Abstract
Introduction:
Urosepsis is a serious potential complication of ureteroscopic procedures for stone disease, yet the risk factors for this complication are not well characterized. The purpose of this systematic review with meta-analysis was to identify potential risk factors for urosepsis after ureteroscopy (URS) for stone disease.
Materials and Methods:
We performed systematic searches of Medline, Embase, and the Cochrane Central Register of Controlled Trials for studies reporting at least one prospectively defined risk factor for urosepsis after URS. Studies that only reported rates of isolated fever, urinary tract infection (UTI), or pooled infectious complications were excluded. The risk factors evaluated in this review were age, sex, body mass index, diabetes mellitus, ischemic heart disease, recent UTI, pyuria, hydronephrosis, stone history, stone size, preoperative stent placement, preoperative positive urine culture, and procedure time. A random effects meta-analysis model with inverse variance weighting was used where the statistic of interest was the odds ratio for dichotomous variables and the mean difference for continuous outcomes.
Results:
In 13 studies (5 prospective) with 5597 patients, the pooled incidence of postoperative urosepsis was 5.0% (95% confidence interval: 2.4–8.2). Six risk factors were statistically associated with increased postoperative urosepsis risk—preoperative stent placement (odds ratio = 3.94, p < 0.001, 6 studies), positive preoperative urine culture (odds ratio = 3.56, p < 0.001, 6 studies), ischemic heart disease (odds ratio = 2.49, p = 0.002, 2 studies), older age (mean difference = 2.7 years, p = 0.002, 6 studies), longer procedure time (mean difference = 9 minutes, p = 0.02, 1 study), and diabetes mellitus (odds ratio = 2.04, p = 0.04, 6 studies).
Conclusions:
Current evidence suggests that among patients undergoing URS for treatment of stone disease, the risk of postoperative urosepsis was 5.0%. Older age, diabetes mellitus, ischemic heart disease, preoperative stent placement, a positive urine culture, and longer procedure time were associated with increased postoperative urosepsis risk. These results will assist urologists with preoperative risk stratification before ureteroscopic procedures.
Introduction
Approximately 10%
URS is the most common interventional treatment for ureteral and renal stones. Ureteroscopic treatment options include using a basket to extract stone fragments and/or using a laser to dust stone fragments. Due in part to high success rates with these techniques, 2 URS is increasingly being used in higher-risk patients, which may increase procedural risk and the likelihood of postprocedural infectious complications. Previous meta-analyses have identified risk factors for infectious complications after URS for stone disease. 3,4 However, the definition of infectious complications in these reviews included a spectrum of diagnoses ranging from isolated fever or urinary tract infection (UTI) to urosepsis. Since patient prognosis and therapeutic strategies differ by diagnosis where urosepsis confers the greatest patient risk with potential for extended hospitalization, unplanned intensive care admission, or death, 5 identification of risk factors specifically for postoperative urosepsis may assist with risk stratification before URS. The purpose of this systematic review with meta-analysis was to identify potential risk factors specifically for urosepsis after URS for stone disease.
Methods
This systematic review and meta-analysis was reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA). 6 A protocol was developed for our review and prospectively registered at research registry website (reviewregistry996).
Two researchers (L.M. and D.F.) experienced in systematic reviews performed searches of Medline, Embase, and the Cochrane Central Register of Controlled Trials from inception to September 30, 2020 to identify studies of patients undergoing URS for renal stones. Manual searches were performed using the Directory of Open Access Journals, Google Scholar, and the reference lists of included articles and systematic reviews. The search strategy was initially developed for Medline (Table 1) and subsequently adapted for the other databases. During screening, we excluded review articles, editorials, commentaries, conference proceedings, case series with <10 patients, gray literature, and studies that reported outcomes from mixed urologic procedures. No language or publication date restrictions were used.
MEDLINE Search Strategy
“*” represents a wildcard symbol used in a search query to represent end truncation.
The same reviewers independently reviewed study records and a final list of studies for full-text review was determined after record comparisons and discussion. We included both cohort and case–control studies that provided data to determine the association between at least one prospectively defined risk factor and the postoperative urosepsis rate. Studies that only reported rates of isolated fever, UTI, or pooled infectious complications were excluded. We pilot-tested a database to align our data extraction methods with data typically reported in the literature. We extracted article metadata, study characteristics, patient characteristics, procedural outcomes, and postoperative urosepsis rates. The risk factors that were evaluated in this systematic review were age, sex, body mass index, diabetes mellitus, ischemic heart disease, recent UTI, pyuria, hydronephrosis, stone history, stone size, preoperative stent placement, preoperative positive urine culture, and procedure time. The risk of bias among included studies was assessed using the Newcastle-Ottawa scale. 7
A statistician author (L.M.) performed a random effects meta-analysis with inverse variance weighting where the statistic of interest was the odds ratio for dichotomous variables and the mean difference for continuous outcomes. Individual study results and pooled meta-analysis data for key outcomes were displayed with forest plots. We used the I 2 statistic to estimate heterogeneity of effects across studies with values of ≤25%, 50%, and ≥75% representing low, moderate, and high inconsistency, respectively. 8 We were unable to assess the potential for publication bias or to explore sources of heterogeneity with metaregression owing to an insufficient number of available studies. All tests were two sided and the threshold for statistical significance was p < 0.05. Statistical analyses were conducted using Stata v. 16.1 and Review Manager v5.3.
Results
Among 251 articles identified in the searches, 13 met the eligibility criteria and were included in the systematic review. 9 –21 A PRISMA flow diagram depicting the study identification and selection results is shown in Figure 1. Among 13 studies (5 prospective) with 5597 patients, mean patient age ranged from 43 to 77 years (median 51 years) and 64% were men. Patient characteristics and risk factors were reported inconsistently among studies (Table 2). Seven studies were judged to have low risk of bias and six were intermediate risk. The most commonly identified risks of bias among studies were attributable to retrospective patient enrollment, inconsistent risk factor reporting, and variability in postoperative urosepsis diagnostic criteria (Table 3).
CONSORT flow diagram.
Study and Patient Characteristics Among Included Studies
Median.
Patients with positive urine culture were excluded from the study.
Patients without preoperative stent were excluded from the study.
Reported using number of procedures in denominator.
BMI = body mass index; DM = diabetes mellitus; HN = hydronephrosis; IHD = ischemic heart disease; P = prospective; R = retrospective; UC = ureteral calculi; UTI = urinary tract infection.
Risk of Bias Assessment with Newcastle-Ottawa Scale Among Included Studies
Selection comprised representativeness of exposed cohort, selection of nonexposed cohort; ascertainment of exposure, and demonstration that outcome of interest was not present at start of study. Comparability comprised study controls for baseline comorbidities and disease severity. Outcome comprised assessment of outcome, was follow-up long enough for outcomes to occur, and adequacy of follow-up of cohorts. Studies classified as high (1–3 stars), intermediate (4–5 stars), or low (6–9 stars) risk of bias.
Mean procedure time ranged from 24 to 97 minutes among studies; length of hospitalization was rarely reported. Postoperative urosepsis rates ranged from 0.2% to 17.8% (Table 4) and urosepsis diagnostic criteria varied among studies (Table 5). The pooled incidence of postoperative urosepsis was 5.0% (95% confidence interval: 2.4–8.2) (Fig. 2). Among three studies reporting urosepsis severity, the pooled incidence was 71% for urosepsis, 5% for severe urosepsis, and 24% for septic shock. Among 13 risk factors evaluated, 6 were statistically associated with increased postoperative urosepsis risk—preoperative stent placement (odds ratio = 3.94, p < 0.001, 6 studies), positive preoperative urine culture (odds ratio = 3.56, p < 0.001, 6 studies), ischemic heart disease (odds ratio = 2.49, p = 0.002, 2 studies), older age (mean difference = 2.7 years, p = 0.002, 6 studies), longer procedure time (mean difference = 9 minutes, p = 0.02, 1 study), and diabetes mellitus (odds ratio = 2.04, p = 0.04, 6 studies) (Table 6). Forest plots displaying the association of patient risk factors with postoperative urosepsis rates are provided for the variables reported in two or more studies (Figs. 3 –7).
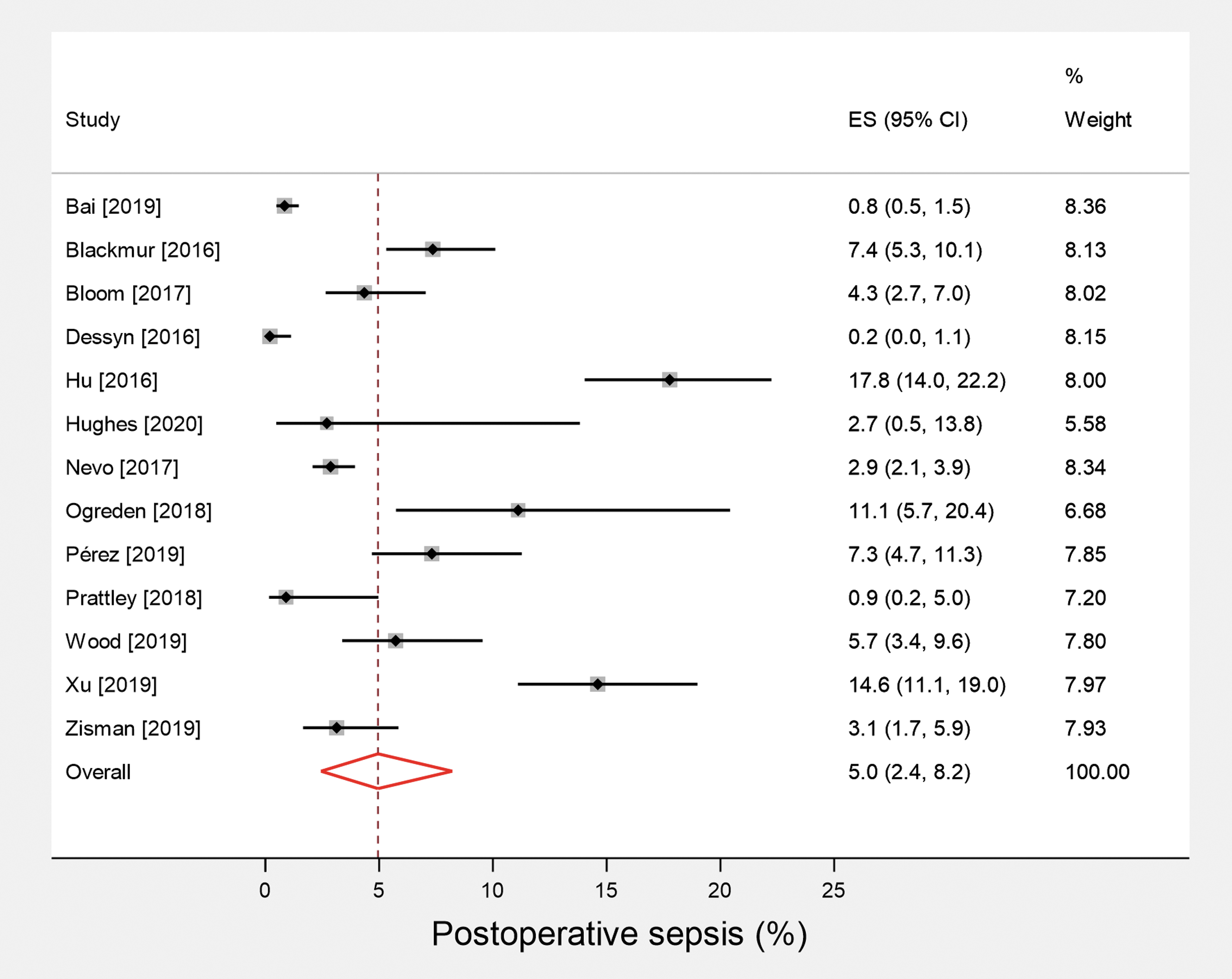
Forest plot of postoperative urosepsis risk in patients undergoing ureteroscopy. The postoperative urosepsis rate and 95% CI are plotted for each study. The pooled mortality rate (diamond apex) and 95% CI (diamond width) are calculated using a random effects model. Pooled urosepsis risk = 5.0%. Heterogeneity: I 2 = 95%; p < 0.001. CI = confidence interval; ES = effect size.
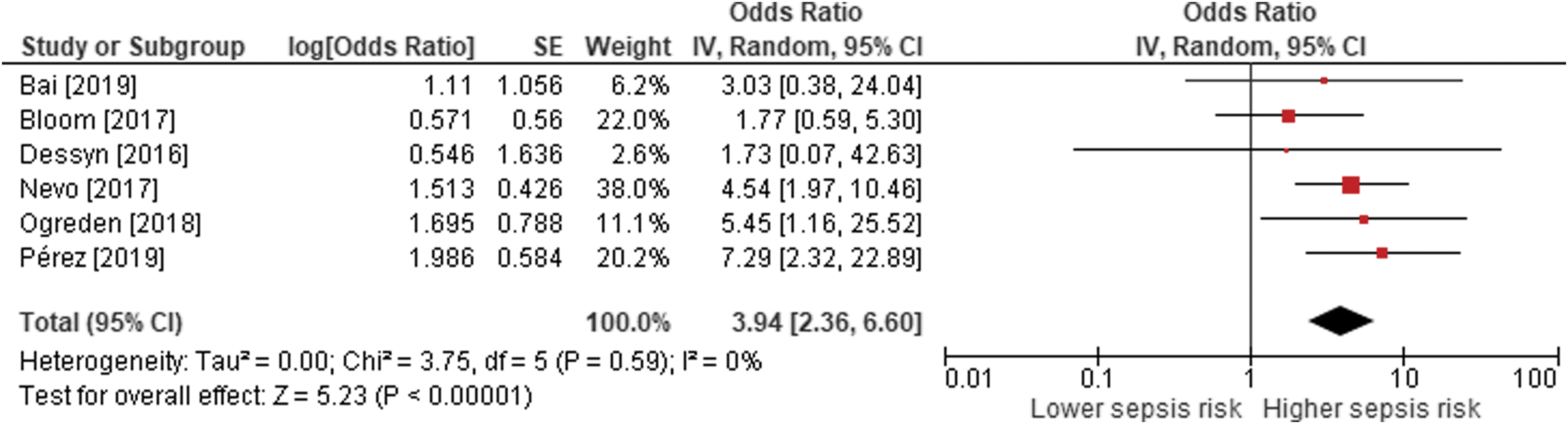
Forest plot of the association between postoperative urosepsis risk in patients undergoing ureteroscopy with preoperative stent. The odds ratio and 95% CI are plotted for each study. The pooled odds ratio (diamond apex) and 95% CI (diamond width) are calculated using a random effects model. Pooled odds ratio = 3.94; p < 0.001. Heterogeneity: I 2 = 0%; p = 0.59. IV = intravenous.
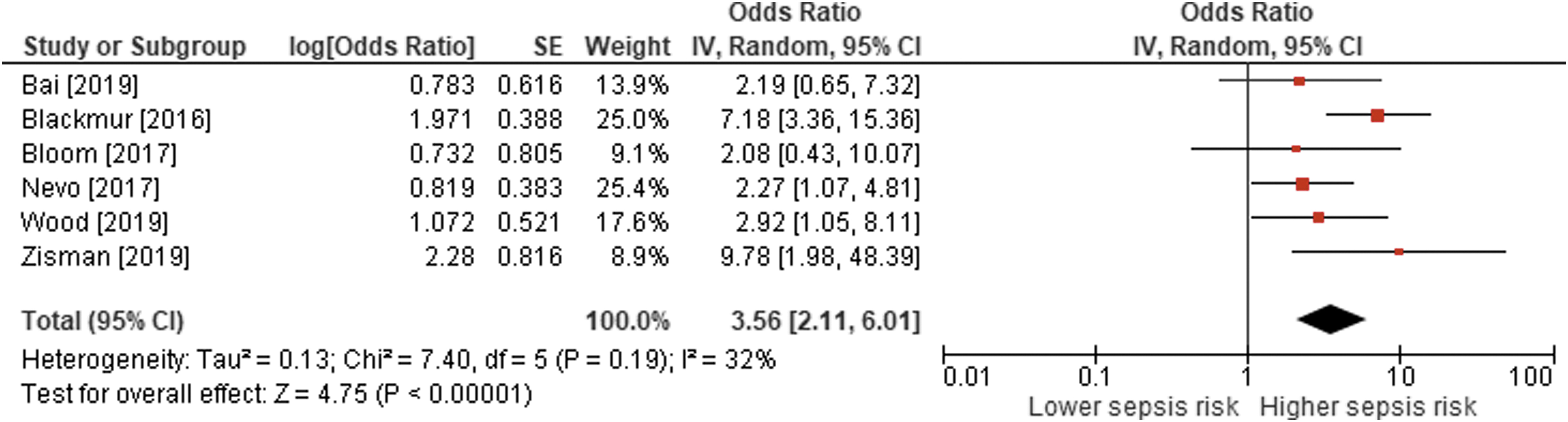
Forest plot of the association between postoperative urosepsis risk in patients undergoing ureteroscopy with preoperative positive urine culture. The odds ratio and 95% CI are plotted for each study. The pooled odds ratio (diamond apex) and 95% CI (diamond width) are calculated using a random effects model. Pooled odds ratio = 3.56; p < 0.001. Heterogeneity: I 2 = 32%; p = 0.19.

Forest plot of the association between postoperative urosepsis risk in patients undergoing ureteroscopy with ischemic heart disease. The odds ratio and 95% CI are plotted for each study. The pooled odds ratio (diamond apex) and 95% CI (diamond width) are calculated using a random effects model. Pooled odds ratio = 2.49; p = 0.002. Heterogeneity: I 2 = 0%; p = 0.51.
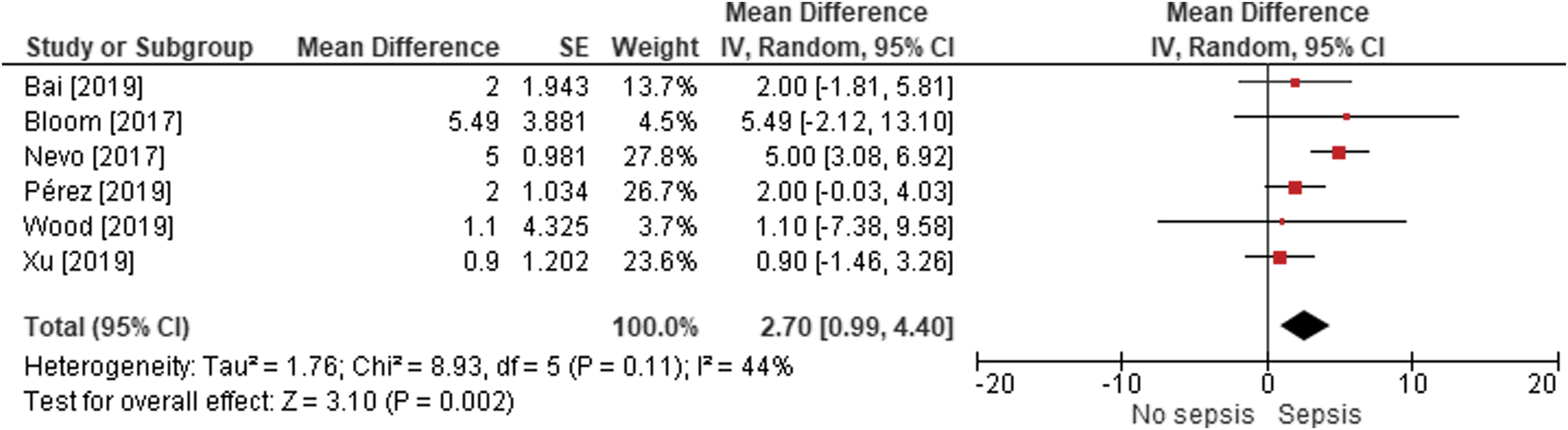
Forest plot of the association between postoperative urosepsis risk in patients undergoing ureteroscopy with patient age. The mean difference and 95% CI are plotted for each study. The pooled mean difference (diamond apex) and 95% CI (diamond width) are calculated using a random effects model. Pooled mean difference = 2.7 years; p = 0.002. Heterogeneity: I 2 = 44%; p = 0.11.
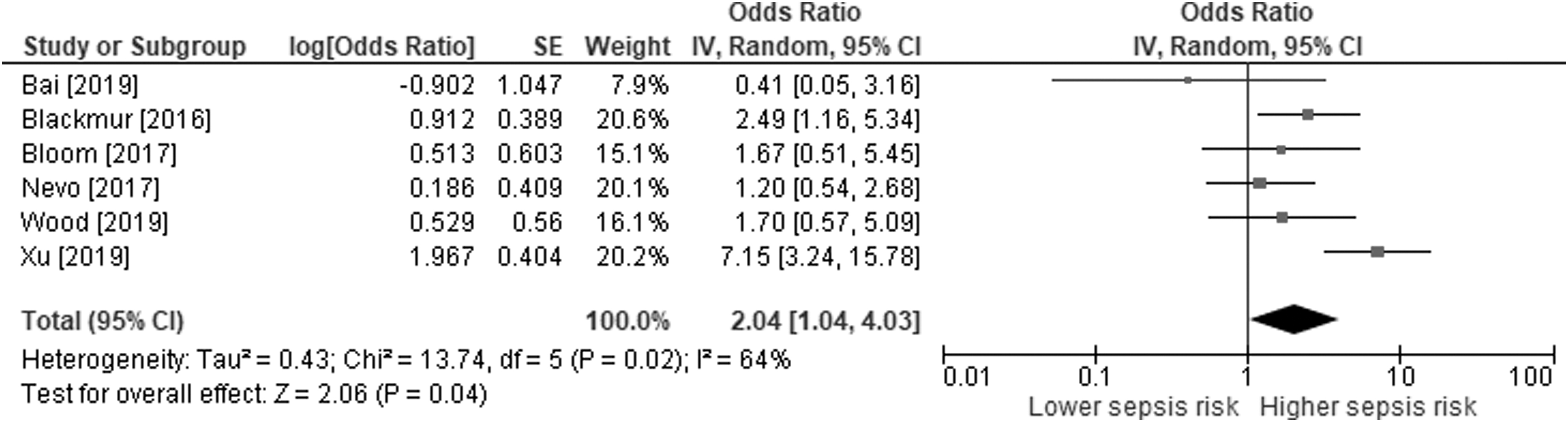
Forest plot of the association between postoperative urosepsis risk in patients undergoing ureteroscopy with diabetes mellitus. The odds ratio and 95% CI are plotted for each study. The pooled odds ratio (diamond apex) and 95% CI (diamond width) are calculated using a random effects model. Pooled odds ratio = 2.047; p = 0.04. Heterogeneity: I 2 = 64%; p = 0.02.
Perioperative Details Among Included Studies
Median.
IV = intravenous.
Urosepsis Diagnostic Criteria Used in Included Studies
Mandatory criterion.
Data unreported.
Urosepsis defined as documented or suspected infection in addition to other criteria set forth by the 2001 SCCM/ESICM/ACCP/ATS/SIS International Sepsis Definitions Conference.
GCS = Glasgow Coma Scale; SBP = systolic blood pressure; WBC = white blood cell.
Summary of Risk Factors for Postoperative Urosepsis in Patients Undergoing Ureteroscopy for Stone Disease
Sorted from lowest to highest p-value for effect size, with statistically significant risk factors in bold font.
Not calculable because of insufficient number of studies.
CI = confidence interval.
Discussion
Because of the considerable clinical and economic burden of urosepsis, identification of risk factors for this complication after URS would be particularly informative to patients, physicians, and health care policy makers. Although others have reported risk factors for generalized infectious complications, 3,4 this is the first systematic review with meta-analysis to specifically investigate risk factors for postoperative urosepsis after URS. The overall risk of postoperative urosepsis was 5.0% in this review. Older age, comorbidities such as diabetes mellitus and ischemic heart disease, preoperative stent placement, positive urine culture, and longer procedure time were independently associated with increased postoperative urosepsis risk. This information is important for urologists as it can assist with preoperative risk stratification to provide individualized treatment recommendations for each patient as well as to inform postoperative surveillance regimens. Furthermore, it is plausible that early identification and appropriate management of high-risk patients may decrease postoperative morbidity and reduce health care utilization, a hypothesis deserving of further study.
The current systematic review is unique in that we identified risk factors for urosepsis after URS, whereas others have reported risk factors for generalized infectious complications that included less severe complications such as isolated fever or UTI. 3,4 However, the risk factors for urosepsis identified in this review are similar to those previously identified for generalized infectious complications. Sun and colleagues 4 reported that a positive preoperative urine culture was the strongest risk factor (odds ratio = 2.95, p < 0.01) for generalized infectious complications after URS, with female sex, diabetes mellitus, preoperative stent placement, and longer procedure time also demonstrating statistical significance. Ma and colleagues 3 performed a similar review where they identified female sex, preoperative stent placement, diabetes mellitus, positive preoperative urine culture, and longer procedure time as the key determinants of postoperative urosepsis risk. In our review, positive urine culture, preoperative stent placement, and older age were the strongest determinants based on reporting in the greatest number of studies and without influence by significant heterogeneity. Based on the collective results of these three reviews, positive preoperative urine culture and preoperative stent placement appear to be key determinants of generalized infectious complications as well as for urosepsis specifically. Although ischemic heart disease, procedure time, and diabetes mellitus were also associated with increased postoperative urosepsis risk in this review, the strength of this evidence was low because of a limited number of studies for ischemic heart disease and procedure time, and the presence of significant heterogeneity for diabetes mellitus.
Patients with a positive urine culture often received targeted microbial therapy followed by confirmation of a second negative culture before URS; however, reporting among studies in this review was inconsistent. Based on this limited evidence, it is plausible that prior antibiotic exposure may confer antibiotic resistance and greater infection risk after URS. In addition, a second negative test may not indicate absence of infection since negative midstream urine cultures are possible where infected urine is present proximal to the obstructing stone. 22 Although infectious stones (struvite) may play an important role in this specific patient population, stone treatment may lower UTI risk even in nonstruvite stones. 23 In higher-risk patients, it may be prudent to tailor preoperative antibiotics to cover both gram positive and multidrug-resistant organisms and also to employ intraoperative stone cultures since they may more accurately determine bacterial pathogens in infectious complications after endourologic procedures. 24 Although preoperative stent placement facilitates endoscopic management of stones, improves stone-free rates, and reduces intraoperative complications, 25 bacterial colonization and bacteriuria is an inevitable process that begins once a stent is inserted and progresses with prolonged stent dwelling time, particularly when stent dwelling times exceed 30 days. 15 These findings suggest that patients with a recent positive urine culture or ureteral stent may benefit from enhanced intraoperative and postoperative surveillance and, if infectious symptoms arise, early institution of broad-spectrum antibiotics followed by targeted therapy pending specific pathogen identification from culture specimens. Although older age, ischemic heart disease, diabetes mellitus, and longer procedure time were also associated with increased postoperative urosepsis risk, a clear explanation for these associations and implications for patient risk stratification remain unclear.
This meta-analysis has certain limitations pertaining to the quality of studies available for analysis that may influence interpretation. First, patient characteristics were reported inconsistently and, consequently, the number of studies that adequately reported urosepsis risk factors was limited. Second, the definitions of postoperative urosepsis varied somewhat among studies and symptom surveillance ranged from 1 to 30 days. Third, we were unable to evaluate the potential for publication bias, perform multivariate analyses, or perform metaregression to determine whether study-level factors may have influenced the observed associations between risk factors and urosepsis rates owing to an insufficient number of studies. Fourth, preoperative stent placement indications and average dwell times were rarely reported, which hindered our ability to analyze these data in greater detail. Finally, studies that used shockwave lithotripsy or percutaneous nephrolithotomy were excluded from this review; thus, the risk factors for postoperative urosepsis after URS identified in this review may not be relevant to other urologic procedures for stone disease.
Conclusion
Current evidence suggests that among patients undergoing URS for treatment of stone disease, the risk of postoperative urosepsis was 5.0%. Older age, diabetes mellitus, ischemic heart disease, preoperative stent placement, a positive urine culture, and longer procedure time were associated with increased postoperative urosepsis risk. These results may assist urologists with preoperative risk stratification before ureteroscopic procedures.
Footnotes
Authors' Contributions
N.B., L.M., S.B., and B.C. conceived and designed the study; L.M. and D.F. (acknowledged) performed the database search, study selection, and data extraction; L.M. conducted the statistical analysis and drafted the article; N.B., L.M., S.B., B.C., and B.H.C. reviewed and revised the article; all authors interpreted data for the study, read, and approved the final article.
Acknowledgment
We thank David Fay, PhD (Miller Scientific), for assistance with the literature review and data extraction.
Author Disclosure Statement
Drs. Bhojani, Miller, and Chew report consultancy with Boston Scientific. Dr. Bhattacharyya and Mr. Cutone report employment with Boston Scientific.
Funding Information
This study was supported by Boston Scientific.
