Abstract
Background:
Many young people with type 1 diabetes have overweight or obesity (OW/OB). OW/OB increases risks of cardiovascular and kidney disease, leading causes of death in type 1 diabetes. Semaglutide, a glucagon-like peptide receptor agonist (GLP-1-RA), improves body mass index (BMI) and glycemia in adults with type 1 diabetes and OW/OB. However, GLP-1-RA and GLP-1/gastric inhibitory polypeptide (GIP)-RA data are lacking in younger patients with type 1 diabetes.
Methods:
We assessed real-world use of GLP-1-RA and GLP-1/GIP-RA in 33 youth/young adults with type 1 diabetes and OW/OB.
Results:
From baseline to 12 months, HbA1c decreased by 0.32% (±0.12), BMI by 2.2 kg/m2 (± 0.38), weight by 7.0 kg (±1.5), continuous glucose monitoring (CGM) average glucose by 19.2 mg/dL (±6.7) and time above range by 10.8% (±3.8); CGM time in range increased by 11.7%. Insulin dose also decreased by 14.6 units/day (±5.7) at 6 months.
Conclusions:
These findings suggest GLP-1-RA and GLP-1/GIP-RA are promising options for treating type 1 diabetes complicated by OW/OB in youth and young adults.
Introduction
Among youth with type 1 diabetes, overweight and obesity (OW/OB) are common, increasing the cardiovascular risk of type 1 diabetes, and may be more difficult to treat because of the effects of exogenous insulin.1,2 Semaglutide, a glucagon-like peptide receptor agonist (GLP-1-RA), improved body mass index (BMI) and glycemia in adults with combined OW/OB and type 1 diabetes, but GLP-1-RAs remain poorly studied among youth/young adults with type 1 diabetes. 3 We aimed to evaluate real-world experience using GLP-1-RA and GLP-1/gastric inhibitory polypeptide (GIP)-RAs in youth and young adults with OW/OB and type 1 diabetes, describing glycemic control (HbA1c and continuous glucose monitoring [CGM] metrics), insulin dosing, BMI, weight, and blood pressure, with the primary outcome of HbA1c at 6 months.
Methods
We performed a retrospective chart review of the electronic medical records (EMR) in youth and young adults with OW/OB and type 1 diabetes treated at the Barbara Davis Center for Diabetes who began a GLP-1-RA or GLP-1/GIP-RA between 6/14/11 and 3/29/24. This study was determined exempt (category 4, secondary research) by the Colorado Multiple Institutional Review Board (20-2686) with a full waiver of the Health Insurance Portability and Accountability Act of 1996.
Inclusion criteria were (1) age 12–25 years, (2) BMI ≥ 25 kg/m2 (≥18 years of age) or BMI percentile ≥85th percentile (<18 years of age), (3) usage of GLP-1-RA or GLP-1/GIP-RA for ≥3 months, (4) positive type 1 diabetes-associated antibodies or a highly convincing type 1 diabetes clinical presentation determined by a pediatric endocrinologist, (5) ≥1 outcome measure at 6 or 12 (±3) months. Exclusion criteria were (1) bariatric surgery within 12 months of starting a GLP-1-RA or GLP/GIP-RA and (2) pregnancy at any point in the study period.
Fifty-four individuals were identified from the EMR who were prescribed a GLP-1-RA or GLP-1/GIP-RA. After excluding 3 individuals for incorrect diabetes type, 3 for incorrect BMI, 14 not on a GLP-1-RA or GLP-1/GIP-RA for at least 3 months, and 3 lacking outcome measures at 6 or 12 months, the final cohort included 33 individuals meeting all inclusion/exclusion criteria. Of 12 individuals meeting eligibility but not on a GLP-1-RA or GLP-1/GIP-RA for ≥3 months, 3 used the GLP-1 or GLP-1/GIP-RA for <3 months, 7 never initiated treatment because insurance denied coverage, 1 never initiated treatment due to inability to afford the copayment, and 1 discontinued use before 3 months due to medication intolerability. Eighteen individuals were sequentially prescribed more than one type of GLP-1 or GLP-1/GIP-RA. Exact GLP-1-RA or GLP-1/GIP-RA start dates were defined as the start date of the prescription or first documented use and stop dates were defined as the end date of the prescription or last documented use. Additional data collected included other medication use, BMI, weight, HbA1c, total daily insulin dose (units/day and units/kg/day), CGM metrics (time in range, time below range, time above range, and mean glucose), and systolic (SBP) and diastolic blood pressure (DBP) from the visit where the GLP-1-RA or GLP-1/GIP-RA was initiated, 6 and 12 months after initiation (allowing for a window of ±3 months). Reports of severe hypoglycemia and hospitalization for diabetic ketoacidosis or hyperglycemia were also collected. Changes in absolute BMI, SBP, and DBP were compared over time (rather than percentile for age) because 45% of our cohort was ≥18 years of age.
To compare changes in outcome measures from baseline to 6 months, 6 to 12 months, and baseline to 12 months, mixed-effects repeated measures analysis of variance (ANOVA) with the Geisser-Greenhouse correction was used. When appropriate, multiple comparisons were conducted using Tukey’s post-hoc test to adjust for multiple testing. Significance was set at P < 0.05. Analyses were completed on GraphPad Prism version 10.2.1. Missing data were handled with the mixed-effects statistical model, which applies maximum likelihood estimation to account for missing values under the assumption that they are missing at random. This method ensures that all available data contribute to the analysis without requiring imputation or listwise deletion. Our statistical analysis did not differentiate between patients based on their use of GLP-1 or GLP-1/GIP agents. As a result, no distinction in statistical analysis was made for patients who were sequentially treated with two different GLP-1 or GLP-1/GIP medications. A priori power analysis (G*Power version 3.1) was conducted to determine the minimum sample size required to achieve statistical power using previously published data. 4 A sample size of 21 participants was predicted to be needed to achieve 80% power to detect an effect size (f2) of 0.65 to detect statistically significant differences in the primary outcome of change in HbA1c from baseline to 6 months using a t-test with a significance level (alpha) of 0.05.
Results
Baseline characteristics of the study population (n = 33) are shown in Table 1. The study cohort was predominantly non-Hispanic White, with a BMI (ages 18–25 years) or BMI percentile (ages 12–17 years) in the obese range. Semaglutide was the most frequently used GLP-1-RA or GLP-1/GIP-RA.
Baseline Characteristics and Changes in Glycemic and Anthropometric Parameters
*P < 0.05, **P < 0.01, ***P < 0.005.
BMI, body mass index; HbA1c, glycosylated hemoglobin; SBP, systolic blood pressure; DBP, diastolic blood pressure; GLP-1-RA, glucagon-like peptide receptor agonist; GIP-RA, glucose-dependent insulinotropic polypeptide receptor agonist.
Changes in average BMI, weight, HbA1c, total daily insulin dose, CGM, SBP, and DBP are shown in Table 1 and Figure 1. There was a decrease in BMI (mean ± SD) from baseline (36.0 ± 8.1 kg/m2) to 6 months (35.1 ± 8.4, P = 0.018), from 6 months to 12 months (33.9 ± 6.3, P = 0.015) and from baseline to 12 months (P < 0.001). Weight also decreased from baseline (103.8 ± 34.1 kg) to 12 months (96.8 ± 24.0, P < 0.001) and from 6 months (102.7 ± 34.6) to 12 months (P = 0.003).
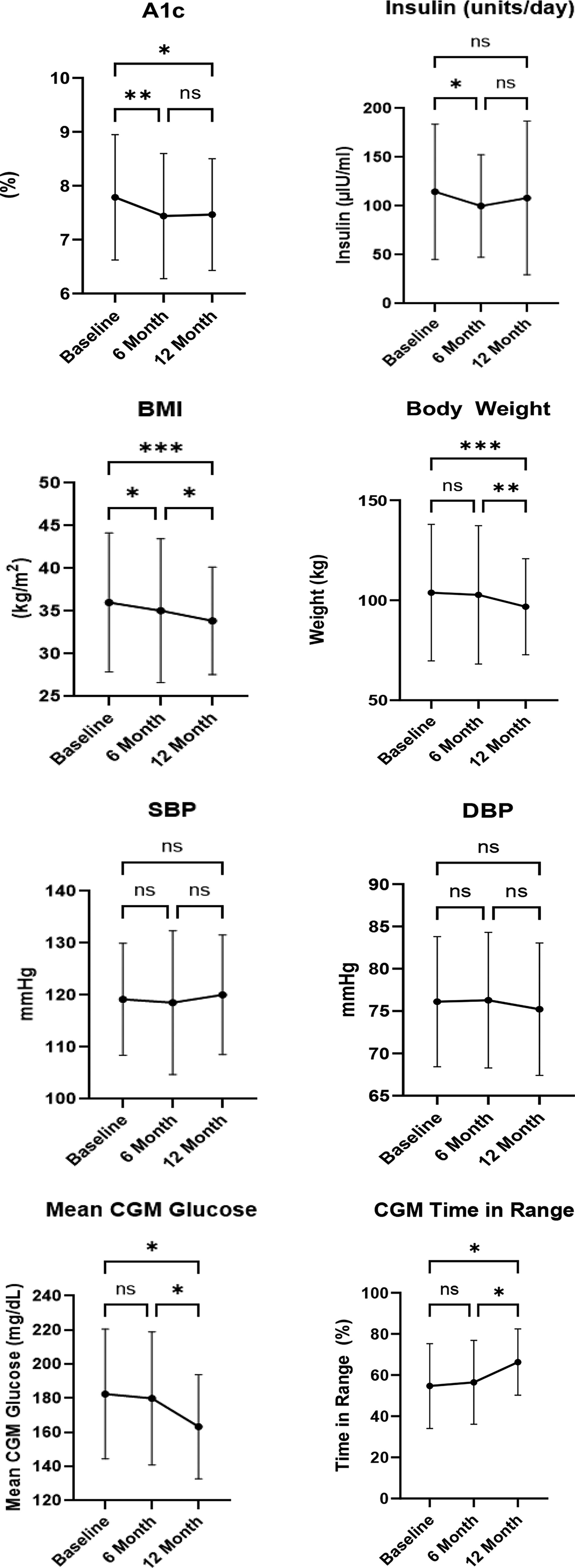
Change in A1c, insulin (units/day), body mass index (BMI), body weight, systolic blood pressure (SBP), diastolic blood pressure (DBP), mean continuous glucose monitor (CGM) glucose, CGM time in range, CGM time above range, and CGM time below range. *P < 0.05, **P < 0.01, ***P < 0.005.
HbA1c decreased from baseline (7.8% ± 1.2%) to 6 months (7.4% ± 1.2%, P = 0.003) and baseline to 12 months (7.5% ± 1.0%, P = 0.036). Insulin dose decreased from baseline (114.7 ± 69.3 units/day) to 6 months (100.1 ± 52.5, P < 0.05). CGM time in range increased from baseline (54.7% ± 20.7%) to 12 months (66.3% ± 16.1%, P = 0.023) and 6 months (56.5% ± 20.4%) to 12 months (P = 0.039). CGM time above range decreased from baseline (42.8% ± 21.6%) to 12 months (32.0 ± 16.5%, P = 0.033). Mean CGM glucose decreased from baseline (182.5 ± 38.1 mg/dL) to 12 months (163.3 ± 30.6, P = 0.031) and 6 months (179.9 ± 39.0) to 12 months (P = 0.020). There were no statistically significant changes in SBP, DBP, insulin dose in units/kg/day, or CGM time below range.
Twelve (36.4%) patients reported gastrointestinal side effects related to GLP-1-RA or GLP-1/GIP-RA use, with the most common being nausea. There were two hyperglycemic events, 4 and 9 months after initiation of a GLP-1-RA or GLP-1/GIP-RA requiring a medical visit, and one episode of diabetic ketoacidosis requiring hospitalization 6 months after initiation of a GLP-1-RA or GLP-1/GIP-RA.
Conclusions
In this retrospective study, GLP-1-RA or GLP-1/GIP-RA use in youth and young adults with type 1 diabetes and OW/OB resulted in a statistically and clinically significant decrease in BMI and improved glycemic control by HbA1c and CGM metrics at 6 and 12 months, a decrease in insulin dose at 6 months, and a decrease in weight at 12 months after initiation. These results are important because of the high frequency of overweight and obesity among youth and young adults living with type 1 diabetes, the paucity of effective treatments for OW/OB, and the fact that only about a quarter of people living with type 1 diabetes in the United States currently reach glycemic targets, with the worst glycemic control occurring during adolescence. 5 Both the current obesity epidemic and the weight gain associated with intensive insulin therapy contribute to the prevalence of OW/OB in youth and young adults with type 1 diabetes.1,6 OW/OB increases the risk of long-term complications from type 1 diabetes, including cardiovascular and kidney disease—with the risk of chronic kidney disease in people with type 1 diabetes even exceeding the risk for people with type 2 diabetes.7,8 Obesity is also associated with social disadvantage as measured by social determinants of health, highlighting the need for insurance coverage of medical and pharmacological interventions.9,10
A retrospective study of adults with type 1 diabetes and OW/OB initiated on semaglutide reported a reduction in BMI of 2.6 ± 0.85 kg/m2 and a reduction in HbA1c of 0.66 ± 0.15% over 1 year. 3 Change in BMI and HbA1c in our cohort were about 50% smaller, likely due to our inclusion of individuals using other GLP-1-RAs (exenatide, liraglutide, dulaglutide) which are associated with less weight loss and may be less effective in reducing HbA1c compared with semaglutide or could be due to age-group related differences in medication adherence. A metanalysis of 12 randomized controlled trials that studied GLP-1RAs as an adjunct to insulin therapy in adults with type 1 diabetes observed a reduction in HbA1c, weight, SBP, DBP, and prandial and basal insulin doses. 10 Our results support the glycemic and insulin dose findings in adults, and augment these data by including CGM data and pediatric data. However, we saw no decrease in SBP or DBP, potentially because of a shorter GLP-1-RA treatment period, or again due to age-group differences. For example, studies of youth with type 2 diabetes receiving GLP-1-RAs have not shown improvements in BMI, lipids, or blood pressure as in adult studies, despite improving glycemia.11–14 Seven individuals, (13% of the potential cohort), were prescribed but not able to receive the GLP-1-RA or GLP-1/GIP-RA because of barriers with insurance coverage. Improving the prescribing process and coverage is an area of opportunity for improvement.
The strengths of this study include novel and real-world GLP-1-RA and GLP-1/GIP use in adolescents and young adults with type 1 diabetes with follow-up up to a year. Weaknesses include the retrospective, single center design, missing data in some individuals, frequent concomitant use of metformin, and lack of a matched control group. Other important measures of cardiovascular health, such as lipid profiling, were not available with enough consistency for inclusion. We used absolute BMI, weight, SBP, and DBP to calculate changes over time rather than percentiles due to the inclusion of young adults, but this was unlikely to impact the results because we compared individual changes over a relatively short time interval.
Despite challenges associated with real-world GLP-1-RA or GLP-1/GIP-RA use (access, side effects, and adherence), BMI and insulin dose both decreased and glycemia improved, suggesting these medications may be important adjunctive agents for managing weight, insulin resistance, and glycemia in young individuals with type 1 diabetes and OW/OB. In turn, these effects may lower risk of cardiovascular and kidney complications. Future research should include longer-term randomized, placebo-controlled trials to assess safety, weight loss, glycemic control, and reduction in long-term cardiovascular and renal complications in youth and young adults with type 1 diabetes across a range of BMI.
Footnotes
Acknowledgments
Personal thanks: The authors thank Bing Wang (University of Colorado Anschutz Medical Campus) for database support.
Authors’ Contributions
K.J.N. and G.T.A. conceptualized the study, researched data, and reviewed and edited the article; G.P.F. conceptualized the study and reviewed and edited the article; C.M.L. wrote the initial draft of the article; C.M.L. and C.Z. researched data and reviewed and edited the article; T.D. assisted with study design, performed statistical analyses, and reviewed and edited the article. K.J.N. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Data and Resource Availability
The datasets generated and analyzed during the current study are available from the corresponding author upon reasonable request.
Author Disclosure Statement
G.T.A. has served on an advisory board for Mannkind; G.P.F. conducts research with Medtronic, Dexcom, Abbott, Tandem, Insulet, Beta Bionics, Sequel, and Lilly and has been a speaker/consultant/ad board member for Medtronic, Dexcom, Abbott, Tandem, Insulet, Beta Bionics, Sequel, and Lilly. K.J.N., C.M.L., T.D., and C.Z. have no conflicts of interest.
Funding Information
Supported by the University of Colorado Diabetes Research Center Clinical Resources Core, National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases (grant P30DK116073).
