Abstract
PM2.5 plays an important role in the physiological and pathological progression of lung cancer. Lentinan exerts antitumor activity in many kinds of human cancers. Plasmacytoma variant translocation 1 (PVT1) exerts antitumor activity in many kinds of human cancers. However, the role and underlying molecular mechanism of PVT1 in the role of lentinan in PM2.5-exposed lung cancer are still largely unknown. Our study confirmed that PM2.5 exposure induced the production of inflammatory factors, epithelial–mesenchymal transition (EMT) and migration of lung cancer cells. Lentinan exerted antitumor effects by inhibiting the production of inflammatory factors, EMT, and migration of lung cancer cells. Lentinan suppressed PM2.5 exposure-induced cellular progression by inhibiting the PM2.5 exposure-induced elevation of PVT1 expression. PVT1 absorbed miR-199a, and miR-199a inhibited caveolin1 expression and thus formed the PVT1/miR-199a/caveolin1 signaling pathway in lung cancer cells. Our study revealed that silencing of the PVT1/miR-199a/caveolin1 signaling pathway affected the role of lentinan in PM2.5-exposed lung cancer cells. Thus, this study first investigated the role of lentinan in PM2.5-exposed lung cancer cells and further displayed the underlying molecular mechanism, providing a potential treatment for PM2.5-exposed lung cancer.
Introduction
As one of the most concerning malignancies worldwide, lung cancer possesses the highest mortality and poor survival (Cheng et al., 2016; Mao et al., 2016). Lung cancer includes nonsmall cell lung cancer (NSCLC) and small cell lung cancer (Schwartz and Cote, 2016). There are many causes of lung cancer, including smoking, occupational and environmental exposure to carcinogens, ionizing radiation, tuberculosis, bronchiectasis and other chronic lung infections, genetics, and air pollution (Cheng et al., 2016). Despite advances in diagnosis and treatment, the 5-year survival rate of lung cancer remains at ∼17% (Bade and Dela Cruz, 2020). Hence, dissecting the molecular mechanism underlying the development and progression of lung cancer would be beneficial for establishing new prevention and treatment methods for the disease.
As a product of rapid urbanization and industrialization, air pollution is increasing rapidly and becoming a major health threat (Orru et al., 2017). PM2.5, defined as particulate matter ≤2.5 μm in diameter, exerts important impacts on the respiratory system, including lung infection, abnormal lung function, and lung cancer (Li et al., 2018a). PM2.5 affects several cellular processes, including cell growth, apoptosis, metastasis, immune response, and epithelial–mesenchymal transition (EMT) (Grzywa-Celinska et al., 2020). However, the role and underlying mechanism of PM2.5 in the physiological and pathological progression of lung cancer remain largely unknown.
Derived from the shiitake mushroom (Lentinula edodes), lentinan plays important roles in various kinds of diseases by enhancing the host immune system (Ren et al., 2012). Lentinan exerts antitumor activity in many kinds of human cancers, including cardiac cancer, gastric cancer, pancreatic cancer, nasopharyngeal cancer, colorectal cancer, non-Hodgkin lymphoma, liver cancer, bladder cancer, ovarian cancer, cervical cancer, and duodenal cancer, as well as lung cancer (Zhang et al., 2019). Yan-rong Liu and colleagues showed that lentinan inhibited cancer cell viability, cell proliferation, cell cycle, cell EMT, and migration (Liu et al., 2018). Clinical data over the past 12 years have shown that lentinan can effectively improve not only the quality of life but also the efficacy of chemotherapy during lung cancer treatment (Zhang et al., 2018). However, there is no research focusing on the role of lentinan in PM2.5-related lung cancer.
Long noncoding RNAs (lncRNAs), dominantly located in the cell nucleus and cytoplasm, are a class of highly conserved RNA molecules with lengths >200 nucleotides lacking protein-coding potential (Kopp and Mendell, 2018). lncRNAs affect various cellular pathways, including proliferation, the cell cycle, metastasis, apoptosis, development, and tumorigenesis, by regulating the number of genes expressed at the transcriptional, post-transcriptional, and translational levels (Orom et al., 2010). lncRNAs are involved in various kinds of human diseases, especially human cancers (Batista and Chang, 2013). Several lncRNAs participate in the role of PM2.5, such as LCPAT1, DQ786227, MALAT1, lncRNA loc146880, and H19 (Li et al., 2018b; Wang et al., 2019). Plasmacytoma variant translocation 1 (PVT1), located at chromosome 8q24, exerts antitumor activity in many kinds of human cancers (Cui et al., 2016). Christiane E Olivero and colleagues reported that p53-activated Pvt1b suppressed tumorigenesis by inhibiting Myc expression (Olivero et al., 2020). However, there is no literature focusing on the role and underlying mechanism of PVT1 in lung cancer under PM2.5 conditions.
As a structural component of caveolae, caveolin-1 exerts crucial function in a variety of cellular functions, consisting of cell growth, adhesion, signal transduction, migration, senescence, development, and tumorigenesis (Boscher and Nabi, 2012; Ketteler and Klein, 2018; Volonte and Galbiati, 2020). Caveolin-1 also played an important role in mechanosensing that reflects the response the mechanical forces (extracellular matrix stiffness, vascular shear stress, and muscle stretching) reaching on the plasma membrane and affects the membrane integrity and repair (Dewulf et al., 2019; Del Pozo et al., 2020; Hubert et al., 2020; Liu, 2020; Singh and Lamaze, 2020). Caveolin 1 involves in the EMT in lung cancer (Garnett, 2016; Shi et al., 2020). A report revealed that caveolin-1 regulated the PM2.5-induced endocytosis in lung cancer (Wei et al., 2020). Several studies demonstrated that caveolin-1 was regulated by miR-199a (Zhang et al., 2015; Zhong et al., 2020). However, the role of caveolin-1 in the inhibitory function of lentinan in lung cancer under PM2.5 conditions is still largely unknown.
In this study, we aimed to dissect the role and the underlying mechanism of lentinan on PM2.5-exposed lung cancer cells. This study revealed that lentinan exerted an inhibitory effect on PM2.5-exposed lung cancer cells by activating the PVT1/miR-199a/caveolin1 regulatory pathway, thus providing a new theoretical basis for studying the occurrence and development of lung cancer.
Materials and Methods
Cell culture and PM2.5 sampling and preparation
The human lung cancer cell lines A549 and H520 were obtained from the Chinese Academy of Sciences Cell Bank (Shanghai, China). A549 cells were cultured in Dulbecco's modified Eagle's medium (DMEM; Gibco BRL, Gaithersburg, MD), and H520 cells were maintained in RPMI-1640 medium (Gibco BRL). All media were supplemented with 10% fetal bovine serum (FBS; Gibco BRL), 2 mM glutamine, and 1% penicillin/streptomycin solution (Gibco BRL). All cells were grown in a humidified atmosphere with 5% CO2 at 37°C.
PM2.5 samples were collected and prepared as described previously. The particles were dissolved in phosphate-buffered saline (PBS) at a concentration of 5 mg/mL and stored at −80°C in our laboratory for further research. For the treatments of PM2.5, cells were exposed to PM2.5 for 24 h at different concentrations (0, 2.5, 5, and 10 μg/mL).
Alteration of gene expression and cell transfections
The construction of ectopic expression of PVT1 and caveolin1 was based on the plvx-puro plasmids. Short hairpin RNA was used to inhibit gene expression and was cloned on the plko-puro plasmids. The miR-199a mimics, miR-199a inhibitors, and their control small RNAs were synthesized from Guangzhou RiboBio Technology Co., Ltd. (Guangzhou, China). The plasmids and small RNAs were transfected into the cells using the Lipofectamine® 3000 transfection kit (Invitrogen, Waltham, MA) for 48 h.
RNA extraction and quantitative real-time PCR
After washing with ice-cold PBS, RNA extraction reagents (Junxin, Suzhou, China) were added to the cells, and the total RNA was isolated according to the manufacturer's instructions. A NanoDrop (Thermo Fisher Scientific, Waltham, Massachusetts, USA) was used to measure the concentration, purity, and integrity of RNA. Total RNA (1 μg) and a cDNA Reverse Transcription kit (Beyotime, Beijing, China) were used for reverse transcription. SYBR green I qPCR mix (Junxin) was used to perform real-time PCR analysis on a 7500 Fast Real-Time PCR System (Applied Biosystems, Waltham, MA). The PCR program consisted of initial denaturation at 95°C for 10 min, followed by 40 cycles of denaturation at 95°C for 15 s, annealing at 60°C for 20 s, and extension at 72°C for 30 s. The expression of β-actin was measured as a reference gene, and the differences in gene expression (fold change) were calculated using the 2−ΔΔCT method. The primers for the target genes and β-actin are given in Table 1.
Sequence of Primers
Enzyme-linked immunosorbent assay analysis
The culture medium (200 μL) was harvested, centrifuged at 1006 g at 4°C for 10 min, and stored at −80°C. Enzyme-linked immunosorbent assay (ELISA; R&D Systems, Minneapolis, MN) was used to analyze the production of interleukin 6 (IL6) and tumor necrosis factor alpha (TNFα) according to the manufacturer's instructions.
Western blot assay
After washing with ice-cold PBS, cells were treated with radioimmunoprecipitation (RIPA) buffer (BOSTER, Wuhan, China), and a bicinchoninic acid (BCA) protein detection kit (Beyotime) was used to measure protein concentration and purity. A sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) separation gel (10%) was introduced to separate the proteins. After transfer to a polyvinylidene fluoride membrane (PerkinElmer, Boston, MA), 5% nonfat milk was used to block the nonspecific proteins on the membrane. Then, the membrane was incubated with primary antibodies overnight at 4°C, followed by incubation with the corresponding HRP-conjugated secondary antibodies. Enhanced chemiluminescence reagents (Junxin) were used to detect the immunoreactive bands. The expression of β-actin was measured as a reference gene. The related primary antibodies were anti-β-actin (Dilution 1:4000; Santa Cruz Biotechnology, Santa Cruz, CA), anti-E-cadherin (Dilution 1:100; Abcam, Cambridge, MA), antivimentin (Dilution 1:1000; Abcam), and anticaveolin1 (Dilution 1:1000; Abcam).
Cell migration assay
Transwell chambers (8.0-μm pore size) (#3422; Corning) were used to detect cell migration according to established protocols. Cells (1 × 104 cells) cultured in the corresponding medium without FBS were plated into the upper chamber, and the corresponding medium supplemented with 10% FBS was added into the lower chamber for 24 h. After the removal of unmigrated cells in the inner face of the chamber, the migrated cells in the outer face of the chamber were stained using 1% crystal violet and were counted by fluorescence microscopy (Olympus, Japan).
Statistical analysis
All data are presented as the mean ± standard deviation. All statistical analyses were performed using SPSS software, version 19.0 for Windows (SPSS, Chicago, IL). Student's t-test and one-way analysis of variance were used to calculate the statistical significance for two- or multiple-group comparisons, respectively. p < 0.05 (*) was considered statistically significant, and p < 0.01 (**) was considered dominantly statistically significant.
Results
PM2.5 induced the production of IL6 and TNFα and promoted the migration capacity of lung cancer cells
To measure the function of PM2.5 on the production of inflammatory factors in lung cancer cells, A549 and H520 cells were exposed to PM2.5 (0, 2.5, 5, and 10 μg/mL) for 48 h, and ELISA, Western blots, and Transwell assays were performed to measure the production of IL6 and TNFα, EMT, and the migration of lung cancer cells. As shown in Figure 1A and B, the production levels of IL6 and TNFα were higher in the PM2.5-exposed culture supernatant than in the control groups. Meanwhile, the EMT and migration of cells were elevated in the PM2.5-exposed cell groups than in the control groups (Fig. 1C, D). Overall, these data proved that PM2.5 exposure promoted the production of IL6 and TNFα, EMT, and the migration capacity of A549 and H520 cells.
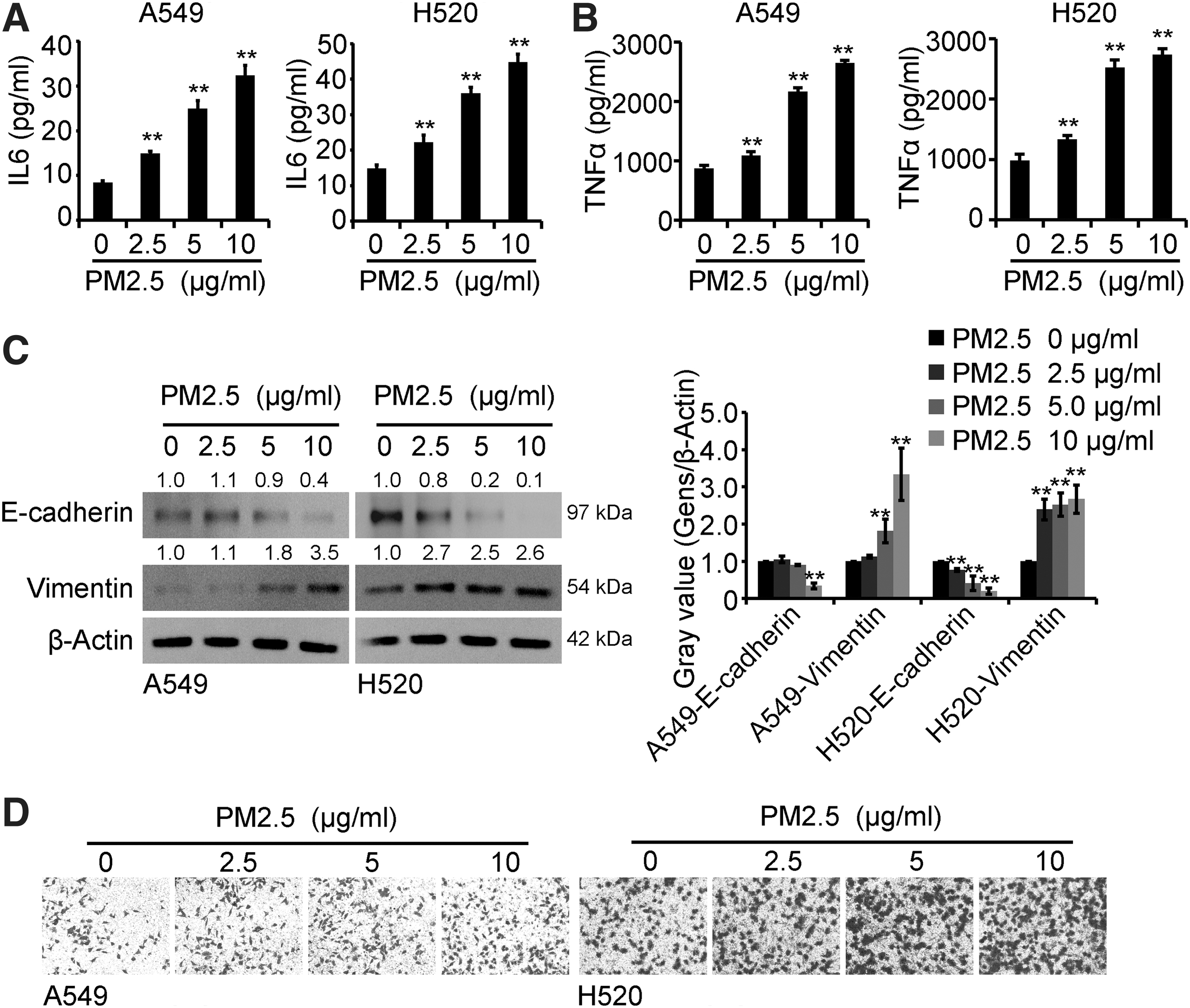
PM2.5 induced the production of IL6 and TNFα and promoted the migration capacity of lung cancer cells.
Lentinan inhibited the production of IL6 and TNFα and the migration capacity of lung cancer cells
To determine the role of lentinan on the production of inflammatory factors in lung cancer cells, A549 and H520 cells were treated with lentinan (0, 100, 200, 400, and 600 μg/mL) for 48 h, and ELISA, Western blots, and Transwell assays were performed to detect the secretion of IL6 and TNFα, EMT, and the migration capacity of lung cancer cells. As shown in Figure 2A and B, the production of IL6 and TNFα was lower in the lentinan-treated culture supernatant than in the control groups. Meanwhile, the EMT and migration capacity of cells were decreased in the lentinan-treated cell groups compared with the control groups (Fig. 2C, D). In general, these results showed that lentinan inhibited the production of IL6 and TNFα, EMT, and the migration capacity of A549 and H520 cells.
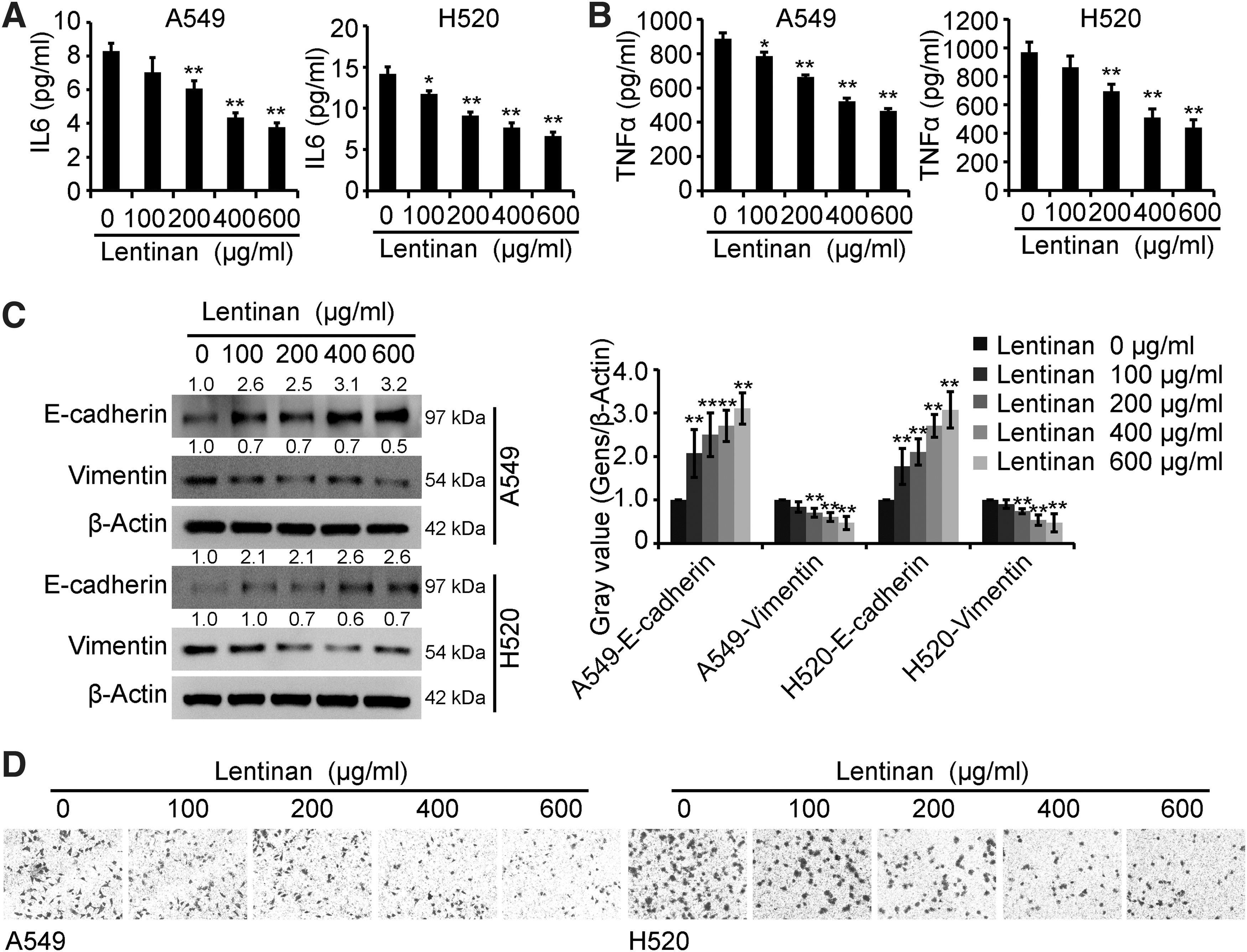
Lentinan inhibited the production of IL6 and TNFα and the migration capacity of lung cancer cells.
Lentinan attenuated the role of PM2.5 in lung cancer cells
To investigate whether lentinan could affect the role of PM2.5 in lung cancer cells, cells were exposed to PM2.5 with or without lentinan treatment for 48 h, and ELISA, Western blots, and Transwell assays were performed to detect the secretion of IL6 and TNFα, EMT, and the migration capacity of lung cancer cells. As shown in Figure 3A and B, lentinan decreased the production of IL6 and TNFα in PM2.5-exposed cells. Furthermore, lentinan inhibited the EMT and migration capacity of PM2.5-exposed cells (Fig. 3C, D). Overall, these observations suggested the inhibitory role of lentinan in PM2.5-exposed cells.
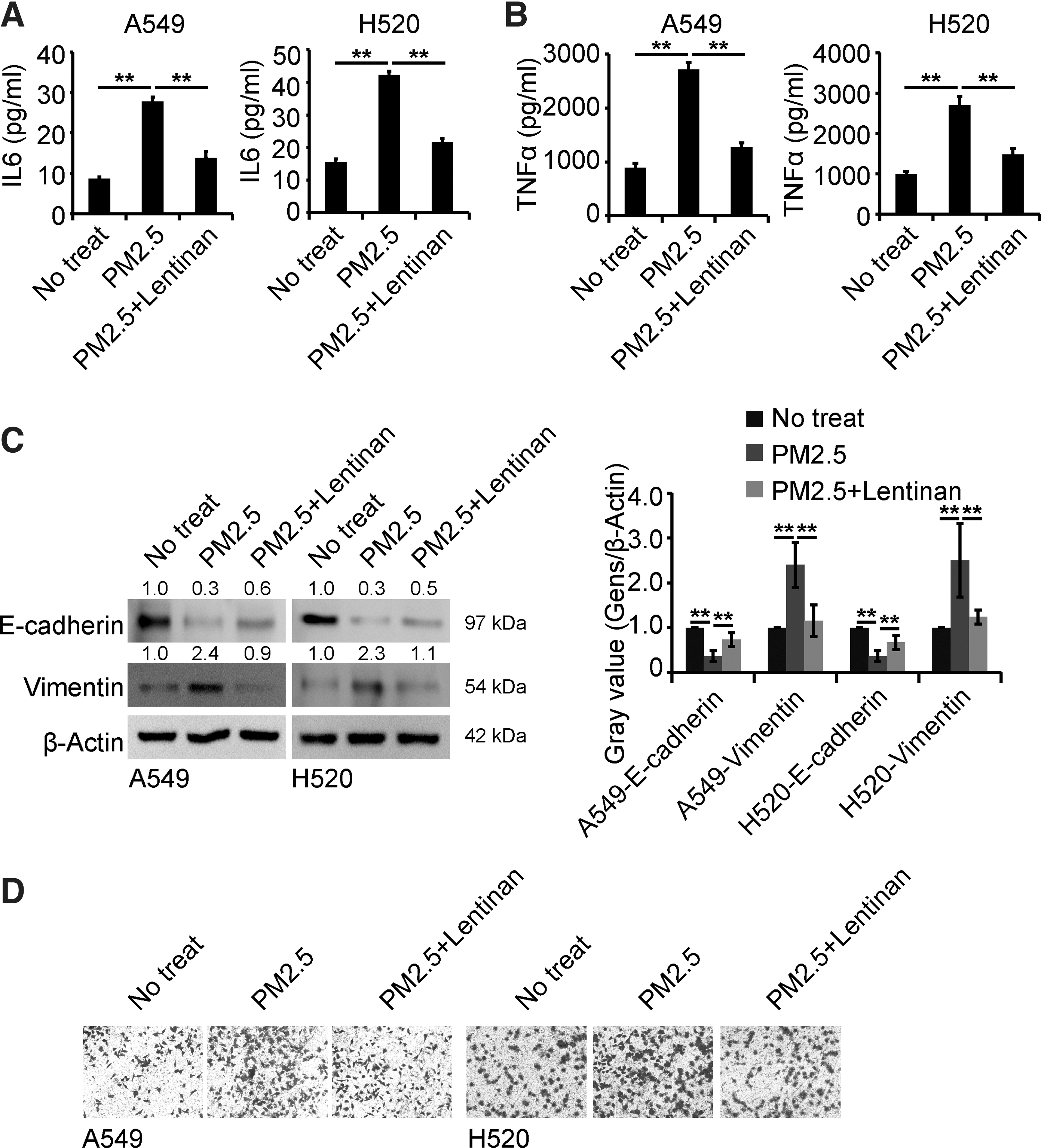
Lentinan attenuated the role of PM2.5 in lung cancer cells.
Lentinan affected the cellular function of PM2.5-exposed cells by inhibiting PVT1 expression
To investigate whether PVT1 was involved in the role of lentinan in PM2.5-exposed cells, PVT1 was measured using quantitative real-time PCR analysis. As shown in Figure 4A, lentinan inhibited the increased expression of PVT1 in PM2.5-exposed cells. As shown in Figure 4B and C, the modulation system of PVT1 expression was successfully constructed. Then, decreased PVT1 expression inhibited the production of IL6 and TNFα in PM2.5-exposed cells, and further, increased PVT1 expression inhibited the role of lentinan in PM2.5-exposed cells (Fig. 4C, D).
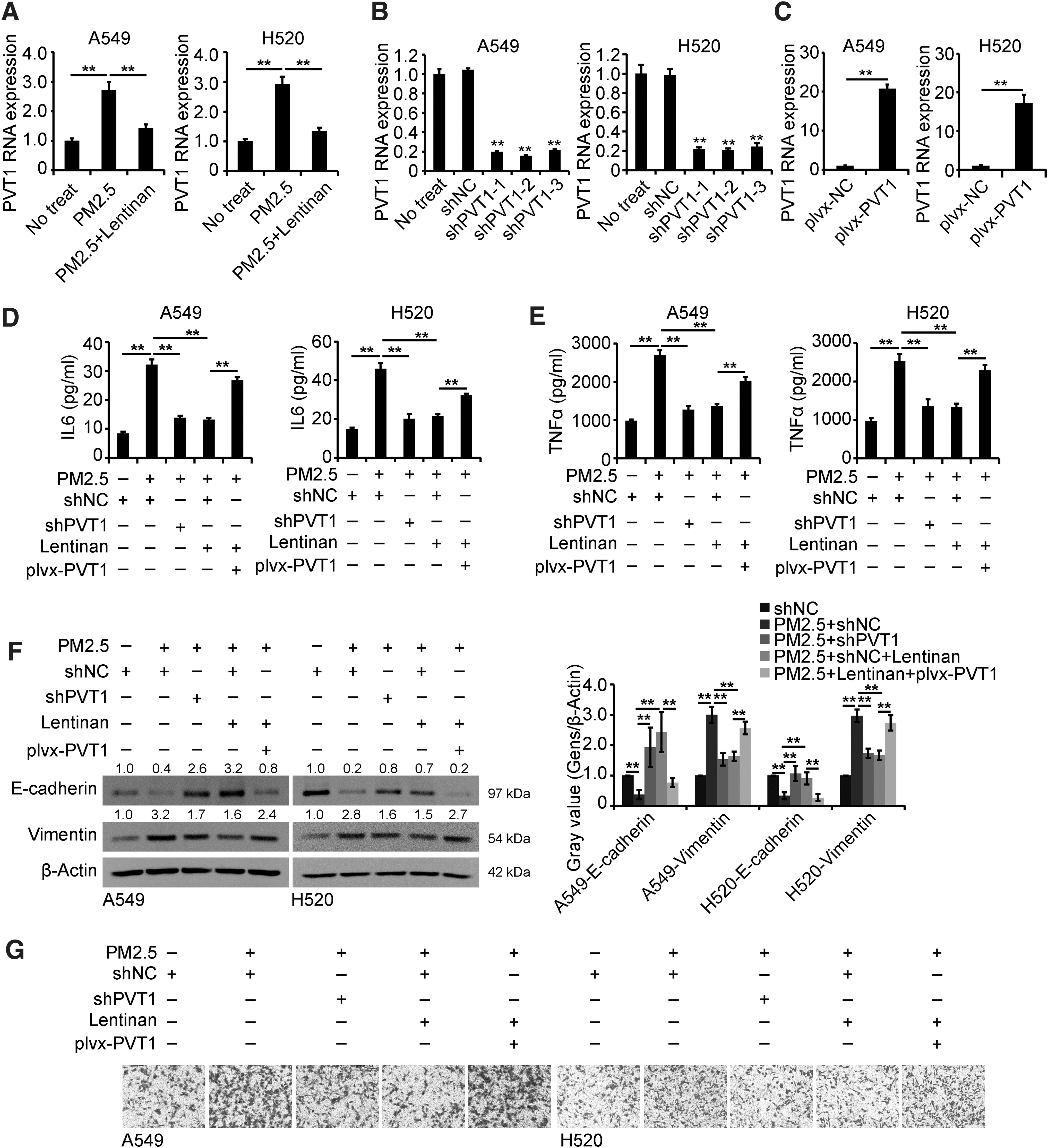
Lentinan affected the cellular function of PM2.5-exposed cells by inhibiting PVT1 expression.
According to the results of Western blots and Transwell assays, decreased PVT1 expression inhibited the EMT and the migration of PM2.5-exposed cells, and further, increased PVT1 expression inhibited the role of lentinan in PM2.5-exposed cells (Fig. 4E, F). Collectively, these data showed that decreased PVT1 mediated the inhibitory role of lentinan in PM2.5-exposed cells.
PVT1 binds miR-199a in lung cancer cells
To investigate whether the “ceRNA” hypothesis was involved in the role of PVT1, the online site “RegRNA2.0” was used to predict the potential downstream miRNA target. As shown in Figure 5A, PVT1 possesses the binding site for miR-199a-5p, and the luciferase reporter assay was used to detect the binding between PVT1 and miR-199a-5p. As shown in Figure 5B, the luciferase activity in the cells transfected with luciferase reporter plasmids inserted with PVT1 containing the binding site of miR-199a-5p (luciferase–PVT1 wt) was inhibited by miR-199a-5p, and luciferase activity was restored in groups transfected with plasmids with the mutant binding site of miR-199a-5p (luciferase–PVT1 mut). Furthermore, lentinan promoted the inhibited expression of miR-199a-5p in PM2.5-exposed cells (Fig. 5C, D).
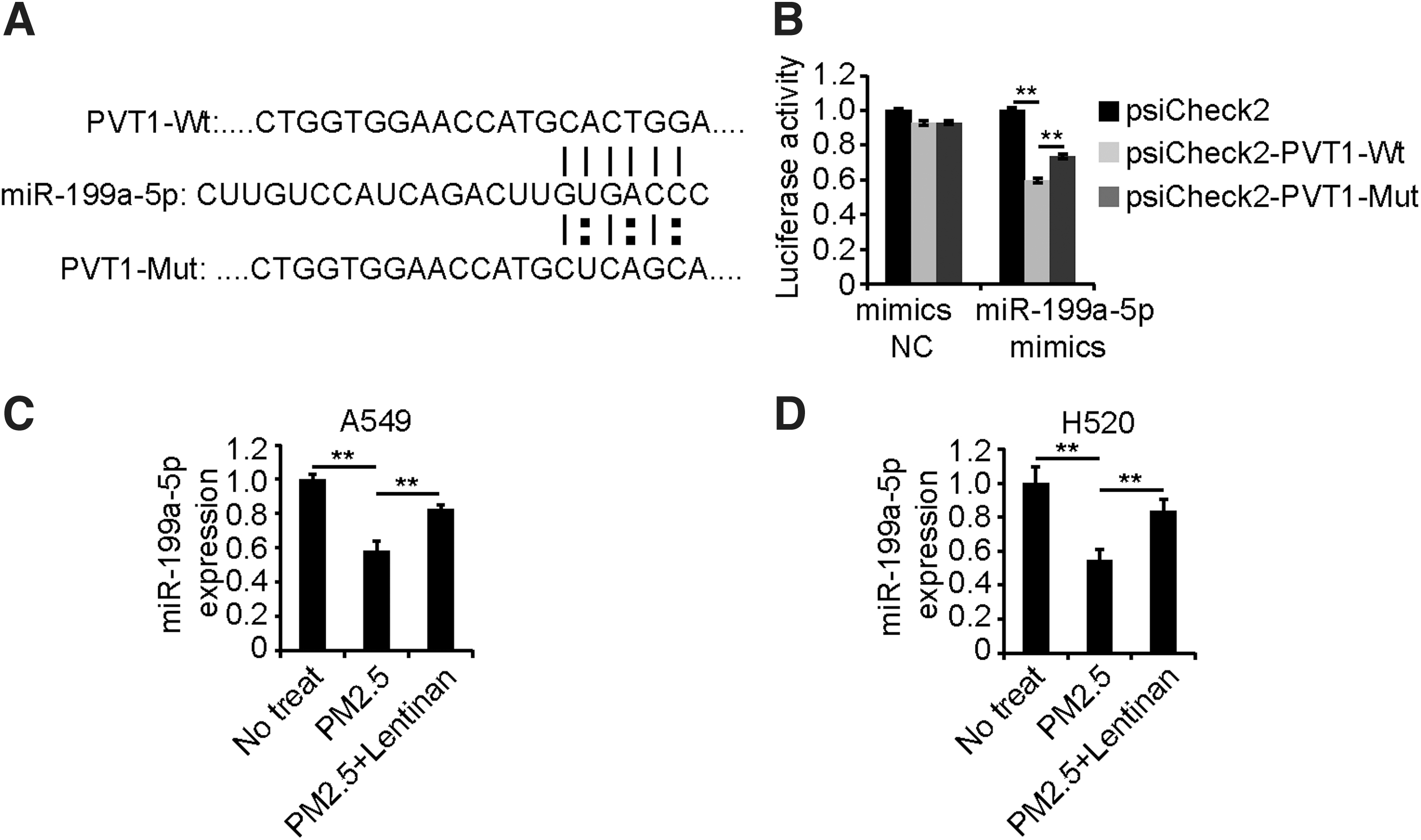
PVT1 binds miR-199a in lung cancer cells.
miR-199a inhibited caveolin1 expression in lung cancer cells
The online site “TargetScan” was used to screen the potential downstream mRNA target of miR-199a, and it was determined that caveolin1 was a potential target of miR-199a (Fig. 6A). The luciferase activity in the cells transfected with luciferase reporter plasmids inserted with caveolin1 containing the binding site of miR-199a-5p (luciferase–caveolin1 wt) was inhibited by miR-199a-5p, and the luciferase activity was restored in groups transfected with plasmids with the mutant binding site of miR-199a-5p (luciferase–caveolin1 mut) (Fig. 6B). As shown in Figure 6C and D, the miR-199a-5p mimics inhibited caveolin1 expression, whereas the miR-199a-5p inhibitor promoted caveolin1 expression in lung cells. Overall, these results indicated that miR-199a inhibited caveolin1 expression in lung cancer cells. To determine whether caveolin1 participated in the role of lentinan in PM2.5-exposed cells, the expression of caveolin1 was measured, and the results showed that lentinan attenuated the PM2.5-induced promotion of caveolin1 expression (Fig. 6E).
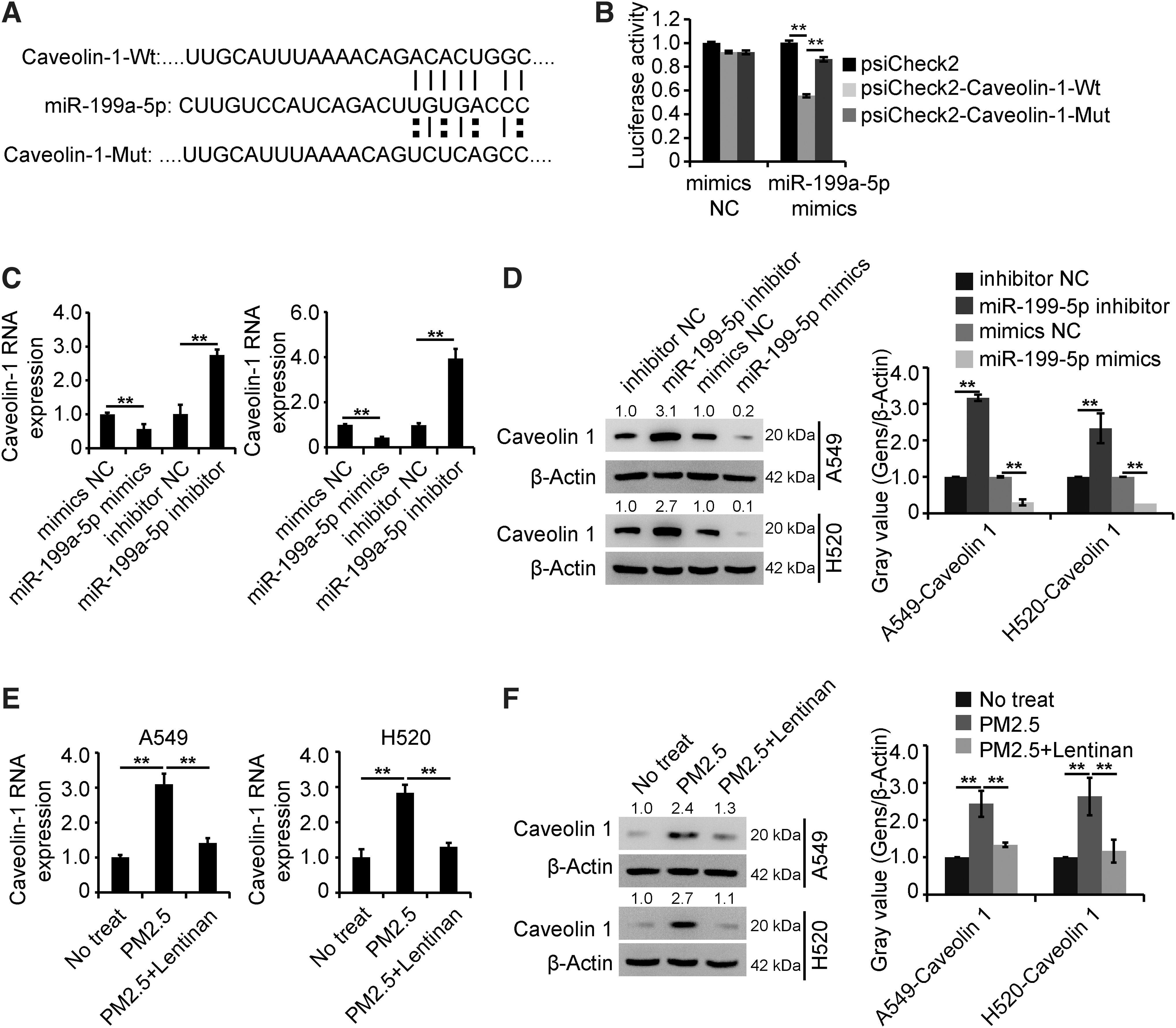
miR-199a inhibited caveolin1 expression in lung cancer cells.
Lentinan exerted its role in PM2.5-exposed cells by regulating the PVT1/miR-199a-5p/caveolin1 pathway
Then, we wondered whether PVT1 regulated caveolin1 expression by affecting miR-199a-5p in PM2.5-exposed cells. As shown in Figure 7A, silencing PVT1 inhibited the miR-199a-5p-induced upregulation of caveolin1 expression. As shown in Figure 7B and C, a silencing system of caveolin1 expression was successfully constructed. Ultimately, the results of ELISA, Western blots, and Transwell assays demonstrated that shPVT1 restored the role of the miR-199a-5p inhibitor in PM2.5-exposed cells treated with lentinan, and further, the ectopic expression of caveolin1 reversed the effect of shPVT1 (Fig. 7D–G). Overall, these results revealed that the PVT1/miR-199a-5p/caveolin1 pathway affected the role of lentinan in PM2.5-exposed cells.
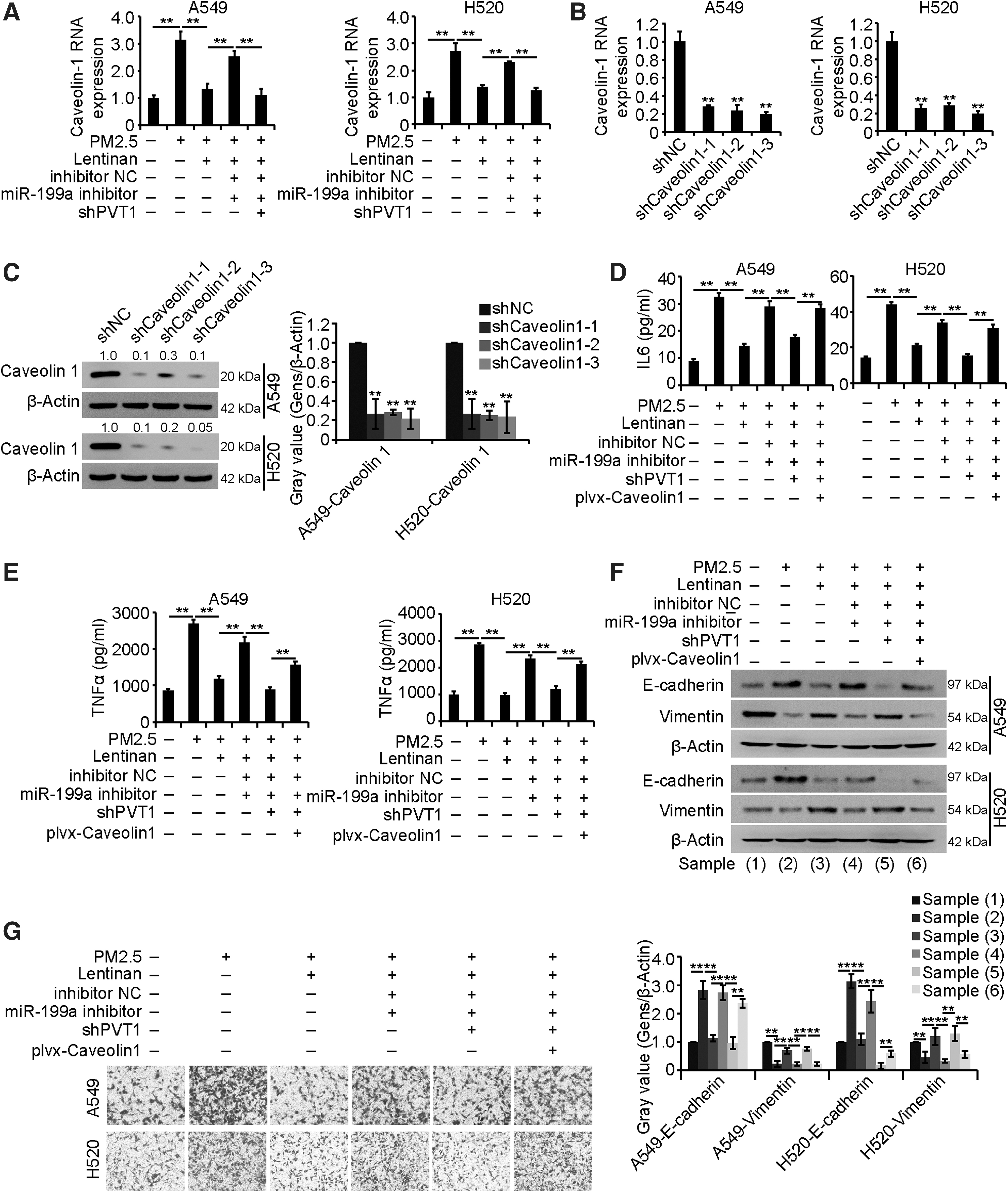
Lentinan exerted its role in PM2.5-exposed cells by regulating the PVT1/miR-199a-5p/caveolin1 pathway.
Discussion
As the main cause of cancer-related death, lung cancer is a major health problem worldwide (Spiro and Silvestri, 2005). According to cancer statistics, the incidence of lung cancer has been increasing worldwide in recent decades (Torre et al., 2016). Thus, it is urgent to further dissect the molecular mechanism of the development and progression of lung cancers.
As a serious kind of air pollution, PM2.5 consists of thousands of chemical compounds, including inorganics, organics, heavy metals, and microorganisms, some of which are well-known carcinogens (Li et al., 2018a). PM2.5 induces reactive oxygen species and exerts cytotoxic effects in many kinds of cellular processes, thus leading to a threat to health, especially pathological respiratory diseases that include chronic obstructive pulmonary disease, asthma, and bronchitis (Fan et al., 2016). However, the role of PM2.5 in cancer is complex; some of research reports a progrowth function, and studies describe an antigrowth function of PM2.5 in different cancer cells, even the same cancer cells (Xu et al., 2017). However, research has reached a consensus that PM2.5 promotes the development and progression of lung cancer. Biao Yang and colleagues revealed that PM2.5 exposure promoted the motility and proliferation of NSCLC (Yang et al., 2016). Xiaobei Deng and colleagues showed that PM2.5 exposure promoted the migration and invasion of lung cancer cells (Deng et al., 2017). Thus, research focusing on the specific mechanisms underlying the role of PM2.5 in the development and progression of lung cancer is essential. Our study revealed that PM2.5 exposure elevated the production of immune factors, EMT, and migration in lung cancer cells.
Lentinan possesses medicinal value, with antidiabetes, anti-inflammatory, and antitumor properties, and has a history of thousands of years in China (Zhang et al., 2018). Chihara and colleagues first reported the antitumor properties of lentinan (Chihara et al., 1987). First approved as a therapeutic adjuvant for gastric cancer in Japan, lentinan has since been approved to treat many kinds of cancer in China (Zhang et al., 2019). Yan-rong Liu and colleagues reported that lentinan conjugated with seleninic acid exerted antitumor effects through inhibition of cell viability, cell cycle, metastasis, and EMT in colon and melanoma cancers (Liu et al., 2018). Shengming Deng and colleagues reported that lentinan inhibited tumor angiogenesis through interferon-γ in lung cancer (Deng et al., 2018). However, there has been no research focusing on the role of lentinan in PM2.5-exposed lung cancer cells. Our study revealed an antitumor function in lung cancer cells and further reversed the PM2.5 exposure-induced promotion of the production of immune factors, EMT, and migration in lung cancer cells.
lncRNAs affect various kinds of cellular processes, such as cell growth, metastasis, EMT, and the inflammatory response, in cancers (Bhan et al., 2017). Several studies have demonstrated the involvement of lncRNAs in PM2.5-exposed cancer cells. A study reported that the PM2.5 exposure-induced lncRNA LCPAT1 promoted EMT in lung cancers (Lin et al., 2018). Another study reported that the PM2.5 exposure-induced lncRNA loc146880 promoted the migration and invasion of lung cancer cells (Deng et al., 2017). However, there is no literature focusing on the role of lncRNAs in the interaction between lentinan and PM2.5 in lung cancer cells. Our study demonstrated that the downregulation of PVT1 attenuated the PM2.5 exposure-induced alteration of cellular progress and that the upregulation of PVT1 alleviated the role of lentinan in PM2.5-exposed lung cancer cells.
Several underlying mechanisms of the role of lncRNAs have been elucidated (Marchese et al., 2017). Currently, the “ceRNA” hypothesis draws intensive attention from scientists (Salmena et al., 2011). The “ceRNA” hypothesis of lncRNAs is defined as lncRNAs absorbing miRNAs, which are negative regulatory factors of mRNA and thus restore the expression of mRNAs. Hence, the “ceRNA” hypothesis of lncRNAs systematically integrates upstream regulatory genes (lncRNAs) and downstream regulatory genes (mRNAs) of miRNAs through miRNA binding sites, thereby forming a regulatory network of lncRNA–miRNA–mRNA (Tay et al., 2014). This study revealed that PVT1 promoted caveolin1 expression by absorbing miR-199a, which is a negative regulatory factor of caveolin1, and thus affected lentinan's role in PM2.5 exposure in lung cancer cells.
Unfortunately, this research still has many shortcomings. More experiments are still needed to confirm the function of lentinan in PM2.5 exposure lung cancer, such as constructing an orthotopic lung cancer model and then detecting the effect of lentinan on lung cancer metastasis caused by PM2.5 exposure. Clinical samples of lung cancer were collected to detect the expression profile of the PVT1/miR-199a/caveolin1 regulatory pathway in clinical samples of lung cancer to provide a new theoretical basis for the clinical diagnosis and treatment of lung cancer.
Conclusion
This study displayed that lentinan exerted inhibitory role in PM2.5-exposed lung cancer cells via modulating the PVT1/miR-199a/caveolin-1 signaling pathway and thus, providing a theoretical basis for the application of lentinan in treatment of lung cancer.
Footnotes
Authors' Contributions
C.X. and H.Q. participated in the design of this study, and they both performed the statistical analysis. H.Q. carried out the study and collected important background information. Y.L. and N.W. collected important background information. H.Q. and C.X. drafted the article. All authors read and approved the final article.
Disclosure Statement
No competing financial interests exist.
Funding Information
This study was supported by the project of Liaoning Provincial Department of science and technology (Grant No. 2017225076).
