Abstract
The prevalence of microplastics (MPs) in the environment is an escalating global concern, driven by their increasing abundance and potential adverse effects. Their detrimental environmental consequences and risks to public health necessitate urgent action to mitigate their impacts. To better understand MP accumulation within the environment, estimate their impacts, and develop effective mitigation strategies, it is imperative to advance our knowledge of MPs’ environmental fate and how this fate is linked with plastics’ intrinsic properties. We highlight the current research on MPs’ accumulation within different environmental sinks and identify the critical knowledge gaps in understanding the flux of MPs between these sinks. We also discuss how the intrinsic properties of MPs, including polymeric backbone, structure, chemical additives, crystallinity, molecular weight, and hydrophilicity, influence their environmental behavior and degradation pathways. Among these properties, the polymeric backbone and functional groups serve as primary drivers of MP stability when exposed to external physical and chemical factors, thereby influencing the pathway and rate of MP degradation. Other intrinsic properties and environmental conditions mostly impact the rate of MP degradation. As MPs undergo environmental degradation, changes in their intrinsic characteristics can lead to further fragmentation. This process potentially enhances environmental harm due to the resulting particles’ increased surface area, greater environmental mobility, and higher potential for contaminant transport. These factors collectively contribute to the complex dynamics of MPs’ fate and impact on the environment.
Introduction
Since the 1950s, plastics have been widely used globally owing to their wide range of utility and easy availability. By 2015, the total accumulated global plastic production was estimated to be 8.3 billion metric tons (Geyer et al., 2017). If current trends in plastic production and waste management persist, it is projected that approximately 12.0 billion metric tons of plastic waste will accumulate in the natural environment or landfills by 2050 (Geyer et al., 2017). The durability of plastics that attracts consumers also leads to long-term persistence in aquatic and terrestrial environments, contributing to subsequent human exposure and presenting substantial health risks (Parveen et al., 2022; Vethaak and Legler, 2021). Although the term “plastic” refers to general synthetic polymeric materials of any size, plastic particles smaller than 5 mm, whether originally produced at this size or formed by the fragmentation of larger objects, are typically referred to as microplastics (MPs). They can enter the human body through ingestion (food and drinking water), inhalation (indoor and outdoor), and skin contact (personal care), with potential accumulation in various organs (Sun and Wang, 2023). These particles may cause physical damage to tissues and can leach harmful chemicals such as bisphenols and phthalates or act as carriers for other environmental pollutants (Yang et al., 2022). Some studies suggest MPs could trigger inflammatory responses, disrupt the endocrine system, or alter the gut microbiome (Sofield et al., 2024). However, while these potential impacts are concerning, the long-term effects of MP exposure on human health are still being researched, and more comprehensive studies are needed to fully understand the risks.
Currently, the majority of end-of-life plastics are discarded in landfills or litter the land, water, and air. For example, in the United States, 35.7 million tons of plastic waste were generated in 2018, accounting for 12.2% of municipal solid waste (EPA, 2018). Only a small fraction, approximately 8.7%, or 3 million tons, of waste plastic is recycled. It is difficult to determine how much plastic enters aquatic systems, but Borrelle et al. (2020) estimated that approximately 11% of global plastic waste in 2016 entered aquatic environments. The impacts of MPs on aquatic and terrestrial ecosystems are extensive and potentially severe. In aquatic environments, marine life can ingest MPs, leading to physical harm and reduced feeding. This ingestion can result in the bioaccumulation and biomagnification of MPs and their associated chemical additives throughout marine food webs (Miller et al., 2020). Additionally, MPs pose a significant threat to biodiversity and overall ecosystem functioning in aquatic systems (Corinaldesi et al., 2021). In terrestrial ecosystems, MPs can alter soil structure, affect water retention, and impact soil-dwelling organisms, potentially disrupting nutrient cycles and plant growth (Jazaei et al., 2022; Yu et al., 2022). Both aquatic and terrestrial MPs can serve as habitats for microbial communities, potentially facilitating the spread of pathogens or altering local microbial ecosystems (Galafassi et al., 2021).
As plastics are released into the environment, they may degrade depending on environmental conditions and their intrinsic properties. The degradation of plastics presents a paradoxical phenomenon. Although it ultimately leads to the breakdown of plastic materials, potentially reducing their long-term environmental presence, it also generates smaller plastic fragments that can pose increased risks. These smaller fragments may have greater potential for environmental damage due to their enhanced toxicity and altered mobility (Beheshtimaal et al., 2024; Jeong et al., 2016). The reduced size facilitates their transport across different environmental systems, spreading contamination more widely. Consequently, plastic degradation simultaneously contributes to the eventual elimination of plastics from the environment and creates intermediate products that may pose significant ecological challenges, underscoring the complexity of addressing plastic pollution.
The intrinsic properties of plastic are defined as the fundamental molecular and structural characteristics of a polymer which determine its physicochemical behavior independent of its size or shape. This paper focuses on major intrinsic properties of MPs including polymeric backbone, structure, chemical additives, crystallinity, molecular weight, and hydrophilicity. Understanding this interrelation could assist plastic manufacturers in designing more environmentally friendly materials and help policymakers develop more effective waste management regulations.
Recent review articles on MPs have primarily covered their sources and distribution across various media (e.g., food, air, marine environments, freshwater, drinking water, wastewater, soil) (Koutnik et al., 2021; Padha et al., 2022; Perumal and Muthuramalingam, 2022), human health (Prata et al., 2020; Rahman et al., 2021) and environmental impacts (Du et al., 2022; Wright et al., 2013), toxicity (Rakib et al., 2023; Verla et al., 2019), removal practices for drinking water, wastewater, and stormwater (Iyare et al., 2020; Padervand et al., 2020; Shen et al., 2020; Stang et al., 2022), and challenges in their quantification and characterization (Jung et al., 2021; Samanta et al., 2022). However, limited attention has been given to understanding the link between the intrinsic characteristics of plastics, and their degradation and fate within the environment received less attention. Therefore, this paper reviews the current literature on MP degradation and identifies the critical knowledge gaps in understanding the links between the intrinsic properties of MPs and their fate. In this paper, we maintain that the paradigm developed in the past decades to characterize element biogeochemical cycles could be adapted to evaluate the fate and transport of MPs. Additionally, studying the influence of intrinsic MPs’ properties and environmental factors on their fate and contaminant uptake highlights the urgent need for comprehensive solutions to mitigate the environmental and health impacts of MPs. We particularly call attention to the connection of plastics’ intrinsic properties with the generation rates of MPs, and their degradation from physical, chemical, and biological processes.
Defining Plastics and Microplastics
Although the word “plastics” is a part of our daily conversation, its meaning is not well-defined. Chemical composition, solid state, and solubility characteristics have been suggested as three primary criteria for classifying plastic (UNECE, 2013; ECHA, 2011). According to the International Organization for Standardization (ISO), a high molecular weight polymer, shaped by flow, is considered a plastic material (ISO 472, 1999). Natural polymers with high molecular weight (e.g., cellulose, silk) are excluded from the definition of plastics, whereas modified natural polymers (e.g., rayon) can be considered synthetic and categorized as plastics. In addition to petroleum-based commodity plastics (e.g., polyethylene [PE], polystyrene), bio-based (e.g., bio-polyethylene), and biodegradable plastics (e.g., polylactic acid), as well as inorganic or hybrid polymers (e.g., silicone) are considered plastics due to their synthetic nature. We will simply define plastic pollutants as insoluble solid debris whose major component is a synthetic high molecular-weight polymer that is not readily decomposed to bioavailable carbon sources.
In defining MPs, size criteria are incorporated alongside the aforementioned features of plastics. MPs are typically characterized as plastic fragments with diameters smaller than 5 mm. Plastic fragments with a size smaller than 1 or 0.1 µm have further been defined as nanoplastics because of their distinct physicochemical and transport characteristics (Gigault et al., 2021); however, nanosized plastics are sometimes lumped to be part of MPs (Lin et al., 2024; Vethaak and Legler, 2021). Primary MPs are intentionally manufactured to be small in size, such as microbeads used in personal care products or pellets used in industrial processes. In contrast, secondary MPs result from the fragmentation of larger plastic materials as they undergo physicochemical and biological processes throughout their life cycle or after disposal.
Although recent plastic pollution studies have mostly focused on particles composed solely of plastic materials, it remains unclear whether particles consisting of a mixture of plastics with other materials such as paper or metals should also be classified as MPs. This ambiguity underscores the need for further investigation into their quantification methods and environmental implications of particles composed of plastic-containing mixtures.
Environmental Fate of Microplastics
One proposed model for describing how MPs move through the environment is a biogeochemical cycle for plastics that tracks the sources, sinks, and fluxes of plastics (Allen et al 2022; Bank and Hansson, 2019; Brahney et al., 2021; Hoellein and Rochman, 2021) (Fig. 1). One important caveat to applying this model of biogeochemical cycling to plastics is that, unlike inorganic contaminants, plastics will ultimately be mineralized to CO2. The timescale of the mineralization of MPs varies significantly depending on plastic type and environmental conditions, and remains an understudied area. Critically though, this “cycle” is not a closed system.
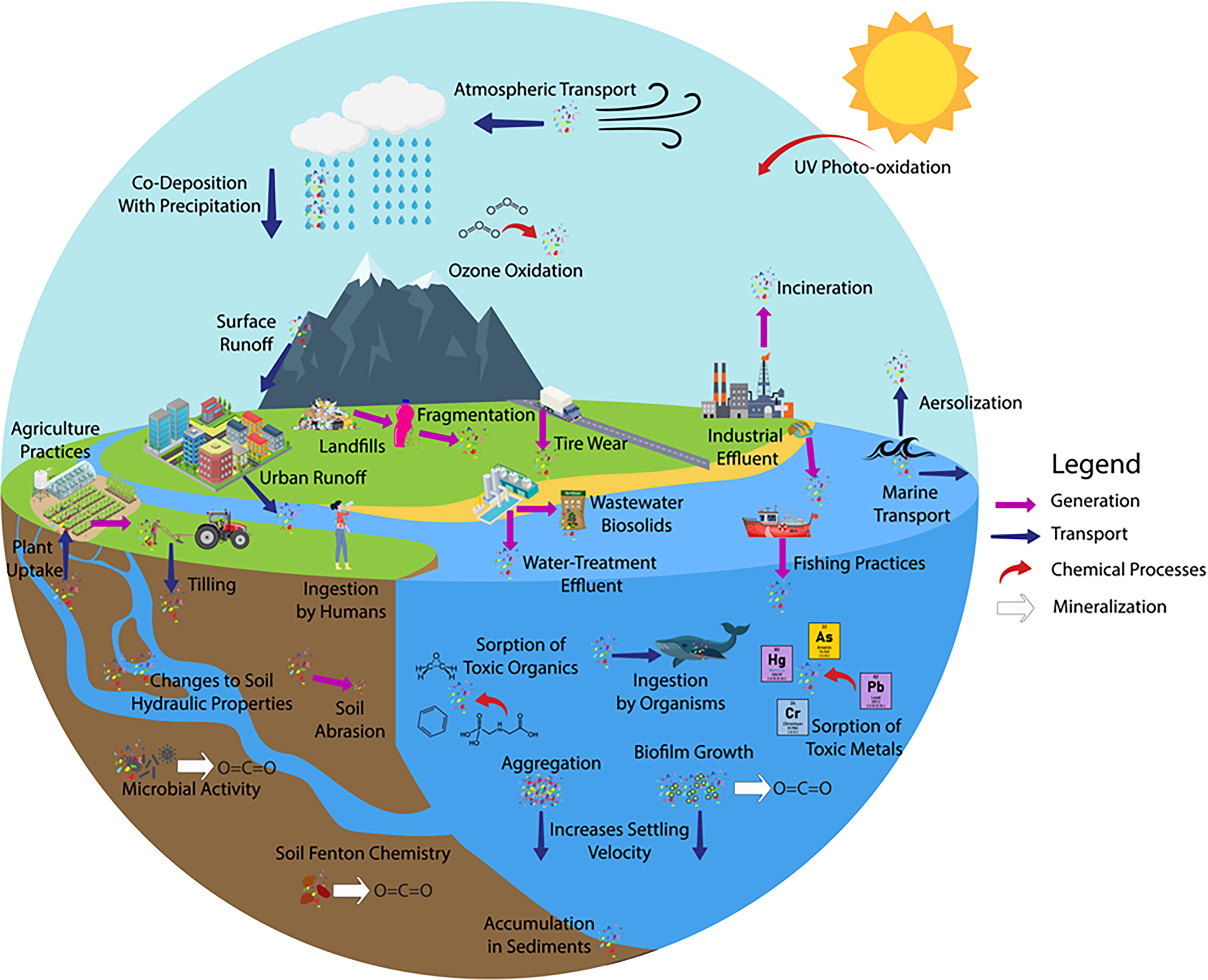
Biogeochemical cycle of microplastics (MPs) in the environment including sources of MP generation and mechanisms for transport, chemical processes, and mineralization of MPs.
MP sources are relatively well understood with numerous published works identifying mechanisms of MP generation (Alimi et al., 2018; Ateia et al., 2022; Bank and Hansson, 2019) and quantifying the number of MPs produced from these sources (Alimi et al., 2018; Allen et al., 2022). Several of these sources of MP generation are shown in Figure 1 (purple arrows). Another very popular topic in the literature is the quantification of the amount of MP in a given environment (sink) ranging from studies of local environments (e.g., number of MPs at a given field site) to efforts to estimate the total amount of MPs in freshwater, marine, terrestrial, and atmospheric systems (Akbari et al., 2024; Bowman et al., 2024; Johnson et al., 2024; Kryl et al., 2024; Setiawati et al., 2024). Although some MP sinks are well-defined (e.g., marine and coastal systems), other environmental sinks are understudied such as terrestrial and atmospheric systems (Allen et al., 2022; Brahney et al., 2021; Horton et al., 2017; Hurley and Nizzetto, 2018). One understudied area related to MP occurrence in different environments is understanding the impacts of high concentrations of MPs on the characteristics of soil systems. For example, high levels of MP in soil can affect soil drainage properties and nutrient retention (de Souza Machado et al., 2019). Additionally, far less is known about fluxes of MP between sinks; there is a need to both identify the nature of MP transport processes and quantify these fluxes (Alimi et al., 2018; Allen et al., 2022; Boyer et al., 2024; Haque et al., 2024; Hasenmueller and Ritter, 2024; Hernandez and Hasenmueller, 2024). Essential factors may include the intrinsic properties of MPs (e.g., density, crystallinity, hydrophobicity) as well as their size and shapes (Horton and Dixon, 2018). Several types of MP transport processes are shown in Figure 1 (blue arrows). Co-occurring with MP transport processes are physicochemical and biological weathering processes (red arrows in Fig. 1) including chemical and biological degradation reactions that change the surface chemistry of plastics and in some instances result in their eventual mineralization within the environment (Alimi et al., 2018; Luo et al., 2022). In addition to the natural processes that result in fluxes of MPs among various environmental compartments, the critical role of anthropogenic processes, such as water and wastewater treatment, in influencing these fluxes and further degradation of MPs should not be overlooked (Crossman et al., 2020; Jeong et al., 2023; Panigrahi et al., 2024; Yu et al., 2024). These engineered systems significantly impact the distribution and fate of MPs in the environment. Furthermore, advancing analytical techniques for the quantification and characterization of MPs could enhance our understanding of their transport across different environmental compartments (Kosuth et al., 2023; Zhu et al., 2024). Improved methodologies would enable more accurate tracking of MP movement and transformation in various ecosystems.
Numerous studies have examined individual mechanisms of plastic degradation, particularly in laboratory settings. Relatively few have focused on the effects that environmental conditions can have on MP degradation pathways. Environmental conditions such as terrestrial versus aqueous environments, salinity, temperature, redox potential, and exposure to sunlight can all affect the mechanisms by which plastics degrade and the rates of their degradation (Andrady et al., 2022; Ren et al., 2021). In an aqueous environment, plastics floating on the surface of the water are exposed to far more UV light and so are more oxidized than denser plastics that fall through the water column (Andrady et al., 2022). Growth of biofilms on MPs (which is more common in aqueous environments) can also slow the rate of oxidative degradation of plastics both by blocking the surface of MPs from UV exposure and by increasing the density of MPs causing them to fall deeper in the water column (Andrady et al., 2022). Biofilm formation may also promote the colonization of microorganisms which can facilitate further degradation, depending on the environmental conditions and the specific microbial communities involved (Debroy et al., 2022). In terrestrial environments, MPs on a soil surface are exposed to far more UV radiation and higher temperatures than those incorporated into the soil through tilling, burrowing by organisms, or vertical transport (Ren et al., 2021). MPs within the soil are exposed to potentially anoxic conditions and high levels of biodegradation compared to MPs on the soil surface or in aqueous environments (Ren et al., 2021). The influence of environmental conditions on MP degradation pathways is complex and while researchers have begun to examine these phenomena, there is much more research to be done.
Plastics’ intrinsic properties and the linkage to their degradation
The intrinsic properties of plastics play a critical role in determining MP performance throughout the use phase and influencing their primary environmental degradation pathways, as outlined in Table 1. Moreover, knowledge of these chemical attributes enables a comprehensive evaluation of MPs’ susceptibility to degradation, aiding the design of environmentally conscious materials. The literature has underscored the polymeric backbone and functional groups as primary drivers of plastics’ environmental degradation characteristics. For instance, extensive research has focused on elucidating mechanisms of photodegradation in plastics with a saturated polymeric backbone such as PE, emphasizing the role of structural defects and impurities introduced during manufacturing or subsequent weathering (Ainali et al., 2021; Fairbrother et al., 2019). Yet, further investigations are needed to identify the type and magnitude of these impurities and the extent of defects generated during weathering processes, which accelerate further degradation of MPs. Understanding their interplay is essential for elucidating the kinetics of MPs’ photooxidation. On the other hand, the presence of unsaturated bonds in polymeric backbones enhances MPs’ susceptibility to oxidation and photodegradation. Plastics with a heteroatom in their polymeric backbone, such as polyethylene terephthalate (PET) and polyurethane (PU), undergo hydrolytic cleavage of ester or amide bonds. This abiotic degradation process not only breaks down the polymeric chains but also creates conditions that can enhance subsequent biodegradation. Plastics containing hydrolyzable covalent bonds (e.g., ester, amide, anhydride, ether, carbamide, or ester amide) may undergo hydrolytic and photodegradation, but the process is slow (Chamas et al., 2020). To expedite these processes, research efforts have been directed toward both modifying the polymer structure (Chen et al., 2022) and promoting biodegradation. Biodegradable plastics are intentionally designed with specific functional groups, such as enzyme-sensitive ester groups, which can be targeted and processed by microorganisms. Enhanced biodegradation can be achieved through the metabolic engineering of microorganisms to enhance the rates of plastic biodegradation (Urbanek et al., 2021). Further investigation is required to examine how undergoing physicochemical degradation in the environment impacts the diversity, composition, and abundance of microbial communities, and thus the biodegradation rates of MPs.
A Summary of the Influence of Microplastics’ Intrinsic Properties on Their Degradation and Their Variations During Degradation
LDPE, Low Density Polyethylene; MP, microplastics; HDPE, High Density Polyethylene; PP, Polypropylene; PVC, Polyvinyl Chloride; PS, Polystyrene; PB, Polybutadiene; PET, Polyethylene Terephthalate; PU, Polyurethane; PEX, Crosslinked Polyethylene; PVDF, Polyvinylidene Fluoride; PEO, Polyethylene Oxide.
The polymer structures, which can be categorized as linear, branched, and crosslinked configurations, can influence the rate of MPs’ degradation. Although research indicates a greater photostability for linear polymers compared to branched polymers, which is attributed to their ability to allow more UV and oxygen penetration as well as greater radical mobility (Herath and Salehi, 2022). However, less information is available regarding how the degree of branching could impact degradation rates. Moreover, the molecular weight of plastics significantly influences their susceptibility to photodegradation. Plastics with lower molecular weights are more vulnerable to degradation. The decrease in molecular chain length also makes MPs more accessible for enzymatic degradation by microorganisms and accelerates their abiotic hydrolysis (Min et al., 2020). Despite the importance of polymeric molecular weight, limited investigations have been conducted to elucidate its critical role in MPs’ environmental degradation and subsequent byproduct release.
The density of MPs significantly affects their environmental fate. Factors such as crystallinity, molecular weight and structure determine the density of MPs, whether they float or sink, and subsequently influence their exposure to environmental factors. Lighter, buoyant MPs remain on the water’s surface, exposing them to aerobic conditions and facilitating longer-distance transport in the atmosphere. Conversely, denser MPs sink into the sediment, potentially undergoing anaerobic biodegradation, and settle more quickly, leading to localized deposition (Beheshtimaal et al., 2024). Future research is needed to examine the variations in MPs’ density as they undergo different degradation processes. This investigation is crucial for enhancing our understanding of their fate and distribution within the environment. Moreover, the hydrophilicity of plastics influences their susceptibility to environmental degradation. Hydrophilic plastics are more prone to hydrolysis and subject to microbial colonization, leading to subsequent biodegradation and additive leaching. However, there has been limited focus on modifying plastics to reduce their hydrophobicity and consequently enhance their biodegradation (Ray et al., 2023).
Most MP environmental degradation studies have been conducted using pure plastic materials; however, the presence of additives within the plastics can significantly influence their degradation kinetics. These additives include a diverse group of compounds, such as plasticizers, slip agents, fillers, light stabilizers, antioxidants, heat stabilizers, biostabilizers, and pigments, that are incorporated into plastics to improve their processability, performance, and durability. The potential role of common plastic additives in degradation is summarized in Figure 2. The intricate role of these additives allows for tailoring plastic formulations to resist the degrading effects of sunlight, ultimately extending the material’s lifespan and functionality during the use phase. Despite their widespread use, there is limited information available regarding the chemical composition and concentration of additives present in commercially available plastics used in everyday life. This lack of data limits our understanding of how additives may influence the environmental degradation of MPs. Additionally, it remains unclear as to what percentage of the additives added during the manufacturing process remains within the plastic structure when they are disposed in the environment. This ambiguity poses a significant challenge in predicting the degradation kinetics of MPs once they are released into the environment. Moreover, in recent years, there has been increasing interest in utilizing additives to enhance the degradation of commodity plastics (Abdelmoez et al., 2021; Selke et al., 2015). However, it is essential to consider the potential environmental impacts of synthetic additives incorporated into the plastics, such as prooxidant/prodegradants designed to accelerate degradation. It remains unclear whether these additives themselves can undergo complete biodegradation. Nevertheless, the development of bioplastics could offer a more sustainable solution for enhancing the degradation of plastics.
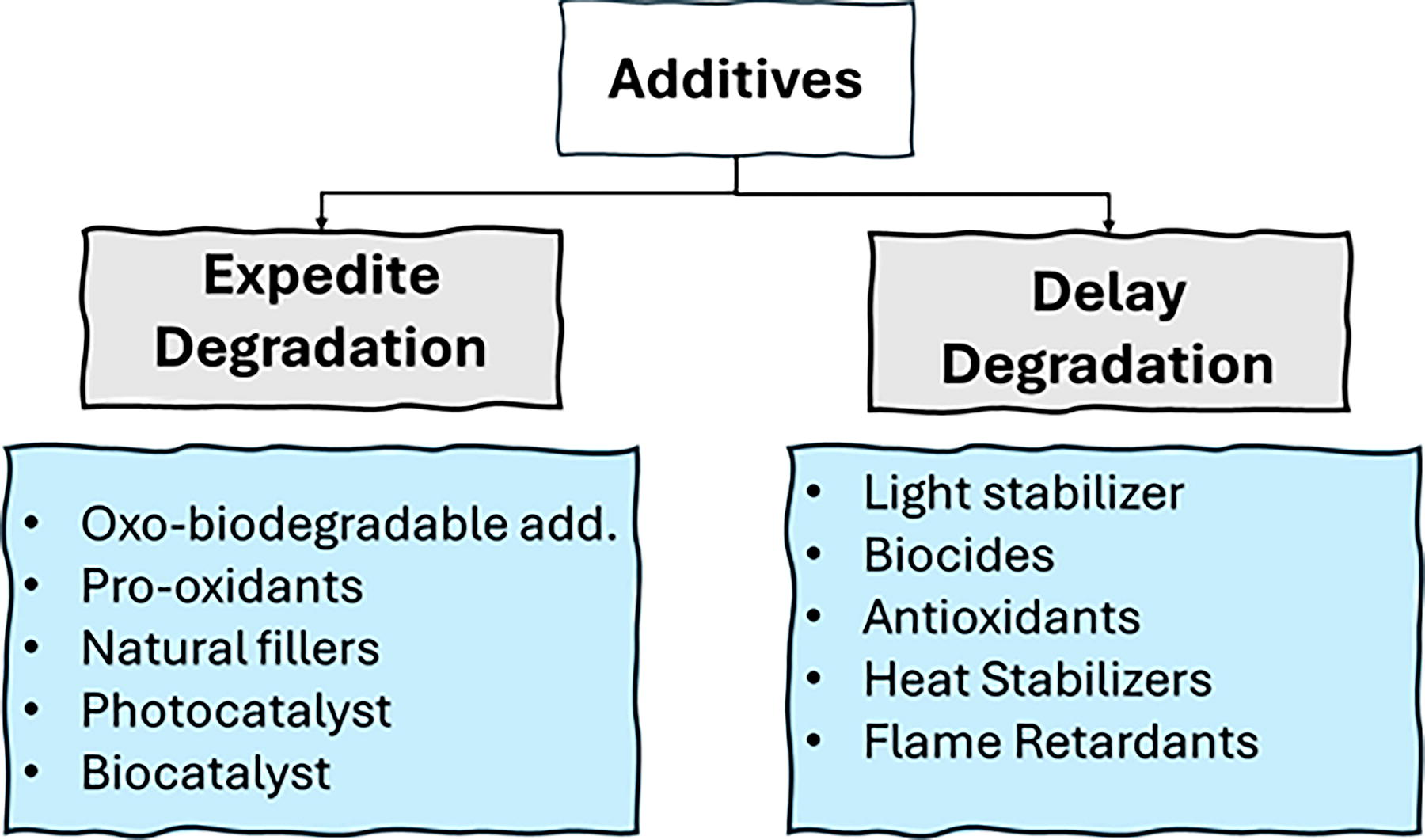
Summary of the potential roles of common plastic additives on degradation.
The effects of environmental degradation on the intrinsic properties of microplastics
The literature has extensively documented changes in MPs’ surface chemistry and morphology due to environmental degradation, including increasing surface roughness, formation of surface cracks, color change, and creation of oxidized carbon functional groups, along with changing hydrophobicity (Aghilinasrollahabadi et al., 2021; McColley and Nason, 2024; Miranda et al., 2021). However, there has been relatively less focus on investigating the variations in the bulk properties and microstructure of MPs. Crystalline zones typically remain intact upon degradation, because cleavage primarily occurs within the amorphous region of the polymer. However, the generation of low molecular weight fragments, their reorientation into crystalline regions, and the crosslinking caused by radicals formed during degradation could alter the molecular weight and crystallinity of MPs (Bonyadinejad et al., 2022; Yousif and Haddad, 2013). Despite their critical importance, the interrelationship of these microstructural changes and the degree of additives and byproducts leaching remains less understood.
As reported in the literature, the degradation of MPs can induce chain scission, thereby weakening the polymer’s overall structure, decreasing its mechanical strength, and facilitating the rapid leaching of additives and degradation/oxidation byproducts. Additionally, mechanical degradation, which enhances surface roughness and fragmentation, contributes to an overall increase in surface area, further accelerating the leaching of additives, unreacted monomers, and oligomers into the environment. The extent of chemical leaching varies depending on environmental conditions, MP properties (e.g., crystallinity, molecular weight), and the structure of the additives. Although more research investigated the leaching of additives from degrading plastics (Luo et al., 2020; Suhrhoff and Scholz-Böttcher, 2016), there has been less investigation into the release of low molecular weight fragments from plastics as oxidation or degradation byproducts (Biale et al., 2022).
Most research on the mechanical degradation of MPs has primarily focused on quantifying the extent of their fragmentation when subjected to various mechanical forces in the aquatic and terrestrial environment or by organisms (Battacharjee et al., 2023; Julienne et al., 2019; Mateos-Cárdenas et al., 2020). However, less attention has been devoted to investigating the linkage between MPs’ intrinsic properties and their mechanical fragmentation. However, the influence of mechanical degradation on the intrinsic properties of plastics could be size-dependent. The strains induced by mechanical forces, including tension, shear, bending, and compression, exerted on polymeric backbones may lead to their breakage at points of physical entanglement. This fragmentation could consequently reduce the average molecular weight of the material (Ravishankar et al., 2018). Moreover, depending on the type of biodegradation including bacterial degradation, fungal degradation, enzymatic degradation, and combined biological degradation, the extent of changes in the intrinsic properties of plastics could be different. Enzymatic degradation results in depolymerization and the formation of shorter molecular chains, oligomers, dimers, and monomers. The small oligomers and monomers could eventually get integrated into the cells by bacteria and be mineralized. Through the biodegradation of plastics, the amorphous regions of the plastics are preferentially attacked by enzymes thus the relative crystallinity of plastic increases following the biodegradation (Aguiar et al., 2024). Although enhanced fragmentation is expected due to increased crystallinity and depolymerization of MPs following biodegradation, further studies are needed to elucidate the links between biodegradation and fragmentation. Such research could offer insights into the long-term transport and ecological impacts of MPs in terrestrial or aquatic systems, particularly considering the reduction in size distribution of MPs resulting from these degradation processes.
Environmental Implications of Microplastic Degradation
Given that environmental persistence is one of the major concerns of MPs as a class of pollutants, it is commonly thought that any degradation of plastics is of environmental benefit. Although the ultimate goal of complete degradation of MPs remains, incremental degradation of plastics can result in materials that have similar or more environmental harm than the initial material. As detailed above, the degradation of MPs results in many changes to their physical and chemical properties, some of which make them more prone to causing deleterious environmental effects. For example, the increased brittleness of plastics degraded through mechanisms such as mechanical abrasion and UV photo-oxidation makes plastics more prone to fragmentation (Andrady et al., 2022). Smaller MPs have a triple threat of an increased surface area to volume ratio (and thus, a higher sorption capacity for pollutants), greater environmental mobility due to their small size, and easier ingestion by organisms due to their small size (Pincus et al., 2023; Ren et al., 2021). MPs, owing to their hydrophobic nature, have a natural tendency to strongly adsorb organic pollutants through multiple mechanisms such as hydrophobic interactions, hydrogen bonding, Van der Waals forces, and π–π interactions (Ren et al., 2021). As MPs degrade, they are often oxidized through mechanisms such as UV photo-oxidation or chemical oxidation, resulting in less hydrophobic more polar surfaces with a higher carbonyl index that has much stronger sorption capacities for inorganic contaminants such as toxic metals and oxyanions (Cai et al., 2024; Herath et al., 2023; Ren et al., 2021). Inorganic contaminants are adsorbed through mechanisms such as electrostatic interactions, surface complexation, precipitation, and coprecipitation (Miller et al., 2024; Quiambao et al., 2023; Ren et al., 2021). Environmental factors such as pH, pe, salinity, and coexistence of other competing molecules in solution can all strongly affect the sorption mechanism and capacity of MPs toward inorganic and organic contaminants (Ren et al., 2021; Wang et al., 2021). Accumulation of contaminants on MPs is of concern due to the increased potential for environmental transport of pollutants, and bioaccumulation and biomagnification of contaminants as MPs are ingested by organisms (Engler, 2012). Thus, it is important to consider that not only are MPs themselves of concern as pollutants but their capacity to serve as vectors for other inorganic and organic contaminants needs to be accounted for as well.
Summary
This paper highlights the critical linkages between MPs’ intrinsic properties and their environmental fate, focusing on degradation processes. The polymeric backbone and functional groups of MPs primarily drive their stability when exposed to environmental factors, significantly influencing degradation pathways and rates. As MPs degrade, their properties undergo substantial changes, affecting further degradation and interactions with contaminants. Most important, the degradation process presents a paradox: while it reduces the duration of MPs’ existence in the environment, the incremental breakdown can result in materials potentially posing greater environmental harm than the initial plastic particles. This complexity underscores the challenges in addressing MP pollution and its long-term environmental impacts. The complex interplay between MPs’ properties, environmental conditions, and degradation processes emphasizes the need for comprehensive research to elucidate mechanisms by which intrinsic properties influence MP degradation under various conditions, investigate the long-term fate of degradation byproducts and their environmental impacts, and develop standardized methods for characterizing changes in MPs’ properties during environmental weathering. This knowledge is crucial for accurately predicting MP’s accumulation in the environment, assessing ecological and health risks, and informing effective policies and technologies to mitigate global plastic pollution.
Footnotes
Authors’ Contributions
M.S.: Conceptualization, investigation, visualization, and writing. L.N.P.: Conceptualization, investigation, visualization, and writing. B.D.: Conceptualization and writing: C.A.P.: Conceptualization and writing.
Author Disclosure Statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Funding Information
Funding for this work was provided by the United States National Science Foundation (NSF) grant CBET-2305189 to the University of Missouri.
