Abstract
Percutaneous nephrolithotomy (PCNL) is a standard treatment for patients with large or complex kidney stones. The procedure has traditionally included postoperative placement of a nephrostomy tube to allow for drainage and possible reentry. This practice was first implemented after complications incurred after tubeless PCNL in a small patient population. Recently, tubeless PCNL has reemerged as a viable option for selected patients, resulting in decreased pain and analgesic use, shorter hospitalization, quicker return to normal activity, and decreased urine extravasation. Gelatin matrix sealants are occasionally used in nephrostomy tract closure. Techniques for delivery of these agents have been ill described, and placement may be performed with varying results. We present a literature review comparing tubeless PCNL to its traditional variant with indications for use of each, as well as a comparison of agents used in closure. Finally, we outline a novel, reproducible technique for closure of the dilated percutaneous renal access tract.
Introduction
P
Efforts to minimize postoperative morbidity associated with the NT while preserving efficacy and safety have included use of small-bore percutaneous catheters. 14 –16 Smaller catheter use has resulted in decreased postoperative pain and narcotic requirements 14,15 and may preclude complications in selected patients; however, placement of these catheters is not benign, regardless of caliber. Adverse events associated with the NT have been well documented and include prolonged hospitalization, 9 urine extravasation, 15 and increased analgesic requirement. 9 Tubeless PCNL has been demonstrated to reduce such complications in selected patients, allowing for a quicker return to normal activity. 9,16 –19
Studies have further shown that standard PCNL does not offer a statistically significant advantage over tubeless PCNL for parameters including postoperative fever, 4 prolonged urinary drainage, 4 ancillary procedures, 4 required reentry, 4 success rates, 8 or morbidity. 8 Such results have prompted many urologists to reevaluate standard protocol in favor of a tubeless approach. 4 –9,11 –13 With mounting evidence in support of this technique, we now routinely conclude PCNL in tubeless or totally tubeless fashion if no indication exists for NT placement (Table 1).
UTI=urinary tract infection; EBL=estimated blood loss.
Increasing use of this technique has led to a new dilemma over optimal closure for dilated percutaneous tracts. Several authors advocate for tubeless nephrostomy tract closure using a gelatin matrix hemostatic sealant (FloSeal, Surgiflo) injected into the tract before primary closure, 20 –24 which many urologists have adopted. Although the adjunctive use of hemostatic agents has been demonstrated as safe and effective in enhancing tubeless PCNL closure, the most appropriate agent and technique have yet to be determined. 22
Through our experience, we advocate for the use of thrombin-soaked absorptive gelatin (Gelfoam), because this provides both tamponade through expansion within the tract and an intrinsic hemostatic property. Although hemostatic agents are increasingly used in tubeless PCNL, descriptions of optimal delivery are lacking. A brief description by Dah-Shyong Yu 23 of packing Gelfoam within the tract provided little technical detail, limiting reproducibility.
We present a novel, reproducible technique for hemostatic plug placement in tubeless PCNL that has been successfully used in more than 200 cases. Notably, this technique needs no additional instruments and is easily adapted into standard practice with materials readily available in the operative suite. Our technique for primary access site closure has resulted in calculated, precise placement of adjunctive hemostatics and a favorable postoperative course.
Technique
Preparation and measurement
1. Before procedure initiation, evaluate preoperative CT to measure distance from the skin to the renal parenchyma/caliceal interface (Fig. 1):
a. This is an estimated length to be confirmed intraoperatively (Step 5).
2. After stone clearance, determine the need for NT placement (Table 1).
3. A 34/30F (outer/inner) clear renal sheath (Boston Scientific M0062601600) is used to allow visualization of the caliceal-parenchymal junction, where the leading edge of the hemostatic plug will be placed (Fig. 2).
a. Do not discard the blue packaging cylinder inside the clear renal sheath on unpacking; keep on the sterile field to be used as a plunger in plug deployment.
4. The sheath is withdrawn, positioning the leading edge at the caliceal-parenchymal junction (yellow/white collecting system meets pink parenchyma).
5. Extracorporeal segment of the sheath (skin to end of sheath) is measured (Fig. 3). This length is subtracted from the total length of the sheath (17 cm) to confirm skin-to-calix length, which will closely match the preoperative CT measurement (Step 1).
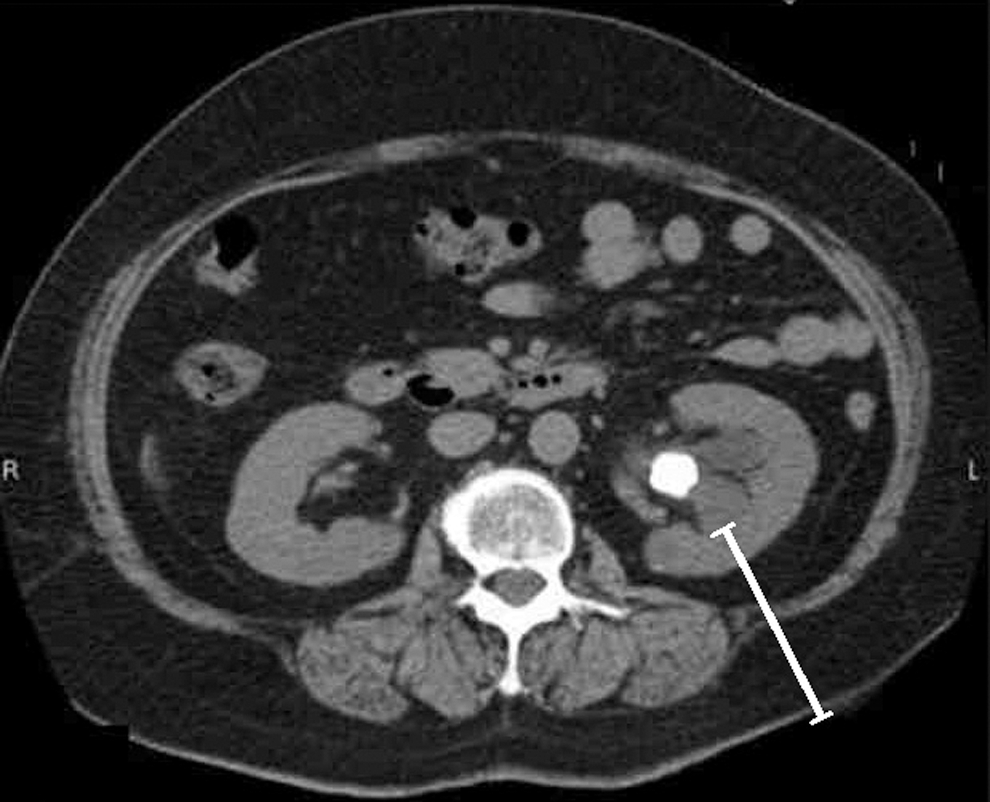
Preoperative CT evaluated in surgical planning determining the zone of safe access and to obtain measurements of skin to renal calix for determining plug length.
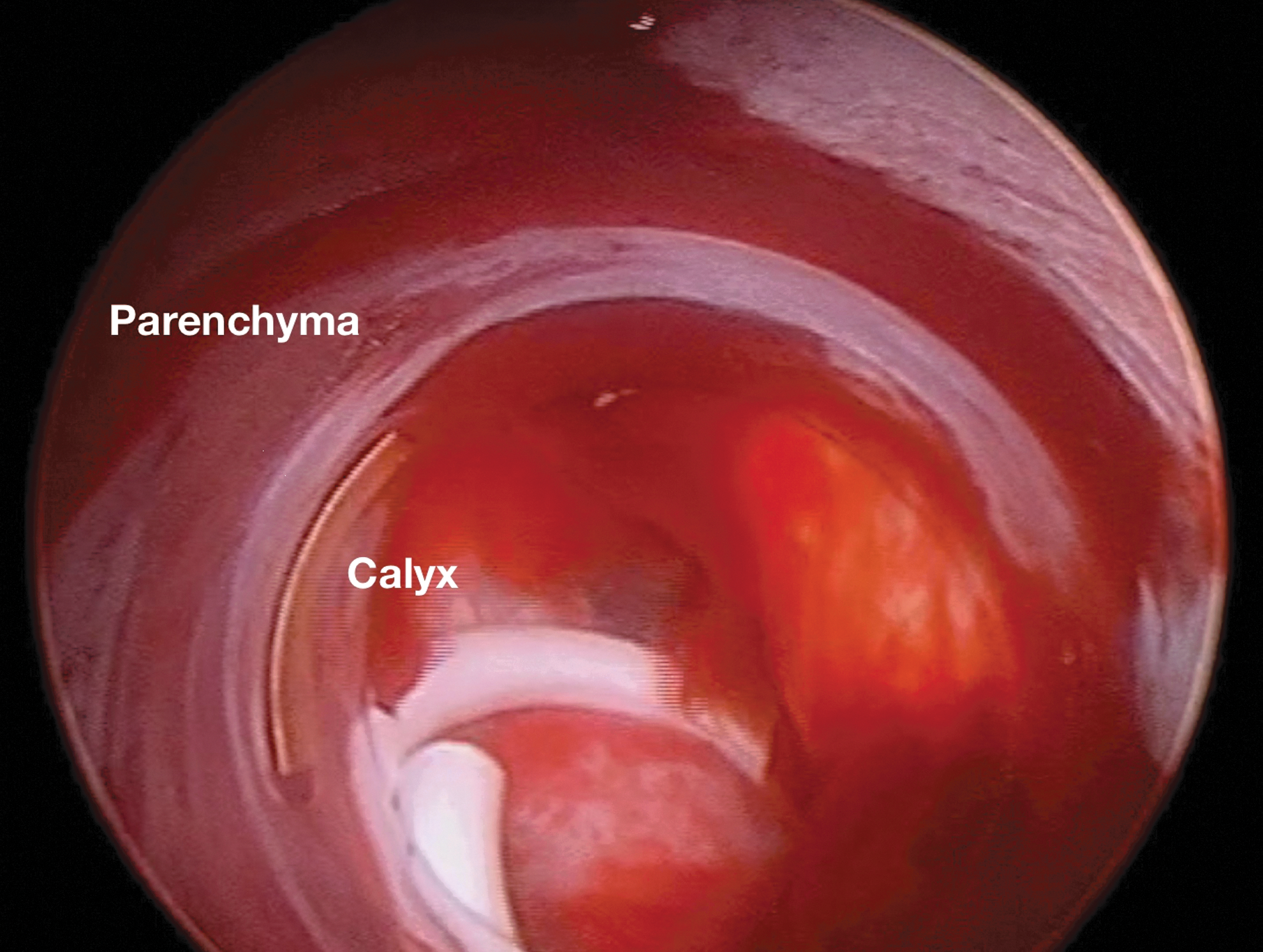
Identification of the calices-parenchymal junction as the clear renal sheath is withdrawn positioning the leading edge of the sheath at this junction.
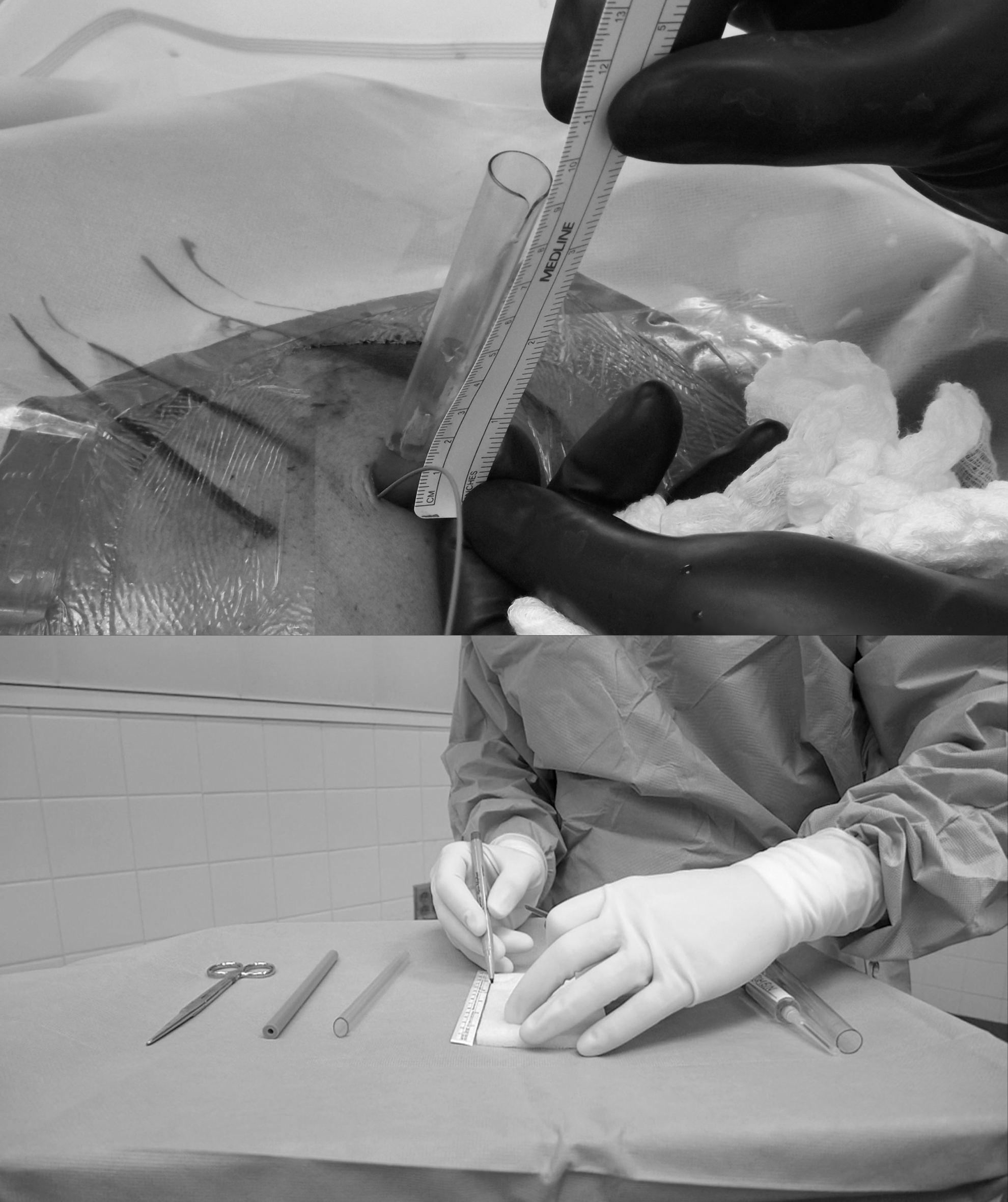
Measurement taken of the external portion of the clear renal sheath. This measurement is then subtracted from the total sheath length (17 cm), determining the exact length needed to create the Gelfoam plug.
Plug placement
6. Gelfoam gelatin sponge is cut to patient-specific skin-to-calix length (Step 5) (Fig. 3).
7. Gelatin foam is rolled on its longitudinal axis in “cigar” fashion (Fig. 4).
8. Gelatin foam is placed inside the clear renal sheath. Inject 5 to 10 mL thrombin into rolled Gelfoam to create plug (Fig. 5A).
9. Advance plug down the sheath using the packaging cylinder as a plunger until the plug is flush with the skin (Figs. 5B, 5C, 5D).
10. Once flush with the skin, maintain plug position by holding the plunger steady while backing out the sheath (modified Seldinger technique) (Fig. 5E).
11. Postplug nephrostography is performed via catheter into the collecting system (Fig. 6) (previously placed retrograde ureteral catheter, catheter over safety wire alongside plugged access site, or secondary no-dilated percutaneous renal access).
12. Safety wire (Amplatz Superstiff) is removed.
13. Primary skin closure is followed by sterile dressing application (Tegaderm).
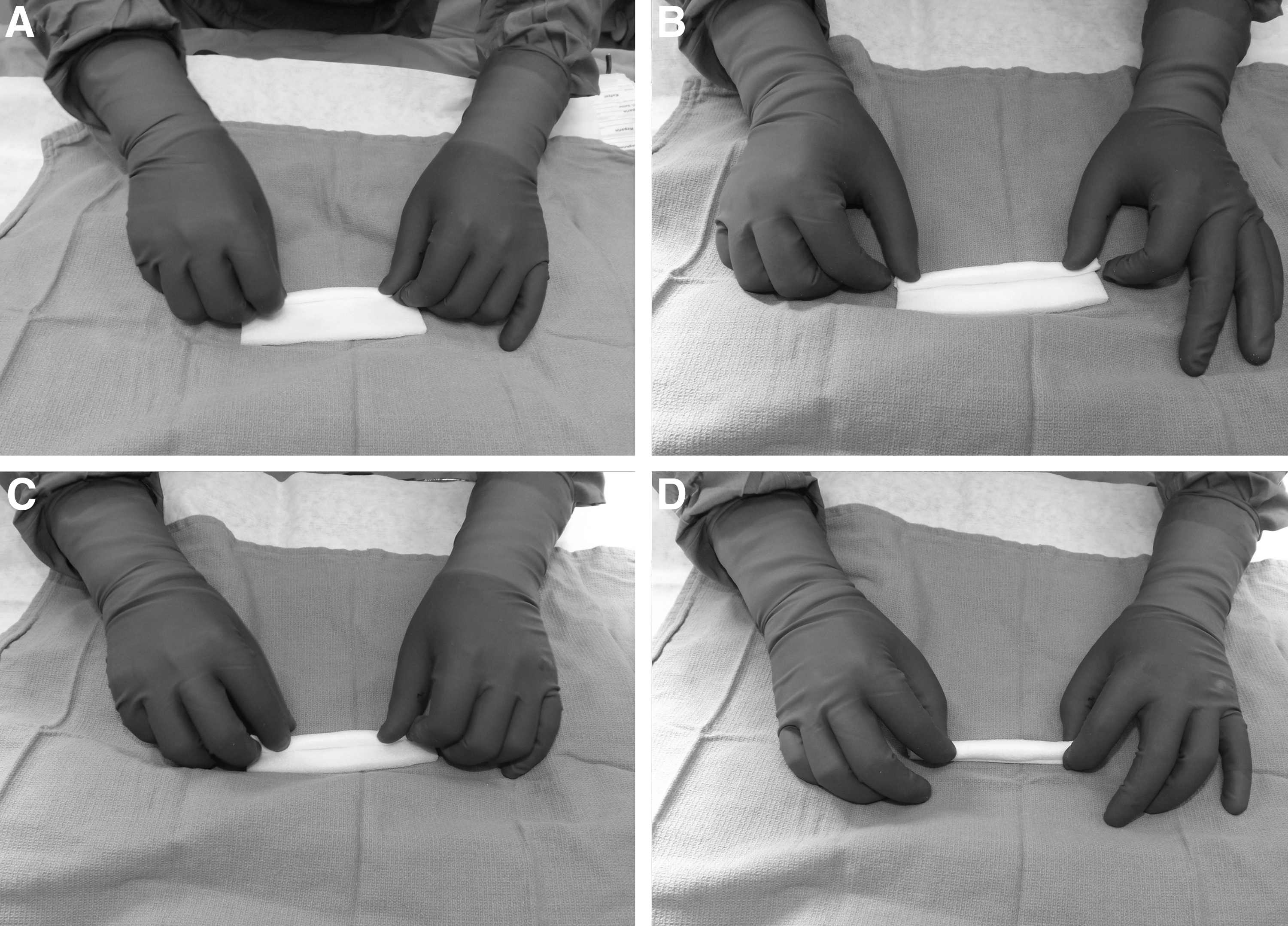
After cut to precise length, Gelfoam is rolled in “cigar” fashion to create the plug.
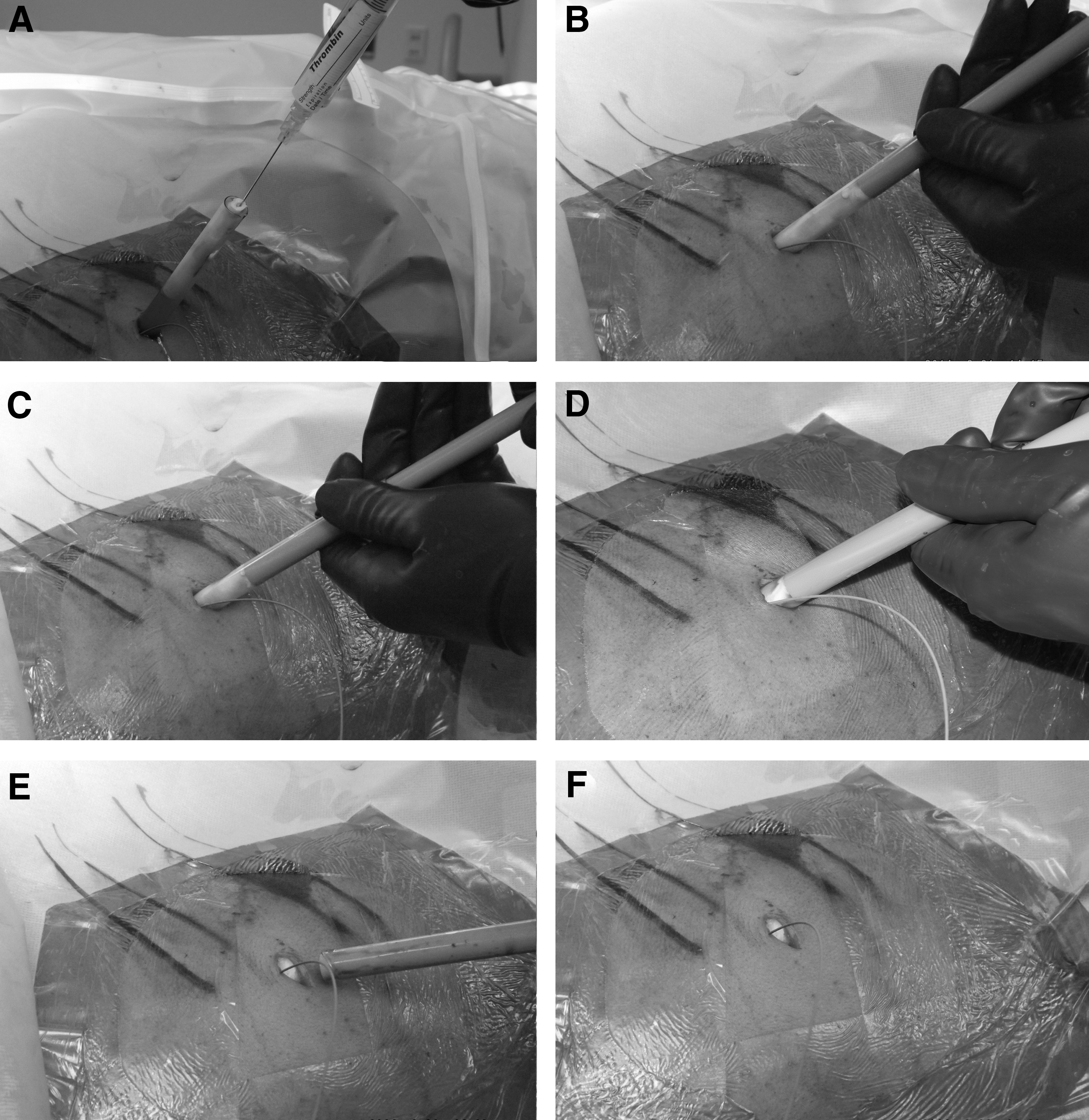
Injection of thrombin into the Gelfoam plug followed by advancement of the plug to the skin edge using the cylindrical packaging insert as a plunger. The plug is deployed by backing out the clear renal sheath in a modified Seldinger technique.
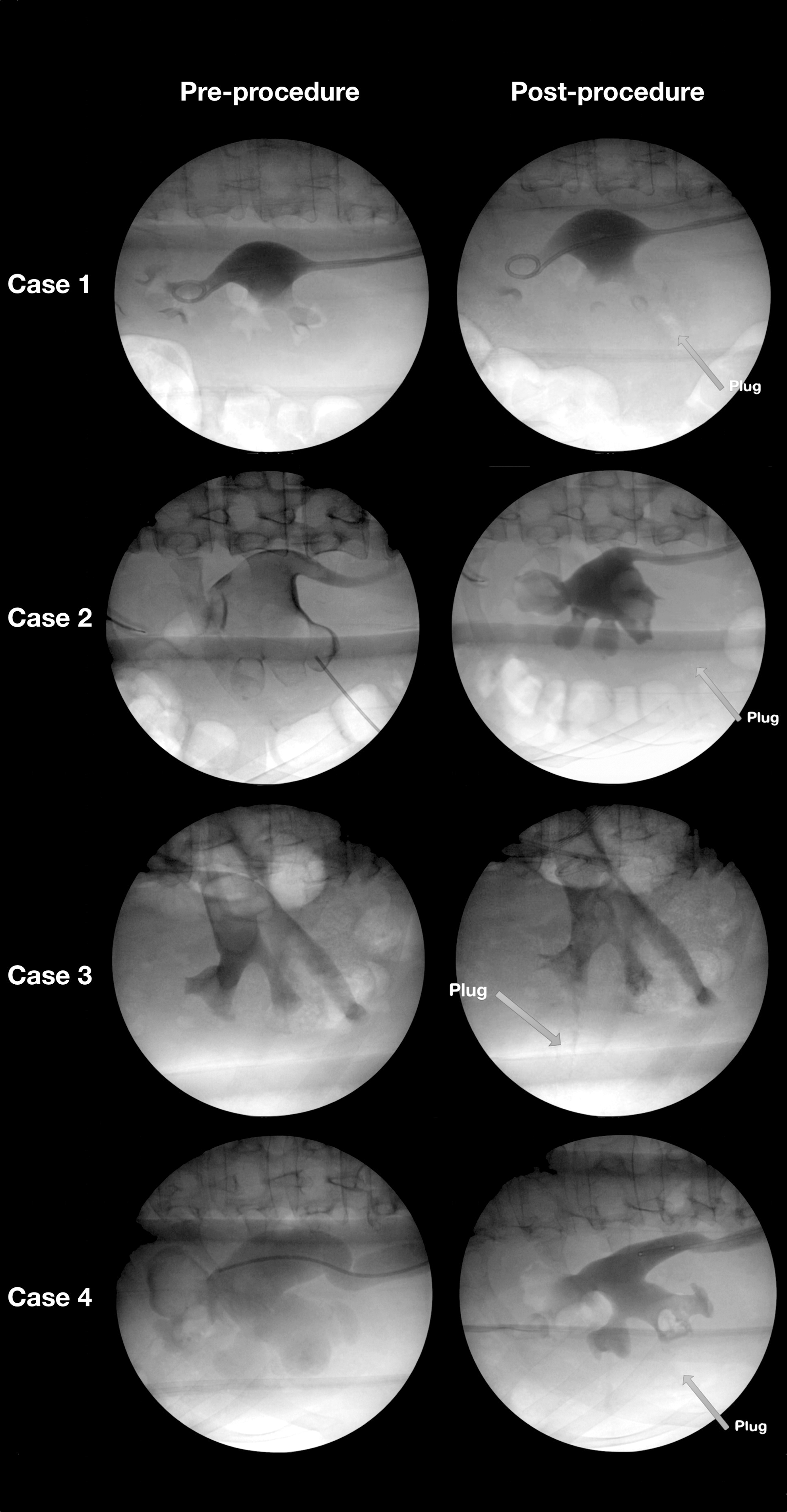
Pre- and postprocedure images of four tubeless PCNL cases. The plug is identified in the postprocedure images. Minimal extravasation is seen with high-pressure contrast injected nephrostogram.
A video demonstrating this technique is available at
Materials/Equipment
• Thrombin 5000-IU
Gelfoam absorptive gelatin 12 cm×8 cm
• Clear renal sheath 34F OD×17 cm (Boston Scientific M0062601600)
Role in Urologic Practice
Our standardized technique for PCNL closure has yielded excellent clinical results. We have noted significant improvement in patient satisfaction including fewer complaints, decreased need for narcotics, shortened length of stay, and improved postprocedure nephrostogram images. Pre- and postoperative nephrostography is routinely performed in our PCNL procedures, and the nephrostograms have shown remarkable equivalency despite manipulation of the renal collecting system (Fig. 6). These patients also undergo routine postoperative CT to assess stone clearance. The plug is notable on CT and appears as low density (similar to fat) occupying the space of the tract between the skin and kidney (Fig. 7).
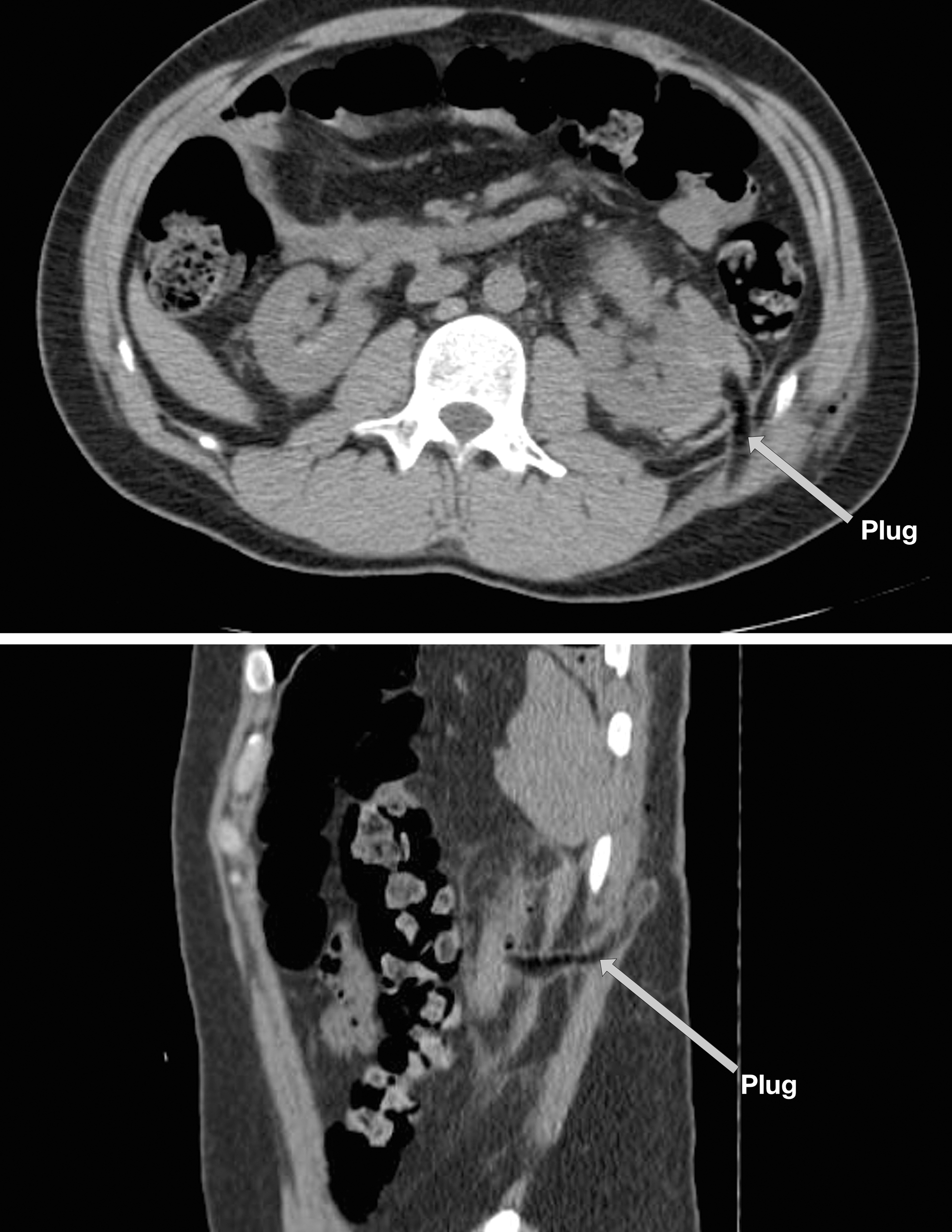
Postoperative day 1 CT scan demonstrates the appearance of the hemostatic plug placed within the tract of the left kidney after percutaneous nephrolithotomy.
In our initial implementation of this closure, we performed retrograde ureteronephroscopy at 4-week intervals to evaluate the collecting system directly. In this series, we observed completely healed calices with no visual abnormalities. We experienced no episodes of stone formation at the plug site, nor any wound or tract infections associated with the plug. We had one episode in which the plug was too long and inadvertently advanced too far into the collecting system. This was noted intraoperatively, and the patient was monitored carefully over a normal postoperative course. This patient underwent retrograde renoscopy on postoperative day 28 to ensure no residual plug remained in the renal collecting system (a theoretic nidus for additional stone formation). No abnormalities were seen, and the plug material had completely dissolved.
Although an overall benefit has not yet been shown, the use of hemostatic agents to augment tubeless PCNL may be of value for patients in the acute postoperative period, without adding to the potential morbidity of the procedure. In a prospective study by Feng and associates, 18 nephrostomy tract bleeding and extravasation of urine into the retroperitoneum were identified as indications for the use of adjunctive hemostatic agents. Although multiple authors advocate the use of adjunctive hemostatic agents to addresses the concerns of nephrostomy tract bleeding and extravasation of urine into the retroperitoneum with resultant morbidity, there are limited data that strongly support the adjunct use of these agents. 18,20 –24
Once a surgeon has opted for a hemostatic adjunct to access site closure, the critical consideration is use of an appropriate hemostatic agent. Two main classifications of hemostatic agents are passive and active, based on their mechanism in promoting hemostasis. Passive agents act to provide direct contact against bleeding sites and act as a matrix for platelet aggregation and clot formation. These include collagens (Avitene), cellulose (Surgicel), gelatins (Gelfoam, Surgifoam), and polysaccharide spheres (Arista). These are advantageous when applied directly to the site of hemorrhage in a patient with a full complement of functional clotting factors.
Active agents act biologically on the clotting cascade and are usually liquids. These include thrombin, thrombin combined with passive gelatin granules (FloSeal, Surgiflo), and fibrin sealants (Tisseel, Vitagel, Cryoseal, Evicel). Thrombin production is the final coagulation step needed to cleave fibrinogen into fibrin, providing a lattice for platelet aggregation and thrombus formation. Thrombin additionally acts on smooth muscle causing vasoconstriction, further aiding hemostasis. Although significantly more expensive, thrombin products have become favorable in most surgical settings and combination agents have been demonstrated as preferred agents for PCNL tract closure. 22
Fibrin sealants are useful in the absence of intrinsic clotting factors due to immediate formation of an artificial clot. Limitations associated with these agents include preparation time, cost, lack of hemostatic effect beneath this fibrin “cap,” and potential dislodgement of the artificial clot. 22
The use of diathermy techniques, an alternative to the aforementioned agents, has also been hypothesized. Jou and colleagues 25,26 and Mouracade and coworkers 27 investigated electrocoagulation of the nephrostomy tract using rollerball or bugbee electrode. These techniques may have a role in providing hemostasis for tubeless PCNL; however, concern has been raised over potential issues with tract healing. Compared with diathermy, adjunctive hemostatic agents seem to provide not only hemostatic control but also a framework on which the tissue healing is promoted and sealing of the urinary collecting system is achieved. 22 Further studies are needed to identify the benefit of one over the other, and the choice ultimately remains the surgeon's preference.
Our initial experience with primary tract closure followed the trend of using FloSeal injected into the tract before skin closure. This closure method typically yielded acceptable extravasation on postprocedural imaging, and patients fared well; however, FloSeal's intrinsic viscosity resulted in suboptimal agent retention within the collapsing tissue of the nephrostomy tract. We have since had greater clinical success and improved appearance of postoperative nephrostograms since transitioning to thrombin-soaked Gelfoam, especially since refining the procedure to the current technique described.
Gelatins like Gelfoam are intrinsically absorptive and as a result can increase up to 40 times in weight and expand 200% to 400% in size. These properties are advantageous in providing mechanical tamponade within a confined space. 22 Gelatin agents lack viscosity, providing control during application to ensure that agent contents remain outside of the collecting tract and flush with the patient's skin. Liquid agents lack this stability, and spillover can occur both intra- and extracorporeally.
Urine extravasation, or extracorporeal leak, is a common complaint associated with standard PCNL, 8 which can be particularly worrisome to the patient. While tubeless PCNL with hemostatic plug placement provides an expectedly substantial decrease in the amount of urinary extravasation, 8 further measures can be undertaken in standard PCNL to minimize this complication. Desai and colleagues 15 found that patients receiving large-bore NT after PCNL experienced significantly prolonged urine extravasation compared with those receiving small-bore NT (21.4 vs 13.2 hours), and even longer rates when compared with those receiving no NT drainage (21.4 vs 4.8 hours), with no difference in other complications between the three arms of the study. This provides added credence to the benefits of tubeless PCNL.
When NT is necessary, a small-bore nephrostomy catheter provides improved comfort to the patient and significantly minimizes urine extravasation. We have found that if placed through an additional, minimally dilated to 10F access (similar to a standard NT), these benefits are maximized. We therefore advocate for small bore NT placement in a subcostal, separate, nondilated access site, which can be achieved during initial access and preserved in case NT is desired at procedure conclusion. All dilated access sites are uniformly plugged and closed in the outlined fashion.
Tubeless PCNL provides numerous advantages to the patient irrespective of physical complaints, including a significant cost advantage achieved through shorter hospital stays and decreased analgesic requirements. The advent of tubeless PCNL has also led to selected procedures being performed in an ambulatory setting, providing considerable savings compared with hospital-based procedures. Our goal is a complication-free closure method; if achieved, this can further reduce cost attributed to follow-up and can aid in the success of the tubeless approach. The only added cost incurred in performing a hemostatic plug closure is that of the nephrostomy tract closure materials including the clear renal sheath (if not already used by the surgeon). The costs of these materials are shown (Table 2), and the cost per case is approximately $102.00.
Many surgeons use a simple skin suture closure, further reducing cost, but not addressing the concern of potential hemorrhage and urine extravasation. Others use FloSeal with an approximate cost of $85.00 for the 5 mL vial (Table 3). Although our technique necessitates additional consumables (renal sheath and Gelfoam), this minimal cost is offset by the potential benefit in patient satisfaction and surgical outcomes with thrombin-induced hemostasis and vasoconstriction plus Gelfoam matrix for platelet aggregation leading to clot formation and tract tamponade. This cost is further offset from a “standard PCNL” in that an 8.3F Percuflex locking loop NT costs $127 (Boston Scientific); thus, plug placement may equate to a cost savings. Thus far, use of our technique for renal access closure has yielded excellent clinical outcomes in more than 200 tubeless PCNL cases, although randomized control studies are necessary to adequately compare against other closure methods.
Surgeons already using a clear renal sheath will not incur additional cost.
Many hospitals are covered with Group Purchasing Organizations to decrease cost.
Conclusion
Tubeless PCNL is an operative technique that has been extensively studied, with strong data supporting its use in selected patients. Tubeless PCNL is associated with decreased length of hospitalization, 9 decreased urine extravasation, 15 and decreased analgesic requirement 9 compared with PCNL with small bore 10F Cope loop NT with stent placement. As urologists adopt the practice of using hemostatic agents in closure of the PCNL tract, an optimal technique and agent must be determined. We have outlined one such option for adjunct hemostatic agent placement for tract closure for any surgeon electing to use hemostatic agents at the conclusion of PCNL. Our technique is reproducible, time efficient, safe, cost-effective, and provides excellent clinical outcomes.
Footnotes
Acknowledgment
Previous abstract publication in the Journal of Endourology. October 2013, 27(S1): P1- A470 (abstract V10–12).
Disclosure Statement
Julio G. Davalos, MD, is a consultant for Boston Scientific.
Abbreviations Used
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
