Abstract
Automated Insulin Delivery (AID) is in a new era of implementation challenges and opportunities for people with diabetes (PWD) and healthcare providers (HCPs). Beyond technologic variation, cost, access, and HCP endorsement/experience lead to uneven uptake of AID technologies, and attenuate universal ease of use. For AID to be broadly implemented, we must prioritize the lived experience for PWD, and consider how to alleviate burden to promote holistic wellbeing. Expectations and education help HCPs and PWD navigate the similarities and differences between AID devices, and help find common ties: users need to give the system time to work, learn to trust it, and not try to “trick” the system. Despite these learnings, disparities in uptake exist, both in clinical trials and in routine clinical care. Strategies to proactively address AID disparities are needed at multiple levels, all of which increase in importance as AID becomes more common for people with type 2 diabetes. Further, broader implementation will require comprehensive healthcare system integration efforts, including new data solutions. Overall, the success of AID requires ongoing transformation of clinical paradigms, with lockstep alignment between PWD and their families, healthcare professionals, researchers, funders, policy makers, and industry partners.
The best automated insulin delivery (AID) system is the one that an individual chooses and can use day after day. While simple in principle, the implementation challenges are complex. Different AID systems provide unique challenges for different people. Beyond physiological and technologic variation, cost, access, and health care provider (HCP) endorsement/experience lead to an uneven uptake of AID technologies, and attenuate universal ease of use.
Research supported by the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK)/National Institutes of Health (NIH) and other funders has been key to developing many currently available, transformational technologies for treatment of diabetes and this article belongs to a collection celebrating NIDDK’s 75th Anniversary. Forums such as the Fifth Artificial Pancreas workshop hosted by the NIDDK/JDRF/Helmsley Charitable Trust in May 2023 have facilitated these collaborative advancements, while additionally promoting forward facing conversations essential to the next era of AID. 1 Equally important to the technological revolution are the considerations that impact the broader ecosystem of diabetes care. How do we get AID technology into the hands of everyone with diabetes? How do we prepare them for success with realistic expectations? This chapter explores the factors necessary to advance AID at the individual, health care, institutional, and payer/systems level.
Human–Machine Interaction and the Lived Experience with AID
AID technology has necessarily evolved with consideration to how people with diabetes (PWD) and HCPs will use the device. The ideal AID system would not require input from the user apart from placement and removal. As we work toward this goal, broader concepts of how humans interact with machines are adapted for the AID paradigm (Fig. 1). 2 Humans are active participants in AID system functions, with the user responsible for perceiving diabetes and device state and determining if action is needed. The AID system is responsible for the intake of information from various sources (including the PWD) and responding accordingly. Between the AID system and the PWD lies the “interface zone.”
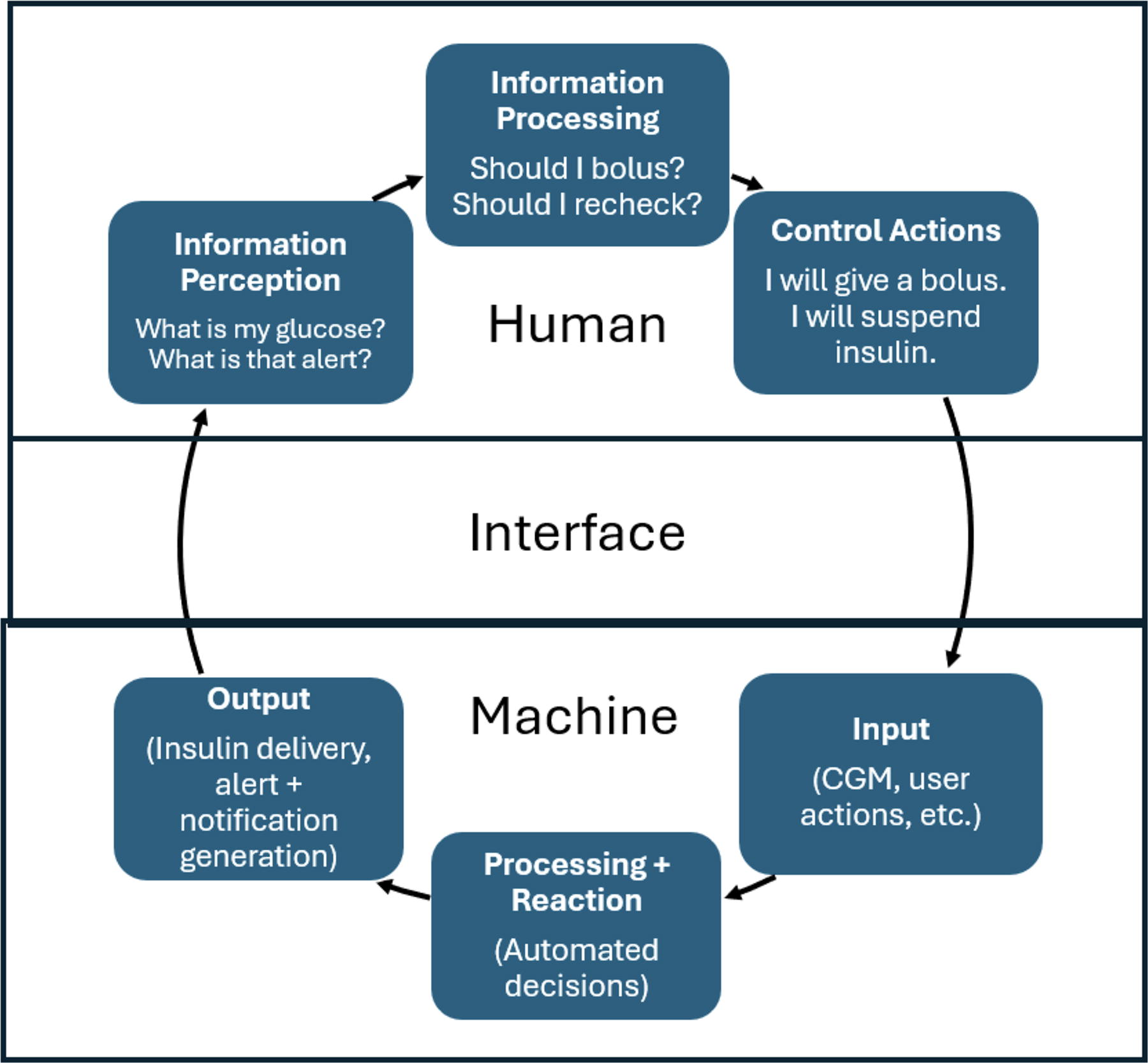
Human–machine interaction model for AID (adapted from Redmill et al 2 ). AID, automated insulin delivery.
Professional disciplines have tackled different aspects of the human–machine interaction, with important work continuing in the “interface zone.” For example, human factors’ work helps determine ways to maximize safety and usability of AID systems, evaluating workflows, device interface, and comprehensive aspects of how the user interacts with the system. 2 Similarly, an entire body of research has been focused on the holistic user experience, which extends beyond the literal human–machine interface. 3 –7 This allows the ability to overlay modern terminology and new concepts into human–machine interaction paradigms; for example, form-factor, cost–benefit trade-off, user burden, and problem-solving, when issues arise, and the potential for de-skilling of basic diabetes management.
For AID to be broadly implemented, we must consider the physical, psychological, and social well-being of its users, and ideally alleviate burden in each of these areas. However, in reality, trade-offs are required to achieve an acceptable balance of burden and benefit. In addition to the large body of literature showing glycemic improvement with AID, there is considerable evidence that AID systems are associated with improved quality of life (QoL) for many users with regard to safety, reduced anxiety around diabetes self-management and hypoglycemia, improved sleep, and lower diabetes distress. 8,9 Challenges persist, however, intrinsic to the human–machine interaction. For example, AID systems increase the visibility of diabetes, which has been reported as a barrier to device uptake in both adults and adolescents with diabetes, and intrusive alarms in social situations can cause embarrassment, distress, and serve as a nuisance. 4,7
Ongoing research with AID must prioritize the lived experience for PWD. This will continue to be assessed and addressed with a broad range of methodologies. Quantitative, survey-based objective measurements offer broad signals for exploration, while subjective measurements, such as interviews, focus groups, and therapeutic writing offer more nuanced exploration of individual experiences with AID. By utilizing these mixed methodologies, it is possible to gather a deeper understanding of important aspects that influence continuation or discontinuation of systems, satisfaction or dissatisfaction, and impact on QoL and functional health status.
Wide-ranging domains of inquiry address different aspects of health status and QoL, including indicators of physical/functional health status, psychological well-being, and social well-being as overarching categories. 3,10 Within these, there are subcategories that affect individuals to a greater or lesser extent depending on their personal circumstances, including depression and anxiety, diabetes distress, fear of hypoglycemia, treatment satisfaction, sleep quality/quantity, self-efficacy, sexual/reproductive health, and the impact of devices and technologies on what it is to be human. Overall, cross-disciplinary research must continue to discover optimal ways to enhance the lived experience of PWD using AID systems.
Expectations and education for AID systems
“It knows you eat pizza for breakfast, and will dose accordingly”—said the endocrinologist, circa 2017, when the first commercial hybrid closed-loop system became available in the United States. At the time, it was unclear how true or untrue this was. The relationship between PWD and diabetes technology starts with their expectations of the system.
Right-sized technology expectations began with continuous glucose monitoring (CGM), which fundamentally shifted the understanding of glycemic excursions. Where people previously interpreted 4–8 glucose readings each day, suddenly 288 data points were available and ready for consumption. PWD needed to learn to live with CGM, to recognize the benefits and the drawbacks, and to avoid becoming overwhelmed by data. 11,12 Early guidance on insulin adjustments based on real-time information, such as trend arrows, and retrospective data analysis grew from the NIH–funded Diabetes Research in Children Network (DirecNet), 13,14 and JDRF CGM trials, 15 as well as early clinical practice, 16 –18 ultimately culminating in an international guideline. 19 CGM education set the stage for future success with AID.
Once CGM transformed the landscape of diabetes, AID changed how to traverse it. AID required new thinking at the ecological scale: regulatory, payer, endocrinology practice, clinic-level, and individual/family engagement—all to accept CGM-informed insulin delivery with reduced user-intervention. With every new AID system, the diabetes community would ride the familiar “hype cycle” of inflated expectations, discouragement, and plateau of reality. 20 Trust became a core requirement for both PWD and clinicians, and was gained in a stepwise manner, applying first to predictive low-glucose suspend, and thereafter to hybrid closed-loop systems. Research showed that trust was context-dependent (e.g., easier at night compared with postprandial), impacted by previous experience with manufacturers, and reliant on perceptions of accuracy and safety. 21 The reality is remarkably consistent across devices: users need to give the system time to work, learn to trust it, and don’t try to “trick” the system. Often PWD must pace their intervention rather than be overly aggressive with insulin dosing while waiting for the system to respond.
While expectations and trust capitalize on similarities between systems, education deciphers the differences. The CARES framework evolved out of NIDDK-funded AID research (Table 1), lending structure to understanding how systems Calculate insulin delivery, settings that are Adjustable, when to Revert to open-loop, necessary Education, and Sensor/Share characteristics. 22,23 Clinicians require knowledge of which “levers to pull” for optimal treatment outcomes in different systems 24 –26 and how to help PWD choose the best system for them. As AID evolves, differences in algorithm specifications may become less relevant, although pillars of structured education for PWD and clinicians will remain paramount, including education around insulin pharmacodynamics.
CARES Paradigm for Differentiating Automated Insulin Delivery Systems
Practical performance will inevitably continue to chase user and clinician expectations, and education will remain critical to achieving optimal glycemic and user-reported outcomes. While current systems may not know whether a person will eat pizza for breakfast, future systems will better handle the challenge when they do.
Technology Use Disparities and Psychosocial Determinants of Health
There has been growing awareness of disparities in AID use, both in the development of the technology and in the integration into routine diabetes management. In clinical practice, disparities are evident within the first year following diagnosis of type 1 diabetes. 27 Racial and ethnic minority groups have been significantly underrepresented in the large-scale clinical trials that led to FDA approval/clearance of AID systems. 28,29 This lack of representation in clinical trials leads to device development that does not consider the needs and preferences of minority groups. It also precludes identification of important barriers to AID use that can inform education for PWD and support programs for onboarding, and subsequently impacting overall outcomes. 30,31 The American Diabetes Association (ADA) is acutely aware of these disparities, calling explicit attention to AID as an agent of equalization in the 2025 Standards of Care in Diabetes, recommending “AID systems should be the preferred insulin delivery method to improve glycemic outcomes and reduce hypoglycemia and disparities in youth and adults with type 1 diabetes.” 32
Two recent large-scale studies confirm the major disparities in the use of AID across racial/ethnic groups (Table 2), with the lowest use found in non-Hispanic Black populations. 33,34 In a review of >48,000 medical records from 15 adult and pediatric type 1 diabetes (T1D) centers, Ebekozien et al. found trends of increased AID use and lower A1c levels over past few years, but that these gains were not equivalent across race/ethnic groups. A second review of over 5000 records found similar results; however, disparities in AID use were even greater in Black children compared with Black adults. 33 These racial/ethnic disparities persist even after controlling for other contributors to health inequities such as type of insurance (e.g., public vs. private). This indicates that it is important to investigate additional contributors, such as implicit provider bias 35 and the technology divide, 36 among other factors.
Percent of Automated Insulin Delivery Use Across Race/Ethnic Groups
It is critical to address disparities because of their impact on medical outcomes. While the incidence rate of T1D is increasing, this rise is occurring more rapidly among racial/ethnic minority groups compared with non-Hispanic Whites. 37 Furthermore, as AID use proves to benefit people with type 2 diabetes (T2D), the impact of these disparities will increase exponentially since, in the United States, more than 80% of individuals with T2D belong to a minority group. 38 In addition to racial and ethnic disparities, gaps in AID access and use across insurance types and socioeconomic (SES) levels are yet to receive the warranted scientific attention, although early research is documenting significantly less technology use and suboptimal glycemic outcomes in lower SES groups. 39
Strategies to address AID disparities must be enacted at multiple levels. HCPs as individuals are responsible for ensuring all PWD are offered AID as standard of care, identifying and reducing their own biases in prescribing AID systems, and promoting shared decision making with PWD. 40 Clinic-based strategies need to include promotion of benchmarking to reduce the impact of bias, 41,42 quality improvement methods, 43 –45 and screening for and addressing social determinants of health. 46,47 Insurance barriers must be addressed; transformational policy changes for AID access will require robust engagement of all relevant stakeholders. 48,49 Future studies should test these and other strategies in rigorous clinical trials specifically designed to improve AID equity.
Implementation Challenges and Health System Transformation
It is challenging for HCPs to keep pace with the technological advances of AID: knowing how to recommend AID systems, navigating insurance coverage, ordering and onboarding sufficiently, and then competently working with multiple systems. There have been efforts over the past decade to identify needs and help equip HCPs with the resources needed to understand current systems and provide education and support to PWD. 50,51 DiabetesWise (https://diabeteswise.org/) is a free online resource funded by the Helmsley Charitable Trust, which provides educational vignettes from PWD and their care partners as well as training resources for PWD and HCPs. 52 The Panther Program (https://www.pantherprogram.org/) offers device-specific overviews, comparison charts, and clinical care worksheets to support HCPs in prescribing and using new technologies. The educational model for this program follows the CARES framework, providing HCPs with concise information on how each device operates. 22,53 While these efforts aim to provide unbiased access to device specifics, ongoing work needs to educate, re-educate with system updates, and support the HCP community as technology transforms.
Broader adoption of AID systems has brought to light the need for new data solutions at the clinic and hospital level. Current CGM and AID systems record a large volume of glucose and insulin dosing data. However, questions regarding ownership, governance, availability, and standardization of access abound. 54 Data are provided through over half a dozen online platforms, each requiring different account management and some requiring subscription plans by the health system. This leads to challenges scaling any data paradigm beyond individual clinics, let alone health care systems and networks. Accessible data vary by manufacturer, ranging from only summary statistics for some and minute-by-minute presentation of algorithm actions for others. HCPs and PWD need to download and review these data to adjust AID system settings or suggest behavioral modifications to optimize glycemia.
Several projects have aimed to streamline these data processes and allow for direct data transfer from a commercial industry partner into a clinical electronic health record (EHR). 55,56 These efforts so far have been limited, allowing for transfer from one commercial entity to one hospital system’s EHR without real-time access. PWD should be afforded access to their own data and permit others, including HCPs, to have unbridled access. In the United States, the 21st Century Cures Act mandates laboratory results be released to individuals through an EHR, unless certain circumstances exist. 57 Thus, precedence exists regarding release of data through EHR, which provides intriguing opportunities when CGM and AID data are more seamlessly integrated. Eliminating these barriers may prompt more HCPs and innovators to embrace diabetes technologies, thereby improving care and autonomy for PWD. Broad availability of data does raise additional considerations, however, including ensuring adequate reimbursement by payers for HCP time required to review AID data, and avoiding unrealistic expectations about how often this can occur.
Within the realm of in-hospital care, there is a need for both real-time device integration and updated ordering around dynamic insulin dosing. At some institutions, PWD admitted to the hospital for diabetes- or nondiabetes-based reasons may continue to use their CGM and AID system, whereas in others, AID and even CGM may not be allowed “by policy.” To permit more permanent adoption, optimization of workflows will require both real-time CGM-based alerts and EHR entry of AID system-based insulin dosing, which is currently far outside the realm of conventional hospital ordering and dosing. Updating communication between the devices and the hospital systems is a necessary step to further integrate AID within hospital-based care. The time is now to ensure present-day tools are leveraged in all necessary care settings and creating the framework needed to achieve these goals remains a critical area of investigation.
Conclusions
While we have come a long way with diabetes technology advances, taking stock of where we are today helps us move forward for the future. The success of AID will be measured not just by time-in-glycemic-target ranges, but in worldwide access, use, and equity. It is only possible to change clinical paradigms with lockstep alignment between PWD and their families, health care professionals, researchers, funders, policy makers, and industry partners.
Footnotes
Acknowledgment
The author group would like to thank the NIDDK for 75 years of supporting research to improve the lives of people living with diabetes.
Authors’ Contributions
L.H.M.: Conceptualization, investigation, writing (original, review and editing), and project administration. G.P.F.: Conceptualization, investigation, and writing (original, review and editing). L.G.-F.: Conceptualization, investigation, and writing (original, review and editing). K.H.: Conceptualization, investigation, and writing (original, review and editing). O.E.: Investigation and writing (original, review and editing). K.B.-K.: Investigation and writing (original, review and editing). L.M.L.: Investigation and writing (original, review and editing). J.L.S.: Investigation and writing (original, review and editing). R.L.: Investigation and writing (original, review and editing). S.A.W.: Investigation and writing (original, review and editing).
Author Disclosure Statement
L.H.M. is an employee and shareholder of Tandem Diabetes Care.
G.P.F. conducts research sponsored by Medtronic, Dexcom, Abbott, Tandem, Insulet, and Beta Bionics and has been a speaker/consultant/ad board member for Medtronic, Dexcom, Abbott, Tandem, Insulet, Beta Bionics, Lilly, and Sequel.
L.G.-F. has received consulting fees/research support from Dexcom, Abbott Diabetes Care, Johnson and Johnson, Merck, and AstraZeneca, and is a partner in HFS-Global LLC, which in partnership with the University of Virginia licenses use of the Hypoglycemia Fear Survey for clinical trials conducted by for-profit pharmaceutical and medical device corporations, with part of these funds supporting ongoing research and education on the problem of hypoglycemia.
K.H. has received consulting fees from Sanofi, MannKind, and Havas Health; and a research grant from Embecta.
O.E. has received research grants through his organization T1D Exchange from Medtronic, Lexicon, Vertex, Lilly, and Sanofi.
K.B.-K. is founder and CEO of Spotlight-AQ Ltd; speaker honoraria from Roche, Tandem, Novo Nordisk, and Sanofi. Research funding from Dexcom, Breakthrough T1D NIH, and Roche.
L.M.L. serves as a consultant or on advisory boards for Boehringer Ingelheim, Medtronic, Tandem Diabetes, Dexcom, Arbor Biotech, Sanofi, Sequel, MannKind, and Vertex.
J.L.S. has conducted clinical trials for Abbott Diabetes Care, Dexcom, JDRF/Breakthrough T1D, Insulet, Medtronic, and Provention Bio and has received in-kind support for research studies from Dexcom and Medtronic. She has consulted for Abbott Diabetes, Insulet, Medscape, Medtronic, Vertex, and Ypsomed. She has been a member of advisory boards for Cecelia Health, Insulet, MannKind, Medtronic Diabetes, StartUp Health T1D Moonshot, and Vertex.
R.L. has received consulting fees from Abbott Diabetes Care, Adaptyx Biosciences, Biolinq, Capillary Biomedical, Deep Valley Labs, Gluroo, PhysioLogic Devices, Portal Insulin, Sanofi, and Tidepool. He has served on advisory boards for Provention Bio and Lilly. He receives research support from his institution from Insulet, Medtronic, Sinocare, and Tandem.
S.A.W. has conducted clinical trials for Abbott Diabetes, JDRF/Breakthrough T1D, Medtronic, Tandem; has received honoraria for consulting or speaking engagements from Abbott, Dexcom, Insulet, Medtronic, and Tandem; and has served on advisory boards for Zealand. He currently receives research support (to his institution) from Abbott Diabetes.
Funding Information
There was no funding provided for this article.
