Abstract
Introduction:
To assess the interest of a new sphincter preserving anastomosis technique for continence recovery after robot-assisted laparoscopic radical prostatectomy (RALP).
Materials and Methods
: We performed a monocentric single-operator study on 187 consecutive RALP. Patients were divided into two groups: Group 1 (standard anastomosis, until December 2017) and Group 2 (subsphincteric anastomosis [SSA], since January 2018). The SSA consisted in respecting the sphincteric sleeve during the anastomosis suturing only the internal layer of the urethra with the bladder and thereby avoiding the loss of sphincteric length induced by the suture. Pre-, intra-, and postoperative data were prospectively collected and compared. Criteria of continence were as follows: no pad use and complete absence of leakage at catheter removal at 1 month and 1 year.
Results:
The two groups were comparable in terms of prostate-specific antigen, gland volume, and Gleason score. In Group 2 (SSA), we observed a complete continence recovery in 75.6% at catheter removal (p = 0.0000035), in 82.9% at 1 month (p = 0.000092), and in 97.5% at 1 year (p = 0.028), independently of bladder neck preservation (p = 0.388). There was also a significant difference between the two groups concerning urinary reeducation requirement (p = 0.0006), pad use, and urinary quality of life (p = 0.0000002). No anastomosis complication was reported.
Conclusions:
The SSA significantly improved the rates of immediate, early, and 1-year continence recovery after RALP. These results need further study among larger numbers of patients.
Introduction
The return of urinary continence and its delay after radical prostatectomy (RP) represents a major criterion for patient's quality of life (QOLu), and even more post-RP continence may change even beyond the first year following surgery. 1 The knowledge of the anatomy of the urethral sphincter complex 2 did not differ much from the descriptions made in the 19th century. In the most recent studies, the main differences concerned the cranial extension of the outer rhabdosphincter and the caudal extension of the inner smooth muscle component in the urethral wall. If the function of the inner smooth sphincter is the continence at rest, the function of the outer striated sphincter is the active continence during stress conditions.
Even longer preoperative membranous urethral length had a significant shorter time to stable postoperative continence in multivariate analysis measured by MRI 3,4 or transrectal ultrasound, 5 surgical techniques have mainly focused on preservation during dissection and reconstruction of the musculofascial structures to restore the length of the urethrosphincteric complex, prevent its caudal retraction, and avoid tension on the anastomosis. 6 –11 Ficarra and colleagues 12 reported in a meta-analysis that posterior musculofascial reconstruction with or without anterior reconstruction was associated with a small advantage in urinary continence recovery 1 month after robot-assisted RP (RARP) and that only complete reconstruction was associated with a significant advantage in urinary continence 3 months after RARP (p = 0.04).
Whitson and colleagues 13 demonstrated the solitary role of the bladder neck for urinary continence after traumatic injury to the rhabdosphincter that led to develop techniques of bladder neck sparing 14 or reconstruction during RP. In 2018, Argun et al. 15 described the use of “stay sutures” that aimed, while suturing the urethra, to avoid its retraction and thus the injury to the striated sphincter.
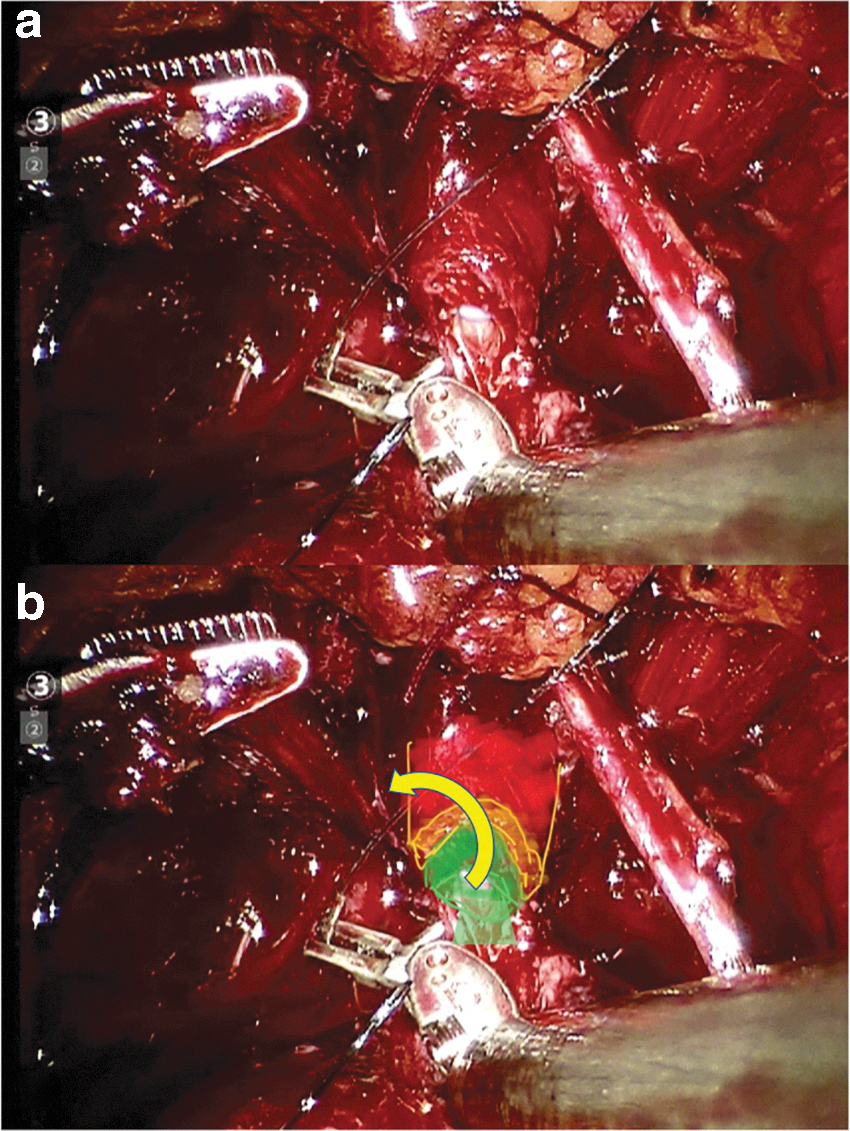
The aim of our study was to assess the impact of a new sphincter preserving anastomosis technique on the immediate (at catheter removal), early (at 1 month), and at 1-year continence recovery stages. This anastomosis was also based on a posterior “stay suture” that allowed the exposition of the internal layer of the urethra (Fig. 1) and its suture with the bladder while respecting the rhabdosphincteric sleeve (external layer). In this way, this suture aimed to avoid the potential loss of sphincteric length induced by the standard suture that concerns the entire thickness of the urethral wall and thus traps some of the proximal sphincteric striated muscular fibers.

Exposition and visualization of the two concentric layers of the urethra. One- Internal layer (concerned by the suture). Two- External layer (that corresponds to the outer striated sphincter and has to be spared by the suture to avoid its injury).
Materials and Methods
We performed an observational retrospective monocentric single operator study. (Exempt from the need for ethical review under French legislation).
Data were prospectively collected during consecutive robot-assisted laparoscopic radical prostatectomies for prostate cancer from May 2010 to March 2019. Da Vinci (Intuitive surgical°) devices were used during the procedures: standard (from May 2010 to March 2016), S (from June 2016 to January 2018), and SI (from January 2018 to March 2019).
Standard urethrovesical anastomosis, using a semicontinuous suturing technique and a double (posterior and anterior) reconstruction and suspension with a 3/0 barbed wire (V-Lock Covidien° [Glycolide, dioxanone, and trimethylene carbonate]) and 17 mm tapercut needles was utilized until December 2017.
Since January 2018, using also a V-Lock Covidien° barbed wire suture, the anastomosis technique has been modified, prepared by a first “stay” stitch placed (outside in) on the posterior part of the urethra before ending its transection. This stitch aimed to avoid the internal invagination of the subsphincteric urethra deeply under the outer striated sphincteric sleeve (external urethral layer) (Fig. 2). The second suture was then used to start the anastomosis on the right side, beginning also with a posterior musculofascial plate reconstruction. The traction on the first stay suture then allowed the passage (inside out) of the needle of the second suture in the subsphincteric mucosal urethra (internal urethral layer). Every passage of the needle in the urethra had to respect the external layer during the anastomosis, aiming to avoid the retraction of the mucosal urethra under the striated sphincter, to expose step by step the internal layer simplifying its suture and to avoid passages of the suture on active muscular sphincteric length (Fig. 3). The suture on the bladder side was not modified with standard outside-in stitches, except for the first stitch (inside-out) of the first suture (on the left side).
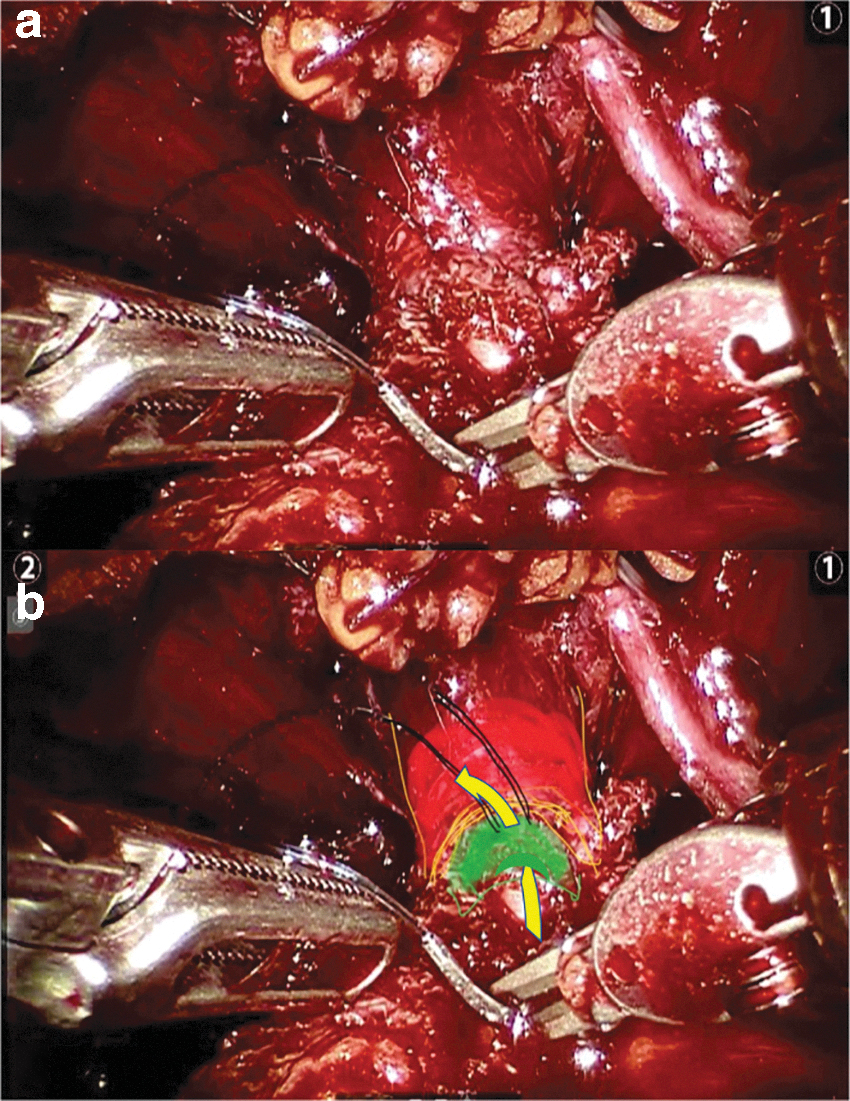
The two sutures ran then anteriorly to be tied on the anterior part of the anastomosis. The subsphincteric anastomosis (SSA) ended by an anterior reconstruction suspension stitch, after watertight anastomosis was ensured by filling the bladder with saline.
Bladder neck was reconstructed in the standard and in the SSA when necessary. A 18F silicon Foley catheter was then placed for eleven days and removed at home by a nurse at 9 am. Every patient was then systematically seen the same day at the clinic by the operator at least 5 hours after to verify the continence status and the postoperative evolution. Cystography was indicated only in case of encountered difficulties while suturing or not perfect watertight anastomosis. In both groups, no urinary reeducation or physiotherapy was realized before surgery, and after surgery, urinary reeducation was only prescribed in case of leakage at the 1 month assessment.
Data collection
Preoperative variables included patient age, preoperative prostate-specific antigen (PSA), biopsy Gleason score, clinical stage, prostate volume, and MRI targets characteristics. Intraoperative parameters were pelvic lymph node dissection, presence of a pudendal artery, nerve sparing, bladder neck preservation, quality of the anastomosis, and difficulties encountered during the procedure. Postoperative variables included length of catheterization, postoperative complications, results of the histologic stage, definitive Gleason score, and surgical margins. Follow-up variables were PSA, time to recurrence, urinary reeducation, time to full continence recovery, number of pads used at catheter removal (immediate continence), and at 1,3, and then every 6 months during 2 years postsurgery.
The criteria of continence were as follows: no pad use and the complete absence of leakage reported by the patient during his clinical evaluation. A urinary-related QOLu analogic scale ranging from 0 (no discomfort) to 10 (unbearable discomfort) was also completed at the 1-year follow-up in response to the question “How bothered do you feel by your actual urinary status?”
The patients were divided into two groups: Group 1 (standard anastomosis, until December 2017) and Group 2 (SSA, since January 2018).
Statistics
Categorical variables were analyzed using chi-square test or Fisher's exact test as appropriate and continuous variables were analyzed using Student's t-test. The limit of statistical significance was defined as p < 0.05.
Results
In this study, 187 patients underwent a robot-assisted laparoscopic RP by a single surgeon. The standard urethrovesical anastomosis concerned 146 patients vs 41 for the SSA. The results (groups' features and evolution of the continence) are resumed in the Table 1.
Population Features and Continence Results, Respectively, for Group 1 with Standard Anastomosis and Group 2 with Subsphincteric Anastomosis
PSA = prostate-specific antigen; QOLu = quality of life.
The mean follow-up was 5.67 years (1.44–10.32) and 1.83 years (1.24–2.32) for Group 1 and 2. The two groups were comparable in terms of PSA (Group 1: 8.4 ng/mL [1.3–52.6] vs Group 2: mean 8.8 ng/mL [2.1–22.47]; p = 0.41), gland volume (Group 1: 42.1 cc [15–140] vs Group 2: mean 45.5 cc [25–153]; p = 0.23), except concerning the age of the patients with a statistical more elderly population in the Group 2 (SSA) (mean 65.9 years vs 67.9 years, p = 0.006), and the histologic findings with 33.6 vs 56.1% of pT3 stage in Groups 1 and 2 (p = 0.0087).
The overall positive surgical margin rates were 8.7 and 22.2% in pT2 and pT3 and the overall mean Gleason score was 6.8. The neurovascular bundles were preserved in 78% and 90% in the Groups 1 and 2 (p = 0.08).
In Group 1 (standard anastomosis), we observed a complete continence recovery in 34.9% at catheter removal, in 48.6% at 1 month, and in 84.24% at 1 year. Bladder neck preservation represented 63% of the procedures in this group with a significant role on continence recovery the day of catheter removal (p = 0.0018) and at 1 year (p = 0.0188) (Fig. 4), and on the potential risk of requiring a secondary incontinence surgery (exclusively by male suburethral sling in this study) (p = 0.017). The causes of no preservation of the bladder neck were prior prostatic surgery (transurethral resection of the prostate …), large median lobe, bladder neck dissection difficulties, and basal tumor location (to limit the risk of positive surgical margins). We obtained 129 responses to the 1-year QOLu analogic scale.
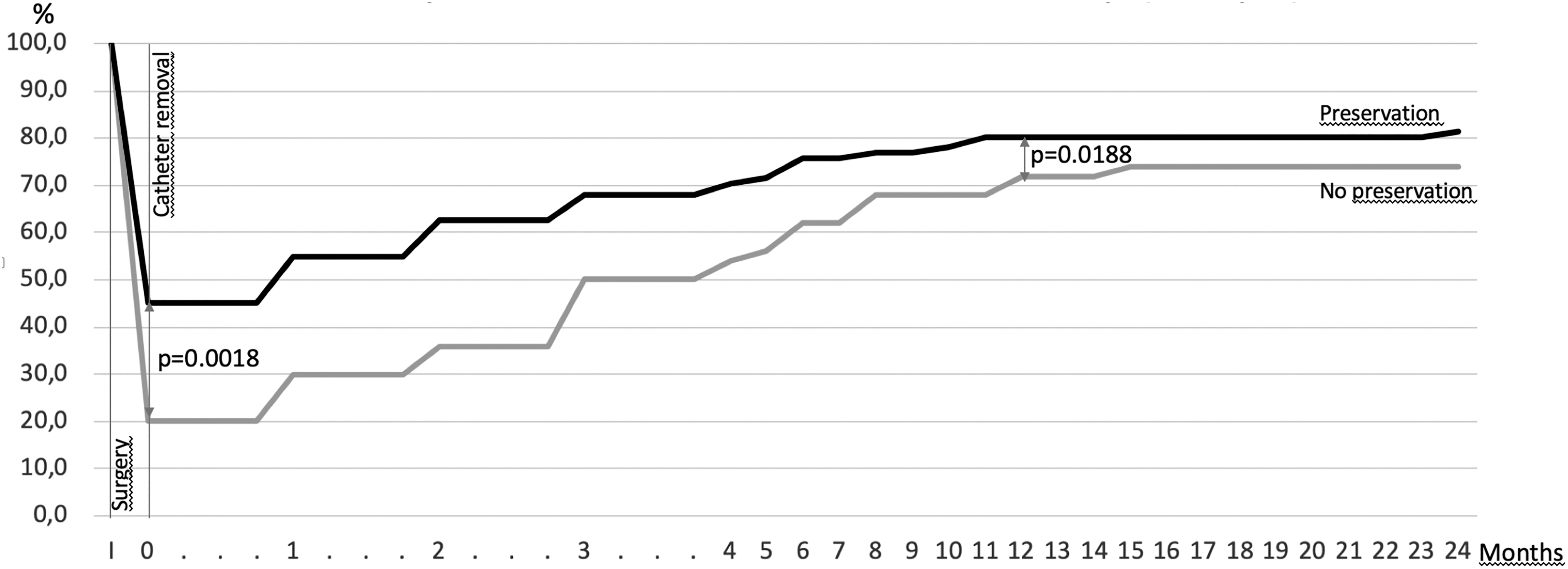
Outcome of bladder neck preservation on the full continence recovery in case of standard anastomosis (Group 1). I: Surgery. 0: Day of catheter removal. Gray: without bladder neck preservation. Black: with bladder neck preservation.
In Group 2 (SSA), we observed a complete continence recovery in 75.6% at catheter removal, in 82.9% at 1 month, and 97.5% at 1 year. Bladder neck preservation represented 60.9% in this group and had no statistical role on immediate continence recovery in this group (p = 0.388). We obtained 33 responses to the 1-year QOLu analogic scale.
Comparison between the two groups revealed a statistical difference for continence recovery: immediate (p = 0.0000035), at 1 month (p = 0.000092), at 1 year (p = 0.028) (Fig. 5), for urinary reeducation requirement (p = 0.0006), and at 1-year follow-up concerning the number of pads used per day and the results of the QOLu analogic scale (p = 0.0000002).
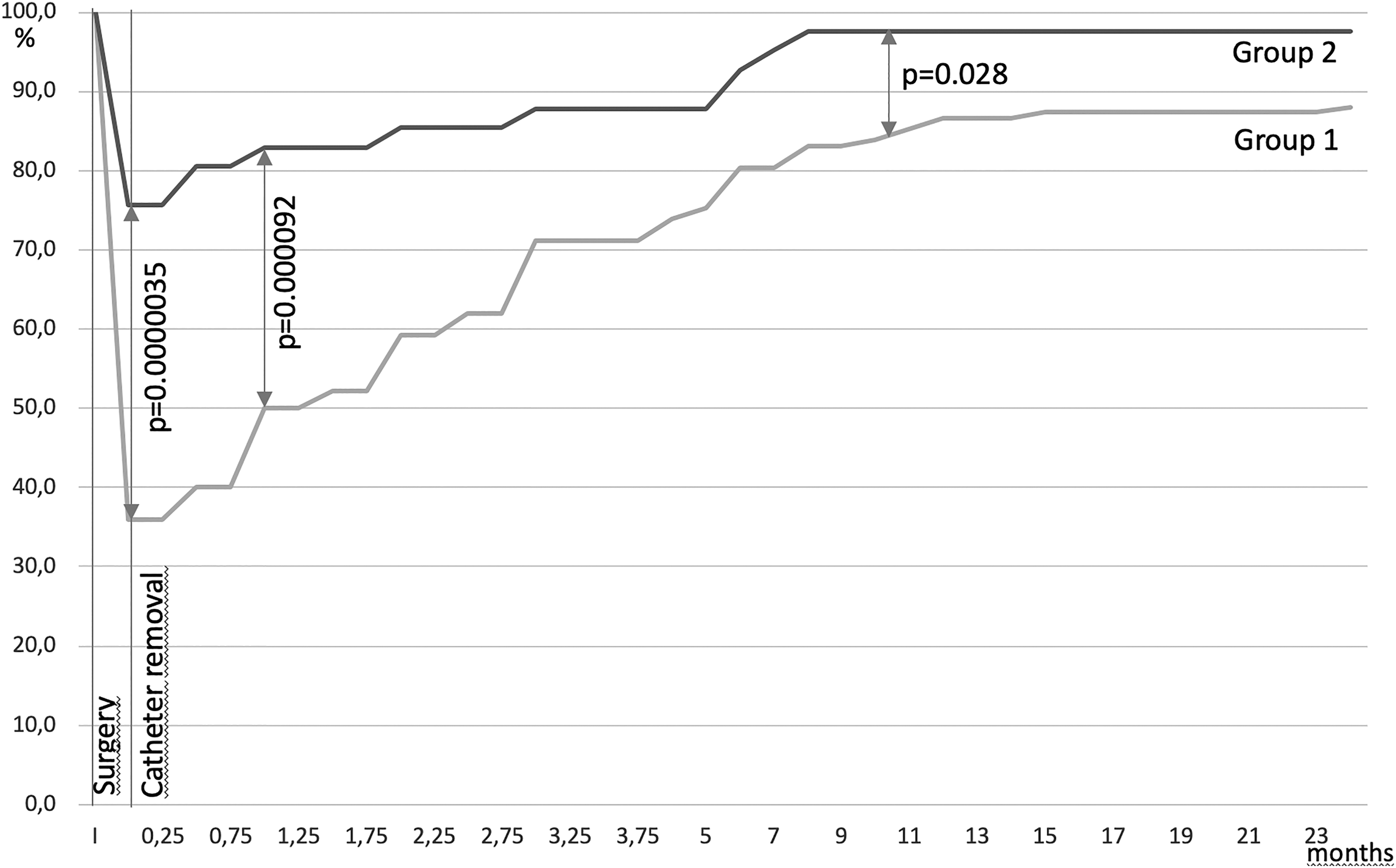
Full continence recovery comparison between Group 1 (standard anastomosis) and 2 (SSA). I: Surgery. 0: Day of catheter removal. Gray: Group 1. Black: Group 2.
SSA was impossible in only 1 case, because of bad conditions of anastomosis (excessive tension on the suture), that required a standard technique with a watertight result. No Cystography was required. No anastomosis complication (stenosis, urinoma, fistula …) in relationship with the SSA technique has been reported.
Discussion
The return of urinary continence and its delay after RP represents a major criterion for patient's QOLu.
Walsh and colleagues 16 described pad-free continence rates of 54, 80, and 93% at 3, 6, and 12 months, respectively, after open radical retropubic prostatectomy (RRP).
In his meta-analysis, Ficarra et al. 12 showed a better 12 months urinary continence recovery after RARP in comparison with RRP (p = 0.03) or laparoscopic RP (p = 0.006). With a ≥ 6 months follow-up, the urinary incontinence rates in robot-assisted laparoscopic RP (RALP) series ranged from 85% to 98%. Patel and colleagues 17 reported no-pad continence rates of 27% immediately after catheter removal, 47% at 1 month, 82% at 3 months, 89% at 6 months, 92% at 9 months, and 98% at 1 year.
Tewari and colleagues 18 demonstrated a faster return to continence in the RALP group compared with the open RRP group (50% continence rate at 44 vs 160 days).
Early continence recovery was evaluated by Ahlering et al. 19 with 33% pad-free at 1 week after catheter removal, 50% at 1 month, 80% at 3 months, 85% at 6 months, and 92% at 1 year.
Most evidence supports the idea that postoperative urinary incontinence is primarily due to sphincteric dysfunction, as it was observed urodynamically in 85–90% of the incontinent patients. 20,21 A potential cause could be represented by the injury of the neural branches leading to the rhabdosphincter that can play a role in the guarding reflex pathway during RP. If the preservation of the neurovascular bundles seem to have a positive role on long-term continence recovery, 22 this hypothesis is supported by the significantly higher sensory threshold in incontinent patients observed by John and colleagues 23 after RP and by the continence improvements observed when Retzius-sparing surgery has been realized 24,25 with 92% of immediate continence and 96% after 1 year (with the use of more than one safety pad as definition of incontinence).
Even longer preoperative membranous urethral length had a significant shorter time to stable postoperative continence in multivariate analysis measured by MRI 3,4 or transrectal ultrasound, 5 surgical techniques have been developed focusing on preservation during the dissection and reconstruction of the musculofascial structures to restore the length of the urethrosphincteric complex, prevent its caudal retraction, and avoid tension on the anastomosis. 6 –11 Despite the improvement of the dissection 26 –28 and the better visualization of the urinary sphincter complex, the continence rate was 42.6% at catheter removal, 46.3% at 1 month, 70% at 3 months, and 94.4% at 6 months after RALP. A significant enhancement of the continence rates was reported by Tunc with the improvement of the dissection and the preservation of the bladder neck and of the apex. 14,29
Argun and colleagues 15 was the first to report of a positive impact of stay sutures on early continence recovery (73% vs 35% p < 0.0001 at 1 month, and 83% vs 53% p = 0.0004% at 3 months), allowing more precision during placement of the urethral sutures and thereby avoiding retraction of the urethra and injury to the striated sphincter while suturing.
The results of our study prove the major impact and the logic of preserving the sphincteric outer striated muscular sleeve during the anastomosis with a complete continence recovery in 75.6% at catheter removal, in 82.9% at 1 month, and in 97.5% at 1 year despite a more elderly population with more severe lesions in the SSA group. As expected, urinary reeducation was less required in case of SSA (p = 0.0006), with less pad use (p = 0.0000002) and a best QOLu analogic scale result (p = 0.0000002).
As demonstrated by Argun et al., 15 the key point is probably to put the first stay stitch before ending the section of the urethra to avoid the retraction of the internal urethral layer underneath the sphincter. This modification allows the exclusive suture of the internal layer of the urethra to the bladder that is exposed step by step by the progressing suture, respecting the external layer that corresponds to the sphincteric sleeve. The suture is thereby so precise and fragile that needs a double (posterior and anterior) reinforcing suspension and surely cannot be performed by another technique than using the robotic assistance. Despite this fragile nature, no complication due to the SSA technique has been reported.
According to Argun and colleagues, 15 who only identified the “stay suture” technique and the younger age as independent variables in multivariable analysis, we also observed in the SSA group no significant impact of the bladder neck preservation, while its preservation improved significantly the continence in case of standard urethrovesical anastomosis. Both groups had posterior and anterior reconstruction or suspension that allowed the comparison of the two groups independently of this technical trick. Moreover, the SSA technique provided an immediate positive effect on early continence recovery that could also suggest a short learning curve for skilled robot operators. The duration of the SSA technique was not measured nor compared with the standard one. Obviously, it should be probably a little bit more time-consuming at the beginning of the experience because of the delicate gesture that is required.
However, our study has some limitations. Despite significant statistical results, the main limitation was certainly the self and prospective evaluation by a single surgeon (scant number of cases and potential self-evaluation bias). The learning curve and the evolution of the robots during this study should be also evoked as a bias factors that could be balanced by a real “cut-off” point concerning the continence results when the SSA technique has been routinely applied with no statistical impact of the bladder neck preservation and, in addition, in a more elderly population with more severe lesions. We also assessed the learning curve effect on early continence recovery during the first 73 cases and the next 72 cases in the Group 1 (standard anastomosis) and found no statistical difference between these two subgroups in term of 1 month continence recovery (41 and 54%; p = 0.11).
The continence results and the feasibility of the SSA need further study among larger numbers of patients and validation among a wider urologic community. The ideal technique should probably include the best quality of dissection to preserve the anatomic structures, 30 the reconstruction of the surrounding fascial structures, and also the preservation of the sphincter during the suture as described above.
Conclusions
The SSA significantly improves the rates of continence recovery independently by preserving the bladder neck after robot-assisted laparoscopic RP. These results, the feasibility, and the reproducibility of the SSA technique need further study among larger numbers of patients and validation among a wider urologic community.
Footnotes
Authors' Contributions
C.A.: Project development, Data collection, Data analysis, Article writing; C.T., A.S.: Data collection, Data analysis; J.-B.B.: Article writing; G.L.: Data analysis; J.R.G.: Data collection; and G.P.: Project development, Data analysis, Article writing.
Ethical Standards
The ethical standards have been respected. It was not a prospective research on animals or patients. No informed consent was required.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
No funding was received for this article.
