Abstract
Chromium (Cr) (VI) is a toxic, mutagenic, and carcinogenic water pollutant. The standard ion chromatography (IC) method for quantification of Cr (VI) in water samples is Environmental Protection Agency Method 218.7, which requires postcolumn derivatization with 1,5-diphenylcarbazide and UV-Vis spectroscopy detection. Method 218.7 is Cr (VI) specific; thus, it does not allow detection of co-occurring natural and anthropogenic anions in environmental media. In this study, we developed an isocratic IC method with suppressed conductivity detection, a Metrohm Metrosep A Supp 7 column, and sodium carbonate/acetonitrile as mobile phase for simultaneous quantification of Cr (VI),
Introduction
Chromium (Cr) (VI) is a toxic, mutagenic, and carcinogenic water pollutant (Cohen et al., 1993; Costa, 1997; Salnikow and Zhitkovich, 2008). The World Health Organization (WHO) set a maximum allowable limit of 50 μg/L for Cr (VI) in groundwater and drinking water (WHO, 2003; El-Shahawi et al., 2011). In the United States, the drinking water maximum contaminant level (MCL) set by the Environmental Protection Agency (EPA) is 100 μg/L total Cr (US EPA, 2010). At the state level, the MCL can be even lower (e.g., 50 μg/L as total Cr in California) (California State Water Quality Control Board, 2020). The standard ion chromatography (IC) method for quantification of chromate ion (CrO42−), the most common Cr (VI) anion, in water samples is EPA Method 218.7 (Zaffiro et al., 2011). Method 218.7 involves the separation of CrO42− (referred henceforth as Cr [VI]) using a high-capacity anion exchange separator column, a postcolumn derivatization with Cr (VI)-specific reagent 1,5-diphenylcarbazide, and a UV-Vis detection of the colored complex at 530 nm (Zaffiro et al., 2011). The Cr (VI)-specific reagent diphenylcarbazide and UV-Vis detection allow sensitive quantification of Cr (VI) at low microgram per liter concentrations by avoiding interference from other anions like sulfate ion (
Cr (VI) often co-occurs with one or more common inorganic anions such as chloride ion (Cl–),
A limited numbers of studies achieved separation and detection of Cr (VI), As (V), and Se (VI) in the presence of common inorganic anions using anion exchange columns and conductivity detection (Bruzzoniti et al., 1999; Kończyk et al., 2018). However, linearity, precision, and accuracy of the co-detected analytes were not reported in these studies (Bruzzoniti et al., 1999; Kończyk et al., 2018), limiting the methods' applicability to environmental samples commonly analyzed in academic or other research-focused laboratories. In this work, we developed an isocratic IC analytical method with suppressed conductivity detection for simultaneous quantification of Cr (VI) and eight other environmentally relevant anions: fluoride ion (F–), Cl–, nitrite ion (
Materials and Methods
Instrumentation
All analyses were performed using a Metrohm AG 930 compact IC flex system (Herisau, Switzerland). The IC was equipped with a chemical suppressor (Metrohm suppressor module [MSM]) and a conductivity detector. An 800 dosino regeneration system was used to deliver the chemical suppressor solution to the MSM. The Metrohm CO2 Suppressor removed the carbonate (as CO2) produced during the chemical suppression reaction in the MSM. The anions were separated using a Metrosep A Supp 7 analytical column (250 × 4 mm; Metrohm) and a Metrosep A Supp 5 Guard column (5 × 4 mm; Metrohm). A Metrohm AG 919 IC autosampler plus was used for sample injection. The volume of the sample injection loop was 1000 μL. The data acquisition and processing were performed with the MagIC Net 3.2 Metrodata software.
Chemicals and reagents
Reagent water, LC-MS Ultra CHROMASOLV (Honeywell, Charlotte, NC), was used to prepare the standards and for the sample dilutions. Cr (VI) standards were prepared using K2CrO4 (Sigma-Aldrich, St. Louis, MO). As (V) and Se (VI) standards were prepared using Na2HAsO4 • 7H2O (J.T. Baker, Phillipsburg, NJ) and Na2SeO4 (ACROS Organics, Geel, Belgium).
The eluent and the MSM suppressor solutions were prepared using deionized and purified water using a PURELAB Ultra (ELGA LabWater, United Kingdom) with a specific resistance ≥18.2 MΏ-cm. The eluent (mobile phase) contained 10.8 mM Na2CO3 (3% [v/v] of Metrohm's A Supp 7 eluent 100 × concentrate) and 35% (v/v) gradient grade acetonitrile (Sigma-Aldrich) in deionized water. The pH of the eluent as prepared was 11.9 ± 0.02. The MSM suppressor solution contained 500 mM H2SO4 in deionized water.
The 10% (v/v) H2SO4 and 10% (v/v) H3PO4 solutions for colorimetric determination of Cr (VI) were prepared from concentrated H2SO4 (95–98% solution; VWR, Randor, PA) and concentrated H3PO4 (85% solution; Alfa Aesar, Haverhill, MA), respectively. The complexing reagent contained 5 g/L of 1,5-diphenylcarbazide (Sigma-Aldrich) in acetone.
Analytical methods
The IC method used a constant eluent flow rate of 0.8 mL/min and a constant column/oven temperature of 55°C. The MSM stepping interval was 10 min and the conductivity detector was set at 2.3% per °C. At these conditions, the back pressure was 12 ± 0.4 MPa. The pump start-up time was at 45–60 min during the equilibration of the instrument. Calibrations for the anion analytes were established by injecting quadruplicates of 1, 5, 10, 25, 50, 100, and 200 μg/L standard mixture. The upper limit of quantification (LOQ) for Cl–,
EPA Method 7196A was used to quantify Cr (VI) in a contaminated surface water sample and compare the concentrations obtained by the IC method. Cr (VI) concentration was determined colorimetrically at 540 nm using the diphenylcarbazide method (US EPA, 1992). In brief, 0.1 mL of sample or standard was added to a 10 mL test tube followed by addition of 1 mL each of 10% H2SO4 and 10% H3PO4. Then, 0.1 mL of 5 g/L diphenylcarbazide in acetone was added to the test tube. The mixture was then vortexed and incubated at room temperature for 5 min. Absorbance of the magenta color was analyzed using a Varian Cary 50 UV-Vis spectrophotometer (Agilent, Santa Clare, CA) at 540 nm. The spectrophotometer was calibrated using the standard Cr (VI) solution. The calibration range for the colorimetry method was 0.5–75 mg/L Cr (VI) and the detection limit was 0.25 mg/L.
Resolution, limit of detection, LOQ, and accuracy
Resolution of two peaks (R), defined as the ratio of the difference in retention times between two peaks and the average baseline width of two peaks (Harris, 2010), was determined using Eq. (1):
where TR1 and TR2 are the retention times of adjacent peaks (analyte 1 elutes before analyte 2) and wb1 and wb2 are the widths of the peaks at baseline.
The limit of detection (LOD), defined as the lowest concentration of analyte in a sample that can be readily distinguished from the absence of that analyte (a blank value) (McNaught and Wilkinson, 1997; Inczedy et al., 1998; Allegrini and Olivieri, 2014), was determined using Eq. (2):
The LOQ, defined as the smallest concentration of analyte in a sample that can be quantitatively determined with suitable precision and accuracy, was determined using Eq. (3):
In Eqs. (2) and (3), Sa is the standard deviation of the response estimated by the standard error of y-intercepts of the regression lines and b is the slope of the calibration curve (Shrivastava and Gupta, 2011). A calibration curve with concentrations between 0.3 and 25 μg/L was used to obtain LOD and LOQ of all analytes.
Accuracy, defined as the closeness between a measured value and either a true or accepted value, was evaluated from precision and trueness values of each analyte (Munch et al., 2005). The precision was determined by calculating the relative standard deviation (RSD) using Eq. (4). Trueness was determined by calculating the recovery using Eq. (5).
Environmental samples
Tap water from the city of Tempe and reverse osmosis grade water (US Water Systems, Indianapolis, IN) were collected at the Biodesign Institute, Arizona State University (Tempe, AZ). Tap water from the City of Mesa was collected from a domicile in Mesa, AZ. Three groundwater samples were obtained for testing. One groundwater sample was from Phoenix Goodyear Airport-North Superfund site (AZ) (Rangan et al., 2020). The other samples were collected from two confidential sites in the Southwestern United States. Cr (VI) contaminated surface water was collected from Tamil Nadu Chromates and Chemicals Ltd. (TCCL), an abandoned chromate manufacturing facility in Ranipet, Tamil Nadu, India. The wastewater samples used in this study were received from a power station in the Eastern United States and from the Northwest Water Reclamation Plant (Mesa, AZ).
Laboratory microcosm experiments
The developed IC method was applied to monitor anions in soil and culture-only batch microcosms. Soil laboratory microcosms (Ziv-El et al., 2011; Rangan et al., 2020; Joshi et al., 2021) focused on abiotic and microbiological Cr (VI) reduction were established in 160 mL glass serum bottles with 25 g of Cr (VI)-contaminated soil and 100 mL anaerobic mineral medium as described elsewhere (Delgado et al., 2012, 2017). The soil was collected from 0 to 0.25 m depth at the TCCL site, India, and was homogenized in the anaerobic glove chamber (Coy Laboratory Products, Inc., Grass Lake, MI) under 3.5% H2 and 96.5% N2 atmosphere. Two grams per liter yeast extract and 10 mM lactate (870 mg/L) were added to the microcosms as electrons and carbon sources for the microorganisms. The initial Cr (VI) concentration in the soil microcosms was ∼90 mg/L.
Culture-only microcosms (Delgado et al., 2014, 2016) focused on microbiological Cr (VI) reduction were established in 160 mL serum bottles containing 100 mL anaerobic mineral medium as used in soil microcosms. The inoculum (4% v/v) was a mixed culture grown on Cr (VI) and lactate. The culture-only microcosms were amended with 1 g/L yeast extract and 3 mM lactate (∼260 mg/L). The initial concentration of Cr (VI) was 15 mg/L. All (soil and culture-only) microcosms were established in triplicates, were incubated in the dark at 30°C, and were shaken on a platform shaker at 125 rpm. Liquid samples from the soil microcosms were collected for IC analysis during the experiment at 0.2, 3.7, and 8.2 h. Liquid samples from the culture-only microcosms were collected at 0, 2, 7, 10, 11, 14, and 17 days. The liquid samples were filtered using 0.2 μm syringe filters (mdi Membrane Technologies, Inc., Harrisburg, PA) and analyzed for anions by IC.
Results and Discussion
In this study, we report an isocratic IC method with suppressed conductivity detection for simultaneous quantification of Cr (VI), F–, Cl–,
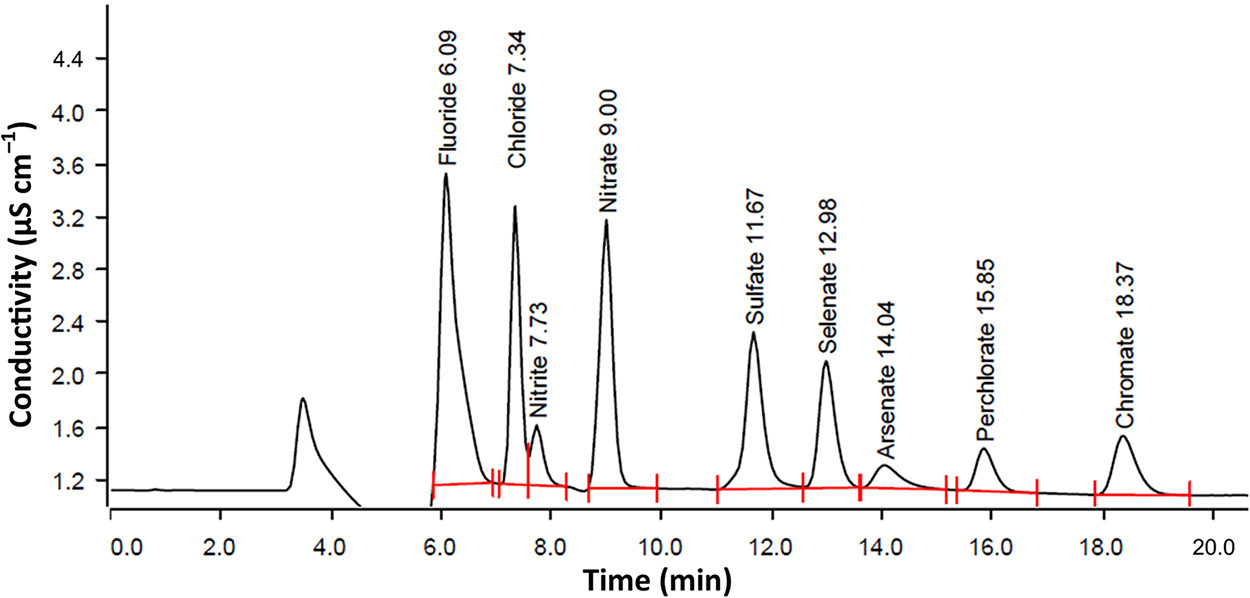
IC chromatogram of a mixture of 50 μg/L each of Cr (VI),
Resolution, Regression Equation, Determination Coefficient, Quantification Range, Limit of Quantification, and Limit of Detection of Nine Analytes Using the Method from This Study
R values >1.5 are baseline resolutions.
Cr, chromium
A comparison of published IC methods for measurement of Cr (VI) in aqueous samples is given in Table 2. The contribution of our method over previously published IC methods for Cr (VI) quantification is that
Comparison of Various Ion Chromatography Methods for Chromium (VI) Quantification in Aqueous Samples
Values from carbonate/bicarbonate eluent system.
Linear regression equation, precision, and trueness were not reported.
EPA, Environmental Protection Agency; NR, not reported.
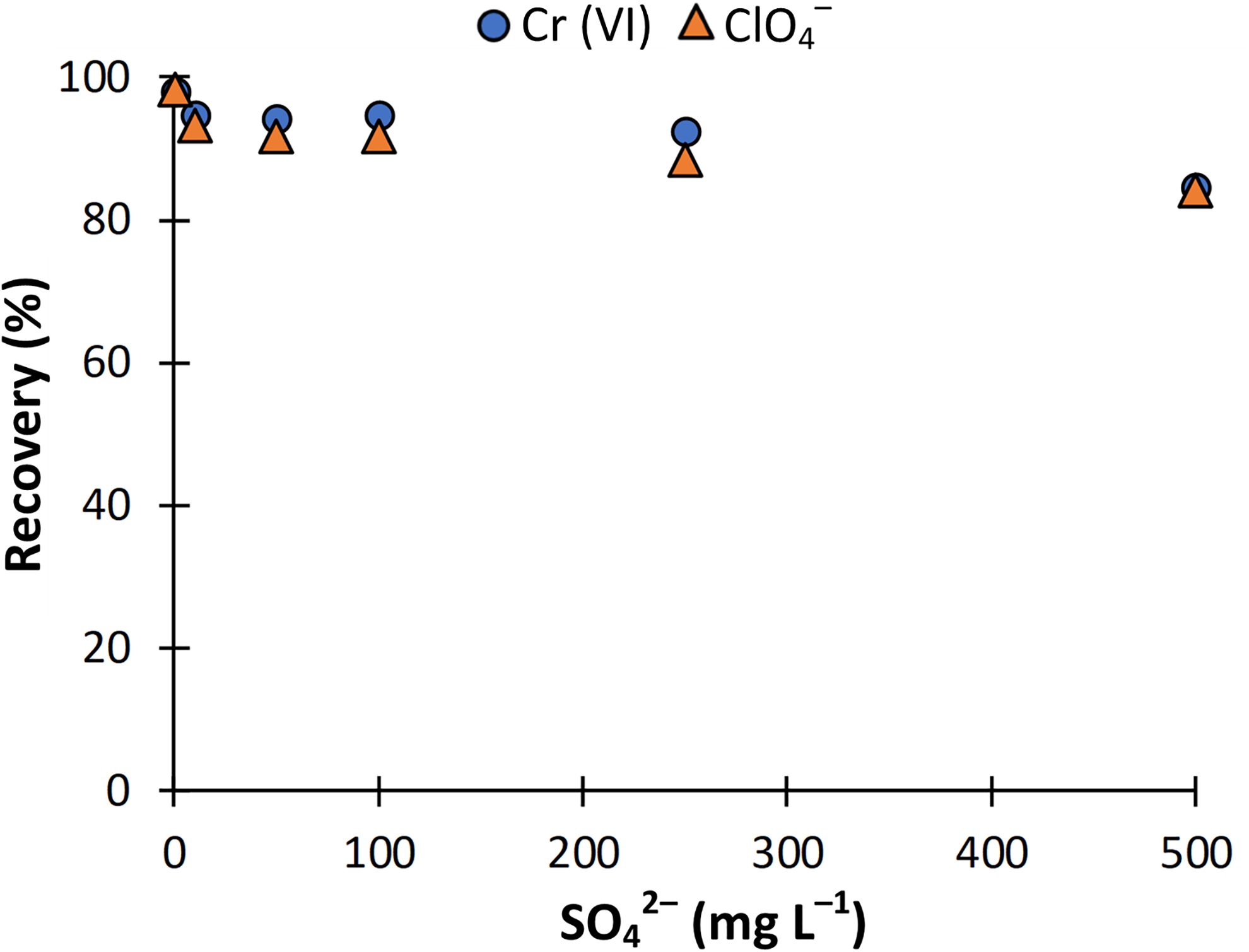
Effect of
We evaluated the analytical accuracy (precision and trueness) for quantification of the anions at three concentration levels (2, 10, and 100 μg/L) using the developed IC method. In reagent water or DI water, US EPA's acceptance criterion for RSD is ≤10%. The acceptance criterion for recovery is 80–120% for mid-level check standards (US EPA, 1996; Hautman et al., 1999). The acceptance criterion for recovery is 50–150% at concentrations close to the LOD of the analyte (low-level check standard) (US EPA, 1996). Table 3 documents the recovery of all anion analytes. At 100 μg/L, all analytes were quantified with RSD <2.3% and the recovery was in the range of 96.2–107.9%, showing precision and trueness (accuracy) for quantification (Table 3). At 10 μg/L, the RSD and recovery for F– and Cl– were substantially affected (RSD values >10% and recovery of 47.5–90.6% [Table 3]). These results are expected as 10 μg/L is within a factor of 3 from the LOD of F– and Cl– (US EPA, 1996). All other analytes were quantified with RSD <7.4% and recovery of 92.6–105.3% using 10 μg/L standard (Table 3). At 2 μg/L concentration, all analytes except
Analyte Accuracy of Quantification Using the Method From This Study
NA, not applicable (concentration below limit of detection); RSD, relative standard deviation.
To test the applicability of the developed IC method on environmental aqueous samples, we evaluated the recovery of all analytes in deionized water, tap water, surface water, groundwater, and wastewater. The US EPA's acceptance criteria for recovery of analytes in environmental samples is 80–120% (Hautman et al., 1999). As given in Table 4, the recovery of Cr (VI) in all environmental samples tested was in the range of 97.2–102.8%. The recovery of the other analytes was within the acceptable recovery criterion in most environmental samples (Table 4). These data support the applicability of this method for simultaneous quantification of the analytes in environmental aqueous samples.
Recovery of All Anion Analytes in Environmental Samples
The data are averages with standard deviation of triplicates. The spiking concentration for all anions was 100 μg/L.
ND, not determined; RO, reverse osmosis.
We evaluated the trueness of Cr (VI) concentration in the surface water sample measured with our IC method by comparing it with the measured value using the EPA method 7196A (diphenylcarbazide-based colorimetry method). The concentration of Cr (VI) in the surface water was 20.6 ± 0.2 mg/L using the diphenylcarbazide method (EPA Method 7196A). Assuming this was the true Cr (VI) concentration, the recovery of Cr (VI) concentration using the IC method was 100.2 ± 3.4% (data not shown), demonstrating trueness for Cr (VI) quantification in the surface water sample. For Cr (VI) quantification using the IC method, the surface water was diluted 1000 times with reagent water to fit the Cr (VI) concentration within the calibration range.
We further applied the IC analytical method to simultaneously track concentrations of anions in typical batch microcosms used commonly in laboratory settings. The microcosms in this study were focused on abiotic and microbiological Cr (VI) reduction. Figure 3 shows the time course concentrations of Cr (VI) (naturally present and spiked) and
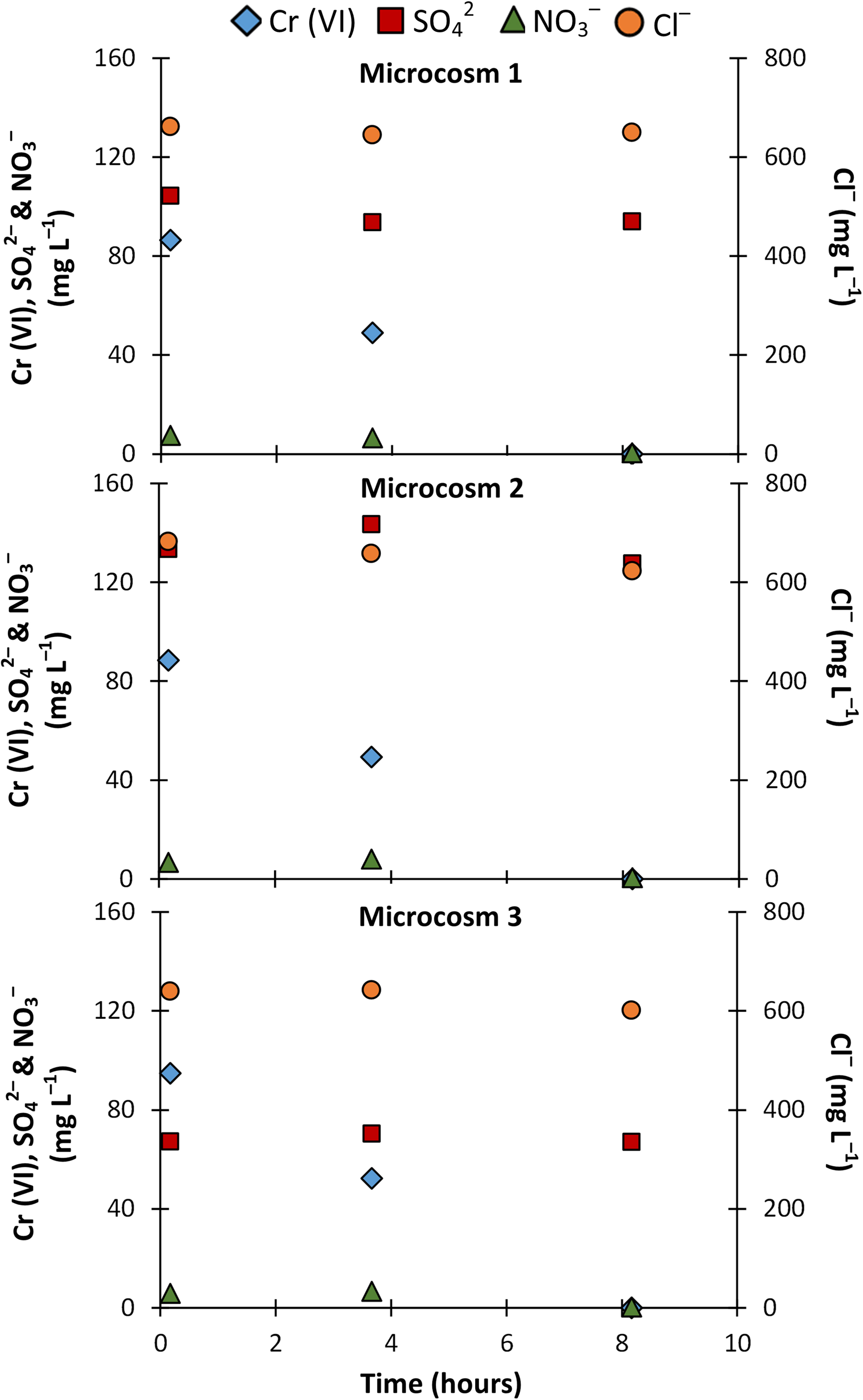
Concentrations of Cr (VI),
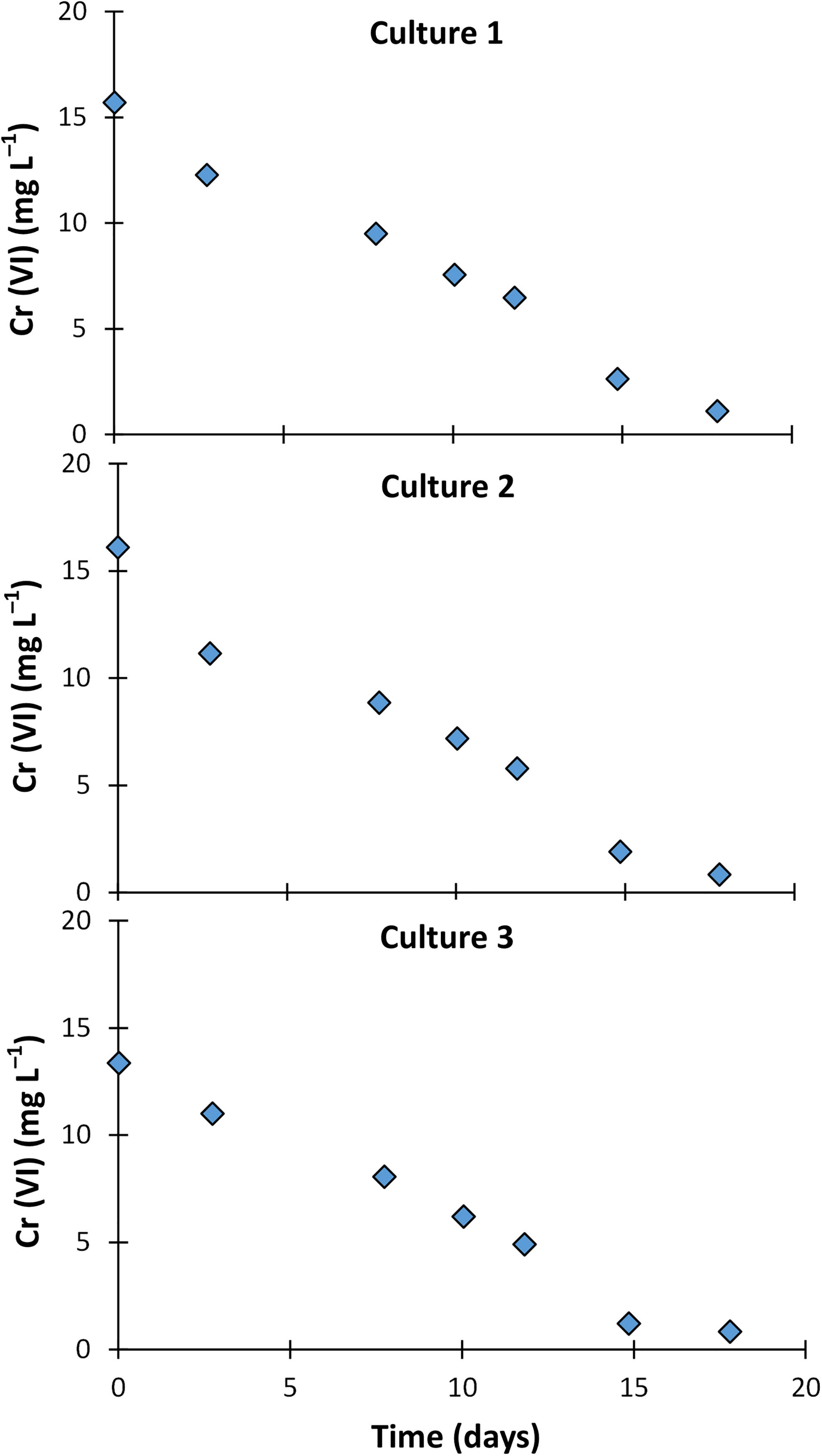
Concentrations of Cr (VI) during incubation in replicate culture-only microcosms.
Owing to the capability of quantifying several anions simultaneously, the IC method developed in this study is useful to environmental practitioners, academic and research organizations, and other industries that routinely measure Cr (VI) and co-occurring anions. An ion chromatograph equipped with a suppressed conductivity detector is a common instrumentation that many laboratories possess for quantification of common inorganic anions (e.g., Cl–,
Footnotes
Acknowledgments
The authors acknowledge Ibrahim Ibrahim and Anton Sachs for their assistance in conducting the laboratory microcosm experiments.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This material is based upon work supported by the National Science Foundation (NSF) under NSF CA no. EEC-1449501. Any opinions, findings, and conclusions or recommendations expressed in this material are those of the authors and do not necessarily reflect those of the NSF.
