Abstract
Corneal fibrosis is a complication of severe corneal injury, one of the major causes of vision loss. The formation of myofibroblasts has emerged as a key stimulative factor of corneal fibrosis. In the current study, we focused on the role of LINC00963 in regulating corneal fibrosis. Transforming growth factor β1 (TGF-β1) was used to induce human corneal stromal cells differentiating into corneal myofibroblasts, and the significant increase of α-smooth muscle actin (α-SMA) was verified by quantitative real-time PCR (qRT-PCR), western blot, and immunofluorescence, respectively. LINC00963 was identified to be one-half decreased compared with nonstimulated human corneal stromal cells, indicating that it might play a role in corneal fibrosis. Interestingly, overexpression of LINC00963 resulted in decreased formation of myofibroblasts indicating that it might exhibit an inhibiting effect. Moreover, bioinformatics tool was applied to predict the downstream target of LINC00963. We investigated that LINC00963 suppressed α-SMA induced by TGF-β1 in corneal fibroblasts, at least in part, by downregulating the expression of miR-143-3p. In addition, either LINC00963 promotion or miR-143-3p inhibition could significantly decrease myofibroblast contractility and collagen I and III secretion, which are the key to contribute to corneal fibrosis. Taken together, our study identified LINC00963 as a promising therapeutic target.
Introduction
Excessive healing reaction after corneal injury easily leads to the formation of corneal fibrosis scars, which seriously affects vision (Naito et al., 2014). Transforming growth factor β1 (TGF-β1) stimulating the transdifferentiation of corneal stromal cells into myofibroblasts is considered to be the ultimate reason of corneal scar formation (Wei et al., 2019). At present, there is no effective treatment method for vision loss caused by corneal fibrosis scar formation. Clinically, steroid hormones, antimetabolites, surgical treatment, and other treatment methods are used, all of which have unsatisfactory curative effects and large side effects (Yang et al., 2011; Milani et al., 2013; Dvashi et al., 2014).
Lately, noncoding RNA gene therapies using noncoding RNA have emerged as new therapeutic approach for fibrotic diseases, which are more effective than traditional drugs or antibody therapy (Abdel-Al et al., 2018; Peng et al., 2018; Zhao et al., 2018). Noncoding RNAs are a class of RNAs that rarely encode proteins, which include long noncoding RNAs (lncRNAs), circular RNAs, piwi-interacting RNAs, guide RNAs, microRNAs (miRNAs), etc. lncRNAs are often with more than 200 nucleotides length, which are rarely protein-coding potential and are involved in a variety of pathophysiological processes.
The regulatory mechanisms of lncRNAs are mainly as follows: (a) lncRNAs can bind to DNA methyltransferase (DNMT), thereby inhibiting promoter methylation and promoting gene expression (Qi et al., 2016). (b) lncRNAs can regulate histone modification and chromosome reconstruction. (c) lncRNAs can regulate gene transcription by interacting with transcription factors (Kugel and Goodrich, 2012). (d) lncRNAs act as a precursor of some miRNAs to regulate gene expression (Zou et al., 2016). (e) lncRNAs can act as miRNA sponge and then upregulate the translation of its targeting mRNAs, which is the most common regulatory mechanism (Yan et al., 2015).
MiRNAs are a class of small single-stranded noncoding RNA molecules in length of 18–22 nucleotides. They can regulate gene expression at transcriptional (promoting mRNA degradation by binding to 3′-UTR of the target mRNA) and post-transcriptional (inhibiting protein translation) levels. Studies have reported that dysregulation of noncoding RNA is associated with poor treatment of common ophthalmic diseases (Funari et al., 2013; Huang et al., 2015). So far, lncRNA NEAT1 (Bai et al., 2018), miR-206 (Li et al., 2015), miR-21 (Zhang et al., 2017), miR-145 (Ratuszny et al., 2015), and miR-184 (Park et al., 2017) have been reported to participate in the mechanism of scar formation or neovascularization after corneal fibrosis.
miR-143 showed strong expression in TGF-β1-induced subconjunctival fibrosis, and inhibition of miR-143 expression decreased myofibroblast formation to reverse the fibrosis (Hwang et al., 2019). LINC00963 has been reported to affect renal fibrosis (Chen et al., 2018) and participate in the development of multiple tumors such as osteosarcoma (Zhou et al., 2019), melanoma (Jiao et al., 2018), and hepatocellular carcinoma (Wu et al., 2018). However, no research has shown that it participates in corneal fibrosis. Thus, whether lncRNAs are involved in corneal fibrosis remains to be elucidated.
Recently, lncRNAs have been proposed to be involved in the complex processes, such as fibrosis and scarring. This study focused on LINC00963 to study its role in corneal fibrosis. We also investigated that miR-143-3p, which was associated with cardiac fibrosis (Li et al., 2019), was negatively regulated by LINC00963. Based on our findings, LINC00963 relieved corneal fibrosis by competitively binding to miR-143-3p.
Materials and Methods
Cell culture and treatment
Human corneal stromal cell line was gifted from Qingdao Eye Hospital. Cells were maintained in Dulbecco's modified Eagle's media: Nutrient Mixture F-12 (DMEM/F-12) with 10% fetal bovine serum and antibiotics (100 IU/mL penicillin and 100 mg/mL streptomycin) in a humidified atmosphere of 5% CO2 at 37°C. This experiment does not involve animal or human experiments and does not require IRB approval.
Quantitative real-time PCR
Total RNA was extracted from human corneal stromal cells and fibrotic corneas or normal corneas using TRIzol reagent (Invitrogen, Carlsbad, CA) according to the manufacturer's instructions. Reverse transcription (RT) of mRNA and lncRNA to cDNA was synthesized with random primers by PrimeScript RT Reagent Kit (Takara Biomedical Technology, Dalian, China), whereas the reverse primer of miRNA is synthesized by BGI Genomics (Shenzhen, China). The SYBR Premix Ex TaqII Kit (Takara Biomedical Technology) was used for quantitative real-time PCR (qRT-PCR) analysis.
The primers for α-smooth muscle actin (α-SMA), GAPDH, U6, and miR-143-3p are listed in Table 1. All data analyses were operated by qRT-PCR system (Bio-Rad Biosystems). U6 and GAPDH, respectively, served as the internal control for miRNA and mRNA quantification. All qRT-PCR experiments were performed in triplicates.
Primer Sequences for Quantitative Real-Time PCR
miRNA, siRNA reagents, and their application
miRNA agomir or antagomir and siRNA were synthesized by Gene Pharma (Shanghai, China). All the sequences are shown in Table 2. Lipofectamine RNAiMAX (Invitrogen) was used for transfection of miRNAs in vitro. Human corneal stromal cells were planted in wells or plates 24 h before use with 40–60% confluence.
Sequence of Transfection
Plasmid construction
According to the sequences of LINC00963 in Genebank, plasmid containing full length of LINC00963 and negative control (NC) plasmid were synthesized by BGI (Shenzhen, China).
Dual-Luciferase Reporter Assay
The wild-type fragment or mutants of the predicted binding site of miR-143-3p on LINC00963 transcript were synthesized and constructed into the pGL3-control reporter vector (Promega, Madison, WI). 293T cells were harvested at 24 h after transfection and assayed for luciferase activity by the Dual Luciferase Reporter Assay Kit (Promega).
Western blot analysis
Anti-α-SMA antibody and anti-GAPDH antibody were purchased from ABclonal (Wuhan, China), and horseradish peroxidase (HRP)-labeling Goat anti-Rabbit IgG and HRP-labeling Goat anti-Mouse IgG were purchased from EpiZyme (Shanghai, China). RIPA lysis buffer was purchased from Solarbio Life Sciences (Beijing, China). RIPA lysis buffer supplemented with phenylmethylsulfonyl fluoride (Roche) was used to lysis cell or tissue samples. The concentrations of all protein samples were assayed using a BCA Protein Assay Kit (Merck, China). Add equal amounts of protein samples to the gel wells and separate them by 10% SDS-polyacrylamide gel electrophoresis gels.
After electrophoresis and transfer of the protein band to the polyvinylidene fluoride membrane, the membrane was blocked with 5% skim milk dissolved in Tris-buffered saline for 1 h. Then membranes were incubated with the following primary antibodies: α-SMA (1:1000 dilution; ABclonal) and GAPDH (1:50,000 dilution; ABclonal) for 1.5 h at room temperature or shaking at 4°C overnight. Blots were washed thrice for 10 min and incubated with goat-anti-mouse/rabbit secondary antibody (1:3000 dilution; EpiZyme) for 1 h at room temperature. Visualization was performed using Omni-ECL™ Femto Light Chemiluminescence Kit (EpiZyme).
Immunofluorescence
Immunofluorescence staining was performed on human corneal stromal cells grown in 24-well (ExCell Bio.). Cells were washed using phosphate buffered saline (PBS) thrice and fixed with 4% paraformaldehyde for 10 min, permeabilized with 0.1% Triton X-100 in PBS for 5 min, followed by a blocking step with PBS containing 2% bovine serum albumin for 90 min at room temperature. Subsequently, cells were stained with rabbit anti-human α-SMA antibody for 90 min at room temperature or shaking at 4°C overnight, washed, and incubated with FITC goat anti-rabbit IgG (ABclonal) for 60 min. The slides were mounted in a closure containing DAPI (Solarbio Life Sciences) and placed on a confocal microscope equipped with a FITC or DAPI filter set (using a 63 oil objective).
Cell contraction assay
Corneal stromal cells were transfected with LINC00963 or miR-143-3p antagomir or NC for 24 h and then stimulated with TGF-β1 for 48 h to differentiate into myofibroblast. Then cells were seeded into collagen gels; after 48 h, gel contraction was assessed according to the protocol of the kitCell (Biolabs, Inc.).
Collagen secretion assay
Cells were seeded into 48-well plates and further transfected with pcDNA3.1-LINC00963 or miR-143-3p agomir or antagomir for 24 h and then stimulated with TGF-β1 (2 ng/mL) for 48 h. The content of collagen I and collagen III in supernatants was detected by Enzyme-Linked Immunosorbent Assay (ELISA) Kits (Shanghai Jianglai Biotech, Shanghai, China). Optical density was measured at 450 nm.
Statistical analysis
Data analysis was performed using SPSS 22.0 (SPSS, Chicago) statistical software. The data were represented as
Results
TGF-β1 promoted human corneal stromal cells to transdifferentiate into myofibroblasts
Human corneal stromal cells were treated with 2 ng/mL TGF-β1 for 6, 12, and 24 h, respectively. De novo expression of α-SMA, a widely accepted myofibroblast marker (Tomasek et al., 2002), showed that human corneal stromal cells successfully transdifferentiated into myofibroblasts (Fig. 1a–c).
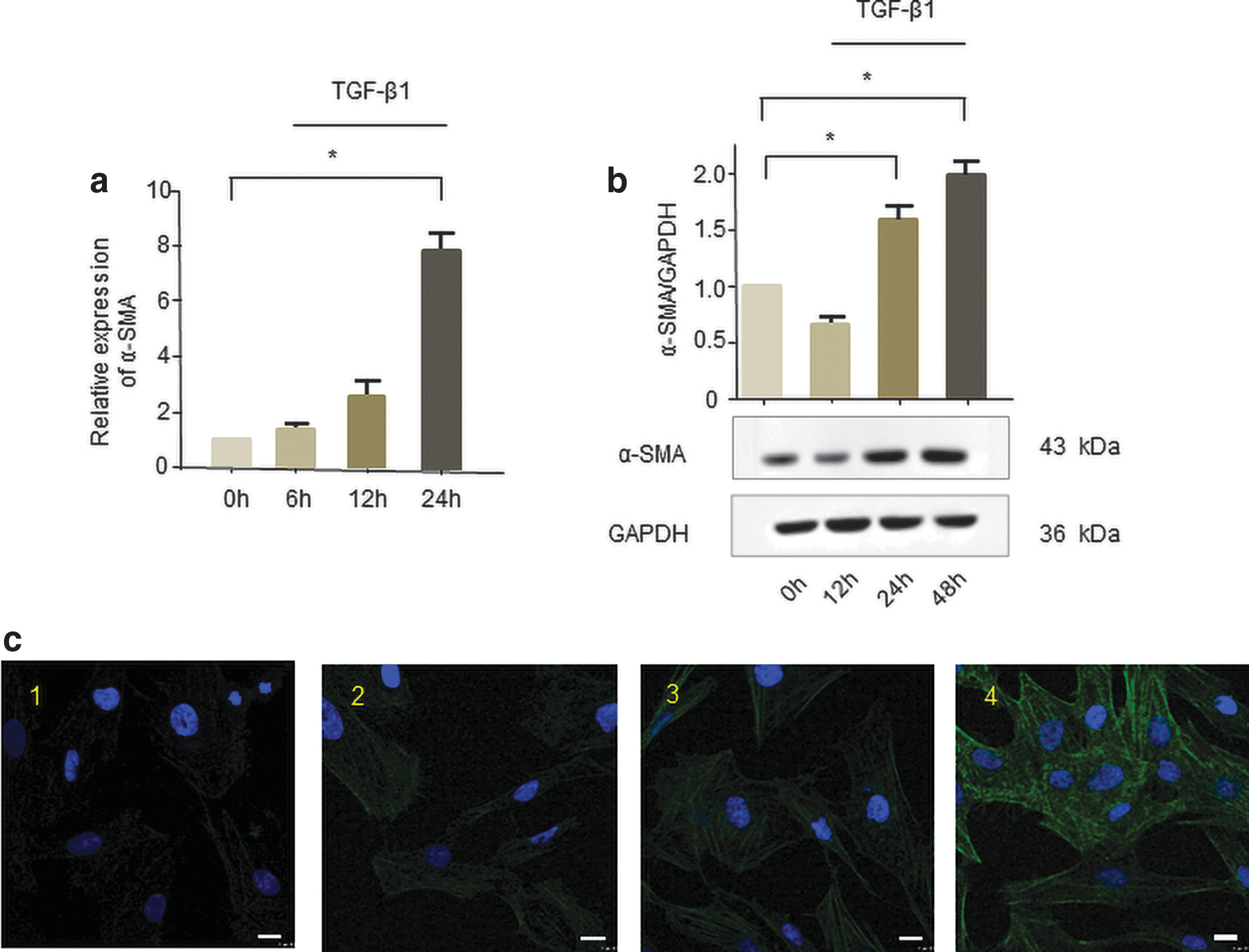
The role of TGF-β1 in promoting the transdifferentiation of human corneal stromal cells into myofibroblasts in vitro. Human corneal stromal cells were treated with TGF-β1 (2 ng/mL) for 6, 12, and 24 h.
Candidate lncRNAs involved in scarring of the cornea
Through extensive literature searches, we have discovered some lncRNAs that may play a role in fibrosis, such as GAS5 (Tao et al., 2017), NORAD (Kawasaki et al., 2018), and LINC00963 (Chen et al., 2018). We detected the expression of candidate lncRNAs in human corneal stromal cells treated with TGF-β1 in a time-dependent manner. The results showed that GAS5 (Fig. 2a) and NORAD (Fig. 2b) had less change under the stimulation of TGF-β1. However, LINC00963 expression was gradually decreased after TGF-β1 treatment (Fig. 2c), indicating that LINC00963 may be involved in the occurrence of corneal fibrosis.
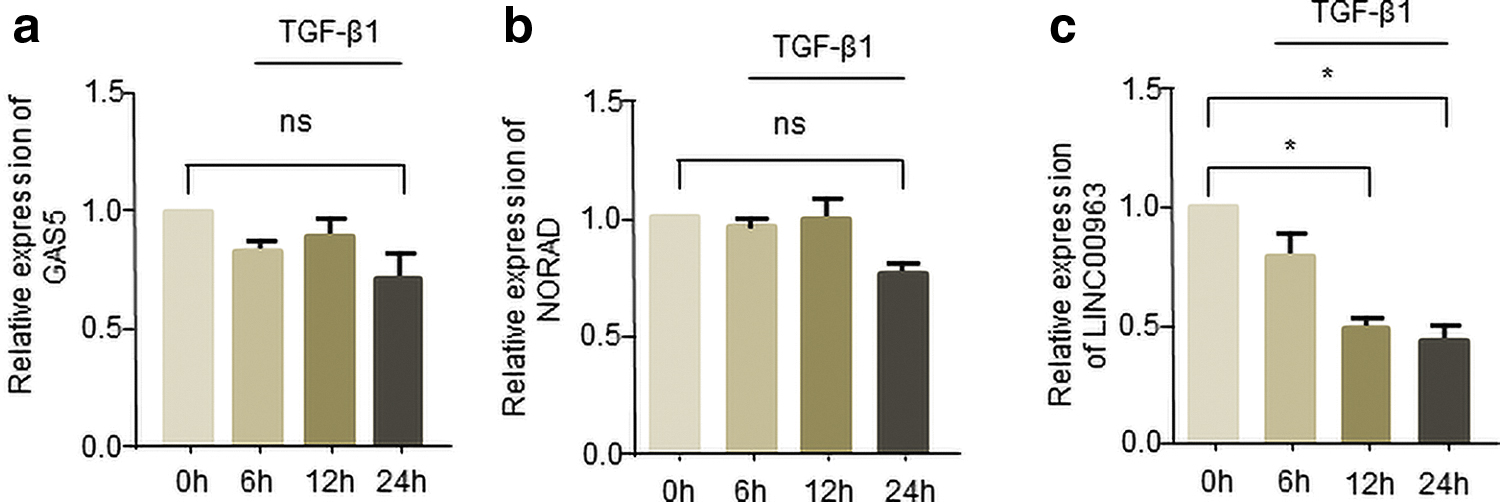
Candidate lncRNAs potentially involved in corneal fibrosis.
Overexpression of LINC00963 prevented human corneal stromal cells from transdifferentiating into myofibroblasts
To assess whether LINC00963 was critical for myofibroblast function, we constructed LINC00963 overexpression plasmid and analyzed changes in the expression of α-SMA, a common marker of myofibroblast. qRT-PCR demonstrated that LINC00963 levels increased twofold compared to NC plasmids (Fig. 3a). And α-SMA mRNA and protein levels were upregulated in TGF-β1-stimulated human corneal stromal cells, but significantly decreased in LINC00963 enforced myofibroblasts compared with NC (Fig. 3b–d). Next, three-dimensional collagen gels were used in corneal stromal cell contraction studies. We can see that overexpression of LINC00963 can significantly reverse the increase of cell contractility treated by TGF-β1 (Fig. 3e). Moreover, LINC00963 can also reverse the collagen I (Col I) and Col III secretion caused by TGF-β1 (Fig. 3f).
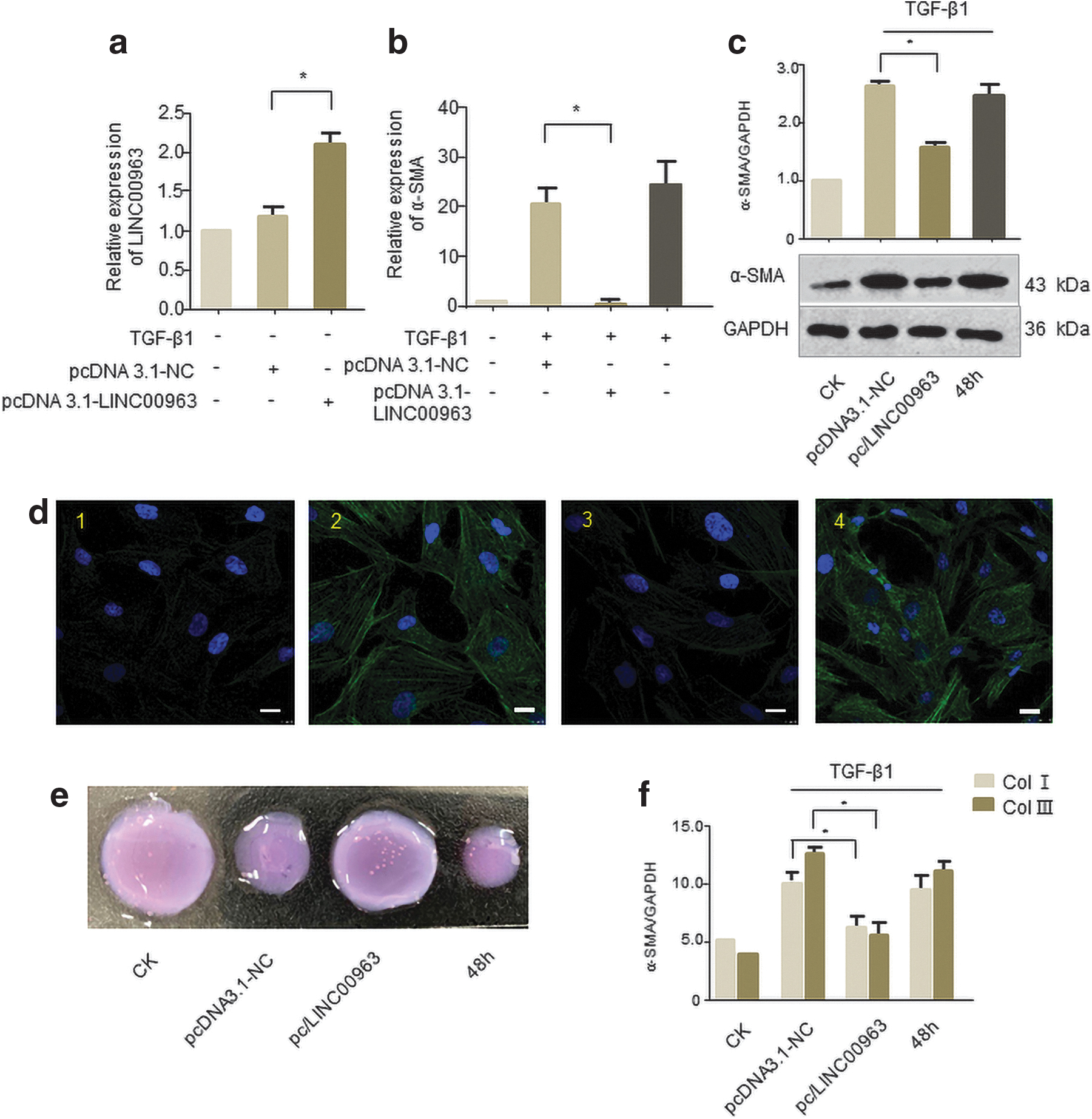
Overexpression of LINC00963 repressed human corneal stromal cells to transdifferentiate into myofibroblasts.
LINC00963 directly regulates the expression of miR-143-3p
Since one of the functional mechanisms of lncRNAs is to act as miRNA sponge (Song et al., 2017; YiRen et al., 2017), we applied Starbase website to predict the downstream target miRNA of LINC00963. Finally, we selected miR-143-3p to verify their interactions, which has been reported to be linked with heart fibrosis (Li et al., 2019). However, the role of miR-143-3p in corneal fibrosis still remains to be explored. We found that enforced LINC00963 reduced miR-143-3p level in human corneal stromal cells (Fig. 4a). In addition, downregulation of LINC00963 increased the expression level of miR-143-3p (Fig. 4b).
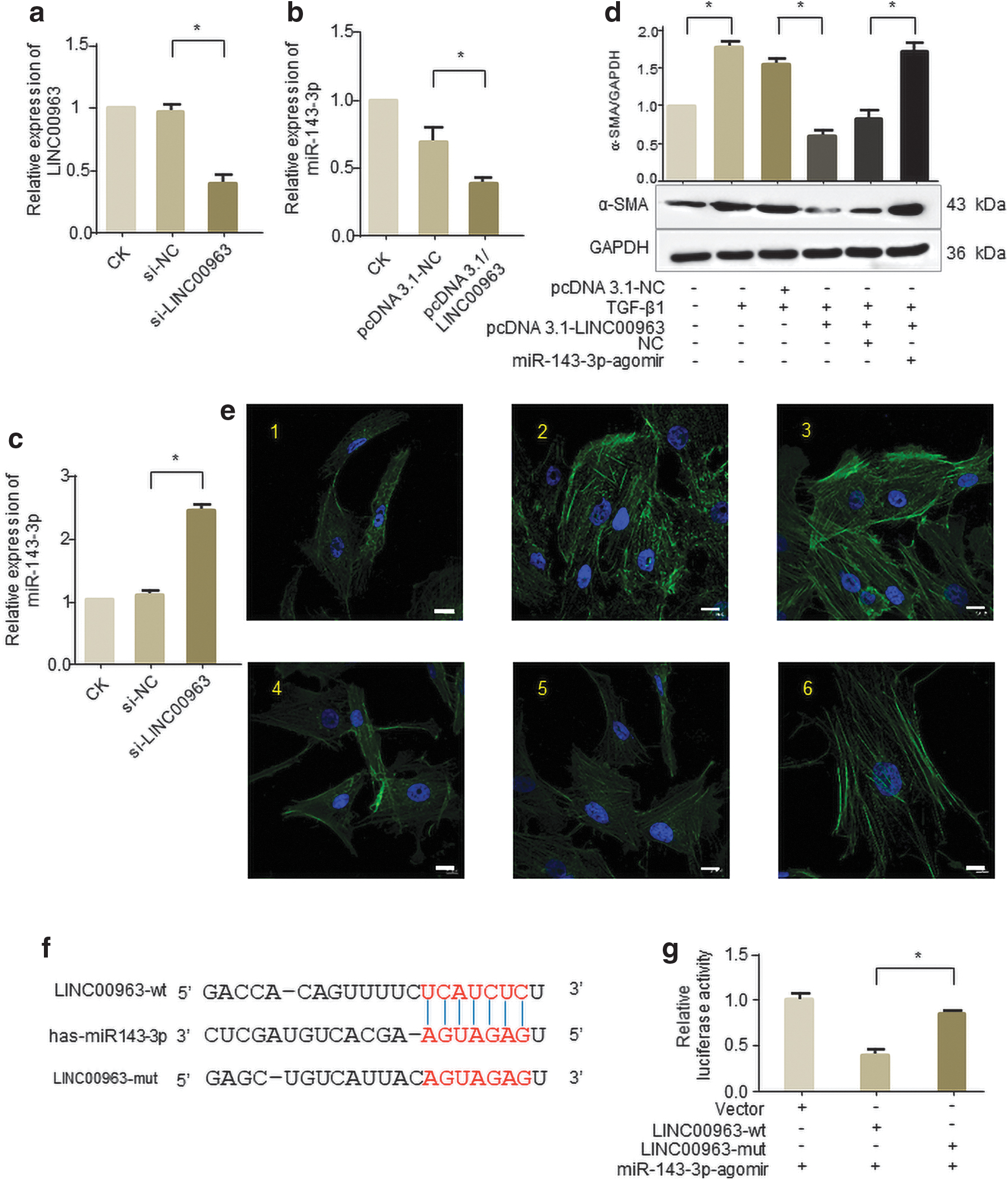
LINC00963 is physically associated with miR-143-3p. Cells were transfected with pcDNA3.1/LINC00963 or siRNA targeting LINC00963 for 24 h.
Then, we examined whether LINC00963 attenuated corneal fibrosis through sponging miR-143-3p. Overexpression of LINC00963 inhibited TGF-β1 induced the high expression of α-SMA, while forced expression of miR-143-3p could abrogate this inhibition (Fig. 4c). Representative images indicated that human corneal stromal cells are slim and have weak fluorescence expression, but cells become fat and have stronger fluorescence with TGF-β1 treatment. Overexpression of LINC00963 inhibited TGF-β1 induced this change, while forced expression of miR-143-3p abolishes this inhibition (Fig. 4d).
To further confirm the endogenous direct binding between miR-143-3p and LINC00963, we constructed luciferase reporters containing the 200-nt of 3′ LINC00963, which includes wild-type (wt) or mutated miR-143-3p binding sites (Fig. 4e). It shows that overexpression of miR-143-3p inhibited the luciferase activities of the wt reporter vector but not empty or mutant ones in human corneal stromal cells (Fig. 4f), suggesting that LINC00963 may function as miR-143-3p sponge in human corneal stromal cells.
miR-143-3p facilitated human corneal stromal cells transdifferentiating into myofibroblasts
Then we investigated whether miR-143-3p participated in corneal fibrosis formation. As shown in Figure 5a, miR-143-3p expression was gradually upregulated under TGF-β1 treatment in human corneal stromal cells. Next, cells were transfected with miR-143-3p agomir and antagomir, respectively. And qRT-PCR demonstrated that miR-143-3p levels significantly increased (Fig. 5b) or decreased (Fig. 5c) compared to the NC. The miR-143-3p agomir efficiently promoted the expression of fibrosis marker α-SMA in the level of transcription (Fig. 5d) and translation (Fig. 5e), respectively. Meanwhile, miR-143-3p antagomir can eliminate the rise of α-SMA in TGF-β1 treatment human corneal stromal cells (Fig. 5f, g).
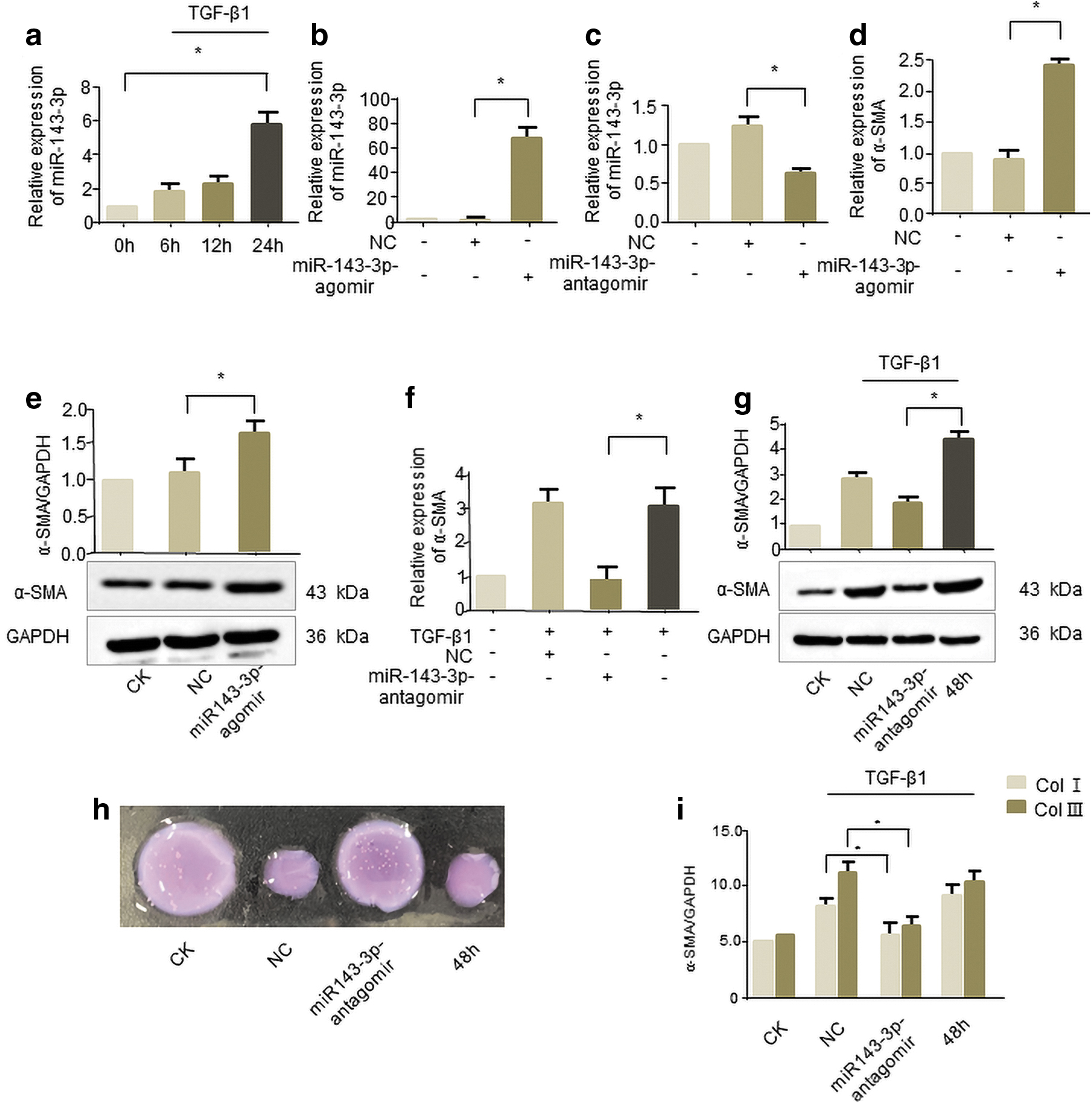
miR-143-3p promotes human corneal stromal cells transdifferentiating into myofibroblasts
Consistent with the α-SMA expression, we observed that miR-143-3p antagomir not only reverses the increase in cell contraction (Fig. 5h) but also reverses Col I and Col III secretion caused by TGF-β1 (Fig. 5i). These results indicate the involvement of miR-143-3p in TGF-β1 treated human corneal stromal cells, which can facilitate the corneal fibrosis.
Discussion
The formation of corneal fibrosis scar can cause corneal blindness, which is the second blinding ocular disease in the world (Flaxman et al., 2017). The process of corneal fibrosis scar formation is very complicated, mainly because TGF-β1 secreted from the wound area after corneal injury promotes the transformation of corneal stromal cells into myofibroblasts, which can be identified by its specific marker α-SMA (Blalock et al., 2003; Wilson, 2012).
However, there is no effective treatment for visual loss caused by corneal fibrosis so far. There are still no Drug Administration approved drugs that specifically reduce the expression of corneal fibrosis related genes. Mitomycin C is often applied in some ocular surgeries, but it has side effects such as death of endothelial cells (Zhao et al., 2008), stromal melting, and conjunctival thinning (Raviv et al., 2000; de la Fuente et al., 2008).
Gene therapy has been a research hotspot in recent years, and it is more effective than traditional drugs or antibody therapy. siRNA target drugs have already been used for treatment of ocular diseases. For example, bevasiranib targets the VEGF gene to reduce VEGF production and is used to treat age-related macular degeneration (Garba and Mousa, 2010). Therefore, it is urgent to find a gene therapy for corneal fibrosis scar.
As new therapeutic approach for fibrotic diseases such as cardiac fibrosis (Leisegang, 2018), pulmonary fibrosis (Lu et al., 2018), and renal fibrosis (Wang et al., 2018), lncRNA-based gene therapies have emerged recently. However, the function of lncRNAs in human corneal fibrosis still requires a great deal of exploration.
To identify those lncRNAs that may be involved in the differentiation of human corneal stromal cells, we narrowed down our target range by searching extensive literature. In that TGF-β1 can effectively promote the transdifferentiation of cells into myofibroblasts (Knuppel et al., 2018; Chen et al., 2019). Therefore, 2 ng/mL TGF-β1 was used to stimulate human corneal stromal cells that transdifferentiate to construct a cell model of corneal fibrosis scars and detected the expression of α-SMA by qRT-PCR and western blot (compared with the control group, the expression increased by more than two times) to verify the success of the model. The results showed that the expression of α-SMA was significantly upregulated after TGF-β1 stimulated for 24 h (transcription level) and 48 h (translation level).
The above experiments show that 2 ng/mL TGF-β1 can induce the transdifferentiation of corneal stromal cells into myofibroblasts, which proves that the cell model of corneal fibrosis scar has been successfully constructed. Then the expression level of lncRNAs that may be involved in fibrosis regulation in the literature was detected in this cell model.
LINC00963 was reported to promote melanoma malignant progression by elevating expression of NACC1 through inhibiting miR-608 (Jiao et al., 2018). It also has function in renal interstitial fibrosis and oxidative stress of chronic renal failure using the forkhead box O (FoxO) signaling pathway (Chen et al., 2018).
To date, there was no research uncovering its role in corneal fibrosis. Our experimental evaluation of candidate lncRNAs showed that LINC00963 is significantly downregulated in TGF-β1-induced corneal myofibroblasts in a time-dependent manner, which may be a signal that it can act on the formation of corneal fibrosis scars. Moreover, overexpression of LINC00963 could eliminate the increase of α-SMA caused by TGF-β1. We also observed a strong decrease in myofibroblast contractility and decreased secretion of collagen I and III, revealing a novel mechanism for regulating corneal scars.
lncRNA acts as miRNA sponge, which can disrupt the binding of miRNA to its target mRNA, which consequently promotes mRNA translation. For example, lncRNA-MIAT can upregulate CD47 expression by inhibiting miR-149-5p expression in advanced atherosclerosis (Ye et al., 2019). LINC00346 inhibits BRD4 expression to promote pancreatic cancer growth and gemcitabine resistance, primarily by sponging miR-188-3p (Shi et al., 2019).
In this context, we found that LINC00963 might act as a ceRNA for miR-143-3p using the bioinformatics program Starbase. This view has also been verified by our in vitro results. In addition, miR-143-3p was reported to promote human cardiac fibrosis by targeting sprouty3 after myocardial infarction (Li et al., 2019) and promote Col III expression in interstitial fibroblasts (Naito et al., 2014), which provides the probability of its role in corneal fibrosis. Our studies confirmed the findings that LINC00963 could directly bind to the miR-143-3p and suppressed its expression, thereby reversing the effect of miR-143-3p on promoting corneal fibrosis.
In summary, our study identified a lncRNA LINC00963 that inhibits the development and progression of corneal fibrosis. We demonstrated the relationship between LINC00963 and miR-143-3p in TGF-β1-induced corneal fibrosis. Our study may provide theoretical support for a promising treatment strategy for corneal fibrosis suggesting the use of LINC00963 alone or in combination with other anti-scar or inflammatory drugs. However, the involvement of miRNAs and/or molecules in ceRNA networks involving LINC00963 are still next issues to be addressed. Taken together, our research provides new ideas and theoretical support for the treatment of corneal fibrosis, and LINC00963/miR-143-3p axis may provide promising therapeutic approach for the prevention and treatment of corneal fibrosis.
Footnotes
Disclosure Statement
No competing financial interests exist.
Funding Information
This study was financially supported by the National Natural Science Foundation of China (81770900), the Qingdao Science and Technology Key Project (20-3-4-43-nsh), the Science and Technology Development Foundation of Shandong Province (2014GHY115025), and the Qingdao Science and Technology Plan fund (16-6-2-28-NSH).
