Abstract
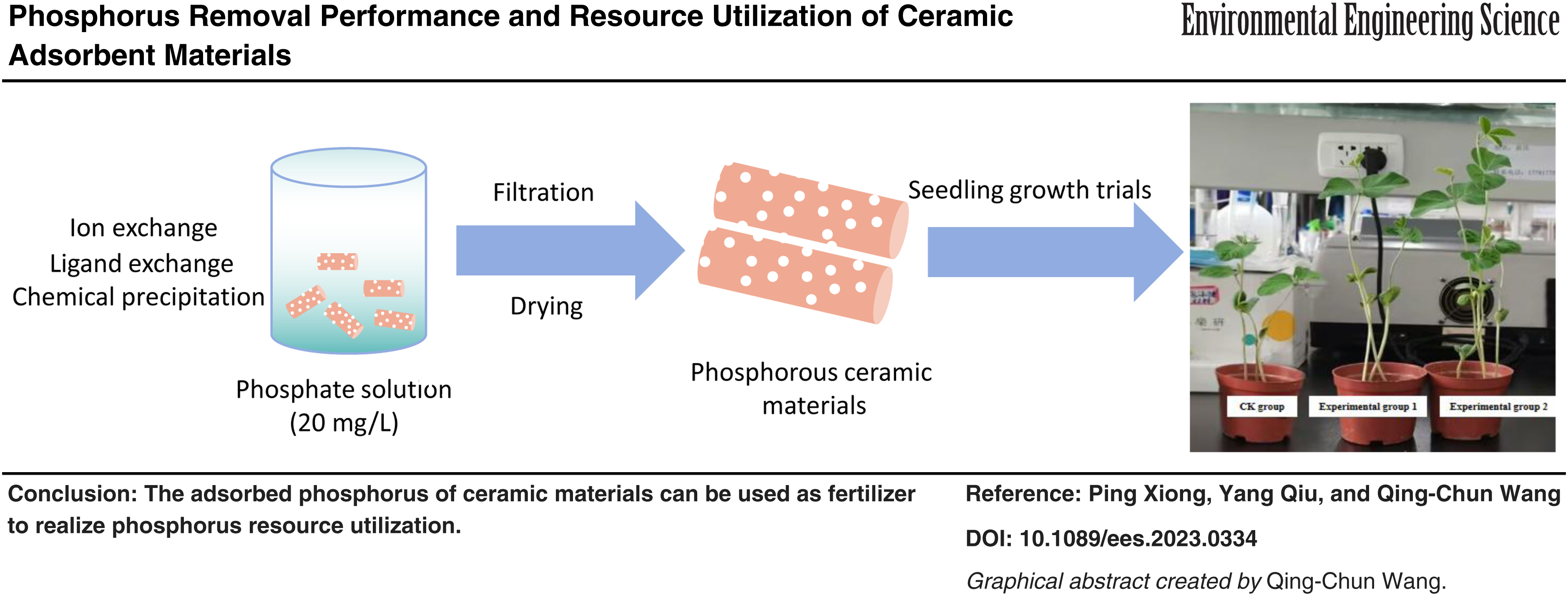
Significant progress has been made in the study of efficient phosphorus adsorption materials. However, more studies need to be on implementing resourceful recycling of materials after adsorption and phosphorus removal. Therefore, this study analyzed ceramic adsorbent materials prepared from clay supplemented with Al2O3, CaCO3, and their use in the treatment of phosphorus-containing wastewater to minimize phosphorus residues, and the feasibility of its direct use as soil. Many adsorption experiments showed that the optimal conditions for removing phosphorus were pH 6, the dosage of 0.15 g/100 mL, and adsorption time of 16 h, and the total phosphorus removal rate reached 96.77%. Meanwhile, ceramic material has good selective phosphate adsorption under the interference of various anions. The phosphorus removal process of ceramic particles was consistent with quasi-secondary kinetics and Langmuir modeling, and it was the monomolecular layer chemisorption with a maximum adsorption capacity of 21.98 mg/g. Phosphorus removal is achieved by ion exchange, ligand exchange, and chemical precipitation between the metal and phosphate in the ceramic material. The phosphate in solution was mainly combined with aluminum and calcium in the ceramics (Al-P: 63.52%, Ca-P: 33.58%), which plants could utilize accounted for 91.68% (Ca2-P, Ca8-P, Al-P, and Fe-P) of the total phosphorus, indicating that the ceramic materials after adsorption of phosphorus had the potential to be used as a phosphate fertilizer. The seedling growth test showed that the adsorbed phosphorus ceramic grain can increase the available phosphorus content of the soil, enhance the activity of alkaline phosphatase, and promote the growth of soybeans, and humic acid can enhance the phosphorus release ability of the ceramic material. It shows that the adsorbed phosphorus of ceramic materials can be used as fertilizer to realize phosphorus resource utilization.
Get full access to this article
View all access options for this article.
