Abstract
Dynamic mutations in the 5′ untranslated region of FMR1 are associated with infertility. Premutation alleles interfere with prenatal development and increase infertility risks. The number of CGG repeats that causes the highest decrease in ovarian reserves remains unclear. We evaluated the effect of FMR1 CGG repeat lengths on ovarian reserves and in vitro fertilization (IVF) treatment outcomes in 272 women with alleles within the normal range. FMR1 CGG repeat length was investigated via PCR and capillary electrophoresis. Alleles were classified as low-normal, normal, and high-normal. Serum levels of follicle-stimulating hormone and anti-Mullerian hormone (AMH) in the follicular phase of the menstrual cycle were measured, and antral follicles (AFC) were counted. IVF outcomes were collected from medical records. Regarding FMR1 CGG repeat length alleles, 63.2% of women presented at least one low-normal allele. Those carrying low-normal alleles had significantly lower AMH levels than women carrying normal or high-normal alleles. Low-normal/low-normal genotype was the most frequent, followed by low-normal/normal and normal/normal. A comparison of ovarian reserve markers and reproductive outcomes of the three most frequent genotypes revealed that AFC in the low-normal/normal genotype was significantly lower than the low-normal/low-normal genotype. The low number of FMR1 CGG repeats affected AMH levels and AFC but not IVF outcomes per cycle of treatment.
Introduction
Infertility affects 17.5% of the population in their reproductive age, representing one in every six couples worldwide (WHO, 2023). Genetic variants can be associated with infertility factors, including the dynamic mutation in the FMR1 gene (fragile X messenger ribonucleoprotein 1), which has been associated with premature ovarian failure (POI) (Cordts et al., 2011).
Oberlé et al. (1991) identified an unstable and expandable sequence of CGG trinucleotides in the 5′ untranslated region (UTR) of FMR1. This gene, located at Xq27.3, is expressed in different tissues, especially in the brain, testes, ovaries, and epithelia. The encoded protein FMRP (Verkerk et al., 1993) regulates the proper functioning of synapses and plays a role in the transport and translation of target mRNAs and protein production within cells (Noto et al., 2016). It is also expressed in the oocyte pool during prenatal development (Rifé et al., 2004).
The American College of Medical Genetics has identified four possible FMR1 alleles: (1) normal: 5–44 CGG repeats; (2) intermediate: 45–54 CGG repeats; (3) premutation: 55–200 CGG repeats; and (4) full mutation: >200 CGG repeats (Maddalena et al., 2001). The presence of more than 200 triplet CGG repeats results in hypermethylation of the FMR1 promoter region, consequently silencing the gene and leading to the absence of the FMRP protein (Christofolini et al., 2006). Men carrying the full mutation show fragile X syndrome (OMIM 300624), the most common cause of inherited intellectual disability and autism in men (Tassone et al., 2012).
The premutation allele interferes with the prenatal development of the oocyte pool, reducing the number of viable oocytes. Changes in mRNA generated by the premutated allele lead to follicular atresia. Furthermore, 20–28% of women carrying the premutation allele may experience POI, whereas the incidence of POI in women carrying the normal allele varies between 0.1% and 1.0%. In addition, the premutation allele is associated with a higher risk for menstrual cycle changes, such as short menstrual cycles and menstrual irregularities, as well as a higher frequency of infertility (Allen et al., 2007; Cordts et al., 2011).
The clinical conditions associated with the mutation allele (full or premutation) of FMR1 are well described in the literature. However, certain ranges in the number of CGG repeats have been identified among those alleles that are classified as normal. Based on the variation in CGG repeat length, normal alleles are recategorized into three groups: low-normal (<26), normal (26–34), and high-normal (35–44) (Banks et al., 2016; Gleicher et al., 2010; Kushnir et al., 2014). CGG repeats below 26 or above 45 may lead to subtle changes in ovarian reserve because of decreased FMRP translation (Morin et al., 2016; Noto et al., 2016). Women who have regular menstrual cycles but show decreased anti-mullerian hormone (AMH) levels (<1.1 ng/mL) or low antral follicle counting (AFC, <5) are considered as having decreased ovarian reserve (Ferraretti et al., 2011; Grisendi et al., 2019; Humaidan et al., 2016).
Pastore et al. (2019) conducted a meta-analysis to evaluate the impact of CGG repeat lengths of FMR1 in women carrying normal alleles who were undergoing in vitro fertilization (IVF) treatment. They reported that a majority of studies (8 out of the 11 articles evaluated) had been conducted in the United States and found no consensus in the literature.
The first and crucial step in IVF treatment is controlled ovarian stimulation (COS) to obtain mature oocytes (Roque et al., 2019). This step is dependent on the ovarian reserve, which is defined as the pool of ovarian follicles that are available for follicular recruitment, thus reflecting the functional potential of the ovaries, defined by the number and quality of oocytes (Maheshwari et al., 2006). Different studies using various definitions and populations have suggested an association between decreased ovarian reserve and infertility. However, the association between CGG repeat length of FMR1 and diminished ovarian reserve (separate from POI) as well as the number of repeats that confer the greatest risk remains unclear (Pastore et al., 2019).
Based on these elements, we aimed to evaluate the impact of the CGG repeat length of FMR1 on the ovarian reserve and reproductive outcomes of women carrying alleles within the normal range and undergoing IVF treatment.
Materials and Methods
Study design
We conducted a cross-sectional study of women undergoing their first IVF treatment at the Instituto Ideia Fértil–Center for Human Reproduction and Genetics of the Centro Universitário FMABC, Santo André/SP, Brazil, between April 2020 and April 2023. The project was approved by the Research Ethics Committee of the Centro Universitário FMABC (CAAE 16221519.5.0000.0082), and the participants provided written informed consent for their voluntary participation in the study, allowing data collection and analysis for research purposes. The design, analysis, interpretation of data, drafting, and revisions conform to the Declaration of Helsinki, the Committee on Publication Ethics guidelines (http://publicationethics.org/), and the Strengthening the Reporting of Observational Studies in Epidemiology Statement (von Elm et al., 2007), validated by the Enhancing the Quality and Transparency of Health Research Network (www.equator-network.org). The study was not advertised. No remuneration was offered to the patients to give consent to be enrolled in this study.
Patients
Women who took part in the study met the following criteria: in the process of undergoing IVF treatment; age ≤40 years; follicle-stimulating hormone (FSH) levels of ≤15.0 mIU/mL; thyroid-stimulating hormone between 0.5 and 4.0 IU/L; prolactin level of ≤30.0 ng/mL within the normal parameters; both ovaries without morphological abnormalities; ovulatory cycle lasting 21–35 days; body mass index (BMI) of ≥18.5 ≤ 30 kg/m2; and FMR1 alleles with <45 CGG repeats. Women who had undergone previous ovarian surgery and who had undergone radio/chemotherapy or been diagnosed with POI, polycystic ovarian syndrome, or pelvic inflammatory disease were excluded.
Infertility investigations included a comprehensive medical history, hormonal and biochemical profile, tests for sexually transmitted infections, imaging tests (transvaginal ultrasound, hysterosalpingography, and hysteroscopy), and semen analyses (ACOG, 2019; ASRM, 2015).
Data and sample collection
Data related to clinical history and IVF treatment were collected from the medical records of the participants.
On the second or third day of the menstrual cycle, and immediately before the start of COS with gonadotropins, 15 mL of peripheral blood was collected in an ethylenediaminetetraacetic acid -containing tube for genomic DNA analysis and in a tube containing a separating gel and a clot activator for FSH and AMH measurement. After collection, the tube meant for hormone measurement was centrifuged for 10 min at 3000 rpm, and the serum was stored in a −80°C freezer. The tube meant for DNA analysis was stored in a refrigerator at 8°C until extraction.
Hormone measurement
FSH and AMH measurements were performed using a commercial ELECSYS® Electrochemiluminescence kit (Cobas®, Roche, Switzerland).
Investigation of FMR1 CGG repeat lengths
DNA was extracted in an automated manner using a Prepito® DNABlood250 kit (PerkinElmer, MA, USA) with a Chemagic Prepito® (PerkinElmer, MA, USA). The quantity and quality (purity) of the extracted DNA were assessed using a NanoDrop 2000 spectrophotometer (Thermo Scientific, CA, USA). FMR1 CGG repeat length was analyzed using an AmplideX® PCR/CE FMR1 kit (Asuragen, TX, USA). Capillary electrophoresis aimed at evaluating the fragments was performed using 3500 Series Genetic Analyzer® equipment (Applied Biosystems, CA, USA), whereas fragment analysis was conducted using GeneMarker® software (SoftGenetics, LLC, PA, USA). FMR1 CGG repeat alleles were classified as low-normal (<26 CGG repeats), normal (26–34), high-normal (35–44), intermediate (45–54 repeats), and premutation (55–200 repeats) (Banks et al., 2016; Gleicher et al., 2010; Kushnir et al., 2014; Maddalena et al., 2001).
IVF treatment
Transvaginal ultrasound was used to evaluate the ovaries before the onset of COS, on the second day of the menstrual cycle, by an experienced sonographer using conventional 7 Mhz two-dimensional ultrasound (Philips, Low Countries). AFC counts were performed in each ovary, all follicles between 2 and 10 mm being considered.
COS was performed with recombinant FSH (rFSH) via fixed, daily doses of 150 or 200 IU, which were administered from the second or third day of the menstrual cycle for 8–14 days on average. Follicular growth was monitored via transvaginal ultrasound. When the largest follicle reached 14 mm, administration of the gonadotropin-releasing hormone antagonist (Orgalutran®, Schering Plow, USA) or associated oral progestins was started. When the follicles reached a diameter of ∼17–20 mm, human chorionic gonadotropin (hCG; Choriomon®, UCB Biopharma S/A, Belgium) was administered at a dose of 5000 IU, and 34–36 h after hCG administration, oocytes were retrieved.
Intracytoplasmic sperm injection was performed on metaphase II (MII) oocytes on the day of ovarian retrieval. In general, embryos were cultured until the blastocyst stage, if more than two high-cleavage embryos were available. Embryos were also transferred on day 6 if no high-quality blastocysts were available on day 5. Embryos were scored according to the Istanbul consensus (Alpha Scientists in Reproductive Medicine and ESHRE Special Interest Group of Embryology, 2011). A maximum of two embryos were transferred per transfer, guided by ultrasound. Pregnancy was confirmed by beta hCG measurement (>25 mIU/mL) on the 12th day after embryo transfer.
Statistical analysis
All analyses were carried out in Python 3.9, via Jupyter Notebook. Normal distribution was checked with the Shapiro–Wilk test. Comparison for numerical data was performed with the Mann–Whitney U test and for categorical variables with the chi-square test. Variables with more than two categories were compared pairwise, without p-value correction. Relationships between the FMR1 CGG repeat lengths with FSH, AMH, and AFC were presented in scatterplots (biplots) and linear regression curves with 95% confidence interval (95% CI).
Linear regression tests were performed for continuous numerical variables and estimation of linear regression coefficient (R2). Unadjusted models were used to evaluate the number of FMR1 CGG repeats as continuous variables or allele groups as biallelic categories. Poisson regression was performed using CGG repeats in FMR1 as the independent variable against FSH, AMH, AFC, follicles visualized, rFSH dose, oocytes retrieved, number of MII, embryos transferred, and age.
Statistical significance was set at p < 0.05. The sample size was adopted for convenience according to the number of patients treated at the service and those who agreed to participate in the study, after assessed by inclusion and exclusion criteria.
Results
Two hundred and seventy-nine women who underwent IVF treatment were screened for this study. Seven women, including four carrying an intermediate allele and three carrying a premutated allele, were excluded. The frequency of FMR1 CGG repeat length genotypes is shown in Figure 1. The clinical, hormonal, and reproductive characteristics of the 272 patients included in the study are presented in Table 1. Of the 265 cycles (excluding canceled ones), 13 showed no embryo for transfer and 31 had not yet undergone embryo transfer. Thus, of the 221 women who underwent embryo transfers, 42.5% had a biochemical pregnancy.
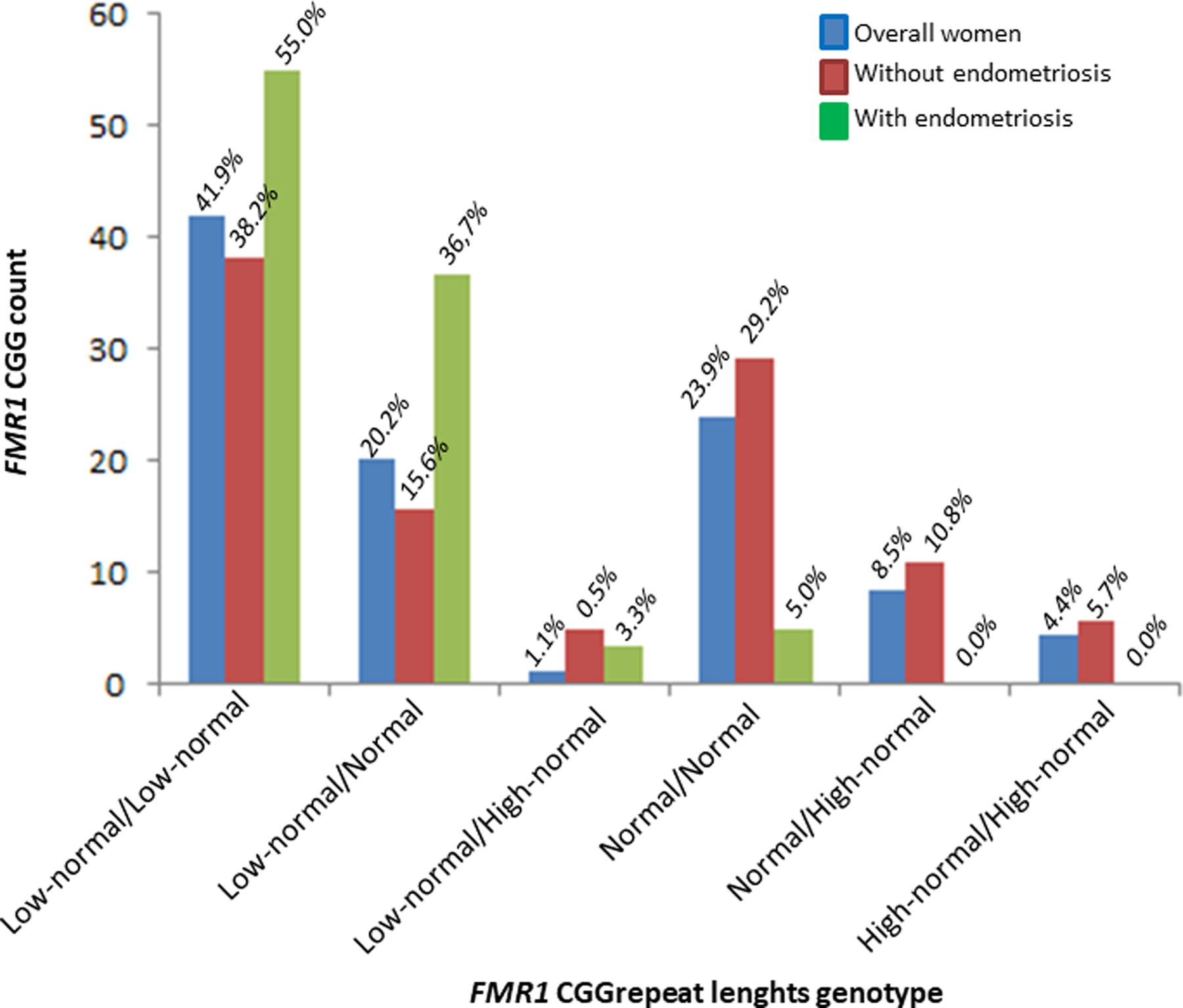
Frequency of FMR1 genotypes, based on the number of CGG repeats in the 5′ UTR region of the gene, in women undergoing IVF. IVF, in vitro fertilization; UTR, untranslated region.
Clinical, Hormonal, and Reproductive Characteristics of the Studied Women
Qualitative variables were presented by absolute and relative frequency and quantitative variables by median and interquartile range (IQR).
AFC, antral follicle counting; AMH, anti-Mullerian hormone; BMI, body mass index; COS, controlled ovarian stimulation; FSH, follicle-stimulating hormone; LH, luteinizing hormone; MII, metaphase II; rFSH, recombinant FSH.
Considering the patients who had the cycle canceled, four of them presented low-normal/low-normal genotypes, one low-normal/normal, one normal/high-normal, and one high-normal/high-normal. Moreover, two of these women presented AMH <1.1 ng/mL and AFC <5. Regarding patients with no embryo for transfer, one of them presented low-normal/low-normal genotype, three low-normal/normal genotype, four normal/normal genotype, and three normal/high-normal genotype. Furthermore, one of these women presented with decreases in both AMH and AFC.
Regarding FMR1 CGG repeat length in the overall group of women undergoing IVF, 63.2% of women carried at least one low-normal allele, and of these, 17.4% presented with AMH <1.1 ng/mL, 15.7% with AFC <5, and 5.2% with decreases in both AMH and AFC (Table 2). Women carrying at least one low-normal allele had significantly lower AMH levels than women carrying at least one normal or high-normal allele. The percentage of women with AMH <1.1 ng/mL was more than twice that of women carrying normal or high-normal alleles. Furthermore, 100% of women with decreases in both AMH and AFC carried at least one low-normal allele. Considering only women without endometriosis, we did not observe any statistical difference in the variables analyzed (Table 2).
Comparison of Ovarian Reserve Markers and Reproductive Outcomes Corresponding to FMR1 Alleles in Women Undergoing IVF
Qualitative variables were presented as median and interquartile range and qualitative variables as absolute number and percentage.
Mann–Whitney U test.
Chi-square test.
The comparison between ovarian reserve markers and reproductive outcomes corresponding to FMR1 CGG repeat length genotypes is shown in Figure 2. A comparison between ovarian reserve markers and reproductive outcomes of the three most common FMR1 genotypes (low-normal/low-normal, low-normal/normal and normal/normal) in overall women is presented in Table 3 and Figures 3 and 4. Women carrying the low-normal/low-normal genotype exhibit a time of years of infertility greater than that exhibited by women carrying the normal/normal genotype. Furthermore, AFC was significantly lower in women carrying the low-normal/normal genotype compared with women carrying the low-normal/low-normal genotype. However, considering only women without endometriosis, no statistical difference was observed in the variables analyzed (Table 4).
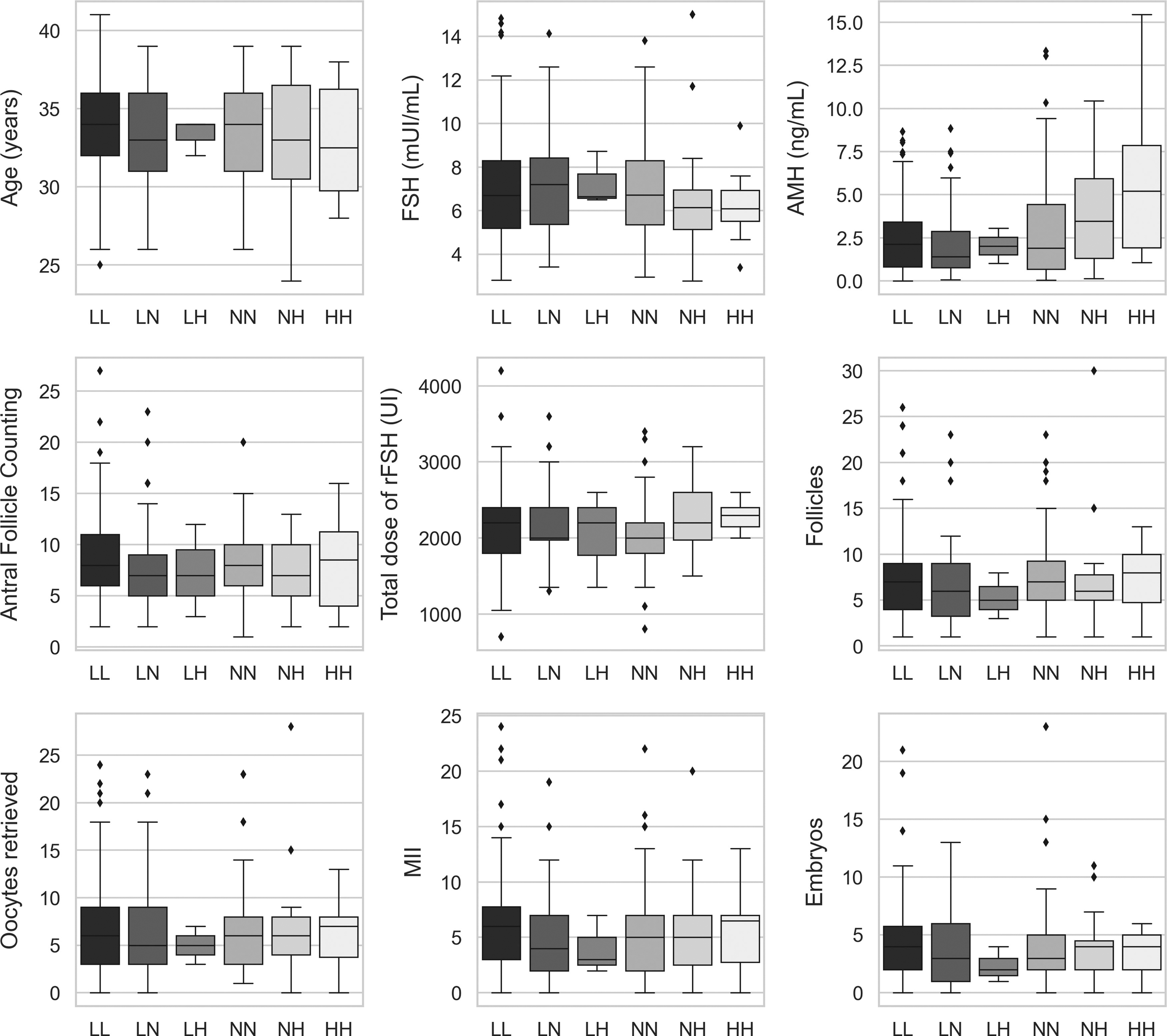
Boxplot depicting the comparison of ovarian reserve markers and reproductive outcomes corresponding to FMR1 genotypes. AFC, antral follicle count; AMH, anti-Mullerian hormone; FSH, follicle-stimulating hormone; MII, metaphase II. Genotypes considering the number of FMR1 CGG repeat length: HH, high-normal/high-normal; LA, low-normal/high-normal; LL, low-normal/low-normal; LN, low-normal/normal; NA, normal/high-normal; NN, normal/normal.
Comparison of Clinical Characteristics, Markers of Ovarian Reserve and Reproductive Outcomes of Overall Women Undergoing IVF, According to the Most Frequent Genotypes, Based on the Number of FMR1 CGG Repeat Lengths
Bold values represent statistical p-value.
Qualitative variables were presented by absolute and relative frequency and quantitative variables by median and IQR.
Mann–Whitney U test.
Chi-square test.
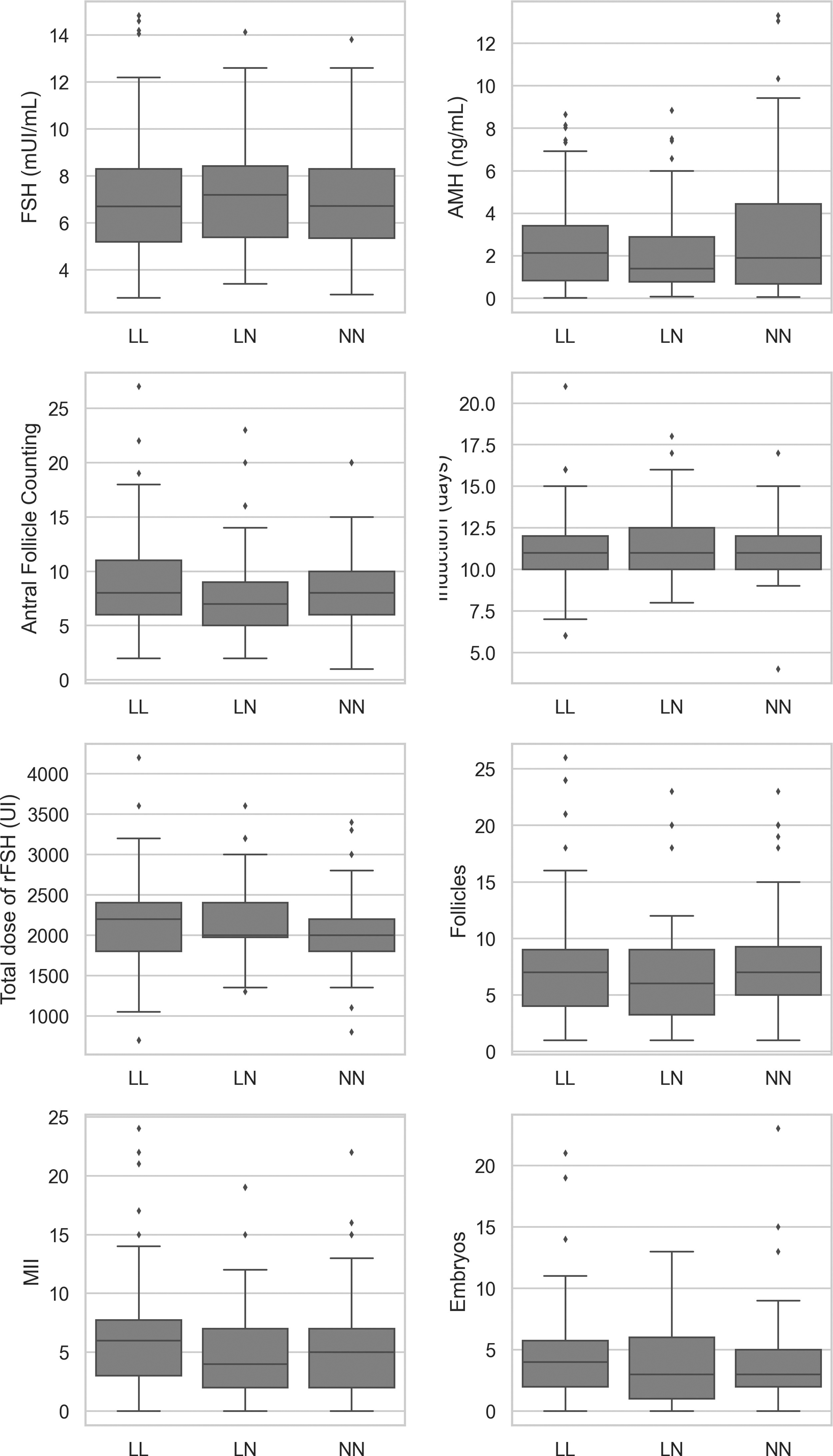
Boxplot depicting the comparison of ovarian reserve markers and reproductive results corresponding to the most frequent genotypes of FMR1. COS, controlled ovarian stimulation.
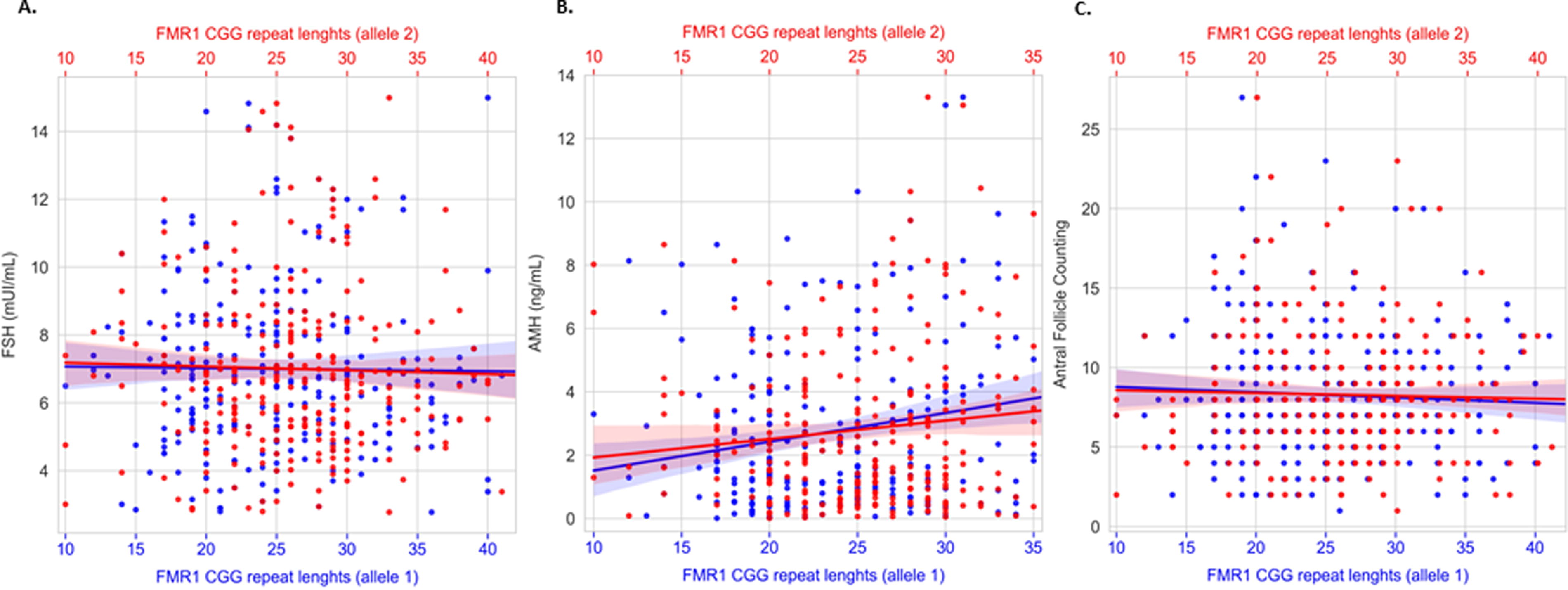
Scatter plot depicting FMR1 CGG repeat lengths and FSH
Comparison of Clinical Characteristics, Markers of Ovarian Reserve and Reproductive Outcomes of Women Without Endometriosis Undergoing IVF, According to the Most Frequent Genotypes, Based on the Number of FMR1 CGG Repeat Lengths
Qualitative variables were presented by absolute and relative frequency and quantitative variables by median and IQR.
Mann–Whitney U test.
Chi-square test.
Of the women included in this study, 22% presented endometriosis: 33.3% had minimal/mild disease (grade I or II), and 66.7% had moderate/severe endometriosis (grade III or IV). Of these women, 43.3% showed a decrease in some marker of the ovarian reserve: 35.0% presented with AMH <1.1 ng/mL, 23.3% with AFC <5, and 15.0% with decreases in both AMH and AFC. Furthermore, 3.33% had their cycle canceled, 3.33% did not have embryos for transfer, 8.33% had not yet undergone embryo transfer, and the biochemical pregnancy rate was 43.1% per cycle of treatment. With respect to FMR1 CGG repeat lengths, 95.0% of these women carried at least one low-normal allele and 55% carried a low-normal/low-normal genotype. A comparison between clinical characteristics, reproductive outcomes, FMR1 alleles, as well as genotypes of women with and without endometriosis is shown in Table 5. Women with endometriosis had significantly higher FSH and luteinizing hormone and lower AMH levels, as well as lower AFC than women without endometriosis; however, there were no statistical differences between days of COS, total dose of rFSH, the number of MII, oocytes and embryos, and pregnancy rates per cycle of treatment of women with or without endometriosis.
Comparison of Clinical Characteristics, Hormonal Profile, Reproductive Outcomes, and FMR1 Alleles Corresponding to Genotypes in Women with or Without Endometriosis, Undergoing IVF
Bold values represent statistical p-value.
Qualitative variables were presented as median and IQR and qualitative variables as absolute number and percentage.
Mann–Whitney U test.
Chi-square test.
In the overall women undergoing IVF, we observed a weak linear coefficient between AMH levels and the FMR1 CGG repeat lengths (R2 = 0.0192 and p = 0.002), considering both alleles simultaneously, in addition to a weak inversely proportional relation between CGG repeat length and the number of embryos (R2 = −0.0170 and p = 0.021). Regarding women without endometriosis, we also observed a weak linear coefficient between AMH levels and the FMR1 CGG repeat lengths (R2 = 0.0331 and p = 0.008).
To verify how the CGG repeat length relates to the FSH and AMH levels, AFC, and reproductive outcomes, a generalized linear model regression considering a Poisson distribution was performed. The number of FMR1 CGG repeat lengths and AMH levels observed showed a direct and statistically significant decrease of 0.177 ng/mL of AMH per 10 CGG repeats (coefficient = 0.0177, z = 3.700, p < 0.001).
Discussion
To the best of our knowledge, this is the first Brazilian study to evaluate the impact of FMR1 CGG repeat length on ovarian reserves and IVF outcomes in women carrying FMR1 CGG repeat lengths within normal range, as well as the first study to evaluate the impact of FMR1 CGG repeat length on ovarian reserves and IVF outcomes in women with endometriosis. The results showed that women carrying a low-normal allele presented a higher frequency of low ovarian reserve and significantly lower AMH levels than women carrying a normal or high-normal allele. Women carrying low-normal/normal genotype had significantly lower AFC. We also observed a weak linear association between AMH levels and FMR1 CGG repeat lengths, as well as a weak inversely proportional relationship between FMR1 CGG repeat lengths and the number of embryos. Despite this, no influence of CGG repeats on reproductive outcomes per cycle of treatment was observed. In addition, women with endometriosis presented more frequently low-normal allele and decreases of ovarian reserve marker than women without endometriosis; nevertheless, IVF outcomes were not different between groups.
The origin of the classic CGG repeat categories arose in the context of the definition of fragile X syndrome, in the maternal risk of anticipation and expansion of the CGG repeat. This premutation was later found to also confer the risks of POI as well as the fragile X-associated tremor and ataxia syndrome. Investigations into markers of ovarian function based on the family histories of the mothers of fragile X syndrome carriers detected a relationship between this premutation and FSH and AMH levels. Initial attempts to stratify FMR1 CGG repeat lengths in the context of ovarian function resulted in a new structural arrangement with <26 repeats, 26–34 repeats, and 35–44 repeats being proposed as low-normal alleles, normal alleles, and high-normal alleles, respectively (Gleicher et al., 2010).
Gleicher et al. (2009) observed that women who were younger than 38 years of age and carried <35 FMR1 CGG repeats produced significantly more oocytes than women carrying ≥35 CGG repeats. In a 2012 study, the same group observed that FMR1 CGG repeat length did not impact oocyte production in women over 40 years of age, but when only women with lower ovarian reserves (AMH <1.05 ng/mL) were considered, those carrying a normal/high-normal genotype had greater oocyte production than women carrying a normal/low-normal genotype (Gleicher et al., 2012). Schufreider et al. (2015) found no significant association between FMR1 CGG repeats and the ovarian reserve markers, FSH, AMH, or AFC. Similarly, Morin et al. (2016) did not observe any association between FMR1 CGG repeat lengths and oocyte production.
Gleicher et al. (2011) showed that IVF pregnancy rates were significantly higher in women carrying a normal/normal genotype than those carrying a low-normal allele, even after adjusting for maternal age. Weghofer et al. (2012) observed that women carrying an FMR1 CGG allele with <26 repeats (low-normal) or >34 repeats (high-normal) had a greater chance of clinical pregnancy. Gleicher et al. (2013) concluded that the FMR1 CGG repeat length was not a predictor of the chance of pregnancy in models that included FSH and AMH levels. By contrast, a low-normal allele was associated with a significantly lower chance of clinical pregnancy (Kushnir et al., 2014).
Lledo et al. (2012) evaluated the impact of FMR1 CGG repeat length in 204 oocyte donors. The authors did not observe any differences in oocyte production or pregnancy rates due to differences in FMR1 alleles. However, Banks et al. (2016) observed a very weak association between FMR1 alleles and oocyte production, although this was not linked to embryo quality or live birth rates. Batiha et al. (2021) evaluated the possibility of a correlation between FMR1 CGG repeat lengths and FSH or AMH levels in oocytes retrieved from Jordanian women. The authors found that poor responders had a higher frequency of shorter CGG repeats, with women carrying alleles with <26 CGG repeat being twice as likely to have a poor response to COS. Cogendez et al. (2022) found no association between FMR1 CGG repeat lengths and ovarian reserves or responses to COS. Finally, Zhou et al. (2022) conducted a study involving 120 Chinese women who underwent IVF and observed a weak but significant association between high-normal alleles and a lower number of live births.
In the present study, 63.2% of women carried at least one allele classified as low-normal and presented significantly lower AMH levels than women carrying normal/high-normal alleles, notably more frequent in women with endometriosis. The percentage of women with AMH <1.1 ng/mL was more than twice that of women carrying normal/high-normal alleles. Moreover, 100% of women with low ovarian reserves carried at least one low-normal allele. Women with endometriosis had significantly higher FSH and lower AMH levels and lower AFC than women without endometriosis. Furthermore, 43.3% of women with endometriosis showed a decrease in some marker of ovarian reserve compared with 20.3% of women without endometriosis. Despite this, there were no statistical differences between the number of oocytes, embryos, or pregnancy rates per cycle of treatment of women with and without endometriosis.
Endometriosis is associated with infertility in 25−50% of cases. However, the mechanisms underlying the association between endometriosis and impaired fertility remain unknown. Recently, observed a faster decline in AMH concentrations in women with endometriomas who had not undergone surgical or medical therapy, compared with healthy women. The average percentage declines in AMH concentration after 6 months of follow-up in the endometriomas group and the control group were 26.4% and 7.4%, respectively.
Observation of the association between the FMR1 CGG repeat lengths and some aspects of ovarian function indicates that FMR1 may play an important biological role in ovarian physiology. However, our data are limited to evaluate such hypothesis, considering the relatively small sample size. Despite this, the clinical utility of FMR1 for predicting IVF outcomes was not observed. In addition, the women involved were not followed up for an adequate period of time, and AMH levels were not evaluated over the follow-up period, which could reveal a greater decline in women carrying the low-normal allele. Besides, the association among endometriosis, decreased ovarian reserve markers, and high frequency of low-normal allele is still to be explored in further studies.
Conclusions
Low FMR1 CGG repeats affected AMH levels and AFC but not IVF treatment outcomes. Women with endometriosis showed a higher frequency of the low-normal allele than women without the disease. More research is needed to improve our understanding of the biological role played by FMR1 CGG repeats within the normal range in ovarian function and fertility.
Footnotes
Acknowledgment
The autors would like to thank Conselho Nacional de Desenvolvimento Científico e Tecnológico for granting Bianca Bianco and Caio Parente Barbosa a research productivity grant.
Authors’ Contributions
Conceptualization: B.B. Methodology: A.C.V.N., C.M.T., D.M.C., and B.B. Software: A.C.V.N. and D.R. Validation: A.C.V.N., C.M.T., F.A.L., A.T.D., and D.R. Formal analysis: A.C.V.N., E.M., and B.B. Investigation: A.C.V.N., C.M.T., C.P., F.A.L., F.L.A.F., and C.P.B. Resources: C.P.B. and B.B. Data curation: A.C.V.N., E.M., and B.B. Writing—original draft preparation: A.C.V.N. and B.B. Writing—reviewing and editing: A.C.V.N., C.P.B., A.S.L., E.M., and B.B. Supervision: B.B. Visualization: B.B. and E.M. Project administration: B.B. Funding acquisition: B.B. All authors critically reviewed the manuscript and approved the final version of the manuscript. All authors contributed to the article and approved the submitted version.
Disclosure Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding Information
This work was supported by the São Paulo Research Foundation-FAPESP research grant #2020/08643-1 and #2024/08776-2 Coordenação de Aperfeiçoamento de Pessoal de Nível Superior by granting A.C.V.N. a student’s Master scholarship, and Conselho Nacional de Desenvolvimento Científico e Tecnológico by granting F.A.L. a student scholarship.
