Abstract

Asian Americans comprise 8.2% of the U.S. population and are the fastest growing ethnic population in the United States, projected to double to 34.4 million by 2060. In this perspective, we would like to highlight challenges that exist for evaluating, risk stratification, diagnosing, and treating obesity in South Asians in the United States.
The limitations of body mass index (BMI) in managing and categorizing obesity are particularly evident in the Asian population. Asians have a higher percentage of body fat than Caucasians of the same age and sex, resulting in a greater disease burden. Recognizing this, the WHO, in 2004, recommended using BMI thresholds of ≥23 kg/m2 and ≥27.5 kg/m2 as public health action points, but did not formally revise BMI cut offs used for obesity classification. 1 Several professional societies such as the American Diabetes Association uses a BMI of ≥23 kg/m2 as the threshold for diabetes screening in Asian Americans, 2 and the American Society of Metabolic and Bariatric Surgery and International Federation for the Surgery of Obesity and Metabolic Disorders (IFSO) define obesity at a BMI of ≥25 kg/m2 and consider for metabolic and bariatric surgery at a BMI of ≥27 kg/m2 as. 3 However, the FDA continues to follow the formal BMI cut offs of 25 kg/m2 for overweight and 30 kg/m2 for obesity which also guides research and development of obesity medications. 4 A newly proposed guidance document currently open to comments maintains the cut offs while acknowledging that supplementary anthropometric measurements, such as waist circumference, may be appropriate in certain cases. 5
The misalignment of BMI thresholds can lead to misdiagnosis and misclassification of Asians potentially preventing them as a higher risk population from receiving appropriate obesity care. In addition, it creates confusion among our primary care providers which are often the only touch point for many patients delaying counseling, referrals and interventions.
Despite multiple past federal initiatives to establish new guidelines which focused on disaggregation of Asians in research studies, inclusion and categorization of Asians in the landmark clinical trials remains poor (FIG. 1). 6 Current existing data in the in the obesity and other disease states either categorizes race at the aggregated level of “Asians” or focuses on only one Asian American subgroup. 7
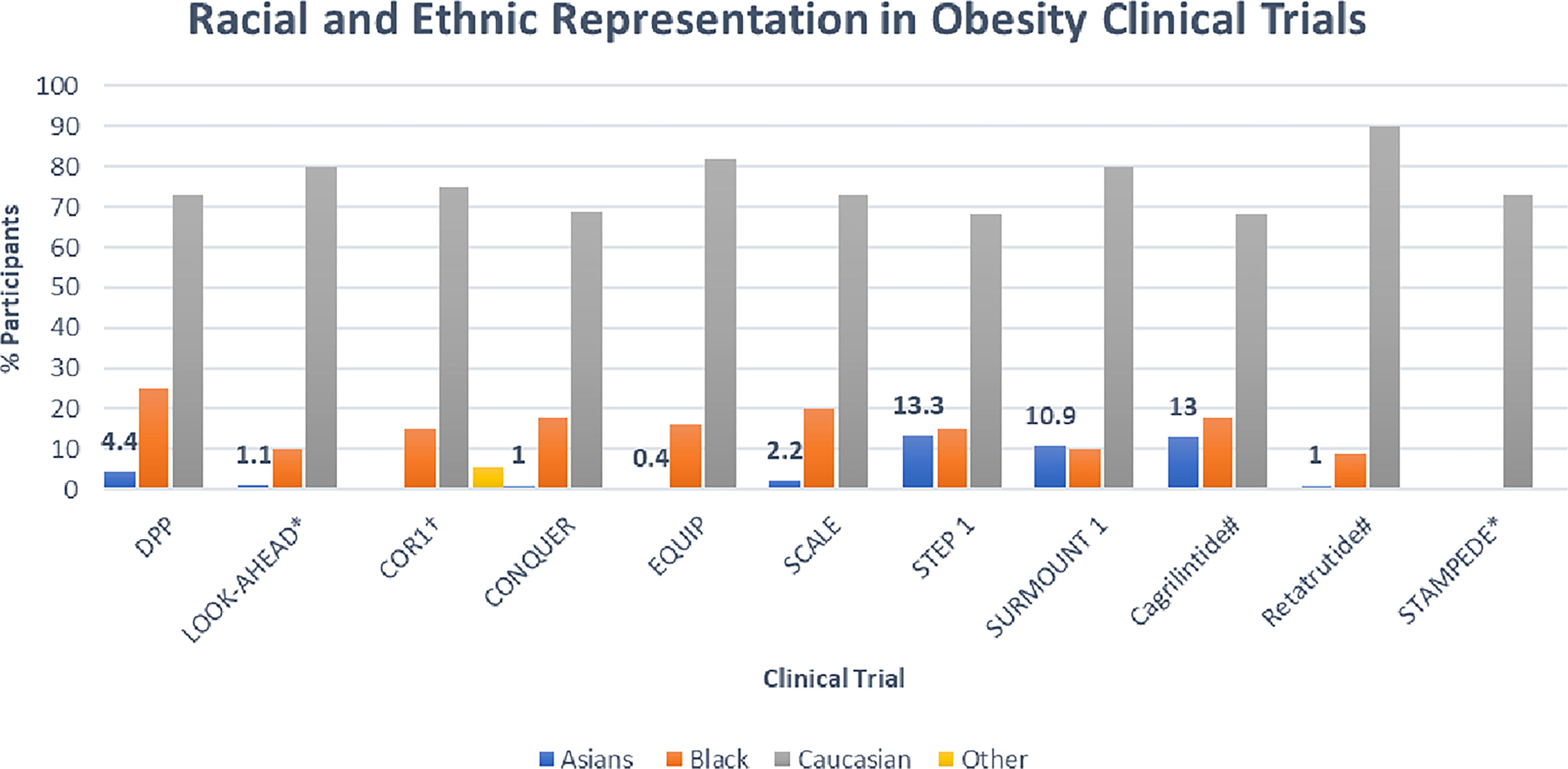
Racial and Ethnic Representation in Select Obesity Clinical Trials. *Did not report Asians as a separate subgroup †Grouped Asians and Pacific Islanders together #Unapproved obesity medications, Phase 2 data. DPP, Diabetes Prevention Program; LOOKAHEAD, Look Action for Health in Diabetes; COR1, Contrave Obesity Research-1; CONQUER, Effects of low-dose, controlled-release, phentermine plus topiramate combination on weight and associated comorbidities in overweight and obese adults; EQUIP, Controlled-Release Phentermine/Topiramate in Severely Obese Adults: A Randomized Controlled Trial; SCALE-Satiety and Clinical Adiposity-Liraglutide Evidence; STEP1, Semaglutide Treatment Effect in People with Obesity-1; SURMOUNT1-Tirzepatide Once Weekly for the Treatment of Obesity; STAMPEDE-Surgical Therapy and Medications Potentially Eradicate Diabetes Efficiently.
Representation of South Asians in obesity lifestyle and pharmacotherapy trials is woefully poor. In the Look- AHEAD trial, had about only 1.2% of the randomized population was categorized as Asian/Pacific islander with no further disaggregation. 8 A meta-analysis of lifestyle intervention trials in the United States showed Asians being included in only 30.8% of the 94 studies comprising just 5% of the total. Despite more promising representation in recent clinical trials of third generation anti-obesity medications (AOM) and direct recruitment for these newer agents in Asia, challenges persist. Inconsistencies in BMI cut offs for enrolment further complicate the applicability of these studies to South Asians in the US as some trials use FDA guidelines while others use alternative BMI thresholds. 9
The STEP-6 trial which enrolled patients from Japan (90%) and Korea (10%) had lower mean body weight (87.5 kg) and BMI (31.9 kg/m2) compared to other STEP trials (99.8–107.2 kg and 35.7–38.5 kg/m2). Its enrollment criteria also differed, requiring a BMI of ≥27.0 kg/m2 with ≥2 weight-related comorbidities or a BMI of ≥35.0 kg/m2 with ≥1 weight-related comorbidity. The trial reported slightly lower weight reductions (13.2%) compared with 14.9%–17.4% in other STEP trials, excluding STEP 2 which focused on patients with diabetes. 10 Notably, STEP 6 demonstrated significant reduction in visceral fat (≥20%) suggesting that GLP-1 receptor agonists would be particularly beneficial in the South Asian population. However, these findings underscore the need for caution in assuming similar efficacy across different ethnic groups.
In metabolic surgery, the landmark STAMPEDE trial reported that 73% of its patients were Caucasian but other ethnicities were not disaggregated. Only one other clinical trial in metabolic and bariatric surgery reported has recognized East Asian ethnicity in its data. It remains to be seen if the recently released ASMB and IFSO clinical guidelines will influence clinical practice and improve diversity of participants in future clinical trials.
In clinical practice, providing nutrition recommendations can be difficult for South Asians due to diversity of different cuisines as well as degrees of acculturation. A study comparing Gujaratis, who migrated to the United Kingdom with their counterparts in rural India, found that immigrants had increased total energy intake, with a greater percentage of calories (kcal) from fat. 11 Similarly, data from the National Health and Nutrition Examination Surveys (NHANES 2011–2018) showed ultra-processed foods accounted for approximately 39% of total daily energy among Asian Americans, including South Asians. Further analysis revealed that acculturation played a role in the increased consumptions of ultra-processed food (48.8% vs. 34.3% of total kcal, P < 0.05). 12 Studies comparing diet patterns and composition of South Asian immigrants with Westerners have produced mixed results, influenced by factors such as first versus second generation immigrant status, length of residence, and the ethnic composition of study populations. 13 Community-based, culturally adapted interventions may help these challenges. For example, the Diabetes, Research, Education, and Action for Minorities (DREAM) initiative in Atlanta provides an educational support for South Asians with type 2 diabetes and hypertension, while the South Asians Active Together in Chicago pairs sedentary South Asian mothers with their daughters in a group exercise program.14,15 Faith-based interventions also hold promise, for example, a New York program focused on blood pressure management demonstrated significant improvements within 6 months. 16 At an individual level, culturally tailored dietary resources, encouraging physical activity and, when possible, matching patients with ethnically concordant healthcare providers may improve health outcomes.
Current exercise recommendations (≥150 min/week of moderate exercise) might not confer the same cardiovascular benefit for South Asians. 17 A study of 75 South Asian and 83 European men without a history of coronary heart disease, cerebrovascular disease, peripheral vascular disease, or diabetes found that South Asian men required 266 min of exercise per week to achieve the same cardiovascular benefit as European men completing 150 min/week. 18 Similarly, there is limited data on the efficacy of AOMs specifically for Asian populations, underscoring the need for more targeted research.
Continued underrepresentation of Asians in behavioral, pharmacological, and surgical obesity interventions, along with lack of data disaggregation, and poor utilization of Asian specific BMI cutoffs, not only hinders appropriate obesity management but limits the development of ethnicity-specific obesity guidelines and equitable appropriate patient care. More inclusive clinical trials are essential to more accurate assess obesity therapies enabling clinicians to make more evidence-based decisions.
Footnotes
Authors’ Contributions
V.M. conceptualized the paper. V.M. and W.B. co-wrote the original draft as well as reviewed and edited. Both authors reviewed and approved the final version.
Author Disclosure Statement
The authors declare no competing financial interest.
Funding Information
The authors did not receive any support for the work submitted in the form of grants and/or equipment and drugs.
