Abstract
Introduction:
The Diabetes Control and Complications Trial (DCCT) clearly documented long-term beneficial effects on both micro- and macrovascular complications associated with type 1 diabetes (T1D) by using intensive insulin therapy (IIT) via multiple daily injections or insulin pumps more than 30 years ago. IIT, both during the DCCT and with translation into clinical practice, has been demonstrated to increase the risk of severe hypoglycemia and weight gain. Automated insulin delivery (AID) systems have become the standard of care in T1D management in the developed countries.
Materials and Methods:
We reviewed the registration and real-life studies for different AID systems reported to date. Many of the registration studies were sponsored by the National Institute of Diabetes and Digestive and Kidney Diseases. A systematic literature search was conducted using the MEDLINE (PubMed) database. Studies with the longest duration and/or with the largest number of participants were included.
Results:
In the last decade, the introduction of many AID systems for patients with T1D has shown improvements in glycemic metrics as documented by hemoglobin A1c values, time in range, time below range, and quality of life. Most of the registration and real-life studies have shown safe and effective use of AID systems for all age groups living with T1D.
Conclusions:
In this review, we summarize the registration and real-life studies of U.S. Food and Drug Administration-approved AID systems. Real-life studies confirmed the glycemic outcomes of AID systems reported from registration studies.
Introduction
More than 40 years ago, management of type 1 diabetes (T1D) usually consisted of 2–3 insulin injections administered subcutaneously daily. 1 Importantly, targets for treatment were debated with questions of whether achievement of lower glucose levels, based on a hemoglobin A1c (HbA1c), would be advantageous for people with diabetes. The Diabetes Control and Complications Trial (DCCT), sponsored by the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK), concluded in 1993 and revolutionized the care of individuals with diabetes. The DCCT clearly showed that intensive insulin therapy (IIT) using multiple daily injections (MDI) or insulin delivery via continuous subcutaneous insulin delivery (CSII; insulin pump) along with frequent self-monitoring of blood glucose (SMBG) improves glycemic control as measured by HbA1c. 2 In addition, the group randomized to IIT had significant reductions in long-term microvascular complications, including diabetic kidney disease (DKD), diabetic retinopathy, and diabetic peripheral neuropathy, which are all commonly associated with T1D. Indeed, the results were so compelling that the study was discontinued early because it would be unethical to continue the current standard of care. 2,3 At the conclusion of the DCCT in 1993, the tools used to manage diabetes were rather rudimentary compared with today’s methods. At that time, insulin pumps had fewer features, and continuous glucose monitors (CGMs) to adjust insulin dosing were not available.
In people without diabetes, the beta-cells of the pancreas release insulin continuously (as well as in response to rising blood glucose levels) in a pulsatile form directly to the liver through portal circulation. For delivery of exogenous insulin, it is not possible to deliver this insulin directly to the liver, thus it is typically infused subcutaneously via MDI or CSII. To better mimic the physiological delivery of insulin, 40 years ago, MiniMed, Inc. (Northridge, CA) introduced the first wearable small insulin pump, MiniMed 502, in 1983. 4 From the inception of this device, Alfred Mann (1925–2016), then CEO of MiniMed, had a vision of creating an artificial pancreas (AP) system. 5
Nearly a quarter of a century ago, CGMs were approved by the Food and Drug Administration (FDA) as adjunctive to SMBG, however, their accuracy was suboptimal (mean absolute relative difference; MARD > 15%–20%). 6,7 Over the next 10 years, CGMs became more accurate, smaller and stand-alone. 6 Eventually they were deemed by regulatory agencies to be safely used nonadjunctively. During a similar time frame, multiple pump companies (including Smith’s Medical®, BD®, Eli Lilly®, Roche®, Animas®, Beta Bionics®, Disetronic®, Insulet®, MiniMed®, Sequel®, SOOIL®, Tandem®, and Ypsomed®) improved their insulin delivery devices. 8,9 At the same time, most of them developed or licensed different algorithms to automatically activate or halt insulin delivery based on sensor glucose values. 5,10 Multiple studies were conducted to demonstrate the safety of sensor-augmented pumps (SAPs) that could suspend insulin delivery (low-glucose suspend; LGS, threshold suspend; TS) after hypoglycemia, or in advance of predicted hypoglycemia (predictive LGS/TS systems). 11 –15 These advances were followed by studies that included automatic delivery/stoppage of insulin based on sensor glucose concentrations in the clinic/research settings. The first automated insulin delivery (AID) system (Medtronic 670G) was approved by the FDA in 2016. 16 Since then, many more AID systems have been approved by the FDA and/or the European Medicine Agency, including Medtronic 780G, Tandem Control-IQ, Omnipod 5, iLet Bionic Pancreas, do-it-yourself loop, and CamAPS FX. In this review, we focus on registration and real-life studies on these approved AID systems.
Methods
We conducted a systematic literature search using the MEDLINE (PubMed) database using keywords of FDA-approved AID, hybrid closed-loop (HCL) systems, and T1D. We only reported the studies with the longest duration and/or highest participant numbers. Randomized controlled trials (RCTs) were prioritized over single-arm studies, however, if we found no RCTs, then single-arm or crossover trials were included. Tables 1 and 2 show the summary of registration and real-life studies of AID systems, respectively. Figure 1 illustrates each AID system with its own registration and real-life study results, where available.
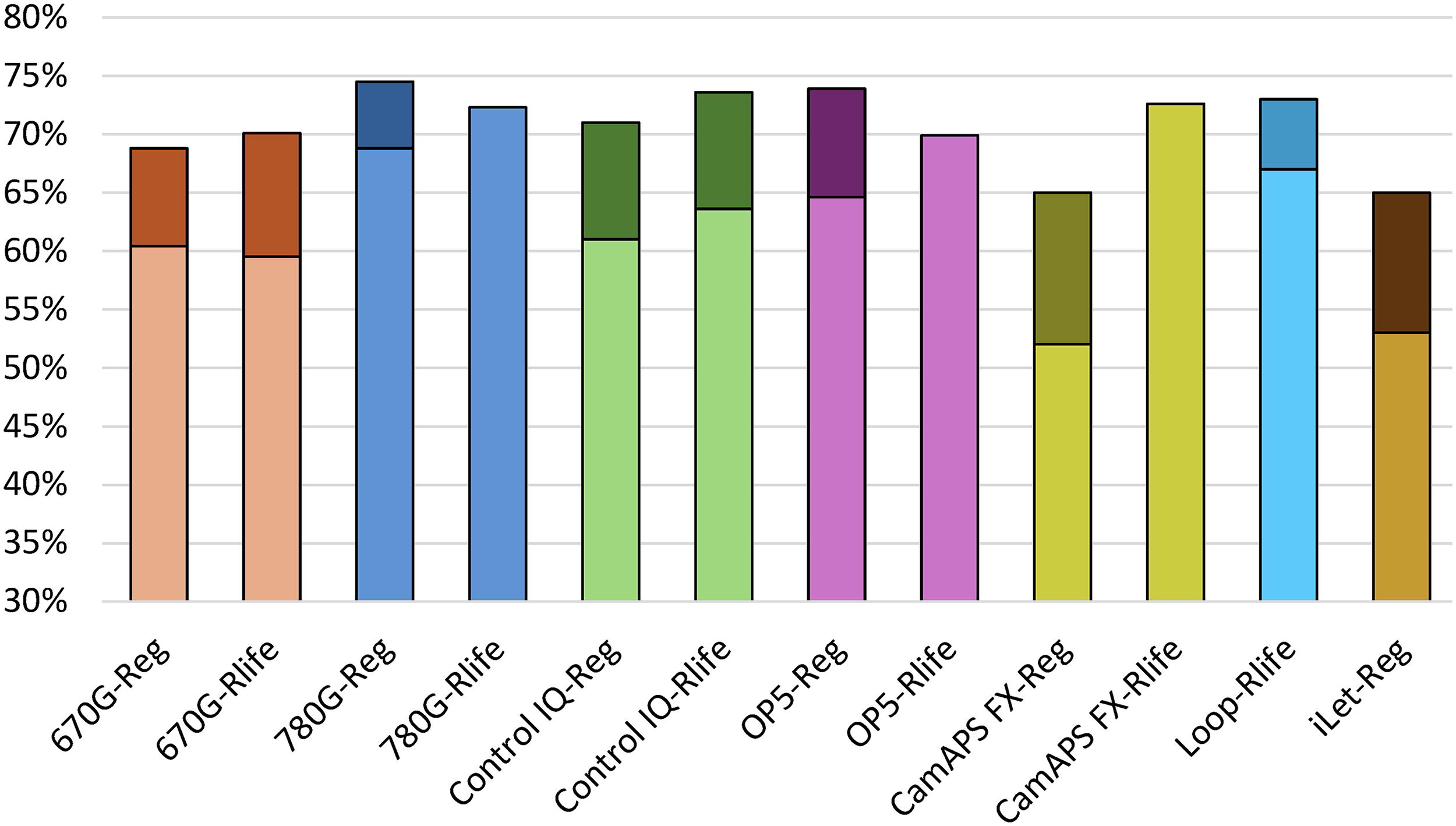
Time in range (70–180 mg/dL) automated insulin delivery (AID) system from registration and real-life studies in people with type 1 diabetes*. Reg; Registration Study, Rlife; Real-life Study. * This is not intended to be a direct comparison between systems (and is for illustration purposes only). Change from baseline time in range was shown in darker colors for each system. In some real-life studies, change from baseline was not reported. The AID system, population, and age range were different in the studies that were included. Loop only had a real-life study and iLet only had a registration study.
Registration Studies for Food and Drug Administration-Approved Automated Insulin Delivery Systems a
This is not for a direct comparison among systems. Automated insulin delivery (AID) system, diabetes type (type 1 or 2), study type (single-arm, randomized controlled trial), study inclusion criteria, baseline hemoglobin A1c (HbA1c), population, and age range differed in the studies included. Extension studies are included.
CGM, continuous glucose monitor; CSII, continuous subcutaneous insulin delivery; MDI, multiple daily injections; RCT, randomized controlled trial; SAP, sensor-augmented pump.
Real-Life Studies for Food and Drug Administration-Approved Automated Insulin Delivery Systems a
This is not for direct comparison among systems. AID system, baseline HbA1c, methodology, population, and age range differed in the studies included.
GMI, glucose management indicator; PLGS, Predictive low glucose suspend.
Medtronic MiniMed 670G/780G
MiniMed 670G, the first AID system approved by the U.S. FDA, was evaluated in 124 adolescents and adults (14–75 years old) with T1D in a multicenter single-arm pivotal study. 17,39 The primary aim of the study was to evaluate the safety of the HCL system in patients with T1D. The 670G system was used during a 2-week run-in phase without HCL or auto mode (control period). 39 Auto mode was enabled during a 3-month study phase. 39 From baseline run-in to the end of the study phase at 3 months, HbA1c levels decreased significantly from 7.7% ± 0.8% to 7.1% ± 0.6% and from 7.3% ± 0.9% to 6.8% ± 0.6% (P < 0.001) in adolescents and adults, respectively. 39 Time in range (70–180 mg/dL) increased from 60.4% ± 10.9% to 67.2% ± 8.2% in adolescents and from 68.8% ± 11.9% to 73.8% ± 8.4% in adults (P < 0.001). 39 In another study, MiniMed 670G was compared with CSII in a 6-month RCT in 151 children, adolescents, and adults (2–80 years old) with T1D. 40 Participants were grouped according to their baseline HbA1c levels. For participants with high baseline HbA1c (>8%), there was a significant mean (95% confidence interval [CI]) difference in HbA1c change in the HCL group (−0.8% [−1.1% to −0.4%], P < 0.0001). 40 For individuals with lower baseline HbA1c values (<8%), there was also a significant decrease in HbA1c of −0.3% [−0.5% to −0.1%] favoring the HCL group (P < 0.0001). 40 The TIR difference was 12% [8.8% to 15.1%] in favor of 670G system. 40 Time below range (<70 mg/dL) (TBR) decreased in both high HbA1c (−2.2% [−3.6% to −0.9%]) and low HbA1c groups (−4.9% [−6.3% to −3.6%]; P < 0.0001 for both). 40
In a real-life study with MiniMed 670G involving 127 adults with T1D (21–68 years old), TIR increased from 59.5% ± 1.1% to 70.2% ± 1.2% and 70.1% ± 1.1% at 3 and 6 months, respectively (P < 0.001). 32 Several patients discontinued using 670G due to the need for many calibrations and multiple alarms, especially at night. 41
MiniMed 780G is commonly referred to as the advanced HCL system (AHCL) that gives auto-correction microboluses up to every 5 min when the maximum basal rate is reached and sensor glucose is >120 mg/dL. 9 780G system was evaluated in a 3-month single-arm pivotal trial with 157 patients with T1D (14–75 years old). 19 The 780G use was compared with a run-in phase in which patients used SAP ± PLGS or auto basal enabled for ∼14 days. 19 Compared with run-in phase, in the AHCL phase, HbA1c decreased from 7.5% ± 0.8% to 7.0% ± 0.5%, TIR increased from 68.8% ± 10.5% to 74.5% ± 6.9%, and TBR decreased from 3.3% ± 2.9% to 2.3% ± 1.7% in 3 months (P < 0.001 for all). 19 Using the glucose target setting of 100 mg/dL and an active insulin time of 2 h, the TIR further increased to 78.8% without a change in the TBR. The system was evaluated in a crossover trial of two 12-week comparisons of AHCL (780G) with MiniMed 670G in 113 patients with T1D (14–29 years old). 18 The mean HbA1c level was 7.9% ± 0.7% before randomization and decreased to 7.6% ± 0.6% by the end of the study period in the 670G arm and 7.4% ± 0.8% in the AHCL arm (P = 0.03). TIR increased from the baseline 57% ± 12% to 63% ± 8% in the 670G group and 67% ± 8% in the AHCL group (P < 0.001). 18 In the same study, glucose monitoring satisfaction subscales for emotional and behavioral burdens improved significantly (P < 0.01) over time with the use of AHCL versus MiniMed 670G. However, patient-reported outcomes (PROs), including distress, technological attitudes, and hypoglycemia confidence, were not different. 42
Most of the early real-life study data from 780G are available from Europe due to delay in FDA approval in the United States. In a large real-life study, data from 1,01,629 users (34 countries) were analyzed. 34 The mean duration of the observation period post-AHCL initiation was 287 days. 34 The mean TIR achieved was 72.3% and increased to 78.8% when optimal settings (glucose target of 100 mg/dL and active insulin time of 2 h) were consistently used. The mean glucose management indicator (GMI) was 7% without an increase in TBR. 34
In another real-life study with AHCL in the United States, individuals with T1D were grouped by age (≤ 15-year olds and > 15-year olds). 33 Those who were ≤ 15 years old (N = 3211) achieved a TIR of 73.9% ± 8.7% and those who were > 15 years old (N = 8874) achieved a TIR of 76.5% ± 9.4% post-HCL initiation without an increase in TBR. 33 Participants maintained time in AHCL > 94% at 6 months in both groups. 33 In a subgroup analysis, patients using recommended settings achieved TIR of 78.9% and 81.3% in the group with ≤ 15-year olds and in the group with > 15-year olds, respectively, with no significant difference in TBR. 33
Tandem X2 with Control-IQ technology/Mobi
The Tandem Control-IQ system algorithm works by automatically increasing the programmed basal insulin delivery rate and/or administering automated correction boluses when glucose levels are predicted to exceed 160 mg/dL. 9 A 6-month RCT compared Control-IQ with SAP in 168 patients with T1D (14–71 years old). 20 The two groups started with an identical mean HbA1c of 7.4%. The difference in HbA1c after 6 months was −0.33% (95% CI, −0.53 to −0.13; P = 0.001). TIR increased from 61% ± 17% at baseline to 71% ± 12% at 6 months in the Control-IQ group and remained unchanged at 59% ± 14% in the control group (P < 0.001). 20 A 16-week RCT compared Control-IQ with SAP in 101 children with T1D (6–13 years old). TIR increased from 53% ± 17% at baseline to 67% ± 10% in the Control-IQ group and from 51% ± 16% to 55% ± 13% in the control group (P < 0.001). 21 Extended use for 12 weeks 35 and use for over 3 years in a similar cohort in France showed continued HbA1c reduction and TIR improvements. 43 A 13-week RCT compared Control-IQ with the standard of care (MDI or CSII with CGM) in 102 children with T1D (2–6 years old). 22 The HbA1c difference at 13 weeks was −0.42% (Control IQ 7.0% and control 7.5%, 95% CI, −0.62 to −0.22; P < 0.001). TIR increased from 56.7% ± 18.0% at baseline to 69.3% ± 11.1% after 13 weeks (P < 0.001). The standard care group had similar TIR at 13 weeks (54.9% ± 14.7% to 55.9% ± 12.6%). 22
A meta-analysis of these three RCTs including 369 participants with T1D (2–72 years old) compared Control-IQ with various control groups, all of whom, by design, were using CGM. 44 In the Control-IQ group (n = 256), TIR increased from 57% ± 17% at baseline to 70% ± 11%, and in the control group (n = 113), TIR increased from 56% ± 15% at baseline to 57% ± 14% (P < 0.001). 44
In a real-life study in the United States with 9451 individuals (6–91 years old) with T1D using Control-IQ, TIR increased from 63.6% at baseline (interquartile range [IQR]: 49.9%–75.6%) to 73.6% (IQR: 64.4%–81.8%) at 12 months. 45 Notably, the increase in TIR was noted shortly after adoption of Control-IQ and was sustained over the 1-year period of data that were analyzed. Another real-life study evaluated 4243 Medicare and 1332 Medicaid patients with T1D using Control-IQ. 36 In Medicare patients, TIR increased from 64% at baseline to 74%, and GMI decreased from 7.3% to 7.0% (P < 0.0001). 36 In Medicaid patients, TIR increased from 46% at baseline to 60%, and GMI decreased from 7.9% to 7.5% (P < 0.0001). 36
Tandem Mobi is the smallest durable tubed AID system that uses the Control-IQ algorithm and offers multiple wear options, including an on-body sleeve. 46 In a 6-week real-life study of Mobi with 1280 participants with T1D, prior MDI users (n = 235) achieved a median (IQR) GMI of 7.0% (6.6%–7.4%) and a TIR of 72.1% (62.5%–83.8%). Prior non-Tandem users (n = 208) achieved a GMI of 7.0% (6.6%–7.3%), and a TIR of 72.5% (65.0%–81.3%). 47 Prior Tandem pump users (n = 837) had a nonsignificant decrease in GMI from 7.0% (6.7%–7.4%) to 6.9% (6.6%–7.3%) and TIR increased significantly from 70.5% (60.5%–79.0%) to 73.0% (63.8%–81.3%) (P < 0.001 for both). 47
Omnipod 5
Omnipod 5 (OP5) is the only FDA-approved tubeless AID system in the United States that adjusts insulin delivery every 5 min based on the preset target glucose (110–150 mg/dL), which can be varied by time of the day. 46 OP5 was evaluated in a 3-month, single-arm prospective study with 235 participants with T1D (6–70 years old). 48 HbA1c decreased in children by 0.71% (7.67% ± 0.95% to 6.99% ± 0.63%) and in adults by 0.38% (7.16% ± 0.86% to 6.78% ± 0.68%, P < 0.0001 for both). 48 TIR increased from standard therapy by 15.6% ± 11.5% in children and by 9.3% ± 11.8% in adults (P < 0.0001 for both). 48 In an extension study that lasted up to 2 years after the initial 3-month pivotal trial, 224 patients (95% of the participants) with T1D opted to continue OP5. 23 HbA1c was maintained at 7.2% ± 0.7% in children and 6.9% ± 0.6% in adolescents and adults after 15 months (P < 0.0001). 23 TIR was maintained at 65.9% ± 8.9% in children and 72.9% ± 11.3% in adolescents and adults during the extension phase (P < 0.0001). 23 Another single-arm prospective study evaluated OP5 in 80 young children (2–6 years old) with T1D in 13 weeks. 49 HbA1c decreased by 0.55% and TIR increased by 10.9% (P < 0.0001 for both). 49 OP5 was compared with CSII with CGM (open-loop) in 194 adults with T1D (18–70 years old) in a 13-week RCT. 24 There was a greater decrease in HbA1c from baseline compared with the control group (−1.24% ± 0.75% vs. −0.68% ± 0.93%, respectively; P < 0.0001), and TIR increased from 43.9% ± 14.0% baseline to 61.2 ± 11.2% in the intervention group and from 41.3% ± 14.6% to 43.8% ± 14.5% in the control group in 13 weeks, with an adjusted mean difference of 17.5% (95% CI 14.0%–21.1%; P < 0.0001). 24
A real-world study with OP5 involving 69,902 individuals with T1D showed TIR of 68.8%, 61.3%, and 53.6% for users with glucose targets of 110, 120, and 130–150 mg/dL, respectively. 50 For the group with a glucose target of 110 mg/dL, the median TIR was 65% in children and adolescents (2–17 years) and 69.9% in adults (≥18 years). 50 Users with Medicaid insurance (n = 1064; median age 21 years) and Medicare insurance (n = 1503; median age 67 years) using target glucose of 110 mg/dL had TIR of 62.2% and 72.3%, respectively, after 6 months. 50
OP5 recently received FDA approval for use in people with insulin-requiring type 2 diabetes (T2D). OP5 was evaluated in an 8-week small pilot study in 24 people with T2D. 25 Before enrollment, participants had a baseline HbA1c of ≥ 8% and were using either basal-only or basal-bolus insulin injections, with or without CGM. 25 HbA1c decreased by 1.3% ± 0.7% and TIR increased by 21.9% ± 15.2% (P < 0.0001 for both). 25 In an extension phase of this study, 22 participants continued using OP5 for another 26 weeks. 51 HbA1c decreased by 1.6% ± 1.2% and TIR increased by 22.4% ± 19.2% (P < 0.0001, for both) at the end of the extension phase. 51 The SECURE-T2D trial evaluated OP5 in 305 participants with T2D using MDI or CSII. After 14 days of standard therapy, the study participants used OP5 for 13 weeks. HbA1c decreased from 8.2% to 7.4% and TIR increased from 45% to 66%. 26 In patients with T2D, there was a significant decline in the insulin dose at the end of the study.
iLet bionic pancreas
The iLet bionic pancreas is an AID system with three adaptive algorithms that collectively automate 100% of all insulin doses, and do not require carbohydrate counting by the user. The system is initialized with only the user’s weight. The user only needs to announce the type of meal as “breakfast,” “lunch,” or “dinner,” and the size of the meals as “usual for me,” “more,” or “less” than usual. 46 In a 13-week pivotal RCT, supported by the NIDDK, iLet (n = 219) using insulin aspart or insulin lispro was compared with standard of care (n = 107) (including other HCL systems) in participants with T1D (6–79 years old). 27 HbA1c decreased from 7.9% at baseline to 7.3% in the iLet group at 13 weeks, however, it did not change (7.7% at both time points) in the standard care group. 27 The mean baseline-adjusted between-group difference in the HbA1c at 13 weeks was −0.5% (95% CI, −0.6 to −0.3; P < 0.001). 27 The baseline-adjusted between-group difference was −0.5% separately for both children 6–17 years old (95% CI, −0.7 to −0.2; P < 0.001) and adults 18 years and older (95% CI, −0.6 to −0.3; P < 0.001). 27,52,53 The baseline-adjusted between-group difference in TIR was +11% (95% CI, 9–13; P < 0.001), with TIR increasing from a baseline of 51% ± 20% in both groups to 65% ± 9% in the iLet group and 54% ± 17% in the standard care group. The baseline-adjusted between-group difference in TIR was +10% (to 60%) over 13 weeks in the iLet group in children 6–17 years old (95% CI, +7 to +13; P < 0.001) and +11% (to 69%) over 13 weeks in the iLet group in adults 18 years and older (95% CI, +8 to +13; P < 0.001). 27,52,53 In a single-arm extension study for 13 weeks immediately following the RCT, 90 of 107 patients in the standard-of-care group used the iLet system. 28 HbA1c decreased from 7.7% ± 1.0% at baseline to 7.1% ± 0.6% at 13 weeks (mean change –0.55% ± 0.72%, P < 0.001), and TIR increased by 12.0% ± 12.5% (from 53% ± 17% to 65% ± 9%, P < 0.001). 28 Concurrently in the pivotal RCT, 114 adults 18 years and older used the iLet with fast-acting insulin aspart and had glycemic improvement similar to that of the iLet with insulin aspart or insulin lispro. The baseline-adjusted between-group difference in HbA1c versus standard of care was −0.5% (95% CI, −0.7 to −0.3; P < 0.001). 27,52,53 The baseline-adjusted between-group difference in TIR was +14% (to 71%) over 13 weeks in the iLet group with fast-acting insulin aspart (95% CI, +11 to +17; P < 0.001). To date, no real-life data have been published on the use of this system.
CamAPS FX
The CamAPS FX algorithm was recently approved by the FDA but currently it is not yet commercially available in the United States. The glucose target is customizable from 80 to 198 mg/dL. 9 It is the only FDA-approved AID system for use during pregnancy in the United States. 54 –56 In a 3-month RCT, the CamAPS FX algorithm was compared with SAP in 86 participants with T1D (> 6 years old) with elevated HbA1c. 29 HbA1c decreased from 8% ± 0.6% at baseline to 7.4% ± 0.6% in the CamAPS FX group and from 7.8% ± 0.6% at baseline to 7.7% ± 0.5% in the SAP group (mean difference in change 0.36%, 95% CI 0.19–0.53; P < 0.0001). 29 TIR was higher in the CamAPS FX group (65% ± 8%) compared with the SAP group (54% ± 9%) at the end of the study. 29 Another study compared closed-loop insulin delivery to usual care with CSII in a 6-month RCT. In 46 children with T1D (6–18 years old) with elevated baseline HbA1c, using CamAPS FX 30 HbA1c decreased from 7.9% ± 0.9% at baseline to 6.8% ± 0.5% in the CamAPS FX group and from 8.0% ± 0.6% at baseline to 7.9% ± 0.8% in the SAP group (mean difference in change 1.05%, 95% CI 0.04–0.59; P = 0.02). 30 TIR was higher in the CamAPS FX group (63% ± 9%) compared with the SAP group (49% ± 13%) at the end of the study (15%, 95% CI 8–22.1; P = 0.0001) 30 In two 16-week crossover RCTs with 74 children (1–7 years old), CamAPS FX was compared with SAP. 31 TIR was 8.7% (95% CI, 7.4–9.9) higher, and HbA1c was 0.4% (95% CI, −0.5 to −0.3) lower in CamAPS FX group. 31
In a real-life study with 1805 participants with T1D, TIR was 72.6% ± 11.5% for all users and increased by age from 66.9% ± 11.7% for users ≤ 6 years old to 81.8 ± 8.7% for users ≥ 65 years old. 37
Loop
Open-source automated insulin dosing algorithms (also known as do-it-yourself) run on a Smartphone and are largely designed, maintained, and curated by people with diabetes and their loved ones. 57 There are two main available algorithms, the OpenAPS algorithm and the Loop algorithm. Both have been implemented on a variety of platforms. Tidepool Loop, a variant of Loop was recently approved by the FDA in the United States. A prospective real-world observational study was conducted with 558 participants with T1D (1–71 years old) who initiated Loop and provided data for 6 months. 38 HbA1c decreased from 6.8% ± 1.0% at baseline to 6.5% ± 0.8% at 6 months (mean difference 0.33%, 95% CI 0.26–0.4, P < 0.001). 38 TIR increased from 67% ± 16% at baseline to 73% ± 13% at 6 months (mean difference 6.6%, 95% CI 5.9%–7.4%; P < 0.001). 38
Discussion
In the last 3 decades, several advances in therapies, including newer insulin analogs, and technologies such as CGMs, CSII, and AID systems have facilitated achievement of more targeted glucose levels for many individuals with T1D and have simultaneously reduced the burden of this chronic medical condition. Publications before widespread adoption of more advanced diabetes technologies have demonstrated that individuals with T1D have only a decade less life because of increased mortality. 58,59 Whether these gaps will be closed by technological advances remains to be seen. In addition, T1D prevalence is increasing, with the diagnosis being made more frequently in adults. 60
Unfortunately, today nearly 2/3 of the patients in the United States with T1D are overweight or obese, 61,62 Weight gain was first reported in the IIT group >30 years ago from the DCCT trial. 3 Excessive weight gain results in features of T2D, commonly referred to as double-diabetes. Excessive weight gain adds risk for insulin resistance, cardiovascular disease, and DKD. 63 The exact reasons for weight gain in patients with T1D are speculative (potentially including lifestyle changes, higher insulin dose secondary to IIT). 61 Unlike for T2D, there is no other adjunctive therapy approved by the FDA for people with T1D, besides pramlintide. However, its use is limited due to cost, number of injections per day, increased risk of hypoglycemia, and other gastrointestinal adverse effects.
A recent study reported an exponential increase in the off-label use of SGLT 2 inhibitors and GLP-1RA analogs in patients with T1D, especially those with cardiovascular disease and DKD. 64 Several recent off-label uses of GLP-1RA and SGLT2-inhibitors in people with T1D have shown promising results in improving glucose control and cardiorenal biomarkers. 65 –70 A small study even reported using GLP-1RA in new-onset antibody-positive patients with T1D. 71 While a highly selected group, these individuals noted that after a year of GLP-1 RA therapy showed significant reduction and/or elimination of the need for prandial and even basal insulin. 71 Studies to quantify how these agents may be used are critical, 72 however, we do not endorse stopping insulin at any time in people with T1D.
Use of SGLT 2 inhibitors in patients with T1D from several registration studies has reported higher risk of diabetic ketoacidosis (DKA) especially in those on insulin pumps, and thus, none of them has been approved by the FDA for people with T1D due to safety concerns. 65,73 –75 Unless proper mitigation of DKA can be shown with the use of SGLT2 inhibitors, the regulators in the United States are unlikely to approve their use for people with T1D. Newer technologies such as continuous ketone monitors may be used in addition to DKA mitigation plans to support an indication in the T1D population. 76,77 While minimal weight loss (2%–4%) was reported with SGLT2 inhibitors in participants with T1D from registration studies, whether these agents could afford other cardio- and renal protective benefits demonstrated in those with T2D remains to be seen. 65
Use of newer weekly GLP-1RAs in patients with T1D has been reported to cause significant weight loss ranging from 25 to 60 pounds (15%–25% of baseline body weight) over 1–2 years. 66 –69 The expected weight loss from GLP-1RA results in significant changes in insulin dose (prandial and basal) and thus increasing the risk of both hypoglycemia and DKA, however, to date, small retrospective study results have been reassuring. 66,67,69,78 –80 Because of a differential decrease in prandial versus basal insulin dose, it is possible that AID algorithms may need to be tweaked if newer GLP-1 RAs are used in people with T1D. 72,81
Summary and Conclusions
The benefits of AID systems in registration and real-life studies have been well documented in the past decade. In registration studies, study designs varied from single-arm to RCT studies. Primary endpoints in most of the studies included changes in HbA1c or GMI and TIR with secondary endpoints of TBR and other CGM metrics. The lack of a control group in some of the single-arm studies limits the ability to compare the outcomes observed with RCTs.
Real-life study designs were more complex with varying inclusion criteria, duration, and mostly done by the sponsoring companies using their databases. This also limits the comparisons within different AID systems. However, most real-life studies confirmed the registration study data as it relates to improvement in glucose control as measured by HbA1c and TIR. Real-life studies are important to learn about lived experiences and challenges with AID systems and to determine the effectiveness of a new technology once the constraints of inclusion and exclusion criteria are removed as are the guardrails of the frequent contacts and support provided in the clinical trials.
Patient selection, training, and education are the mainstay to achieve optimal diabetes management while using AID systems. In addition to breakthroughs in glycemic control, AID systems improve quality of life, decrease diabetes burden, and fear of hypoglycemia. Moving forward, toward fully AID systems, new-generation AIDs will integrate not only CGM data but also other physiological inputs that may alter glycemia. Some of the future directions for AID systems include novel algorithms (neural network), no meal announcements, alternative routes of insulin delivery, and faster acting insulins.
The important issues of wider access and acceptability and their use include challenges in implementation, lack of support system, and cost. Because of increasing weight in people with T1D, the role of adjunctive therapies such as GLP-1RAs and SGLT2 inhibitors needs to be evaluated for their safety and efficacy in people with T1D. Many of the AID system studies in the last decade were supported by grants from NIDDK. The critical nature of the funding provided by the NIDDK and the commitment of the program scientists, investigators, study participants, and industry to collaborate to reach this first stage in the age of automation have been essential. Continued partnerships will drive the field forward.
Footnotes
Authors’ Contributions
All authors contributed equally to this article.
Author Disclosure Statement
H.K.A. reports research support and honorarium through the University of Colorado from Medtronic, Dexcom, Roche, and Tandem Diabetes Care. S.K.G. has served on advisory boards and received consulting fees from Medtronic, Eli Lilly, and Novo Nordisk. Through the University of Colorado, he has received research grants from Eli Lilly, Medtronic, Dario, Lexicon, NCI, and Dexcom. B.P.K. reports research grants handled by the University of Virginia from the National Institutes of Health, Novo Nordisk, Dexcom, and Tandem Diabetes Care. In addition, B.P.K. has a number of patents with royalties paid to Dexcom and Novo Nordisk, outside of the submitted study. R.V. is a full-time employee of Medtronic Diabetes. R.H. reports having received speaker honoraria from Eli Lilly, Dexcom, and Novo Nordisk; receiving license fees from Braun; receiving consultancy fees from Abbott Diabetes Care; patents related to closed loop; and being director at CamDiab. R.W.B. reports no personal financial disclosures but reports that his institution has received funding on his behalf as follows: grant funding, study supplies, and consulting fees from Insulet, Tandem Diabetes Care, and Beta Bionics; grant funding and study supplies from Dexcom; grant funding from Bigfoot Biomedical; study supplies from Medtronic, Ascensia, and Roche; consulting fees and study supplies from Eli Lilly and Novo Nordisk; and consulting fees from Embecta, Sequel Med Tech, Vertex, Hagar, Ypsomed, Sanofi, and Zucara. M.D.B. reports research grants handled by the University of Virginia from the National Institutes of Health, Novo Nordisk, and Dexcom, and nonfinancial research support from Tandem Diabetes Care, outside of the submitted study. In addition, M.D.B. has a number of patents with royalties licensed to Dexcom, Sanofi, Tandem Diabetes Care, and Novo Nordisk, outside of the submitted study. M.D.B. finally reports consulting/speakership activities with Dexcom, Tandem Diabetes Care, Roche, Portal Insulin LLC, BOYDSense, and Vertex. S.A.B. has received research support through her institution from Dexcom, Insulet, Roche Diagnostics, Tandem Diabetes Care, and Tolerion and has participated on a data monitoring board for MannKind. J.L.S. has received grants or contracts from Breakthrough T1D, the National Institute of Diabetes and Digestive and Kidney Diseases, Jaeb Center for Health Research, Insulet, Medtronic, Provention Bio, and Abbott paid to her institution; has received consulting fees from Abbott; has received payment or honoraria from Insulet, Medtronic Diabetes, and Zealand Pharma; has participated in advisory boards for Bigfoot Biomedical, Cecelia Health, Insulet, Medtronic Diabetes, Novo Nordisk, and Vertex Pharmaceuticals; and reports owning stock/stock options of StartUp Health T1D Moonshot. R.M.B. has received research support, has acted as a consultant, or has been on the scientific advisory board for Abbott Diabetes Care, Ascensia, CeQur, Dexcom, Eli Lilly, Embecta, Hygieia, Insulet, Medscape, Medtronic, Novo Nordisk, Onduo, Roche Diabetes Care, Tandem Diabetes Care, Sanofi, United Healthcare, Vertex Pharmaceuticals, and Zealand Pharma. D.C.K. is consultant for Afon, Glucotrack, Lifecare, Novo, Samsung, Thirdwayv, and Tingo.
Funding Information
No funding was received for this article.
