Abstract
This study investigated the effects of controlling parameters, temperature, pH, ozone dose, and initial Acid
Orange 6 (AO6) concentration on the degradation of target compounds. Decolorization and AO6 reduction
were completed within 40 min of ozonation time for all examined cases. Significant amounts of total
organic carbon (TOC) still remained by the end of experiments, suggesting that ozone could not oxidize
all organic molecules to CO2. Major findings from this study were as follow: (1) the decolorization
of AO 6 dye was affected by ozone dose and pH; however, the effects of temperature contribution on apparent
rate constant was limited. (2) TOC removal was affected by temperature significantly, but the affects
of pH and ozone dose were limited. (3) The highest 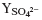 level was about 0.70 as η
TOC was about
0.75, and the increment of
level was about 0.70 as η
TOC was about
0.75, and the increment of 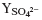 was accelerated as η
TOC > 0.4. (4) The η
TOC was proportional to the
7.64
was accelerated as η
TOC > 0.4. (4) The η
TOC was proportional to the
7.64 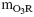 power of exponential term. (5) NO and NO2 were found in off-gas that might provide the explanation
of unbalance of nitrogen mass.
power of exponential term. (5) NO and NO2 were found in off-gas that might provide the explanation
of unbalance of nitrogen mass.
Get full access to this article
View all access options for this article.
