μg of isoprenaline in the 1st minute, appears to be safe and effective in reducing the IRP. Further studies are needed to establish whether the isoprenaline-induced ureteral relaxation will render easier insertion of a ureteral access sheath, reduce IRP during sheathless RIRS, or even promote the practice of sheathless RIRS.
Introduction
Urolithiasis is considered the third most common disease in urology, 1 and changes in lifestyle, diet, comorbidities, climate, and the increased use of abdominal imaging have contributed to its increased prevalence. 2,3 The pendulum has shifted from open procedures to more minimally invasive techniques such as retrograde intrarenal surgery (RIRS). A flexible ureterorenoscope is inserted into the kidney to observe the stone. If the stone is small, it may be snared and removed with a stone-extracting device. In cases wherein the stone is large, or the diameter of the ureter is narrow, the stone can be fragmented or dusted using laser lithotripsy.
Numerous technological advancements in endoscopy have contributed to the growing popularity of RIRS, which is reflected in the recent guidelines of prominent urologic societies, including the European Association of Urology and American Urology Association, both of which have expanded the role of RIRS. 4,5
However, RIRS is associated with complications, many of which are related to intrarenal pressure (IRP). Normal physiologic IRP in an unobstructed kidney ranges from 0 to 15 mmHg (0–20 cmH2O). 6 During RIRS, dangerous IRPs are often encountered, resulting in complications that lead to pyelorenal reflux resulting in the retrograde translocation of uropathogenic bacteria and endotoxins from the urinary system into the bloodstream predisposing to urosepsis. 7,8 Although mortality after ureteroscopy is rare, a recent multi-institutional case series of six post ureteroscopy mortalities revealed that four patients died because of urosepsis. 9
Although the patient's age, comorbidities, stone size, positive preoperative urine culture, and the presence of an indwelling stent contribute to the incidence of urosepsis in RIRS, 10 elevated IRP seems to be as important a risk factor. 10,11 Forniceal rupture is another complication of elevated IRP in RIRS. As the pressure rises within the collecting system, the collecting system ruptures at the renal fornix, causing intraoperative bleeding and, subsequently, poor vision for the surgeon. 12 Postoperative pain, acute kidney injury, and fluid overload are other complications of increased IRP that have been reported, albeit some theoretical. 7,8
To mitigate these complications, surgeons can employ various techniques to control IRP. Perioperative pharmacologic interventions using intravenous or endoluminal isoprenaline, a β-receptor agonist, and intravenous parecoxib, a selective cyclooxygenase-2 inhibitor, have been shown to reduce IRP. 13 –17 However, despite promising results, neither intervention has formed part of clinical practice.
This article describes in detail the design of a novel isoprenaline-eluting guidewire (“IsoWire”) and presents the results of the first in vitro release studies and the first animal studies showing its effect on IRP. Confirmation of coating lubricity, adherence, and durability and guidewire performance of the IsoWire is also presented.
Materials and Methods
Guidewire design
The IsoWire is composed of a central core made of Nitinol (Fig. 1A). This renders the guidewire super elastic; offers good flexibility, stability, and torque while navigating the genitourinary system; and is kink resistant. The body of the wire surrounding the core consists of a high-quality stainless-steel wire wound into a tight coil around the core. The distal tip is a conically grounded core wire that tapers distally with an atraumatic ball at the end. The wires are coated with polytetrafluoroethylene (PTFE) polymer to decrease the coefficient of friction and increase the lubricity. Distally, the hydrophilic coating reduces the friction during deployment and allows easier steering.
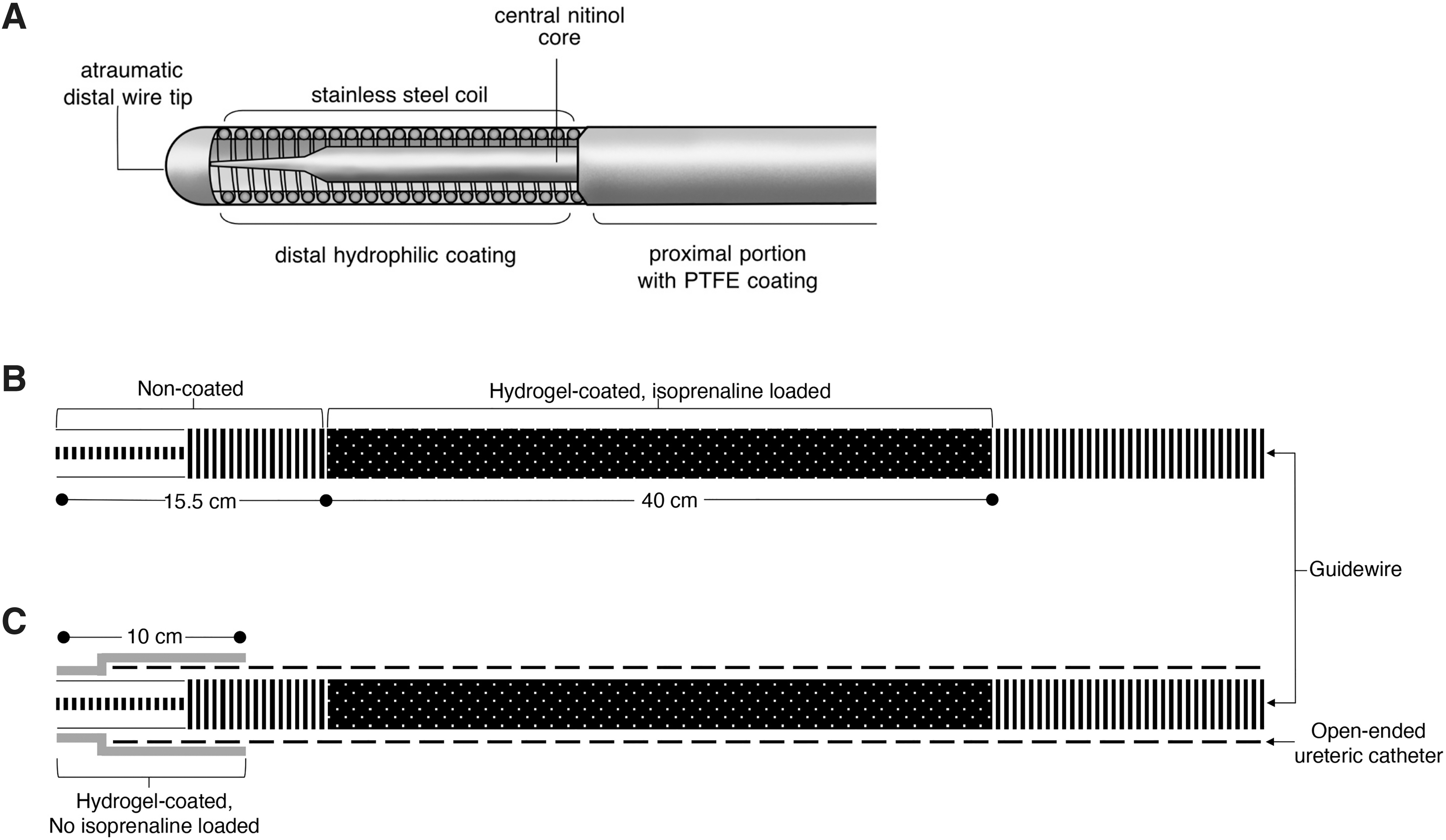
Schematic representation of the structure of the IsoWire
Hydrogel–drug coating synthesis
As mentioned earlier, the stainless-steel wire is wound into a tight coil around the core. The grooves created by the coil provided a reservoir for the hydrogel. The hydrogel coating on the base wire was produced in a series of steps.
Pretreatment of the guidewire
The distal 55.5 cm length of the guidewire was pretreated by wiping it with ethanol and immersing it in hexane for 2 minutes.
Preparation of the first hydrogel coating
The polyvinylpyrrolidone (PVP) hydrogel coating was produced on a 40 cm length of guidewire (Fig. 1B) using a method described by Butruk et al. 18 This length of guidewire, which would be in direct contact with the urothelium, was immersed into a solution of 4% cumene hydroperoxide and 5% ethylene glycol dimethylacrylate in hexane for 10 seconds and then left to dry at room temperature for 2 minutes to allow the solvent to evaporate. It was then immersed in a solution of 3% PVP (average molecular weight 360 kDa), 0.05% iron (II) chloride, and 0.1% ascorbic acid in a solvent mixture of water and propylene carbonate at a volume ratio of 90:10 for 15 minutes. The coated product was washed in deionized water for 10 minutes with agitation and dried at 45°C for 30 minutes.
Drug loading
The hydrogel-coated guidewires were immersed in an aqueous isoprenaline solution for 60 minutes at room temperature to load isoprenaline. They were then left to dry overnight at room temperature. Based on the previously determined release profile, the isoprenaline concentration in the solution was selected to obtain the desired amount of drug released in the 1st minute. Guidewires with hydrogel coatings that released 5.0, 7.5, and 10.0 μg of isoprenaline in the 1st minute were used for animal studies. Only a 40 cm length of guidewire in direct contact with the ureteral mucosa was loaded with isoprenaline.
Determining the release profile of isoprenaline
Ultraviolet–visible spectroscopy was used to determine the release profile of isoprenaline from the hydrogel coating. Fragments of guidewire with a length of 10 cm and hydrogel coating loaded with different amounts of drug were placed in 10 mL of artificial urine at 37°C (98.6°F), which was transferred to a fresh portion of artificial urine every minute for a total of 10 minutes. The artificial urine composition proposed by Königsberger and colleagues 19 was used, and the pH of this solution was adjusted to 5.5.
The concentration of released isoprenaline was then determined by measuring the light absorbance at a wavelength of 291 nm using a SPECTROstar Nano instrument (BMG Labtech, Germany) in 10 × 10 mm optical path quartz glass cuvettes (Hellma Analytics, Germany). The release profile of isoprenaline was determined for samples loaded with the drug from aqueous solutions of isoprenaline at concentrations of (m:v) 1%, 2.5%, 4%, and 5%. Measurements were performed on three independent guidewire samples for each amount of drug loaded (n = 3).
Observation of the guidewire surface
The guidewire surface was observed before and after the synthesis of the hydrogel coating, after loading the coating with isoprenaline, and after drug release using stereoscopic microscopy (Leica M125C; Leica Microsystems, Germany) and scanning electron microscopy (SEM).
Before SEM observations, samples were cut into 1 cm pieces and fixed to microscope stubs with conductive carbon adhesive tape (Agar Scientific). Next, the samples were coated with a 6 nm layer of gold–palladium (Quorum Q150 TS, Quorum Technologies Ltd.). Subsequently, the samples were examined using a Hitachi SU8230 ultrahigh-resolution SEM (Hitachi High-Technologies Corporation) with secondary electron detectors at an accelerating voltage (10 μA). The samples were imaged at a magnification of 80 × .
Determining the friction coefficients of the guidewire
The friction coefficients between the guidewire and porcine ureter were measured using a tribometer developed for this purpose and described elsewhere. 20 In brief, a 20 cm long sample of guidewire was placed between two polymer elements to which the porcine ureter was glued. The device chamber was filled with artificial urine, and measurements were performed at room temperature. Before starting the measurements, each sample was immersed in deionized water for 10 seconds. The product was set to move in a parallel plane at a constant speed of 1 cm/s.
The force of friction between the sample and the animal tissue was registered using a force meter KM202K 3N (Megatron, Munich, Germany) and acquired in a personal computer using an A/D converter DAQPad-6015 (National Instruments, Austin, TX, USA). The signal from the force meter was collected using an application made in the LabView 8 (National Instruments) environment for 3 seconds before and 7 seconds after the start of product movement. The friction coefficient was calculated as the ratio of the measured friction force to the normal force (which is the difference between the force of gravity of the pressing element and buoyancy force). The maximum friction force measured at the beginning of product movement was used to determine the static friction coefficient.
The dynamic friction coefficient was determined using the mean value of the friction force after the peak at the beginning of motion. Three samples of each product were used to determine the coefficient of friction (n = 3). The normality of the distribution of static and dynamic friction coefficient measurement results was tested using the Kolmogorov−Smirnov test (p < 0.05). The difference between the mean values of measured parameters was tested in one-way analysis of variance (p < 0.05) with post hoc Tukey's test for multiple comparisons. Statistical analysis was performed using OriginPro 8 software (OriginLab Corporation, Northampton, MA, USA).
IsoWire assembly
This coated isoprenaline-loaded guidewire was inserted into the 5F open-ended ureteral catheter. A 2 cm length of the atraumatic “floppy tip” was left protruding from the open-ended ureteral catheter. The distal 10 cm, which includes the “floppy tip,” and the junction of the open-ended catheter and guidewire were also covered with hydrogel (Fig. 1C). This prevents the inadvertent release of isoprenaline and reduces friction, allowing easier cannulation of the ureteral orifice and seamless steering into the pelvicaliceal system.
Animal studies
Animal model
Porcine studies were approved by the local animal ethics committee (AEC 020_011). Three female pigs (white, Landrace breed, weight of 50–60 kg) were transported to the study facility and housed for 7 days to allow an adequate acclimatization period. The pigs were fed a standard diet for up to 12 hours before the investigation but had free access to water. They were premedicated with a diazepam patch then induced with zoletil (3 mg/kg IM), medetomidine (0.06 mL IM), and butorphanol (0.15 mg/kg IM)—and intubated and mechanically ventilated (GE Healthcare S5 Avance).
Anesthesia was maintained using isoflurane 1.5% to 3% in oxygen. They were prehydrated with an initial bolus of saline administered at 90 mL/(kg·h) for 5 minutes, and hydration was maintained throughout the procedure with saline at 10 mL/(kg·h) through an ear vein. A central venous catheter and femoral arterial catheter were inserted under ultrasound guidance to allow blood sampling and invasive blood pressure and heart rate (HR) monitoring (Fig. 2).
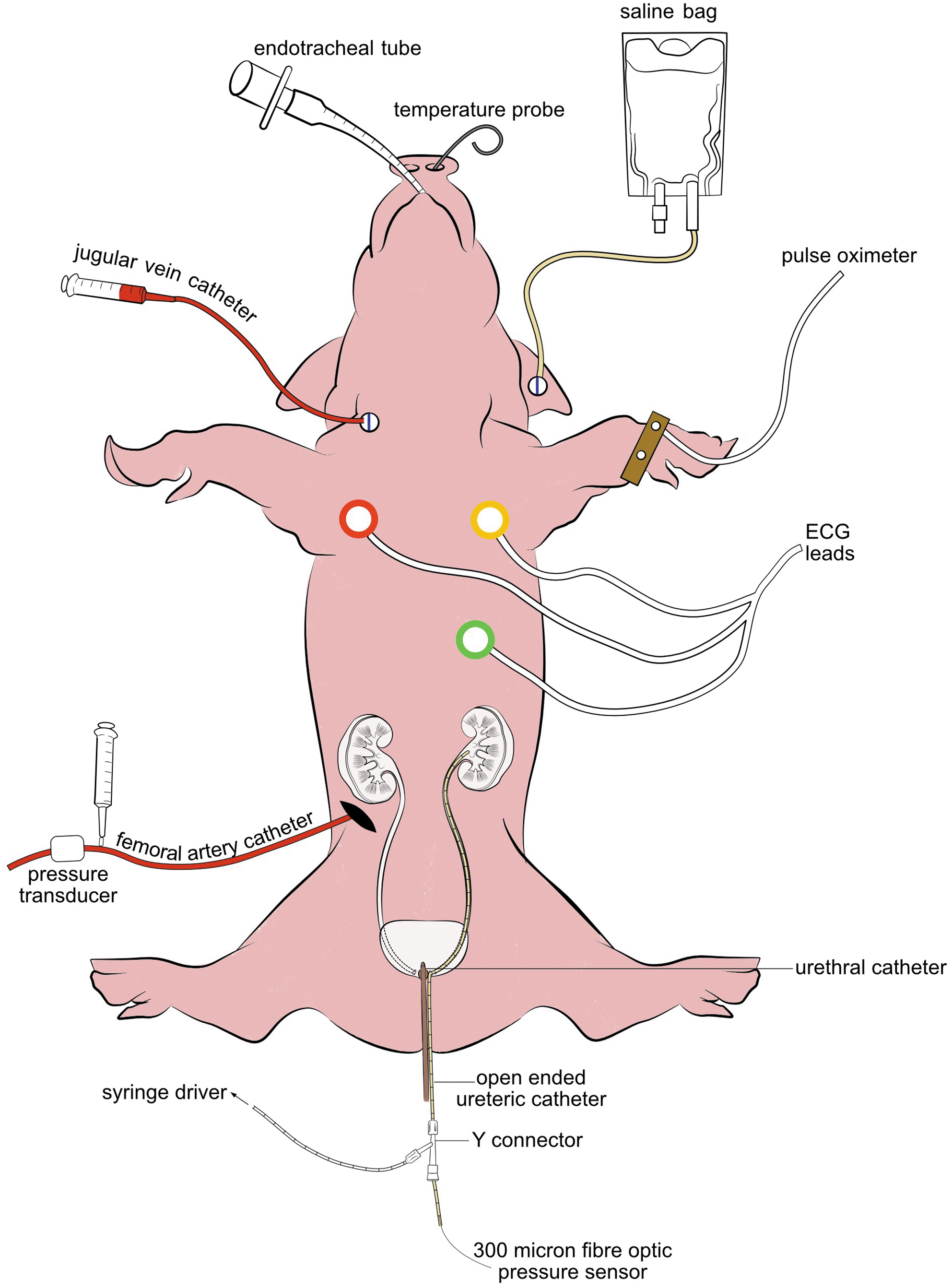
Graphical demonstration of the animal model. ECG, Electrocardiogram.
Experiment
In the lithotomy position, both ureteral orifices were cannulated with hydrophilic guidewires (0.035 in). An open-ended ureteral catheter was inserted into the left renal pelvis over the second hydrophilic guidewire. The second guidewire was removed, and a Touhy-Borst Y-connector was attached to the end of the open-ended ureteral catheter. Saline irrigation was started at 10 mL/min through the first port, and a 300 μm fiber optic pressure sensor (FISO Technologies Inc., Quebec, Canada) was inserted through the second port for continuous IRP monitoring. Size 10 French urethral catheter kept the bladder empty.
As controls, IRP, HR, and mean arterial pressure (MAP) were measured continuously for 6 minutes during constant saline infusion. For the experiment, the standard guidewire was replaced with the IsoWire with the tip of the IsoWire positioned in the renal pelvis, confirmed on fluoroscopy. The protective ureteral catheter over the IsoWire was then removed to enable isoprenaline release. The IRP, HR, and MAP were measured continuously for 6 minutes. Blood samples were obtained to measure the plasma isoprenaline levels every minute after IsoWire insertion. Once the experiment and control were performed on one side, the same procedure was followed on the contralateral side, after 10 minutes. Ureteroscopy was performed bilaterally to evaluate ureteral injuries.
Results
The IsoWire surface before and after synthesizing the hydrogel coating, after loading the coating with isoprenaline, and after drug release is shown in Figure 3. Immersing the guidewire in an artificial urine solution degraded the hydrogel coating, probably because of the ionic strength of the solution.

Stereomicroscopic (upper row) and scanning electron microscopy (lower row) images of guidewire with and without hydrogel coating.
Analysis of the isoprenaline release profile from the hydrogel-coated guidewire (Fig. 4) showed that most (63.9 ± 5.9%) of the loaded drug mass was released in the 1st minute, and almost all the drug was released exponentially in the first 4 minutes. These data allowed us to determine the concentration of isoprenaline solution used to load the drug into the hydrogel coating to obtain a given amount of drug released in the 1st minute.
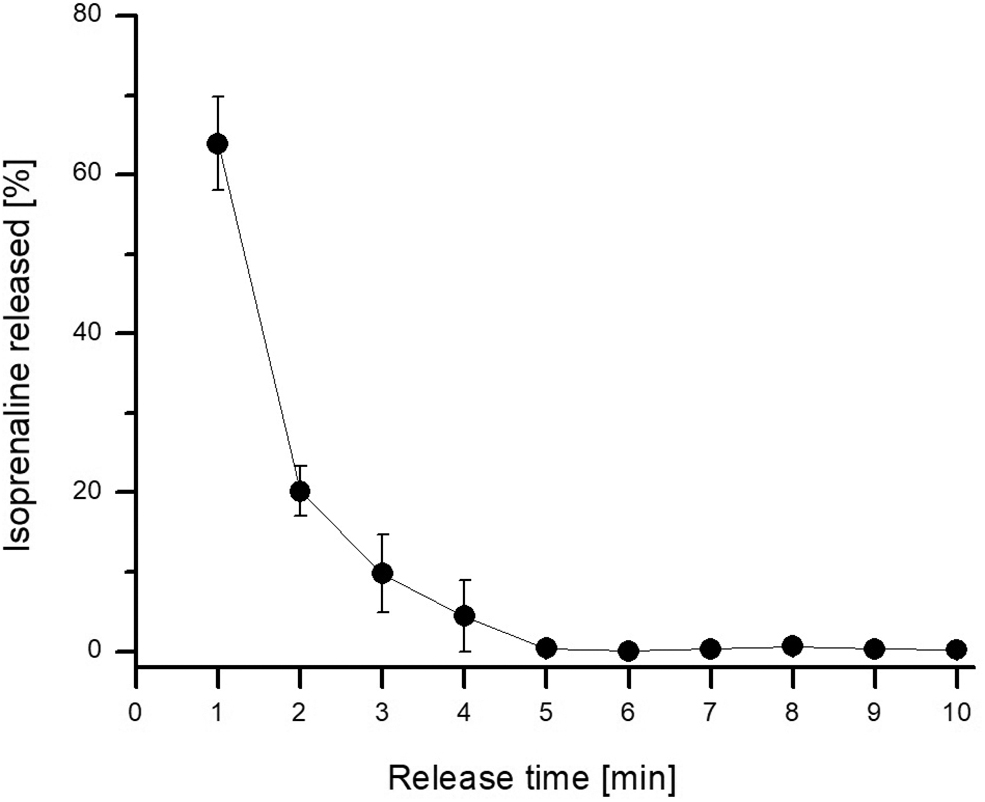
Isoprenaline release profile from hydrogel-coated guidewire into artificial urine solution.
The effect of the hydrogel coating on the friction between the guidewire and urinary tract tissue is shown in Figure 5. The frictional force was significantly reduced when the surface of the product was modified with hydrogel coating. The calculated static and dynamic friction coefficients were 0.084 ± 0.028 and 0.066 ± 0.020, respectively, for the guidewire without coating, and 0.012 ± 0.005 and 0.006 ± 0.002, respectively, for the product with hydrogel coating. Static and dynamic friction coefficients were statistically different (p < 0.05) for the uncoated and coated guidewires. The hydrogel coating led to a 7-fold reduction in the static friction coefficient and an 11-fold reduction in the dynamic friction coefficient.
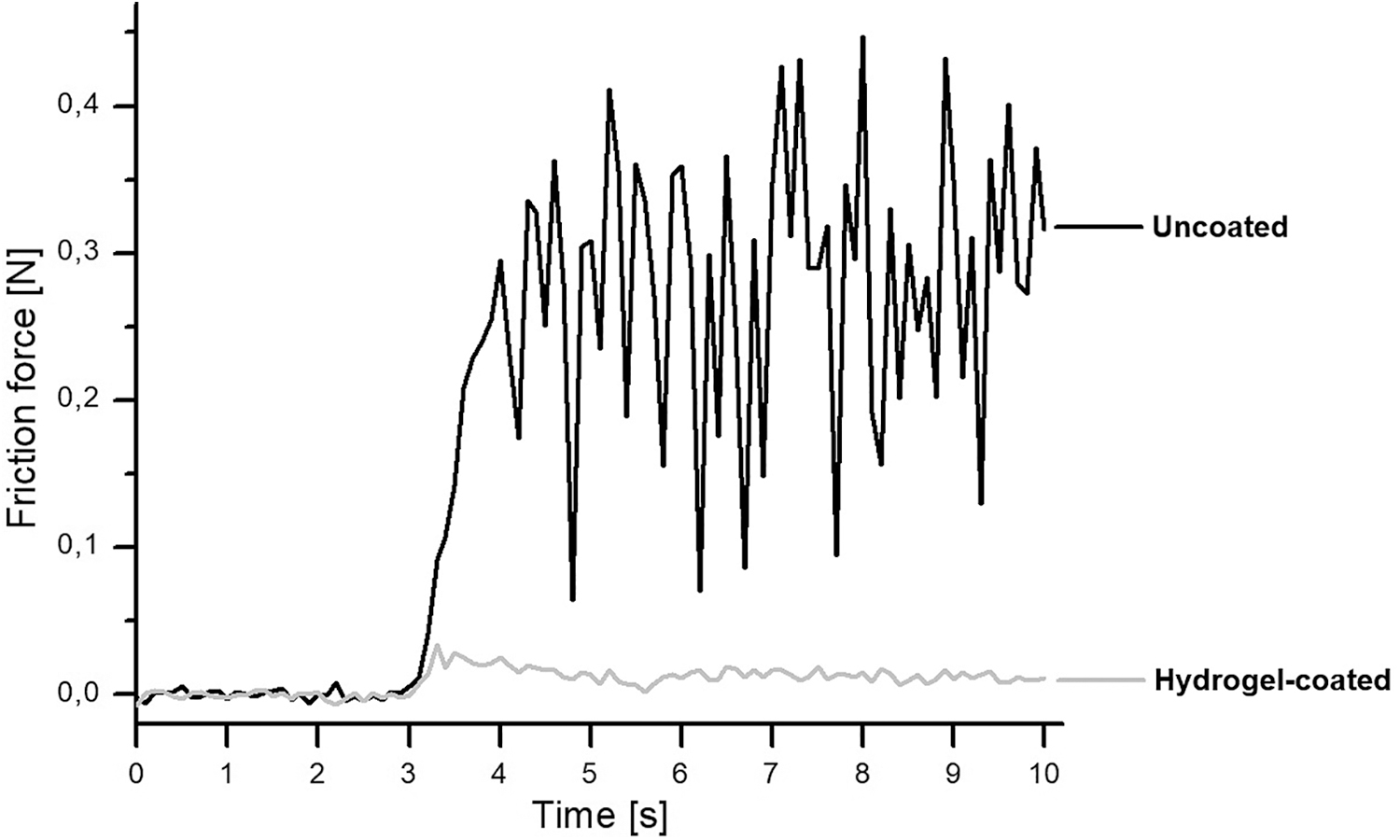
Frictional force between the guidewire and porcine urethra showing guidewire movement commenced in the third second of the experiment. N = newton.
The animal study comprised six renal units. One renal unit was excluded because of failed access to the renal pelvis because of a tortuous proximal ureter. In this case, both the standard guidewire and the IsoWire could not be advanced into the renal pelvis and it was decided not to attempt other described techniques that may inevitably have affected the IRP readings in that renal unit. Therefore, these studies were conducted in five renal units. The use of the IsoWire resulted in a decrease in the IRP in all renal units (Table 1).
Outcome Measures for the Entire Sample
Dose refers to the amount of isoprenaline released from the wire in the first minute.
HR = heart rate; IRP = intrarenal pressure; MAP = mean arterial pressure.
The percentage reduction in IRP was related to the isoprenaline dose. The largest percentage reduction was observed for the IsoWire, which released 10 μg (25.1% reduction). The average percentage reduction in IRP was 8.95% and 21.3% in the IsoWire that released 5 and 7.5 μg of isoprenaline in the 1st minute, respectively. In the IsoWires that released 5 and 7.5 μg of isoprenaline in the 1st minute, no changes in MAP or HR were observed. In the IsoWire, which released 10 μg of the drug in the 1st minute, no change in MAP was observed; however, a marked increase in HR was observed. Plasma isoprenaline levels were all below the level of quantification. No ureteral lesions were identified by ureteroscopy.
Discussion
Guidewires, initially developed for vascular procedures, have become an essential tool in the armamentarium of endourologists. Many guidewires are available commercially, but they all serve a single purpose to function as a track over which catheters, ureteral stents, and endoscopes can be passed into the collecting system of the kidney or bladder. The right guidewire can lead to improved outcomes and reduced procedure times. Although several drug-eluting implants or medical devices have been reported, 21,22 Forman et al. described the only drug-eluting guidewire. The “Adenowire” allowed for the release of pharmacologic amounts of adenosine directly into the microvasculature during a percutaneous coronary intervention procedure. 23
Here, we introduce the “IsoWire”—a platform guidewire to deliver topical isoprenaline to the genitourinary system. Isoprenaline, a drug that has a structural resemblance to epinephrine, is immediately active upon infusion and has a half-life of 2.5 to 5 minutes. 24 Stimulation of β-adrenergic receptors by isoprenaline activates adenylate cyclase and increases cyclic adenosine monophosphate, which in turn activates protein kinase A, causing ureteral relaxation 25 and subsequently lowering IRP.
Although a similar effect on IRP has been observed in other studies with endoluminal administration of isoprenaline, this is the first study to report the delivery of isoprenaline using a drug-eluting guidewire. Danuser and colleagues showed that intravenous and endoluminal infusion of isoprenaline affected the frequency and amplitude of ureteral contractions. 16 In a porcine model, isoprenaline (0.1 mg/mL) added to the irrigation fluid significantly reduced the increase in renal pelvic pressure during ureterorenoscopy.
Jakobsen et al. performed ureteroscopy in pigs while perfusing the renal pelvis at different rates (0, 4, 8, 12, 16, 25, and 33 mL/min) using either saline alone or saline mixed with isoprenaline at 0.1 μg/mL. At all irrigation rates, the perfusion with isoprenaline reduced IRP, with the maximum percentage reduction (52 to 38 mmHg—27%, p < 0.001) at a 4 mL/min infusion rate. 14 A similar effect on IRP with endoluminal isoprenaline (irrigation with isoprenaline at 0.1 mg/mL) was reproduced in humans, without any adverse effects. 26
The in vitro results showed the ability of the hydrogel coating to load isoprenaline and release it quickly. In addition, we have shown that the hydrogel coating reduces the friction between the guidewire and urinary tract tissues. Our animal studies trialed three different doses, and we found that IRP was reduced at all three concentrations. The IsoWire that released 10 μg in the 1st minute showed potential systemic effects, as reflected in an increase in HR after insertion. The IsoWire that released 5 and 7.5 μg in the 1st minute recorded no changes in MAP, HR, Electrocardiogram, or any measurable isoprenaline levels in the blood.
Apart from only reducing IRP, there are several other potential clinical applications for the IsoWire. The majority of endourologists routinely use a ureteral access sheath (UAS) in the management of renal stones. 27 Owing to the failure rates of UAS insertion 28 –31 and the possible ureteral injuries during UAS placement, 32,33 surgeons opt to passively dilate the ureter by prestenting the patient with a ureteral stent and returning 1 to 2 weeks later to manage the stone. The use of our IsoWire may result in ureteral muscle relaxation rendering easier insertion of an UAS in the first surgical sitting and, therefore, reducing the need to prestent the ureter. It may also promote the practice of sheathless RIRS.
Conclusions
The IsoWires, which releases 5 and 7.5 μg of isoprenaline in the 1st minute, appear to be safe and effective in reducing the IRP. The IsoWires have passed bench testing for lubricity, adherence, integrity, and tracking. Further studies are needed to establish how long this promising pressure-modulating effect will be sustained during RIRS and whether the ureteral relaxation will render easier insertion of an UAS, reduce IRP during sheathless RIRS, or even promote the practice of sheathless RIRS.
Footnotes
Acknowledgments
We acknowledge the invaluable contribution of several individuals. (1) Stephan Dijkstra, Peter Magyar, and Russell Maurer of Wismed CC. (2) Peter Zilla, Rose Boltman, and Helen Ilsley of Strait Access Technologies. (3) Janet McCullum, John Chipangura, Tashie Makwavarara, and Thireshni Chetty at the University of Cape Town Research Animal Facility. (4) Michelle Henry, at the Numeracy Centre, Centre for Higher Education Development at the University of Cape Town. (5) Val Myburgh.
Authors' Contributions
All authors contributed to the design of the study, analysis of the results, and drafting and revision of the article.
Author Disclosure Statement
The authors declare that they have no conflict of interest.
Funding Information
This study was supported by the National Center for Research and Development (Project No.
