Abstract
Background:
Potatoes are a staple food, especially in pediatric populations, but they pose distinct challenges for individuals with type 1 diabetes (T1D). This study evaluated glycemic responses in youth with T1D using a second-generation automated insulin delivery system after consuming potatoes prepared by two methods: fried and boiled.
Methods:
The study was conducted during a 5-day school camp for unaccompanied youth with T1D, aged 11–17 years, who had been using the Tandem t:slim X2™ Control-IQ insulin pump for at least 6 months. On two separate days, participants consumed a standardized meal containing 240 g of either fried or boiled potatoes, considered as 38 g of carbohydrates. Continuous glucose monitoring (CGM) data were collected and analyzed for all participants.
Results:
Our study population consisted of 31 children and adolescents (mean age 14.2 ± 1.7 years). Time in range was slightly higher after consuming boiled potatoes compared with fried potatoes, though the difference was not statistically significant (73.7% vs. 67.8%; P = 0.225). Mean glucose changes from pre-meal to 3-h post-meal were comparable between groups (−34.3 vs. −25.4 mg/dL; P = 0.517). Similarly, no significant differences were observed in the area under the curve of glucose levels. However, the percentage of bolus insulin within the 3-h post-meal period tended to be higher after fried potato consumption (20.7% vs. 11.9%; P = 0.075).
Conclusions:
Despite differences in glycemic index and fat content, the Tandem t:slim X2 Control-IQ system effectively maintained satisfactory glucose control within the 3-h post-meal period for both fried and boiled potatoes.
Introduction
The management of type 1 diabetes (T1D) requires an adequate balance between insulin therapy, physical activity, and diet. 1 Particularly, nutrition plays a crucial role in diabetes care. From the onset of diabetes, children and their families need education on maintaining a balanced and healthy diet. 2 For youths with T1D, energy intake varies according to age and development stage, with macronutrient distribution reflecting general healthy eating recommendations for young people without diabetes. Carbohydrates serve as the primary energy source, with requirements depending on factors such as age, gender, physical activity, and previous intake. Clinical evidence suggests that a daily carbohydrate intake including 40%–50% of total energy allows achieving postprandial glycemic targets. 3 To establish the appropriate preprandial insulin dose, individuals with T1D require carbohydrate counting, which has increasingly become a standard of care, particularly with the advent of automated insulin delivery (AID) systems. 4,5 This approach has been demonstrated to positively impact both health quality of life and glycemic outcomes in youth with T1D. 6 However, accurately estimating carbohydrate content can be challenging, especially for certain foods. 7 Some of the latest devices simplify mealtime management by replacing conventional carbohydrate counting with a meal announcement feature that categorizes meals as small, medium, or large. The glycemic index (GI) is a useful tool to minimize postprandial glucose spikes. The GI of a food is influenced by various factors, including its physical state, cooking and preparation methods, starch type, and the quantity of fat and protein consumed with the food. The glycemic load, which takes into account both the GI and carbohydrate content, is another valuable metric for predicting the postprandial glucose response. 8 Among the most commonly consumed foods, particularly in pediatric populations, potatoes present unique challenge for people with T1D. Potatoes are carbohydrate-rich food, low in fat, and provide high content of vitamin C, potassium, and dietary fiber. The GI of potatoes is strongly influenced by the cooking method. Boiled potatoes generally have a lower GI since their starches gelatinize slowly, resulting in a more gradual carbohydrate breakdown and a slower glucose release into the bloodstream. In contrast, fried potatoes have a higher GI due to dehydration and increased fat content during frying, which alters the starch structure and leads to a quicker carbohydrate breakdown. This process can cause a rapid spike in blood glucose levels and, due to the high-fat content, also contributes to delayed gastric emptying, resulting in unpredictable glucose responses. 9
Our study aimed to assess the post-meal glycemic patterns in young individuals with T1D using Tandem t:slim X2™ with Control-IQ technology, following the consumption of potatoes prepared in two different ways: fried and boiled.
Materials and Methods
The study was conducted during a 5-day school camp for unaccompanied youth with T1D, aged 11–17 years, held in Crotone, Italy. All participants had been using the Tandem t:slim X2 Control-IQ insulin pump for at least 6 months. Exclusion criteria included partial clinical remission, defined as an insulin dose-adjusted glycated hemoglobin ≤9%, 10 inability to independently operate the insulin pump, and any concomitant psychiatric disorders. The medical staff comprised 15 physicians, 4 nurses, and 2 educators. The study adhered to the principles outlined in the Declaration of Helsinki. Written informed consent was obtained from all participants and their caregivers prior to the start of any study-related procedures.
On two different days (the second and fourth days of the camp, respectively), all participants consumed a standardized meal, consisting of fried potatoes on the second day and boiled potatoes on the fourth day. The meals, prepared by the same cook, were served at 6.00 p.m. Any hypoglycemia occurring up to 30 min before the start of the potato meal was corrected with simple sugar/glucose at a dosage of 0.1 g/kg according to the national recommendations. 11 Participants used the bolus calculator on their AID system to determine the appropriate insulin bolus based on their individual settings (i.e., ins/carb ratio, insulin sensitivity factor, and glucose target). The insulin bolus was delivered 15 min prior to the meal and announced as a meal containing 38 g of carbohydrates (CHO) corresponding to 240 g of raw potatoes, regardless of the cooking method or the sensor glucose levels. To minimize potential bias, all participants engaged in identical physical activities in terms of time and intensity, both in the morning and afternoon of each study day. The carbohydrate intake during the meals preceding the potatoes consumption was standardized for all participants (30–40 g at breakfast, 20–25 g at morning snack, 85–100 g at lunch, respectively). The diet was prearranged before the start of the camp by two dieticians specialized in diabetes management. All participants were instructed to use the same settings and the same therapeutic management approach, including the use of exercise mode during physical activity, as they had prior to the start of the camp.
For each participant, raw data on continuous glucose monitoring (CGM) metrics across the entire school camp were collected, including mean and standard deviation of sensor glucose, percentage of time spent in the glucose range between 70 and 180 mg/dL (time in range or TIR), between 70 and 140 mg/dL (time in tight range or TITR), above 180 mg/dL (time above range or TAR), between 180 and 250 mg/dL (TAR1), above 250 mg/dL (TAR2), below 70 mg/dL (time below range or TBR), between 54 and 70 mg/dL (TBR1), and below 54 mg/dL (TBR2). Data were retrieved from the Glooko platform. 12
Statistical analysis was performed using SPSS 22.0 (IBM, Armonk, NY) for Windows package. Descriptive statistics were used to describe demographic and clinical characteristics. Numerical data were expressed as mean ± standard deviation, whereas categorical variables were presented as absolute frequencies and percentages. The Mann–Whitney test was performed to compare continuous variables between glucose and insulin delivery metrics during the 3-h period following the consumption of boiled and fried potatoes. The area under the curve (AUC) of glucose levels and the absolute variations from pre-meal to 3-h post-meal of sensor glucose values were also calculated and compared between two different types of potatoes. A threshold of 0.05 was considered as statistically significant.
Results
Our study population consisted of 31 children and adolescents with T1D (mean age 14.2 ± 1.7 years) with a slight predominance of females (51.9%). The mean diabetes duration was 5.6 ± 3.8 years, and participants had been using the Tandem t:slim X2 insulin pump with Control-IQ technology for an average of 1.6 ± 0.8 years. All participants used the Dexcom™ G6 as glucose sensor. During the 90 days preceding the start of the school camp, the mean Glucose Management Indicator of the study population was 7.0 ± 0.4%. The main CGM metrics before the school camp are presented in Supplementary Figure S1. Concomitant celiac disease was present in 9.7% of the cohort. Across the entire school camp, glucose metrics consistently remained within the recommended clinical targets. 13 The mean total daily insulin dose was 46.9 ± 11.9 IU/day, with bolus delivery accounting for a higher proportion compared with basal insulin delivery (58.2 ± 8.9% vs. 41.8 ± 8.9%). Detailed participant characteristics and glucose control metrics across the camp days are provided in Table 1.
Study Participants’ Clinical Characteristics and Glucose Control Indicators Across 5-Day School Camp
Data are expressed as mean and (standard deviation) for numerical variables and frequencies and (percentages) for categorical variables.
CV, coefficient of variation; ICR, insulin-to-carbohydrate ratio; ISF, insulin sensitivity factor; TAR, time above range; TBR, time below range; TIR, time in range; TITR, time in tight range.
Figure 1 summarizes the TIR during the 3-h period following potato consumption. While TIR was slightly higher after consuming boiled potatoes compared with fried potatoes, the difference was not statistically significant (73.7 ± 31.3% vs. 67.8 ± 27.0%; P = 0.225). A similar pattern was observed for TITR (51.4 ± 32.3% vs. 45.9 ± 29.0%; P = 0.225). No significant differences were found in time spent in hypoglycemia or hyperglycemia, with TAR2 remaining <5% in both groups. Pre-meal glucose values were comparable between the two groups (P = 0.613). A significant reduction in glucose levels from pre-meal to 3-h post-meal was observed following the consumption of boiled potatoes (P = 0.002), whereas no significant reduction was detected after fried potato consumption (P = 0.103) (Fig. 2). However, the mean changes in glucose levels between the two groups, from pre-meal to 3-h post-meal, were not significantly different (−34.3 vs. −25.4 mg/dL; P = 0.517) (Table 2). Additionally, no significant differences in the AUC for glucose levels were found between the two potato preparation methods during the 3-h postprandial period (P = 0.374). The percentage of bolus insulin, expressed as proportion of the total insulin dose within 3-h post-meal period, tended to be significantly higher after fried potato consumption compared with boiled potato consumption (20.7% vs. 11.9%; P = 0.075) (Fig. 3). Detailed data on boluses and post-meal basal insulin delivery and autocorrections are summarized in Supplementary Table S1. No manual correction boluses were delivered during the post-meal interval.
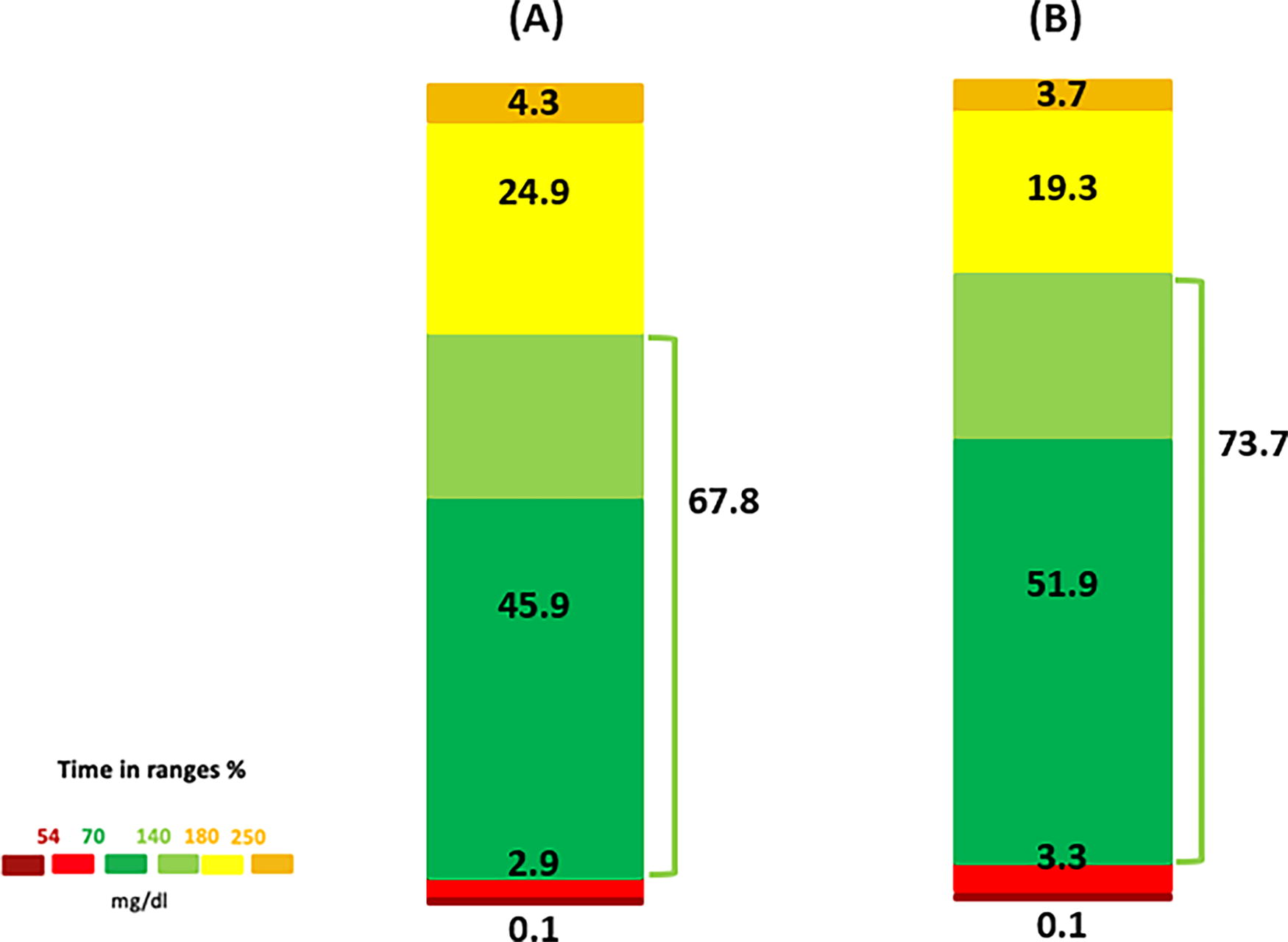
Graphical representation of time in ranges during the 3-h period following the consumption of fried potatoes
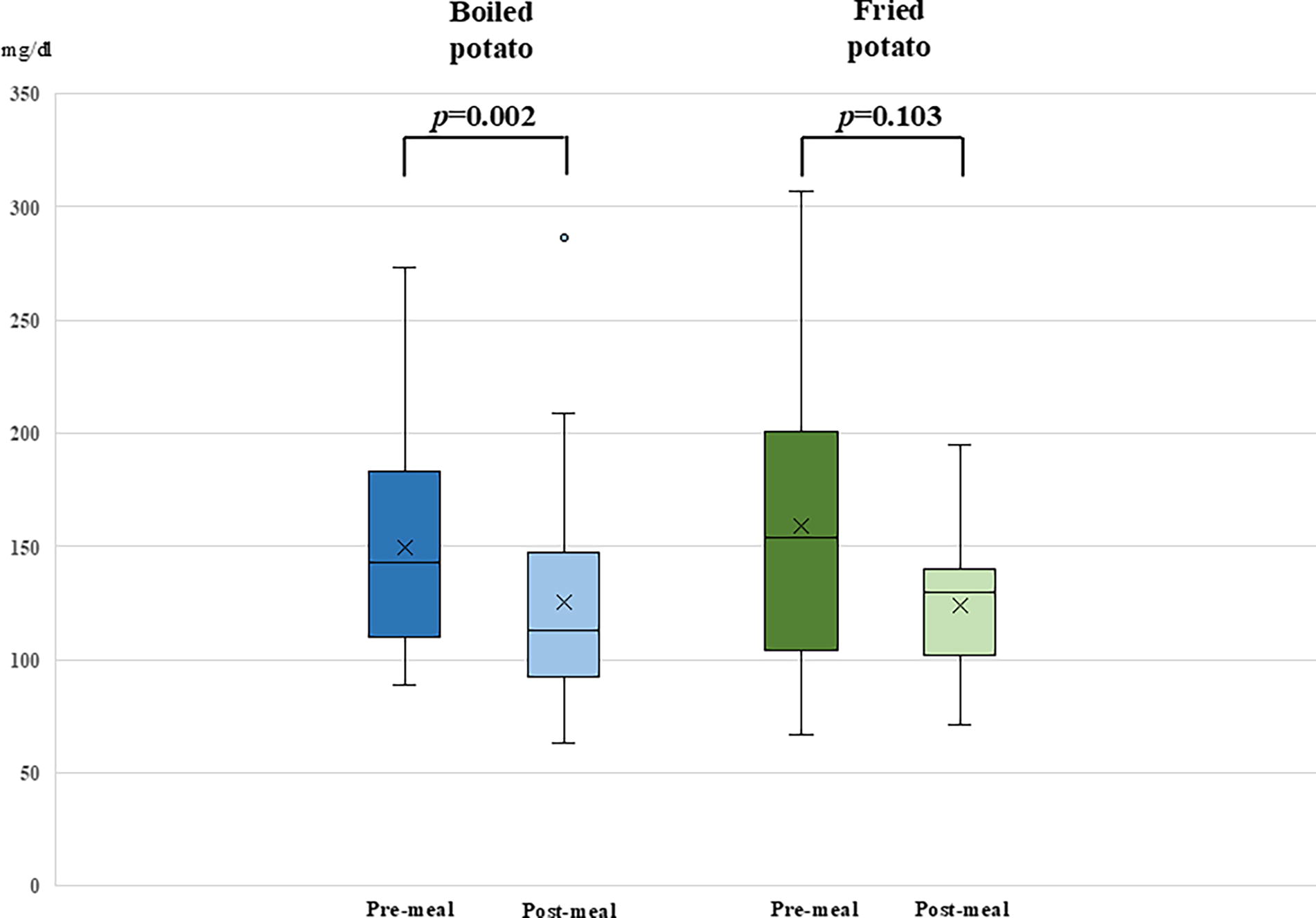
Box plots illustrating differences in glucose levels’ variation from pre-meal to 3-h post-meal between fried potatoes and boiled potatoes.
Comparison of Glucose and Insulin Delivery Metrics During the 3-H Period Following the Consumption of Boiled and Fried Potatoes
Data are expressed as mean and (standard deviation).
SG, sensor glucose.
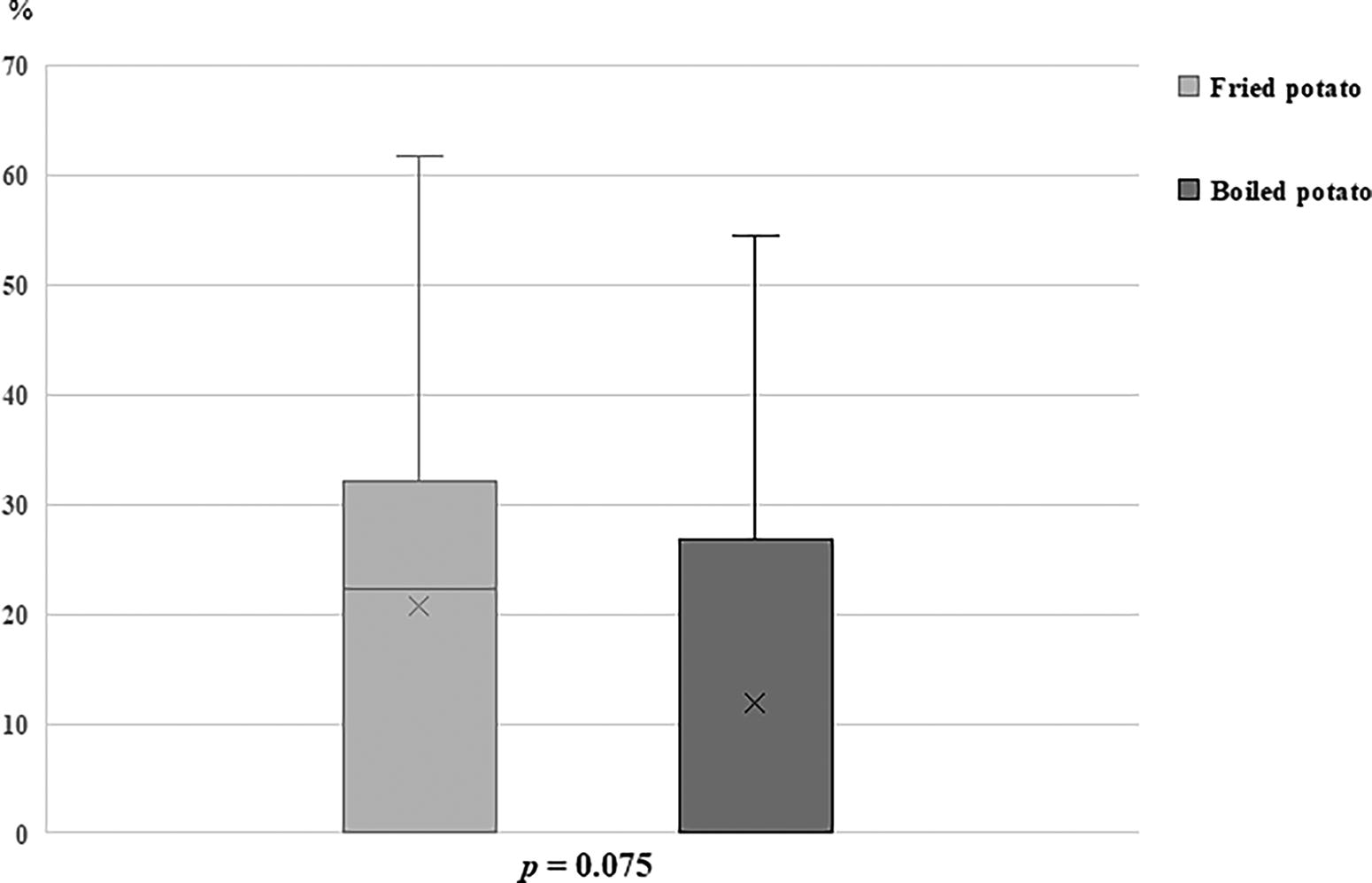
Box plots illustrating the distribution of bolus insulin delivery, expressed as percentage of the total insulin dose within 3-h post-meal period, following the consumption of two different types of potatoes.
Discussion
The primary finding of our study was that the Control-IQ system allowed the maintenance of satisfactory glucose control during the 3-hour post-meal interval, regardless of whether fried or boiled potatoes were consumed, in children and adolescents with T1D.
The Control-IQ technology is a second-generation AID system—also labeled as advanced hybrid closed loop—approved for people aged 6 years or older, which integrates the t:slim X2 insulin pump with CGM systems such as the Dexcom G6 and G7 and allows the automatic adjustment of insulin delivery based on real-time glucose readings. Control-IQ predicts glucose levels 30 min in advance, modulating basal insulin every 5 min and administering automatic correction boluses, up to one per hour, when glucose levels are projected to exceed 180 mg/dL. For safety reasons, the automatic correction boluses are limited to 60% of a full correction bolus, with a target of 110 mg/dL. To mitigate hypoglycemia, insulin delivery is reduced or suspended when low glucose levels are anticipated.
The algorithm is designed to maintain the user’s glucose levels within a target range of 112.5–160 mg/dL. When glucose levels are predicted to exceed or fall below this range, the system automatically adjusts the basal insulin delivery rate to bring glucose back within the desired target. This dynamic adjustment mechanism is crucial in addressing the variability of glycemic responses to different physiological and external factors typical of young age, such as meal composition, physical activity, or stress.
The system also offers customizable activity settings, including Sleep and Exercise modes, which adjust glucose targets to accommodate specific physiological needs. 14
The benefits of Control-IQ technology on glycemic outcomes in children and adolescents with T1D have been extensively demonstrated in both randomized clinical trials and real-world studies. 15,16 Additionally, Control-IQ has been shown to provide meaningful psychological benefits for youths with diabetes and their caregivers by alleviating the emotional burden associated with the daily management of diabetes. 17
Managing post-meal glucose levels remains one of the main challenges in T1D care, even for individuals using most advanced technologies. Certain foods are identified as particularly problematic by youths with T1D and their families due to their tendency to cause postprandial hyperglycemia. Among these, pizza, pasta, and rice are frequently cited, as evidenced by responses to a survey conducted among 100 families attending a pediatric diabetes center. 18 Several studies have evaluated postprandial glycemic outcomes in response to specific foods. An experimental study involving 38 children with T1D using a sensor-augmented pump with a suspend-before-low feature assessed the efficacy of a single bolus insulin strategy in managing postprandial glucose levels up to 11 h after consuming pizza prepared with either short or long dough fermentation. The findings revealed that, despite the use of a sensor-augmented pump, long dough fermentation resulted in better glucose control compared with short dough fermentation. 19 More recently, the potential benefits of AID systems for managing post-meal glucose levels have been explored. A study involving eight youths using the Control-IQ AID system analyzed CGM data over 12 h after consuming white rice prepared as risotto, boiled white rice, or boiled black rice. The study found slightly lower mean glucose values after consuming boiled white and black rice compared with risotto-cooked white rice, with the Control-IQ system achieving satisfactory glycemic control, as defined by International Consensus standards, 13 for all types of rice consumed. 20
Furthermore, second-generation AID systems have demonstrated superiority over previous technologies in managing postprandial glucose levels under real-world conditions. These systems have been shown to reduce hyperglycemia, particularly during the later postprandial period and overnight, reflecting significant advancements in their ability to handle the complexities of mealtime glucose management outside controlled experimental settings. 21
The ability to deliver automatic correction boluses is one of the most significant advancements introduced by second-generation AID systems. 22 This feature plays a fundamental role in maintaining glycemic stability and has proven to be more efficient than solely increasing basal rate, particularly in the postprandial period. It has been demonstrated that, in real-world conditions, complex meals with high lipid content are better managed through fractionated boluses administered over the 3 h following the meal compared with a single bolus. 23 Our analysis showed a higher percentage of automatic correction boluses following the consumption of fried potatoes compared with boiled potatoes, despite the absence of differences in glucose levels between the two cooking methods. This finding highlights the critical role played by automatic correction boluses in managing high-fat meals, such as fried potatoes. Furthermore, it reflects the ability of the Control-IQ system to adapt to varying dietary compositions while maintaining glycemic targets within acceptable ranges.
Our study was conducted under controlled conditions during a school camp, where dietitians provided guidance for accurate CHO counting. Such accuracy is not always achievable in real-world conditions, where difficulties in precisely estimating CHO intake often represent one of the main hindrances to reaching optimal glycemic targets. A study on 74 individuals with T1D demonstrated that CHO intake is underestimated by approximately 28% in real-world conditions, with greater inaccuracies observed in meals containing higher CHO amounts. 24 This underestimation also affects users of second-generation AID systems. Although these devices compensate well for inaccurate CHO counts, it has been shown that glycemic outcomes achieved with approximate CHO estimation are still inferior to those achieved through accurate and precise CHO counting. 25
Our study has several strengths, such as the controlled conditions under which all participants followed the same diet and engaged in identical physical activities, thereby minimizing potential confounding factor. In addition, this is the first study to evaluate the performance of an AID system in individuals with T1D following the consumption of potatoes.
On the contrary, some limitations should be acknowledged. The small sample size represents a constraint, limiting the generalizability of the findings. The presence of unavoidable confounding factors, such as inter-individual differences in pre-meal glucose levels, may have partially influenced the results. Furthermore, the meals analyzed consisted exclusively of potatoes cooked using different methods, which do not adhere to the recommended macronutrient balance for youths. 2
Additionally, although physical activity was standardized for all participants, it may have influenced postprandial glucose values. Another limitation is that the summer camp setting, while providing a controlled environment, restricts the generalizability of the findings to real-world conditions.
Conclusions
In conclusion, this study highlights the effectiveness of the Control-IQ system in managing postprandial glucose levels in a pediatric population with T1D after consuming a potato-based meal, regardless of the cooking method. Automatic correction boluses play a pivotal role in the management of complex, high-fat meals. However, advanced skills in CHO counting remain essential for AID users, particularly when transitioning from controlled settings to real-world conditions.
Footnotes
Acknowledgment
The authors would like to express their sincere gratitude to all the youth who participated in the school camp.
Authors’ Contributions
F.L.: Writing—review and editing; S.P.: Writing—original draft preparation; F.C., S.C., R.D.M., L.F., S.G., N.L., M.L., M.C.L., D.L.P., G.P., E.P., F.S., M.T., and C.Z.: Data curation; B.B.: Formal analysis; D.I., M.P., I.C., S.R., and R.T.: Conceptualization. The article has been read and approved by all the authors, and each author considers that the article represents their honest work.
Author Disclosure Statement
B.B. reports grants from Movi SpA and Abbott. F.L. has received speaker and consultant honoraria from Sanofi and speaking honoraria from Movi SpA. S.P. has received speaker honoraria and report grants from Movi SpA. The other authors declare no conflicts of interest.
Funding Information
The school camp during which the study was conducted was funded by Movi SpA (Milan, Italy).
Supplementary Material
Supplementary Figure S1
Supplementary Table S1
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
