Abstract
Introduction:
Adolescents with type 1 diabetes (T1D) can struggle to reach recommended HbA1c targets more than other age groups. The safety and efficacy of fully closed-loop (FCL) insulin delivery, which does not require mealtime bolusing, have not been assessed in this cohort. We evaluated the use of FCL with faster insulin aspart (Fiasp) in adolescents with T1D whose HbA1c was above recommended targets.
Materials and Methods:
This two-center, randomized, crossover study included 24 adolescents (13–19 years) using insulin pump therapy with above-target HbA1c (mean age 16.2 years, median HbA1c 74 mmol/mol [8.9%]). Participants underwent two 8-week periods of unrestricted living, comparing FCL (CamAPS HX) with Fiasp, with standard nonautomated insulin pump therapy with continuous glucose monitoring (CGM), in random order.
Results:
In an intention-to-treat analysis, the percentage of time glucose was in target range (primary endpoint 3.9–10.0 mmol/L) was higher during FCL than during pump with CGM use (mean ± standard deviation [SD]) 45.2% ± 7.2% vs. 32.3% ± 12.8%, mean difference 12.9 percentage points, 95% confidence interval [CI] 8.5 to 17.3, P < 0.001). Time spent in hyperglycemia >13.9 mmol/L and mean glucose were lower with FCL compared with pump with CGM (median time >13.9 mmol/L 28.7% vs. 39.6%, difference −7.3 percentage points, 95% CI −11.1 to −3.5, P < 0.001; mean glucose 11.1 mmol/L vs. 12.7 mmol/L, difference −1.2 mmol/L, 95% CI −1.8 to −0.5, P < 0.001). Proportion of time with glucose <3.9 mmol/L was similar between interventions (median: FCL 2.78% vs. pump with CGM 2.97%, difference −0.34 percentage points, 95% CI −1.03 to 0.35, P = 0.322). There was no difference in HbA1c after FCL compared with pump with CGM (median: 71 mmol/mol (8.6%) vs. 74 mmol/mol (8.9%), P = 0.227). There was no difference in total daily insulin dose (P = 0.276). No severe hypoglycemia or ketoacidosis occurred.
Conclusions:
FCL insulin delivery with CamAPS HX improved glucose outcomes compared with insulin pump therapy with CGM in adolescents with T1D and HbA1c above target.
Introduction
Adolescents with type 1 diabetes (T1D) have, on average, the highest HbA1c levels when compared with both younger children and adults. 1 Data from England and Wales show that only a minority of children and young people meet the recommended glycemic targets, with only 26.3% achieving HbA1c <53 mmol/mol (7.0%) and 12.8% achieving HbA1c <48 mmol/mol (6.5%). 2 These proportions are even lower in those aged 15–19 years, who have consistently higher HbA1c levels than younger children, with a mean HbA1c of 68 mmol/mol (8.4%) in this age group. 2 Multinational data show that HbA1c universally rises in young adulthood, regardless of country, baseline HbA1c, or use of diabetes technology. 3 Diabetes distress is highest in adolescence, 4 and diabetes management is particularly challenging due to a range of factors, including body image concerns, peer group influences, reduced parental oversight, increased risk-taking behaviors, and increased fear of hypoglycemia. 5
Hybrid closed-loop systems, which automatically adjust insulin delivery rates based on real-time sensor glucose levels, are fast becoming standard of care for young people with T1D, 6 with a wealth of data showing glycemic and quality-of-life benefits. 7 However, most current commercially available systems require users to count carbohydrates and manually initiate insulin boluses before meals for optimal efficacy. Accurate carbohydrate counting and premeal bolusing can be challenging and adds considerably to the burden of diabetes management. It can negatively affect quality of-life for young people with T1D, making them feel more restricted in food choices, and socially anxious when among peers. 5 Missing insulin boluses is especially frequent in adolescents and contributes considerably to suboptimal glucose control. 8 Therefore, a fully closed-loop (FCL) insulin delivery system, which eliminates the need for carbohydrate counting and premeal bolusing, may be beneficial for young people with T1D, particularly for those with higher HbA1c.
We recently reported improved glucose control and quality-of-life outcomes when using a novel FCL system (CamAPS HX, CamDiab, Cambridge, UK) with ultrarapid insulin lispro compared with standard insulin pump therapy with continuous glucose monitoringing (CGM) in adults with T1D and baseline HbA1c ≥ 8.0% (64 mmol/mol). 9,10 Adolescents differ from adults in various aspects, including physiological changes associated with puberty, psychological factors such as elevated diabetes distress, and distinct lifestyle considerations. Those aged 16–19 years are also transitioning from pediatric to adult diabetes services; this is a particularly vulnerable period when HbA1c outcomes decline and we see the highest rates of diabetic ketoacidosis admissions. 11 Therefore, we aimed to determine whether the FCL system demonstrates comparable findings in this higher risk population.
In this study, we evaluated the safety and efficacy of the CamAPS HX FCL system with faster insulin aspart (Fiasp) in adolescents (13–19 years) with T1D and HbA1c above recommended targets (≥7.5% [58 mmol/mol]).
Materials and Methods
Study participants
Eligible participants were recruited from pediatric diabetes clinics at Addenbrooke’s Hospital, Cambridge (with the Royal London Children’s Hospital and Norfolk and Norwich University Hospital as additional participant identification centers) and the Manchester Royal Infirmary Hospital. Inclusion criteria were T1D for at least a year, age 13 to 19 years (inclusive), insulin pump therapy for at least 3 months, and HbA1c ≥ 7.5% (58 mmol/mol). Key exclusion criteria included use of a closed-loop system within the past 30 days, total daily insulin dose of ≥2.0 units/kg/day, and lack of proficiency in English.
Study oversight
Approval was obtained from an independent research ethics committee in the United Kingdom (East of England—Cambridge Central Research Ethics Committee) before study commencement. Participants aged 16 years and older, and parents and guardians of participants younger than 16 years signed informed consent before any study-related activities. Written assent was obtained from participants younger than 16 years. Participants were compensated for their time and reimbursed for travel expenses incurred during the study.
Study design and procedures
This was an open-label, two-center, randomized, two-period, crossover design study comparing FCL insulin delivery with Fiasp (Novo Nordisk, Bagsværd, Denmark), with standard insulin pump therapy with CGM without the use of automated insulin delivery, during unrestricted living conditions.
At enrollment, blood samples were taken for local analysis of baseline HbA1c using an International Federation of Clinical Chemistry and Laboratory Medicine-aligned method and following NGSP standards. Participants subsequently entered a two to three-week run-in period, during which they used their regular insulin pump, insulin and CGM, and wore a masked CGM system (FreeStyle Libre 3 System, Abbott Diabetes Care, CA, USA). Thereafter, participants were randomized to receive either eight weeks of insulin pump therapy with CGM followed by eight weeks of FCL with Fiasp or vice versa, with no washout period between interventions. The order of interventions was randomly determined by permuted block randomization.
At the start of the standard pump therapy with CGM period, participants received training on the use of the study CGM (FreeStyle Libre 3), with competency evaluated through an assessment form. Participants were advised to bolus for meals or snacks as per their usual clinical care. They used their regular pre-study insulin pump and pre-study insulin (rapid or ultrarapid acting) for the next eight weeks at home. There was no automation of insulin delivery during this period.
At the beginning of the FCL period, participants were trained on the use of the study insulin pump (mylife YpsoPump, Orbit infusion set and reservoirs, Ypsomed AG, Burgdorf, Switzerland), CGM (FreeStyle Libre 3), and FCL application (app) (CamAPS HX). Competency on the devices was evaluated through assessment forms. Participants were instructed not to bolus for any meals or snacks but were taught how to bolus on the FCL system if necessary for safety. They were also able to use “Ease-off” and “Boost” modes, although advised to do so sparingly. They were supplied with Fiasp vials and continued the intervention for the next eight weeks at home.
No restrictions were placed on diet, travel, or physical activity. Study personnel contacted participants a week after commencing each intervention. All participants had access to a 24-h telephone helpline to contact the research team in the event of study-related issues.
Closed-loop system
The CamAPS HX app resides on an unlocked Android smartphone, receives continuous glucose monitoring data from the FreeStyle Libre 3 sensor, and uses the Cambridge adaptive model predictive control algorithm
9
to direct insulin delivery on the mylife
Study endpoints
The primary endpoint was the percentage of time with sensor glucose was in the target range between 3.9 and 10.0 mmol/L during the eight-week study periods. Secondary endpoints included percentage of time spent at glucose levels <3.0 mmol/L, <3.9 mmol/L, >10.0 mmol/L, >13.9 mmol/L, and >16.7 mmol/L; mean sensor glucose; glucose variability measured by the standard deviation (SD) and coefficient of variation (CV) of sensor glucose levels; HbA1c; and insulin metrics (total, basal, and bolus amounts). Secondary endpoints were calculated over the whole study period. A subset of endpoints were calculated fortnightly in each intervention period and during daytime (06:00 to 23:59) and nighttime (00:00 to 05:59) periods. Utility evaluation included the percentage of time the sensor glucose was available and the percentage of time the closed-loop system was in operation. Safety evaluation assessed the frequency of severe hypoglycemia, diabetic ketoacidosis, other adverse events, and device deficiencies.
Statistical analyses
This was an exploratory analysis, aiming for 24 participants completing the study. The statistical analysis plan was agreed by investigators in advance. All analyses were conducted on an intention-to-treat basis and included participants with a minimum of 48 h of sensor data in at least one study period. A linear mixed model was used to compare the interventions, adjusting for period as a fixed effect and accounting for the baseline value as a separate period. Highly skewed data were winsorized. Missing data were not imputed. A 95% confidence interval (CI) was reported for the difference between interventions. P values <0.05 were considered statistically significant, and all P values are two-tailed. Correction of P values for multiple comparisons was not done.
Endpoints are presented as mean ± SD for normally distributed values or as median (interquartile range [IQR]) for non-normally distributed values. Endpoints were calculated using GStat software, version 2.3 (University of Cambridge, Cambridge, UK), and statistical analyses were carried out using SPSS version 29 (IBM Software, UK).
Results
From February 2023 to May 2024, 26 participants were recruited and 24 randomized (13 female; mean ± SD age 16.2 ± 2.3 years, duration of diabetes 9.0 ± 4.6 years, baseline time in target glucose range 33.3% ± 13.8%; HbA1c range 7.5%–13.5% [58–124 mmol/mol]; median [IQR] total daily insulin dose 58.7 [40.6, 67.4] units/day [basal insulin 22.5 (18.0, 30.0) units/day, bolus insulin 34.5 (18.3, 41.6) units/day]) (Table 1). Participants used their usual rapid or ultrarapid insulin during the pump with CGM period (NovoRapid n = 15, Humalog n = 1, Fiasp n = 5, missing data n = 3). The flow of participants through the trial is outlined in Supplementary Figure S2. One randomized participant withdrew from the trial two days after commencing the second period (closed-loop) due to a lack of trust in the system components.
Characteristics of Study Participants at Baseline
Data are presented as mean ± SD or median (Q1, Q3) unless otherwise indicated.
Glucose data are based on sensor glucose measurements.
SD, standard deviation.
The primary and secondary endpoints calculated using data from all randomized participants are shown in Table 2. The primary endpoint of the percentage of time with sensor glucose was in the target range between 3.9 and 10.0 mmol/L and was higher during the closed-loop period than during the standard pump therapy with CGM period (mean ± SD: closed-loop 45.2% ± 7.2% vs. pump with CGM 32.3% ± 12.8%, mean difference 12.9 percentage points [95% CI (8.5 to 17.3)], P < 0.001). Supplementary Figure S3 shows individual participants’ time in range during the FCL and pump with CGM periods. The relationship between time in the target range during the baseline period and time in the target range during the closed-loop period is shown in Supplementary Figure S4. The mean percentage of time with sensor glucose was in tight range between 3.9 and 7.8 mmol/L and was higher during the closed-loop period than during the standard pump therapy with CGM period (mean ± SD: closed-loop 29.0% ± 5.9% vs. pump with CGM 19.4% ± 8.2%, mean difference 9.6 percentage points [95% CI (6.3 to 12.8)], P < 0.001).
Glucose Control and Insulin Delivery over Eight Weeks of Fully Closed-Loop with Faster-Acting Insulin Aspart (Fiasp) and Eight Weeks of Standard Pump Therapy with CGM
Data are mean ± SD for normally distributed values, or median (IQR) for non-normally distributed values. Transformation (winsorization) was applied to all highly skewed secondary endpoints before statistical inference.
Primary endpoint.
Based on a linear mixed model, adjusting for period as a fixed effect and accounting for the baseline value as a separate period.
n = 23 for HbA1c as one participant did not finish the fully closed-loop period.
CV, coefficient of variation; IQR, interquartile range.
The percentage of time spent with glucose above the target glucose range was lower during the closed-loop period than during the pump with CGM period across all hyperglycemia thresholds (>10.0 mmol/L mean ± SD: closed-loop 51.8% ± 7.4% vs. pump with CGM 64.3% ± 14.3%, mean difference −12.5 percentage points [95% CI −17.4 to −7.6], P < 0.001; >13.9 mmol/L median [IQR]: closed-loop 28.7% [26.3, 33.0] vs. pump with CGM 39.6% [24.7, 50.1], mean difference −7.3 percentage points [95% CI −11.1 to −3.5], P < 0.001; >16.7 mmol/L median [IQR]: closed-loop 16.8% [14.3, 20.7] vs. pump with CGM 23.5% [12.2, 36.6], mean difference −5.7 percentage points [95% CI −9.3 to −2.1], P = 0.002) (Table 2). The percentage of time sensor glucose was <3.9 mmol/L and <3.0 mmol/L was similar between interventions (median [IQR]: <3.9 mmol/L closed-loop 2.78% [2.06, 3.74] vs. pump with CGM 2.97% [1.97, 5.14], P = 0.322; <3.0 mmol/L closed-loop 0.37% [0.28, 0.76] vs. pump with CGM 0.34% [0.20, 0.74], P = 0.475).
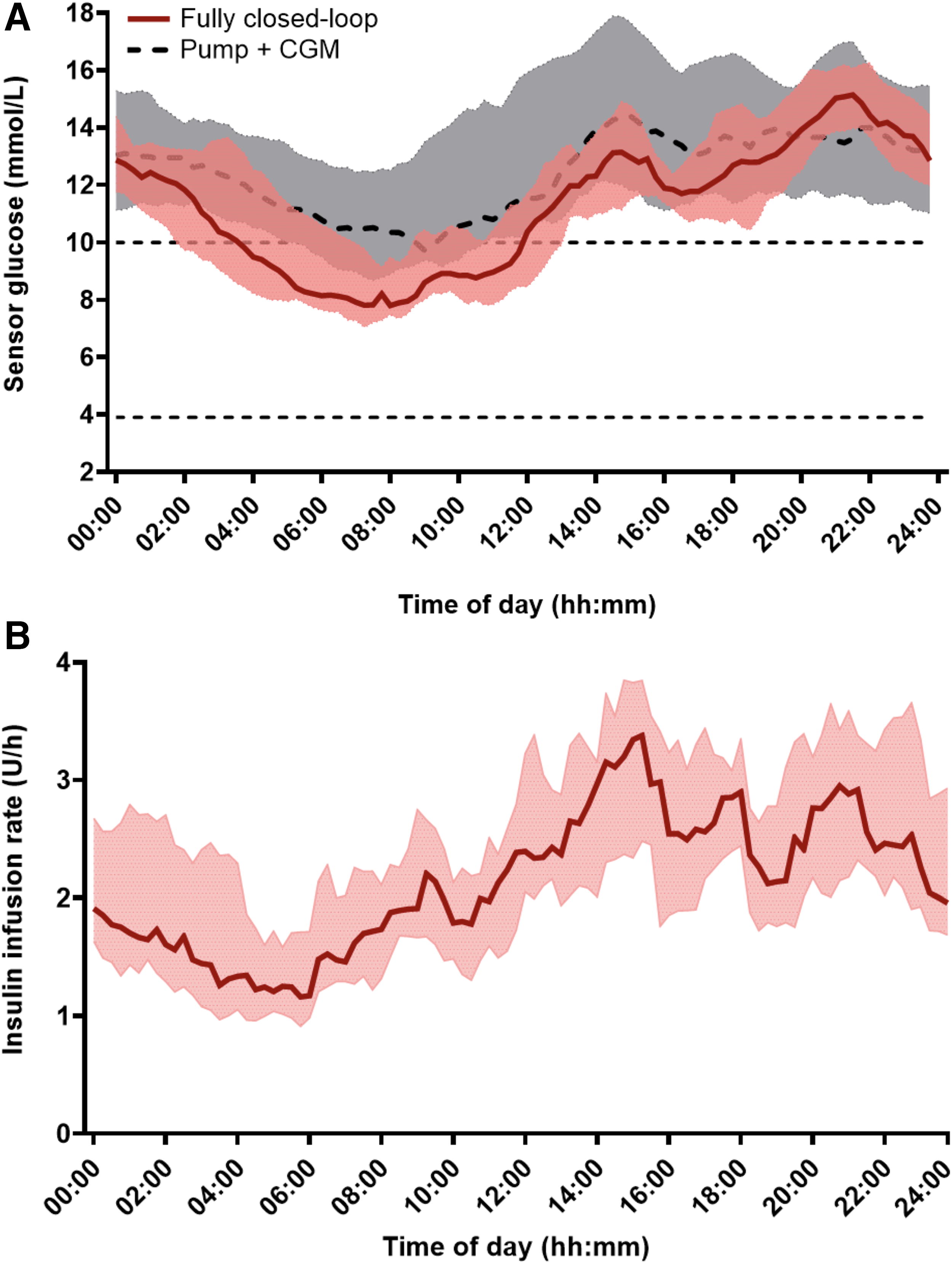
Mean sensor glucose was lower during closed-loop than during pump with CGM (median [IQR]: closed-loop 11.1 mmol/L [11.0, 12.1] vs. pump with CGM 12.7 mmol/L [11.0, 14.3], mean difference −1.2 mmol/L [95% CI −1.8 to −0.5], P < 0.001). There was no difference in glucose SD between interventions (P = 0.391), but the glucose CV was higher during the closed-loop period than during pump with CGM period (mean ± SD closed-loop 48.2 ± 2.8 mmol/L vs. pump with CGM 43.4 ± 5.7 mmol/L, P < 0.001). Figure 1A shows the 24-h sensor glucose profiles for both the intervention periods and Panel B shows the insulin infusion rate during the FCL period.
There was no difference in the median HbA1c after the closed-loop period than after pump with CGM period (median [IQR] closed-loop 71 mmol/mol [68, 77] (8.6% [8.4, 9.2]) vs. pump with CGM 74 mmol/mol [68, 86] (8.9% [8.4, 10.0]), mean difference −2.0 mmol/mol [95% CI −5.4 to 1.3] (−0.2 percentage points [95% CI −0.5 to 0.1]), P = 0.227). Those with a higher HbA1c with pump therapy and CGM showed a greater improvement with the FCL system (Supplementary Fig. S5).
The results from the per-protocol analysis of the primary endpoint, which included participants who used closed-loop for >60% of the closed-loop period (n = 23) and those who used CGM for >60% of the pump with CGM period (n = 22), were consistent with the intention-to-treat analysis findings (Supplementary Table S1). FCL use appeared to lead to the most stable glycemic control during the nighttime, particularly in terms of low levels of hypoglycemia with median time <3.9 mmol/mol 1.8% compared with 3.8% with standard pump therapy and CGM (Supplementary Table S2). Sensor glucose measures remained clinically stable throughout each intervention period (Supplementary Table S3).
Total daily insulin dose was similar between interventions (median [IQR] closed-loop 53.6 units/day [46.7, 62.9] vs. pump with CGM 49.8 units/day [42.7, 67.2], P = 0.276). During the FCL period, the median proportion of insulin delivered as a bolus was 0.8% of the total daily insulin dose compared with 52.3% in the pump with CGM period.
Eight participants experienced smartphone-related connectivity issues (using older Android smartphone models) affecting CGM and closed-loop use. Nevertheless, CGM and closed-loop use generally remained high (Table 2). The median glucose sensor data were available for >96% of the time in both periods. Closed-loop was in use for a median of 91.3% (IQR 85.0, 95.2) of the time. Supplementary Figure S6 shows the correlation between FCL usage and the change in time in range during the FCL period compared with the baseline period.
Safety analyses
Adverse events and device deficiencies are presented in Table 3. There were no severe hypoglycemia or diabetic ketoacidosis events reported during the study. One serious adverse event occurred during the standard pump therapy with CGM period, where the participant was hospitalized due to hyperglycemia associated with an intercurrent illness, although ketone levels were not elevated. Nine other adverse events were reported: three during the run-in period (one each for wrist injury, bacterial sore throat infection, vasovagal episode following venipuncture), four during the FCL period (one each for tonsillitis, two for hyperglycemic episodes, one for headache), and two during the pump with CGM period (one each for migraine and hyperglycemia with ketones 0.8 mmol/L). One adverse event in the run-in period was study-related (vasovagal episode following venipuncture). Another adverse event during the closed-loop period was possibly study-related. This was a hyperglycemic episode without ketones, possibly due to an infusion set failure, and was resolved after the participant performed a set change and resumed closed-loop. All participants recovered fully without clinical sequelae.
Safety Analyses, Adverse Events, and Device Deficiencies
There were ten device deficiencies throughout the study. Early on, it became apparent that the older Samsung S8 smartphones had unreliable Bluetooth connectivity, and this was resolved when participants were changed to newer Samsung S9, S10, or S21 smartphones between March 2023 and June 2023. There was one device deficiency during the run-in period (issues with data flow to Diasend/Cloud requiring smartphone replacement and reinstallation of app), four during the closed-loop period (three were connectivity issues requiring smartphone replacement, one explained below), and five during the pump with CGM period (four were connectivity issues requiring smartphone replacement, one was a broken pump due to the participant accidentally dropping it and required replacement). One device deficiency in the closed-loop period may have led to a serious adverse device effect. The participant experienced symptoms of hypoglycemia, which was confirmed by a finger prick blood glucose test, although the glucose sensor reportedly showed a higher reading. The participant treated the hypoglycemia episode and made a full recovery.
Discussion
This study demonstrates that the CamAPS HX FCL system, not requiring meal announcements or premeal bolusing, with Fiasp significantly improved glycemic control without increasing risk of hypoglycemia in adolescents with T1D and above target HbA1c compared with standard nonautomated insulin pump therapy with CGM.
The FCL system increased time in target glucose range by approximately 3 h per day while reducing mean glucose. The improvement in time in range was primarily achieved through a reduction in time spent above target range, with significant reductions observed at all thresholds of hyperglycemia (>10.0, >13.9, and >16.7 mmol/L). Importantly, there was no increase in hypoglycemia (<3.9 mmol/L) observed with FCL, and time spent in hypoglycemia overnight with FCL was approximately half of that with standard pump with CGM. This is key in a population that frequently has significant fear of hypoglycemia, 12 and makes longer term acceptance of FCL therapy more likely.
Total daily insulin dose remained unchanged, highlighting that the system was modulating the insulin delivery through glucose responsive automation, rather than simply intensifying overall insulin administration. During the pump with CGM period, bolus insulin made up over 50% of total insulin delivery, demonstrating that suboptimal glycemic control was not purely due to missed insulin boluses. This suggests participants were engaging in bolusing behavior, although we did not collect data on the timing, accuracy, or proportion of boluses that were premeal versus corrections.
During the FCL period, both sensor glucose levels and insulin infusion rates rose from 08:00AM with peaks around mealtimes. Sensor glucose levels were highest in the evening; this may be because of algorithm design, which is less stringent towards the end of the day to reduce risk of overnight hypoglycemia.
The mean time in target glucose range achieved with the FCL system of 45% is well below the guideline-recommended target of 70%. 13 However, it still represents a significant improvement in a group that struggles to meet glycemic targets. Baseline HbA1c in our study was high at a median of 74 mmol/mol (8.9%) with mean time in range of 33% during run-in, despite use of standard insulin pump therapy with CGM. This was a high-risk adolescent cohort, facing multiple challenges to optimizing glucose control, including fear of hypoglycemia 12 (leading to setting higher glucose targets on the closed-loop algorithm), erratic eating, and sleeping behaviors 14,15 (possibly making algorithm learning more challenging), and sometimes limited engagement with diabetes management 16 (with occasions when insulin cartridges were not replaced, the phone hosting the algorithm was not charged etc.). Despite this, overall time in FCL was high at median 91.3%, with no diabetic ketoacidosis or severe hypoglycemia events, supporting usability and safety of the system in this group. We anticipate that in this vulnerable cohort achieving 70% time in range using a FCL system with currently available insulins may be unrealistic. The 12.9 percentage point improvement in time in range seen with the FCL system is well above the five percentage points that consensus guidelines define as a clinically meaningful change, 17 and this was achieved while removing the practical and psychological burdens associated with bolusing. Each period lasted eight weeks, which may have contributed to the lack of difference in HbA1c. From a clinical perspective, it is promising that those participants with the highest HbA1c with the pump with CGM had a greater improvement with the FCL system.
The findings from this study are consistent with our previous study using the CamAPS HX FCL system with ultrarapid insulin lispro in adults with above target HbA1c. 9 In this study, we found a similar increase in time in target range, that is, 13.3 percentage points in adults compared with 12.9 percentage points in the present study. The adults had a slightly higher, but still comparable, time in range to adolescents in both the pump with CGM (36.2% vs. 32.3%) and FCL period (50.0% vs. 45.2%). It is encouraging that FCL appears effective in a high-risk adolescent cohort. In addition to the glycemic improvements, qualitative data from the adult study found the FCL system had significant quality-of-life benefits and participants were happy with improved but imperfect glucose control if it came with a break from the day-to-day demands of living with diabetes. 10
Other studies investigating an FCL approach in people with T1D have included adults and have been performed in highly supervised research facilities and/or with use of adjunctive therapies, including glucagon, pramlintide, and sodium–glucose cotransporter 2 inhibitors. 18 –23 These have shown promising results, but differences in population, baseline glucose control, setting, and study design prevent direct comparison. Notably, all studies comparing hybrid closed-loop with FCL systems showed improved outcomes with hybrid closed-loop, unless using an adjunct 19 or some form of meal announcement. 22,23
There have been no studies comparing FCL and hybrid closed-loop in an adolescent population and such future comparisons in a research setting may be of clinical interest. A recent study investigated hybrid closed-loop therapy with Medtronic 780G in a similar high-risk cohort of adolescents with elevated HbA1c despite insulin pump and CGM use. 24 Participants’ mean baseline HbA1c was 9.8%, considerably higher than the 8.9% in our cohort. Over 6 months of advanced hybrid closed-loop use, the intervention group had a 19.1 percentage point increase in time in range compared with the pump and CGM group (51.9% vs. 31.4%). Although the improvements in time in range with FCL in the present study were lower at 12.9 percentage points, comparing the two studies is challenging given the higher baseline HbA1c in the hybrid closed-loop study and additional onus to initiate meal boluses with a hybrid closed-loop system. In a study of the iLet Bionic Pancreas system, the subgroup of participants aged 13–17 years achieved a time in range of 59% over 13 weeks of using the iLet system compared with 50% in the standard care group. 25 However, this was a less vulnerable adolescent cohort than in our study, with higher baseline time in range (44%) despite the fact that over a third of participants were using multiple daily insulin injections at baseline. Furthermore, while the iLet system does not require precise carbohydrate counting, users must qualitatively announce meals and snacks beforehand, adding burden compared with a FCL system.
The strengths of our study include the crossover design, where each participant acted as their own control, as well as the use of an unrestricted home setting and a relatively diverse cohort compared with previous FCL studies in T1D as 29% of participants were from minority ethnic backgrounds. The limitations include the relatively small sample size, shorter study duration, and lack of data collected on exercise, meals, and use of additional features in the CamAPS HX app such as “Boost,” “Ease-off,” and personal glucose targets. We did not adjust for multiple comparisons possibly impacting interpretation of secondary outcomes. It is possible that the use of Fiasp may have improved the performance of the FCL system compared with the pump with CGM period, where participants used their own insulin. The use of the Medtronic 780G hybrid closed-loop system with Fiasp compared with standard insulin aspart in a similar age group improved postprandial time in tight range, although there was no difference between the groups in time in range or time in hyperglycemia. 26,27
Conclusions
FCL insulin delivery using CamAPS HX with Fiasp is safe and effective in adolescents with T1D and HbA1c above target. FCL systems may offer a novel approach to improving glucose control and reducing diabetes distress in this vulnerable population.
Footnotes
Authors’ Contributions
J.W., J.A., C.K.B., and R.H. codesigned the study. N.K., R.L., J.A., J.W., A.T., S.H., H.T., R.H.W., and P.S. were responsible for screening and enrollment of participants, arranged informed consent from the participants, and/or provided patient care. N.K. and R.L. wrote the report. N.K., J.W., M.E.W., and R.H. contributed to data analysis. R.H. designed and implemented the glucose controller. N.K., R.L., C.K.B., M.E.W., J.A., J.W., and R.H. contributed to the interpretation of the results. All authors critically reviewed the report.
Author Disclosure Statement
J.A. reports training fees from CamDiab. C.K.B. has received consultancy fees from CamDiab and speaker honoraria from Ypsomed. M.E.W. reports receiving license fees from B. Braun, patents related to closed-loop, and being a consultant at CamDiab. S.H. reports speaker and advisory board fees from Dexcom, Medtronic, Sanofi & Ypsomed; being director at ASK Diabetes Ltd. and receiving consulting/training fees from CamDiab. H.T. receives consulting fees and speaker honoraria from Eli Lilly, reports having received research support from Dexcom, Inc. and participated in advisory groups for Medtronic and Roche Diabetes. R.H.W. received speaker honoraria from Insulet. J.W. reports receiving speaker honoraria from Ypsomed and Novo Nordisk. R.H. reports having received speaker honoraria from Eli Lilly, Dexcom, and Novo Nordisk, receiving license fees from Braun; receiving consultancy fees from Abbott Diabetes Care, patents related to closed-loop, and being Director at CamDiab. N.K., R.L., A.T., and P.S. declare no duality of interest associated with this article.
Statement of Guarantor
N.K., J.W., and R.H. had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Funding Information
Abbott supplied continuous glucose monitoring devices and Ypsomed supplied insulin pumps and consumables for the study. This research was supported by the National Institute for Health and Care Research (NIHR) Cambridge Biomedical Research Center (NIHR203312) and Novo Nordisk Foundation (NNF23SA0085923). The views expressed are those of the author(s) and not necessarily those of the NIHR, the Department of Health and Social Care, or other funders.
Supplementary Material
Supplementary Figure S1
Supplementary Figure S2
Supplementary Figure S3
Supplementary Figure S4
Supplementary Figure S5
Supplementary Figure S6
Supplementary Table S1
Supplementary Table S2
Supplementary Table S3
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
