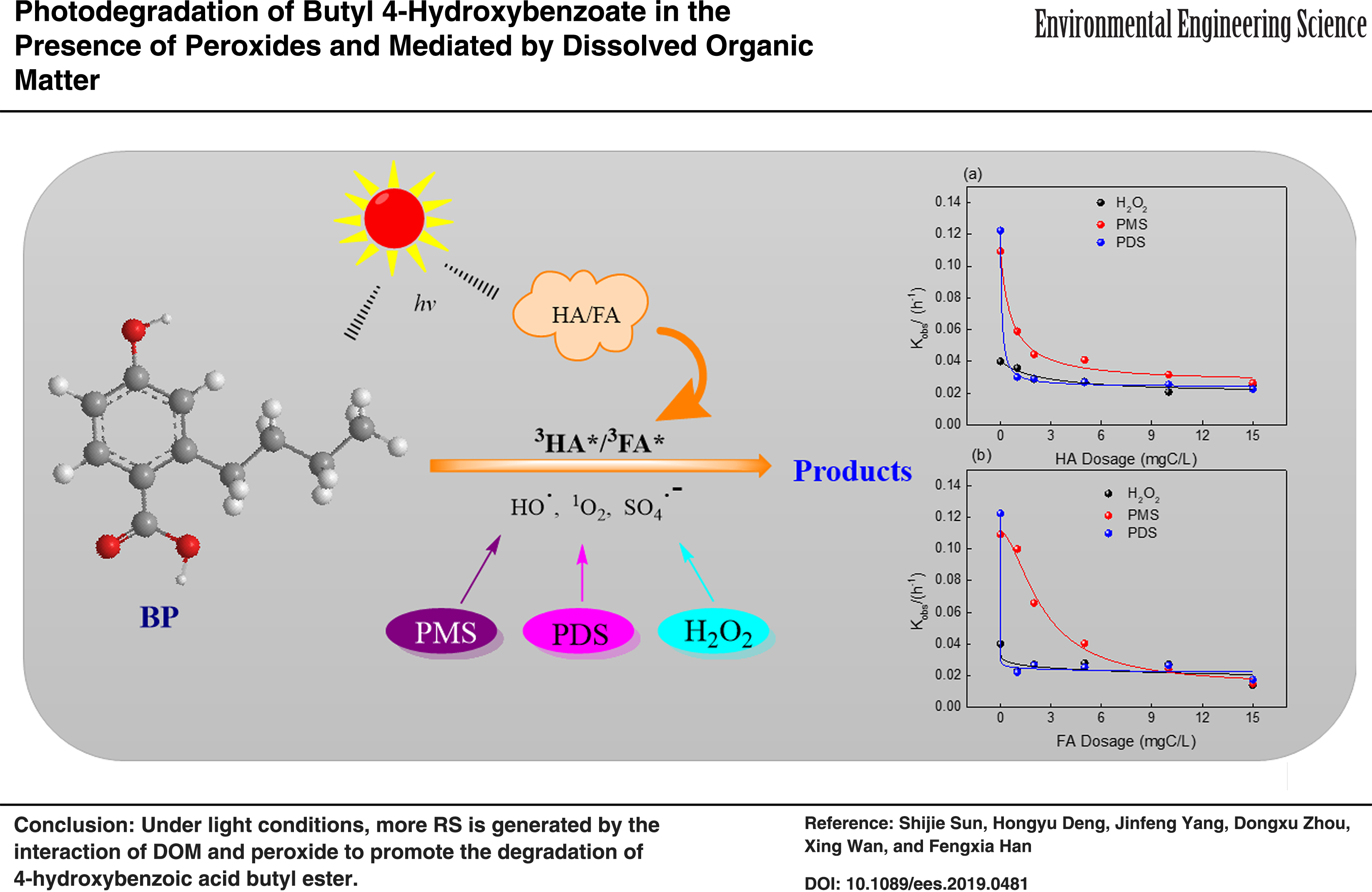
Abstract
Dissolved organic matter can be a photosensitizer in photodegradation processes. It can produce hydroxyl radicals, singlet oxygen, and triplet-state dissolved organic matter, which promote pollutant degradation. This study investigated to what extent the triplet substances and other active substances can, under irradiation, stimulate peroxides to produce more reactive species (RS), which accelerate the degradation of pollutants. The photodegradation of butyl 4-hydroxybenzoate (BP) was studied in the presence of dissolved organic matter (DOM) and persulfate (PDS), peroxymonosulfate (PMS), or hydrogen peroxide (H2O2). The peroxides promoted the photodegradation in the order PDS > H2O2 > PMS. The degradation rates were peroxide > DOM-mediated peroxide > DOM. The interaction of DOM with peroxide produced more RS to promote the degradation. Chlorella vulgaris were used to evaluate the toxicity of the treated effluent and the ecological risk was found to be low. The findings explain the mechanism of the photodegradation of BP in the presence of dissolved organics and peroxides.
Graphical abstract
Get full access to this article
View all access options for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
