Abstract
The aim of this systematic review was to assess whether current best evidence supports the use of topical anti-inflammatory medications for diabetic macular edema (DME). A systematic review, limited to randomized controlled studies published from 2015 onward, was carried out. MEDLINE, EMBASE, PubMed, Scopus, Web of Science Core Collection, and Cochrane Central Register of Controlled Trials (CENTRAL) were searched on October 11, 2024. Subjects with DME treated with topical corticosteroids or nonsteroidal anti-inflammatory drugs (NSAIDs) were included. The effect on central macular thickness was the primary outcome of interest. Seven out of the eight studies included partly/fully support the use of topical anti-inflammatory therapy to reduce macular thickness in DME. Studies were very heterogenous and no meta-analysis was possible. The overall quality of the evidence was deemed to be moderate. Topical corticosteroids and NSAIDs may be a viable therapeutic strategy for DME. However, further studies are required to assess which agents and regimens would be most beneficial, and to allow better patient selection for this approach. Drugs with improved ocular penetration and efficacy may improve the success of this approach.
Introduction
Diabetes mellitus has approximately a 10% lifetime risk of diabetic macular edema (DME), giving a current worldwide prevalence of around 30 million.1,2 Thus, its treatment creates a large burden on health systems and patients. Intravitreal antivascular endothelial growth factor (anti-VEGF) injections are the first-line treatment of DME, but this approach has significant drawbacks, such as the risk of endophthalmitis, the need for repeated injections, and an insufficient therapeutic response; the latter may occur in up to around two-thirds of patients.3–6
Inflammation contributes to the pathogenesis of DME. 7 Therefore, topical anti-inflammatory agents (in this review, this term will be used to encompass steroids and nonsteroidal drugs) may offer a possible treatment modality via their inhibitory effect on intraocular levels of inflammatory mediators. 8
However, delivery of topically applied drugs to the posterior retina and choroid is highly inefficient due to the several barriers between the ocular surface and the target tissue (Fig. 1). 9 Drug passage is affected by complex anatomical and pharmacokinetic factors. The anatomical structures both anterior and posterior to the retina also create multiple clearance pathways. The tear film constitutes the main barrier, and lacrimal drainage removes at least 95% of a topically applied agent. However, there can be a significant systemic absorption of the drug component that reaches the nasal passages, and an unpredictable fraction of this circulates back to both eyes to reach the target tissue hematogenously, with the blood–retina barrier coming into play at this point.
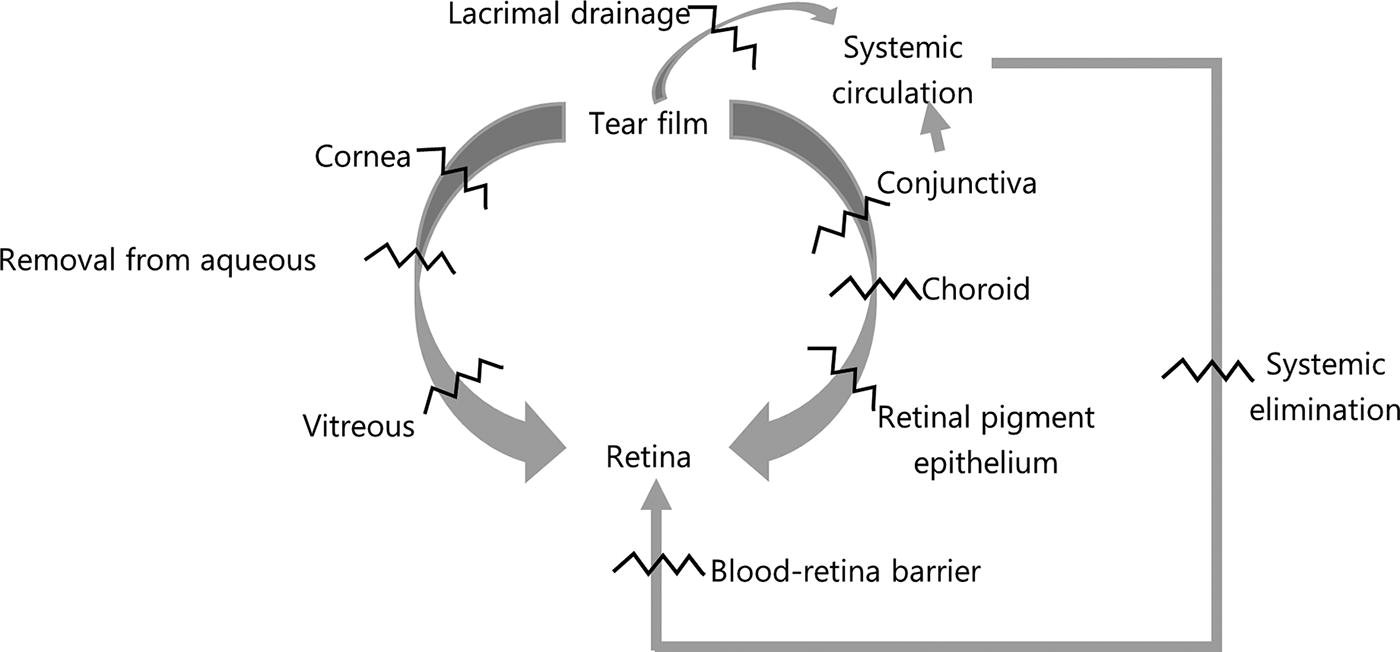
Barriers (zigzag lines) to retinal drug delivery via topical administration. 9
Apart from the transcorneal route to the vitreous, topical hydrophilic (and some lipophilic) drugs also reach the retina and choroid via passage through the conjunctiva and sclera, which have the advantage of a much larger surface area than the cornea. 9 However, the conjunctiva and sclera only allow passage of molecules much smaller (5–10 kilodaltons) than is possible through the cornea (up to 500 kilodaltons). In addition, the retinal pigment epithelium constitutes a major barrier for retinal passage of drugs absorbed trans-scleraly, maintaining the importance of the transcorneal route for retinal delivery of topically applied agents. Lens, zonular, vitreous status, and dimensions and biomechanical features of the various anatomical structures may also affect the passage of drugs.
The inefficiency of topical drug penetration can be compensated by increasing the concentration of the agent and/or optimizing its chemical features. 9 Increasing the drug concentration is limited by the consequent increased risk of potential adverse effects. The effect of the hydrophilic nature of drugs on their passage to the retina is affected by the combination of hydrophilic and hydrophobic barriers along their path, aqueous outflow, active transport, and drug metabolism. For example, lipophilic agents better penetrate the cornea and conjunctiva, and hydrophilic agents pass through the sclera more easily. The negative charge of the cornea and vitreous allows easier diffusion of negatively charged agents.
Hence, the movement of drug molecules from the tear film to the posterior segment is determined by numerous complex and inter-related factors, which make any predictions about drug concentration in the target tissue very uncertain. Studies need to rely on radiolabeling and direct measurement of drug concentration or biomarkers to ascertain drug delivery and effectiveness. 9
There has been significant interest in optimizing the chemical characteristics and form of drugs and vehicles. For example, cyclodextrin nanoparticles, with a hydrophilic surface and a lipophilic core, can carry dexamethasone and increase its ocular penetration. In addition, apart from liquid formulations, topical drugs can be delivered via gels, contact lenses, and forniceal/punctal explants to allow sustained release and reduce the lacrimal elimination of the drug to enhance its ocular penetration.
Topical nonsteroidal anti-inflammatory drugs (NSAIDs) have been shown to reach therapeutic levels in the vitreous.8,10 Topical NSAIDs seem to reduce the risk of pseudophakic cystoid macular edema in diabetics, implying that the drug is reaching the target tissue, and increased aqueous levels of inflammatory cytokines have been measured in DME.11,12 This supports the possible efficacy of NSAIDs in the management of DME.
The potential of currently available topical corticosteroids is questionable from a pharmacokinetic perspective, as steroid delivery to the retina via topical application is very limited and would be expected to hinder any potential therapeutic effect.13,14 However, drug formulations have a significant impact on ocular penetration, and agent selection may be optimized to achieve a therapeutic effect.
DME with a predominant inflammatory component may be more amenable to topical anti-inflammatory therapy. Optical coherence tomography (OCT) biomarkers of inflammation (e.g., large cysts, hyper-reflective foci, and chronic subretinal fluid), might allow better selection of patients that are more likely to respond to an anti-inflammatory treatment approach. 6
A 2015 Cochrane review related to this topic did not yield any studies eligible for analysis. 15 A 2022 review by Thagaard et al. identified two randomized controlled trials (RCTs) pertaining to topical treatment of DME, with the most recent one published in 2018. 16
Our aim was to undertake a systematic review to answer the following research question: Are topical corticosteroids and/or NSAIDs effective for the treatment of DME? The experimental hypothesis was: Topical corticosteroids and/or NSAIDs may be effective in reducing the central macular thickness (CMT) in DME. If effective, topical corticosteroids and/or NSAIDs might offer a useful therapeutic option if conventional strategies are unwarranted, insufficient, or not tolerated by the patient. The results of this review may promote future research in this area.
Methods
A systematic review of the literature was carried out, mostly following the guidelines by The Cochrane Collaboration. 17 Aspects of this review that do not adhere to the Cochrane guidelines include the use of a different risk of bias tool and the omission of a strict “summary of findings” table.
The population of interest was human subjects with DME, excluding those having intraocular surgery or active intra-/peri-ocular inflammation in the previous three months, and ongoing peri-ocular or systemic corticosteroid or NSAID use.
The treatment under consideration was topical corticosteroids and/or NSAIDs, in isolation or in conjunction with any other treatment or intervention. Any comparator was acceptable.
The outcome of interest was the change in macular thickness without definition restrictions, as measures of macular thickness vary but are all generally acceptable. These could include mean central retinal/macular thickness, central subfield thickness, maximum macular thickness, or macular/retinal volume. Visual acuity (VA) was not used as an endpoint as it is prone to negative confounding; for example, cataract, posterior capsular opacification, macular ischemia/scarring, amblyopia, and uncorrected refractive error. This is especially relevant given the association between steroid therapy and cataract risk, and the heterogeneity of the included population.
The following databases were searched: MEDLINE, EMBASE, PubMed, Scopus, Web of Science Core Collection, and Cochrane Central Register of Controlled Trials (CENTRAL). The final searches were run on October 11, 2024. The main model was the Cochrane review by Sahoo et al. 15 In our review, literature indexed prior to 1/1/2015 was excluded, as the searches by Sahoo et al. were finalized in January 2015, and filtering prior literature in this review was deemed unnecessary. The search strategies were customized for this review and for each database (See Supplementary Appendix A1 for search histories).44–54 Table 1 outlines the search schema that was used to improve the sensitivity of the search. Terms for outcome concepts (e.g., outcome, effect, efficacy, treatment, thickness, volume) and/or methods (e.g., randomized, RCT, masked) were to be used only if >250 search results were obtained from a database.
Search Schema
NSAID, nonsteroidal anti-inflammatory drugs; DMO, diabetic macular edema.
References within the ultimately included articles were hand-searched, and Google Scholar was used for forward citation tracking of the most relevant studies. Corresponding authors of pertinent records were contacted to obtain further information and/or check if they are aware of other studies, ongoing work, or unpublished data (see Supplementary Appendix A1).
The inclusion criteria for study selection were: RCTs involving the population, interventions, comparators, and outcome stated above. Studies were not excluded based on language as long as the article could be translated reliably into English text.
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flowchart was used to outline the process. 18 Initial deduplication was carried out using the automatic function in EndNote (Clarivate) comparing titles only. Then the titles were manually screened to exclude articles not meeting the inclusion criteria, some of which were also duplicates missed by the initial software filtering.
The risk of bias of the included studies was assessed using the Joanna Briggs Institute (JBI) critical appraisal checklist (https://jbi.global/critical-appraisal-tools). 19 This tool was used because it includes detailed guidance.
Results
The final searches were run on October 11, 2024. Figure 2 shows the PRISMA 2020 flowchart outlining the selection process. Table 2 shows the main features of the included studies and the relevant results, and Table 3 shows the risk of bias of each study based on the JBI RCT appraisal tool. 19

PRISMA 2020 flowchart.
Summary of Included Studies
Ratios in the “study method” column refer to randomization.
Refer to the individual studies for full description of inclusion and exclusion criteria.
Bd, twice daily; CMT, central macular thickness; CST, central subfield thickness; dexNP, dexamethasone γ-cyclodextrin nanoparticle; DRCR.net, Diabetic Retinopathy Clinical Research Network; g., drops; n/a, not available; (non-)ciDME, (non-)center involved DME; NPDR, nonproliferative diabetic retinopathy; od, once daily; PRP, panretinal photocoagulation; PDR, proliferative diabetic retinopathy; prn, pro re nata (as needed); q4x3, every 4 weeks for 3 consecutive months; qds, four times daily; T1/T2 DM, type 1 or type 2 diabetes mellitus; tds, three times daily.
Risk of Bias Assessment
This mostly related to masking of those treating the subjects. As the subjects themselves applied the drops, this issue was irrelevant.
N/A, not applicable; green, no bias concern; red, bias concern; yellow, uncertain.
A total of eight RCTs published from 2015 onward were included. The studies varied in terms of subject characteristics, treatment, control, methods, and definition of outcomes.20–27 The mean age of the subjects in the studies was around 60 years, apart from the study by Abu Hussein et al., which had a mean subject age of mid-40s.
In a multicenter study, the largest included, Friedman et al. assessed nepafenac 0.1% drops in noncenter-involving DME (non-ciDME). 20 In total, 169 subjects were initially filtered for drop compliance prior to randomization, and one eye per subject was considered. The nepafenac vehicle was used by the control group. Retinal volume and central subfield thickness did not differ significantly at baseline and at the end of the 12 month follow-up period, with the mean and median of both parameters remaining mostly stable throughout the study. The rate of development of ciDMO did not differ between the two groups (11% in the treatment arm vs 14% in the control group). There was a lower rate of ciDMO development in pseudophakic eyes on nepafenac, with 15% of the phakic eyes developing ciDMO versus 5% of the pseudophakic ones, but the difference was not statistically significant. At 12 months, five eyes in each group required rescue DME treatment. There was one case of corneal melting in an eye with severe tear deficiency on nepafenac.
In an open label study, Ohira et al. compared the treatment of chronic DME with dexamethasone 1.5% γ-cyclodextrin nanoparticle drops (which have enhanced ocular penetration), or with subtenon triamcinolone. 21 Participants were followed up monthly for 4 months. CMT decreased by at least 20% in 50% with topical therapy and in 60% with a triamcinolone injection; this was not statistically different. In the dexamethasone group, mean baseline CMT (μm) was 483 ± 141, and decreased by 99 ± 169 after 4 weeks on tds drops, and by 141 ± 211 after eight weeks on bd drops; the difference from baseline was not statistically significant at 12 and 16 weeks on od and no drops, respectively. From a similar baseline thickness, mean CMT decreased significantly by approximately 100 μm with triamcinolone and remained mostly stable till the end of follow-up. The intraocular pressure exceeded 30 mmHg in 25% of subjects on dexamethasone drops, compared with no cases in the triamcinolone group.
Abu Hussein et al. assessed mean CMT and central foveal thickness (CFT) after conservative panretinal photocoagulation (PRP) in patients without DME at baseline. 22 This study was included despite having patients without DME at baseline, as PRP led to an increase in CFT by the first monthly visit, and this was assumed to equate with the onset of early DME. PRP was applied by masked researchers in 4 weekly sessions with a total of 1500–2000 shots with an Argon green laser at 200–500 milliwatts with a 200–500 μm diameter and exposure time of 0.1 s. In the nepafenac 0.1% group, mean CFT (μm) was 192 ± 9 at baseline, reaching a maximum of 211 ± 11 at the end of the 6-month follow-up. In the sham control group, mean CFT at the same time points was 183 ± 10 (p = 0.09) and 228 ± 20 (p = 0.0023), with a linear upward trend, suggesting a prolonged and progressive course of macular edema after PRP.
Evliyaoğlu et al. included eyes with mild clinically significant nonischemic DME without an exact definition, and had the shortest follow-up (6 weeks) of the studies included in this review. 23 Despite the relatively short follow-up, it is reported that mean CMT was significantly reduced in eyes treated with nepafenac 0.1% (p = 0.002); however detailed data were not included in the article as the authors focused primarily on the effect of nepafenac on retinal vascular caliber. A composite measure of retinal arteriolar width was positively correlated with initial and final CMT (r = 0.46, p = 0.012; r = 0.52, p = 0.006).
Nikkhah et al. investigated whether topical ketorolac 0.5% produced an additional benefit when combined with intravitreal bevacizumab 1.25 mg injections (“NSAID + anti-VEGF”), compared with intravitreal injections alone, over 6 months. 24 Bevacizumab was initially given monthly for 3 consecutive months, then if needed, depending on set acuity and CMT criteria. Ketorolac was started after the first intravitreal injection. Mean CMT (μm) at baseline was 464 ± 136 in the NSAID + anti-VEGF group and 450 ± 124 in the anti-VEGF only group (p = 0.69), and at 26 weeks this decreased by 147 ± 124 in the combination group (p ≤ 0.001) and 51 ± 145 in the anti-VEGF only group (p = 0.25), with the difference in the reduction being 97 μm (95% confidence interval [CI] = −182 to −11, p = 0.017). The reduction in mean macular volume from baseline to 26 weeks was 1.3 ± 0.86 mm3 (p ≤ 0.001) and 0.61 ± 1.25 mm3 (p ≤ 0.99) in the combination and anti-VEGF only groups, respectively (p = 0.031). However, at all the time points, the mean CMT and macular volume were not statistically different between the two groups, despite a clear trend toward lower values in the combination group. The mean number of anti-VEGF injections given was around four in each group (p = 0.99).
Gabr et al. also investigated an NSAID + anti-VEGF (bromfenac 0.09% and ranibizumab) combination against anti-VEGF alone. 25 As opposed to Nikkhah et al. in the combination group, Gabr et al. started bromfenac after the initial 3-monthly ranibizumab intravitreal injections, following which further injections were given according to acuity and CMT. Baseline mean CMT (μm) in the combination and the anti-VEGF only groups was 401 ± 73 and 379 ± 77, respectively (p = 0.21). It decreased similarly in the two groups during the loading phase, and it was significantly reduced in both at the end of the 6-month follow-up, especially in the combination group (p ≤ 0.001 vs. p = 0.014). At 6 months, the mean CMT was 315 ± 61 and 370 ± 78 (p = 0.013) in the combination group and anti-VEGF only group, respectively. There was also a significant reduction in the mean average macular thickness (μm) only in the combination group throughout the study period, from 377 ± 53 at baseline to 326 ± 42 at 6 months (p ≤ 0.001). After the loading phase, 6% of patients on combined treatment needed further injections, whereas 23% in the ranibizumab only group did so (p = 0.022). In the latter group, most re-injections were required during the sixth month.
In a multicenter, proof-of-concept phase 2 study, Stefansson et al. investigated the efficacy of a topical dexamethasone 1.5% γ-cyclodextrin nanoparticle drops, similar to the agent used by Ohira et al.21,26 Mean baseline CMT (μm) was 472 ± 140 in the dexamethasone group, and 449 ± 118 in the control group. Mean CMT decreased and was significantly less in the dexamethasone group from week 2 to 12, at which point it was 37 μm less in the treatment group than in the control group (p = 0.012). Around 17% of patients on dexamethasone showed a minimal–mild increase in CMT, around two-thirds of the dexamethasone had CMT reduction of up to around 100 μm, and around 16% of patients had ∼100-560 μm reduction. In those with a baseline VA >65 ETDRS letter, the mean CMT decreased by 42 μm by week 12, whereas in those with a BCVA of ≤65 letters the reduction was of 77 μm. In the control group, around a third had an increase in CMT, and the rest had some improvement, but none had reductions exceeding 200 μm. In the treated subjects, around two-thirds of the mean CMT reduction was lost 4 weeks after stopping dexamethasone drops to reach no statistical difference from the control group.
The study by Tobimatsu et al. was the smallest one included, comparing 10 patients on bromfenac 0.09% with 9 patients on betamethasone 0.1%. 27 There was no improvement in CMT in both groups, however, subgroup analysis found a minimally significant and modest reduction with bromfenac in those with HbA1c <8% (n = 7), with baseline mean CMT 300 ± 31 μm, and a mean CMT reduction of 17 ± 16 μm (p = 0.043).
The included studies may be grouped as follows: A) three studies utilizing nepafenac against no active treatment20,22,23; B) two studies using dexamethasone optimized for ocular penetration21,26; C) two studies looking at an NSAID combined with intravitreal anti-VEGF24,25; D) one study comparing a topical NSAID with a topical corticosteroid. 27
Several reliability concerns were identified, as summarized in Table 3. However, the overall risk of bias was low, with most issues being minor or uncertain. Researcher masking was not applicable in five studies wherein the subjects were self-administering eyedrops, and there were no significant bias issues identified in the studies by Friedman et al. and Gabr et al.20,22,23,25–27 In addition, all studies documented an effort to ensure compliance to drop administration.
The study by Friedman et al. was the largest, most reliable, and the one with the longest follow-up, while Ohira et al. had the most concerns.20,21 The latter authors do not specify the randomization method, and masking during allocation, injection delivery, and outcome assessment were absent; in line with an open label design, sham drops and sham injections could have been used to mask the patients and researchers. It is also unclear if an intention-to-treat analysis was carried out (the corresponding author did not reply to questions via email). Despite the drop group having a smaller proportion of female subjects than the injection group, this was felt not to have a major impact on the relative efficacy of the compared therapies. Hence, the several reliability issues, in addition to the small sample size, reduce the weighting of inferences that can be made from the findings by Ohira et al.
Evliyaoğlu et al. do not describe any subject masking, raising bias concerns; even though this may not be relevant because fellow eyes served as control and the outcome is objective. 23 The authors do not provide detailed CMT data, and hence the appropriateness of the statistical comparison between treated and control eyes could not be verified. In the study by Nikkhah et al., participants lost to follow-up were not included in the final analysis. 24 Even though this raised a bias concern, it should be noted that the attrition rate was low, thus maintaining a good reliability.
Using the GRADE (Grading of Recommendations Assessment, Development and Evaluation) framework, the overall quality of the evidence can be regarded as moderate because subjective downgrading due to possible imprecision and publication bias lowers the initial possible high quality associated with a randomized controlled design. 28 With regard to risk of bias, despite a variable level of possible bias across studies, overall this seems to be low, as shown in Table 3. No downgrading was applied for inconsistency and indirectness, as only one study failed to show a positive treatment response and all studies directly assessed macular thickness with OCT. The wide confidence intervals found in most studies led to a two-point downgrading for imprecision, and because most included studies were small and some were industry-sponsored, a further one-point downgrade was applied for possible publication bias. A one-point upgrade was applied for the “large effects” domain, as the studies utilizing preparations for enhanced retinal drug delivery showed a large treatment effect in some patients.21,26 A further one-point upgrade was applied based on dose response, as this is plausible given the increased in macular thickness seen with reduction or cessation of therapy. Regarding residual confounding, randomization would have limited this and was thus not applicable.
An overall meta-analysis was not possible due to the heterogeneity of the studies. A subgroup meta-analysis of the three studies comparing nepafenac with no active treatment was also not possible due to differing outcome measures and missing data.20,22,23
Discussion
While previous case reports and series and nonrandomized studies already suggested the potential use of topical anti-inflammatory therapy in DME, this review further supports this clinical application.30–34
In this review, seven of eight studies support the experimental hypothesis set in the introduction; only the study by Friedman et al. did not find any therapeutic benefit. 20 Even though it was the most reliable study and the one that had the longest follow-up, it should be noted that it was the only study in which ciDMO (i.e., DME involving the fovea) was an exclusion criterion. It is possible that the potential benefit of topical anti-inflammatory treatment is mostly apparent with central macular swelling. In addition, it can be argued that non-ciDMO, especially if asymptomatic, does not require treatment. Therefore, Friedman et al. provide strong evidence that topical NSAID therapy would not be expected to offer a significant benefit in non-ciDMO. However, this conclusion should not discourage topical NSAID use when CMT is increased given the results with NSAIDs in the other studies.22–25,27
In the study by Ohira et al. some subjects showed major drying of the macula with topical dexNP within the first month, with recurrence of fluid on drop frequency tail down. 21 This implies a possible dose–response relationship and that a maintenance dose above a certain threshold is needed to sustain the initial benefit. This was replicated by Stefansson et al., in which case the treatment effect of topical dexamethasone was lost within 1 month off drops. This provides further evidence to support the therapeutic effect of topical dexamethasone in a formulation, which enhances ocular penetration.
Abu Hussein et al. investigated macular thickening following PRP. 22 This study provides an idea of how recent-onset cases may behave. It also suggests that PRP may trigger DME and prophylactic topical therapy may be beneficial. Interestingly, VA decreased before there was a detectable CMT increase after laser, meaning that a drop in acuity may predict the onset of DME. This study may also show the value of early treatment because the difference between the two groups was highly significant at the end of follow-up. The therapeutic effect of topical nepafenac seems to be delayed by around 2 months according to data in this study.
In the studies looking at combined NSAID and anti-VEGF treatment, both found better CMT reduction in the combination group, but Gabr et al. also found reduced injection requirement with combined treatment, mostly at the end of the 6-month follow-up.24,25 This may mean that combined therapy is synergistic and may allow the initial benefit of the loading phase to be sustained for longer. In addition, from the data in these studies, adding an NSAID to conventional anti-VEGF therapy may be expected to lead to approximately a further 50–100 μm reduction in CMT.
While the studies by Nikkhah et al. and Gabr et al. suggest an additive effect of topical NSAIDs in combination with anti-VEGF injections, a previous retrospective report by Mohan et al. did not find a benefit of combined treatment.24,25,35 Nikkhah et al. did not obtain a statistically significant difference in the mean macular thickness/volume, but the difference from baseline was statistically better when bevacizumab was combined with ketorolac. This implies that combined therapy may allow a quicker rate of reduction in macular thickness, rather than providing more reduction. However, Gabr et al. obtained a significantly larger reduction with combined ranibizumab and bromfenac, and the combination group also required less injections on a prn basis compared to ranibizumab only. The better results obtained by Gabr et al. may be due to the particular agent used (bromfenac), but further studies are needed to support this particular hypothesis.
Hence, a faster and/or better recovery may be obtained with a combined approach. Further studies are required to provide further evidence on the value of combining intravitreal anti-VEGF and topical anti-inflammatory agents. In addition, it would be useful to have head-to-head comparisons between anti-VEGF injections alone and topical anti-inflammatory agents alone, to assess whether the outcomes and safety are comparable, especially in the long-term. 3 No included studies directly compare the efficacy of monotherapy with topical anti-inflammatory drops versus intravitreal anti-VEGF injections.
It is difficult to quantify the expected reduction in CMT, as this is affected by several baseline factors (e.g., severity, duration, glycemic control, comorbidities) and changes in the latter during the course of the treatment, and on the treatment delivery method (e.g., compliance, timing, rescue interventions). However, the study by Nikkhah et al. suggests that an additional thinning of around 100 μm is expected after 6 months when ongoing topical ketorolac is combined with intravitreal bevacizumab. 24 In addition, in this study, the effect of intravitreal anti-VEGF is not sustained and the advantage of additional NSAID drops was apparent only at 6 months, not at 14 weeks. This may imply a change in the underlying pathogenic mediators of DME over time, creating an inflammatory milieu for which topical anti-inflammatory drops may be effective mostly in chronic cases. Adding topical anti-inflammatory therapy after 3–4 months of anti-VEGF injections in persistent DME is a therapeutic strategy that also merits further study.
Similarly, the study by Gabr et al. looked at bromfenac combined with ranibizumab. 25 After the three loading injections, further injections in both groups were given on a prn basis. At 6 months, bromfenac provided an additional 65 μm reduction in CMT from a statistically similar baseline CMT in the two groups. Interestingly, the combination group required significantly less additional injections after the loading phase; this was not corroborated by Nikkhah et al. 24 Thus, whether topical anti-inflammatory therapy has the potential to reduce the number of anti-VEGF injections requires further investigation.
Tobimatsu et al. found a therapeutic effect with bromfenac in patients with good glycemic control only. 27 This may imply that a treatment response is more likely if glycemic control is also achieved; however, the number of patients in the subgroup analysis was very small. Conversely, Friedman et al. did not find a therapeutic effect with nepafenac even after adjusting for HbA1c. 20 Hence, from the data in the included studies, it is not possible to identify potential patient factors, which could be associated with a better response to topical anti-inflammatory medications. Identifying such predictive factors would be challenging due to the wide range of possible variables, such as the duration of diabetes, diabetic retinopathy, and DME. In addition, the studies included in this review failed to select patients on the basis of OCT inflammatory biomarkers. 6 Future studies should optimize the inclusion criteria to allow for better patient selection for this treatment modality based on OCT features and systemic factors. However, as topical anti-inflammatory therapy was found to be effective in a variety of DME subtypes and clinical situations, there may be a significant versatility and very strict inclusion criteria may limit the applicability of results in real-world settings.
The specific agent used may also determine the observed clinical activity, and agents may not be interchangeable. 8 For example, Pastore et al. found that bromfenac and indomethacin reached the highest vitreous levels, with a corresponding superior reduction in vitreous prostaglandin concentration. In addition, lens status may significantly affect drug penetration into the vitreous, with possibly higher levels in pseudophakic eyes.
Drugs are also systemically absorbed and can reach the posterior segment hematogenously, further confounding the effect of the pharmacokinetic profile of the different agents. 13 In addition, interindividual pharmacodynamic variations create further confounding factors that can affect the potential therapeutic activity. 14 Given the diverse therapeutic regimens employed in the included studies, it is difficult to comment on which agent and regimen is best. The required duration of treatment is particularly unclear.
Novel vehicles, drug design, and drug classes may provide better future options for topical treatment, which could replace or aid intravitreal approaches.13,36 A large-scale RCT with a dexamethasone nanoparticle suspension is ongoing (ClinicalTrials.gov identifier: NCT05066997) and has shown promising preliminary data.37,38 Tyrosine kinase inhibitors (which can be administered in oral and topical formulations) are also promising. 39
It is also uncertain whether patient selection for anti-inflammatory drugs is possible. 40 So far, OCT biomarkers are unreliable in predicting treatment response and selection, and other biomarkers may be superior. For example, Brito et al. found a correlation between C-reactive protein, a serum marker of inflammation, and poor response to anti-VEGF. 41 It may be postulated that patients with increased serum levels of inflammatory markers may respond better to DME therapy targeted at inflammatory cascade inhibition. Further studies are required to provide evidence to optimize therapeutic strategies based on systemic markers.
Only one case of corneal melting was reported with NSAIDs in the included studies, however risk factors for keratopathy were common exclusion criteria.20,22,24 The case of corneal melting had pre-existing severe dry eye disease. 20 Apart from possible IOP elevation, no safety concerns relating to topical steroids were identified. 21 Further studies with longer follow-up would allow the likelihood of this and other complications to be better understood, and may provide reassurance about the safety of these topical agents.
This paper builds on two previous reviews related to this topic.15,16 In 2015, Sahoo et al. published a Cochrane review focusing only on the effect of topical NSAIDs in DME, and they failed to identify eligible RCTs. The search strategy used in this review is similar, but Scopus, Web of Science, and PubMed had not been searched by Sahoo et al., and the number of deduplicated records screened in this review is around twice that obtained by Sahoo et al.
Thagaard et al. undertook a wider ranging review on topical therapy for diabetic retinopathy in general. 16 They included nonrandomized studies and did not search Web of Science and CENTRAL. Their inclusion criteria were similar to those in this review, but the scope was not limited to topical NSAIDs and corticosteroids. This review identified an additional six relevant studies compared with Thagaard et al. It should be noted that Thagaard et al. categorize the study by Evliyaoğlu F et al. as nonrandomized. 22 However, Evliyaoğlu F et al. randomly assigned one eye of each participant for treatment, with the fellow eye acting as control, and hence it was included in this review. Thagaard et al. similarly consider the study by Friedman at el. to have a low overall risk of bias, while that by Ohira et al. to carry a significant risk of bias, using the Cochrane risk of bias tool.15,16,19 The conclusion by Thagaard et al. supported the potential therapeutic benefit of topical corticosteroids and NSAIDs for DME, but a meta-analysis was also not possible. They hypothesize that topical anti-inflammatory therapy may be particularly effective in cases of severely increased CMT based on findings by Nakano Goto et al. 31
Repeated anti-VEGF injections are expensive and create a significant logistical pressure for health care systems, and having a noninvasive and readily available option may allow earlier initiation of treatment. 42 Finding a cheaper alternative to intravitreal anti-VEGF injections would constitute a major health economic advantage. However, it would be pertinent to consider how compliant patients would be to administration of long-term topical therapy in nonstudy settings. The maximum attrition rate in the studies included in this review was 11%. 24
The main limitation of this review is the lack of multiple reviewers. For this reason, registration on PROSPERO was not possible. 43 Reviewer bias was mitigated by adhering to the pre-established protocol. Other limitations were: the inability to carry out a meta-analysis due to study heterogeneity, the significant number of contacted authors/researchers that failed to respond, and the possibility of missing relevant studies with the search strategy used, despite every effort to maintain a high sensitivity during the literature searching process.
Suggestions for future research in this area are: further high-quality comparable studies to establish with more certainty if topical therapy is effective to improve macular thickness and VA, using novel formulations and agents in order to maximize ocular surface penetration, and studying compliance issues that may arise with topical therapy for this indication. If topical anti-inflammatory therapy proves to be effective, its role within the current therapeutic armamentarium and algorithms should be addressed.
Conclusions
This is the most recent review on this subject to date. Topical corticosteroids and NSAIDs may be effective and safe for reducing CMT in DME. However, further large-scale studies are required to ascertain the effectiveness of this approach, how best to utilize this modality within the context of the available options, and which patients are most likely to benefit.
Footnotes
Authors’ Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by J.V. The first draft of the article was written by J.V. and all authors commented on previous versions of the article. All authors read and approved the final article.
Author Disclosure Statement
The authors have no relevant financial or nonfinancial interests to disclose.
Funding Information
The authors declare that no funds, grants, or other support were received during the preparation of this article.
Abbreviations Used
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
