Abstract
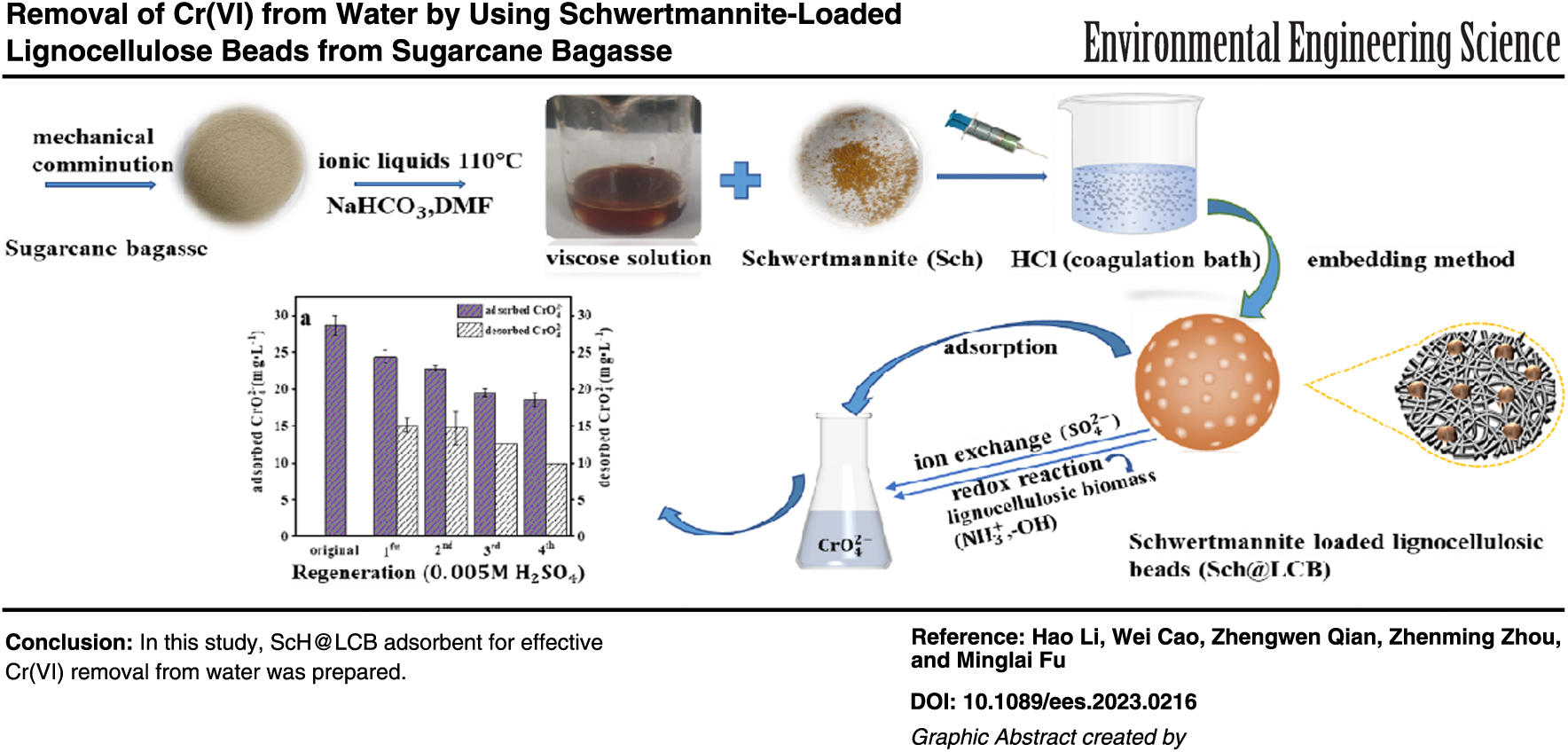
Biosorption is considered a promising method for removing heavy metals from polluted water, benefiting from their abundance, low cost, and environmental friendliness. However, its practical application for Cr(Ⅵ) removal was limited, as the biosorbents often lack uniform size or a stable morphology, and Cr(Ⅵ) oxidation can destroy active adsorption groups. In this study, lignocellulose beads (LCB) with uniform size (∼2 mm) were prepared from sugarcane bagasse using ionic liquids. Additionally, an inorganic structure Schwertmannite (ScH) was synthesized and loaded on LCB, creating permanent active sites for adsorbing Cr(Ⅵ) anions. Characterization studies confirmed that ScH@LCB adsorbent possesses excellent properties, such as homogeneous and stable morphology. Batch experiments indicated that the adsorption isotherms confirmed well with Langmuir model, with the maximum adsorption of Cr(Ⅵ) of 24.62 mg/g. The linear correlation between the released
Get full access to this article
View all access options for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
