Abstract
Objectives:
Pancreas transplantation provides long-term near-normal glycemic control for recipients with type 1 diabetes, but it is unknown how this control compares with an automated insulin delivery (AID) system.
Methods:
In this prospective study, we compared parameters from 31 consecutive pancreas—kidney transplantation recipients versus from 377 people using an AID—either MiniMedTM 780G (n = 200) or Tandem t:slim X2TM Control-IQTM (n = 177).
Results:
Compared with the MiniMed and Tandem AID groups, transplant recipients at 1 month (mean ± standard deviation [SD]: 36 ± 12 days) after pancreas transplantation exhibited significantly lower glycated hemoglobin (38 mmol/mol [36, 40] vs. 55 [53, 56.5] and 56 [54.7, 57.2], respectively), lower mean glycemia (6.4 mmol/L [6, 6.8] vs. 8.5 [8.3, 8.7] and 8.2 [8.0, 8.4], respectively), and spent more time in range (90% [86, 93] vs. 72% [70, 74] and 75% [73, 77], respectively). Time in hypoglycemia did not differ significantly between the groups.
Conclusions:
Overall, compared with AID treatment, pancreas transplantation led to significantly better diabetes control parameters, with the exception of time below range.
Clinical trials registration number is Eudra CT No. 2019–002240-24.
Introduction
Type 1 diabetes is a chronic condition that requires monitoring of blood glucose levels, and disease management to maintain them within a narrow range, to avoid acute hypoglycemia and the development of chronic complications. 1 For patients with type 1 diabetes, pancreas transplantation has been considered the only route to long-term near-normoglycemia without hypoglycemia risk. 2 Islet transplantation is also an effective therapeutic approach for long-term near-normalization of glycemic control. 3,4 Transplantation offers a largely permanent solution by replacing the damaged pancreas with an unaffected organ from a deceased donor. For patients with type 1 diabetes and advanced diabetic kidney disease, the most common procedure is simultaneous pancreas and kidney (SPK) transplantation, 5 which provides excellent long-term glycemic control without the need for exogenous insulin delivery. 6,7 However, transplantation procedures carry risks, including surgical complications, infection, graft loss, 2,5 and the need for long-term immunosuppressive medication.
Recent technological advancements have included the development of automated insulin delivery (AID) systems that offer more streamlined diabetes management. These systems combine continuous glucose monitoring (CGM) with insulin pumps to automatically adjust insulin delivery based on real-time glucose readings. The precise insulin dosing is intended to mimic the function of a healthy pancreas, and to significantly lower the risk of hypoglycemia. 8 Compared with other insulin regimens, 8 –12 AID systems can offer improved glycemic control, quality of life, and cost-effectiveness. 13,14 However, these technological solutions have some limitations—including cost, the need for patient adherence, and technical difficulties. These issues raise questions regarding how the technological solution compares with the surgical approach, in terms of metabolic outcomes. Islet transplant recipients reportedly exhibit more time in range (TIR) and less glycemic variability, compared with individuals using AID technology, 15 but direct comparisons of glycemic control between pancreas transplantation and AID systems are lacking. Comparison of the metabolic control achieved by pancreas transplantation versus by AID may help to better clarify the indications of each method. 16
In this prospective study, we compared glycemic control outcomes in patients with type 1 diabetes who received treatment with SPK transplantation versus with an AID system.
Patients and Methods
Pancreas and kidney recipients
This study included patients with type 1 diabetes who participated in the prospective SIMA SPK study (Eudra CT No. 2019–002240-24) and underwent SPK transplantation between March 2021 and October 2023. The main inclusion criteria for the SIMA SPK study were age of 18–65 years, being in pre/end-stage renal failure with diabetic kidney disease, and being C-peptide negative. All participants signed the informed consent form and were added to the waiting list for a primary SPK transplant. This study was approved by the local ethics committee.
The current comparative analysis included 31 consecutive transplant cases, with no preselection. Glucose profiles were measured at around 1 month after SPK transplantation (Table 1). At this time point, all patients had well-functioning grafts. The mean estimated Glomerular Filtration Rate (eGFR) was 1.2 ± 0.3 mL/s/1.73 m2 at 1 month, and 1.31 ± 0.38 mL/s/1.73 m2 at 3 months. At 1 month (36 ± 12 days) after transplantation, each patient received a glucose sensor that provided data for 18 ± 7.5 days. These patients did not receive insulin or antidiabetic drugs, and exhibited no significant anemia. Pancreas graft function was assessed by glycated hemoglobin (HbA1c) measurement and a mixed-meal test (Supplementary Data), followed by a sensor deployment. Intention-to-treat immunosuppression comprised tacrolimus and mycophenolate mofetil, without oral steroids, at the time of sensor testing. The induction protocol included administration of methylprednisolone and an anti-human T-lymphocyte immunoglobulin. During the time period from the transplant procedure to sensor testing, three patients required reoperation due to early post-transplant infection and bleeding, two patients were treated with methylprednisolone for acute rejection of the kidney graft, eight patients required intravenous antibiotic therapy for infectious complications, and five patients developed neutropenia. No graft loss occurred during the study period. No insulin administration was required at the time of CGM testing (1 month) or at 3 months post-transplantation. During longer term follow-up, pancreas graft failure occurred in two patients. In one of these patients, intra-abdominal infection progressed and caused bleeding of the vascular anastomosis, necessitating graft removal at 4 months after transplantation. The other patient experienced acute cellular rejection of the pancreatic graft at 6 months post-transplantation, and required insulin administration thereafter.
Demographic Parameters of Studied Groups
AID, automated insulin delivery; CI, confidence interval; CKD-EPI, Chronic Kidney Disease – Epidemiology Collaboration equation; SPK, simultaneous pancreas and kidney.
AID users
The comparison group included all patients with type 1 diabetes, who were followed in the prospective institutional registry, and treated using an AID system for at least 3 months. Reports from the AID systems are collected every 3 months, and stored in the institutional database. In this study, for each participant, we used the last 3-month report available before the end of December 2023, which reflected >75% of time using an automated glucose control regimen. We enrolled a total of 377 patients who were using either the t:slim X2TM Insulin Pump with Control-IQTM Technology (Tandem, n = 177) or the MiniMedTM 780G (Medtronic, n = 200). Compared with the SPK group, patients using an AID were younger, had a shorter duration of diabetes, and had better renal function (Table 1). Since the control groups were heterogeneous in terms of number, age, diabetes duration, and renal function, we selected a subgroup of AID users who were matched with the SPK cohort in terms of those characteristics. To our knowledge, no patient elected to return to manual insulin delivery. Some patients suspend the automated glucose control regimen and in manual mode control their diabetes for a limited time period, mainly due to illness or intensive physical activity. For this study, we analyzed the readings that reflected >75% of time using an automated glucose control regimen.
Glucose monitoring
In the SPK group, glucose monitoring was performed using the Dexcom G6 (Dexcom, n = 12) or Freestyle Libre 1 sensor (Abbott, n = 19). In the AID cohort, patients used either the Dexcom G6 (Dexcom, n = 177) or the Guardian™ 4 (Medtronic, n = 200). We collected data regarding the following parameters: TIR with glucose levels at 3.9–10 mmol/L (70–180 mg/dL); time below range (TBR) with blood glucose <3.0 mmol/L (<54 mg/dL) and 3.0–3.8 mmol/L (54–69 mg/dL); time above range (TAR) with glucose levels at 10.1–13.9 mmol/L (181–250 mg/dL) and >13.9 mmol/L (>250 mg/dL); mean blood glucose with standard deviation (SD) and coefficient of variance (CV); HbA1c; and a glycemia risk index (GRI). The GRI score is a composite metric that describes the quality of glycemia in a CGM tracing. 17 For each group, we also calculated the time in tight range (TITR; percentage of time with glycemia of 3.9–7.8 mmol/L; 70–140 mg/dL). Figure 1 presents the TBR, TIR, and TAR, as well as other parameters for each group.
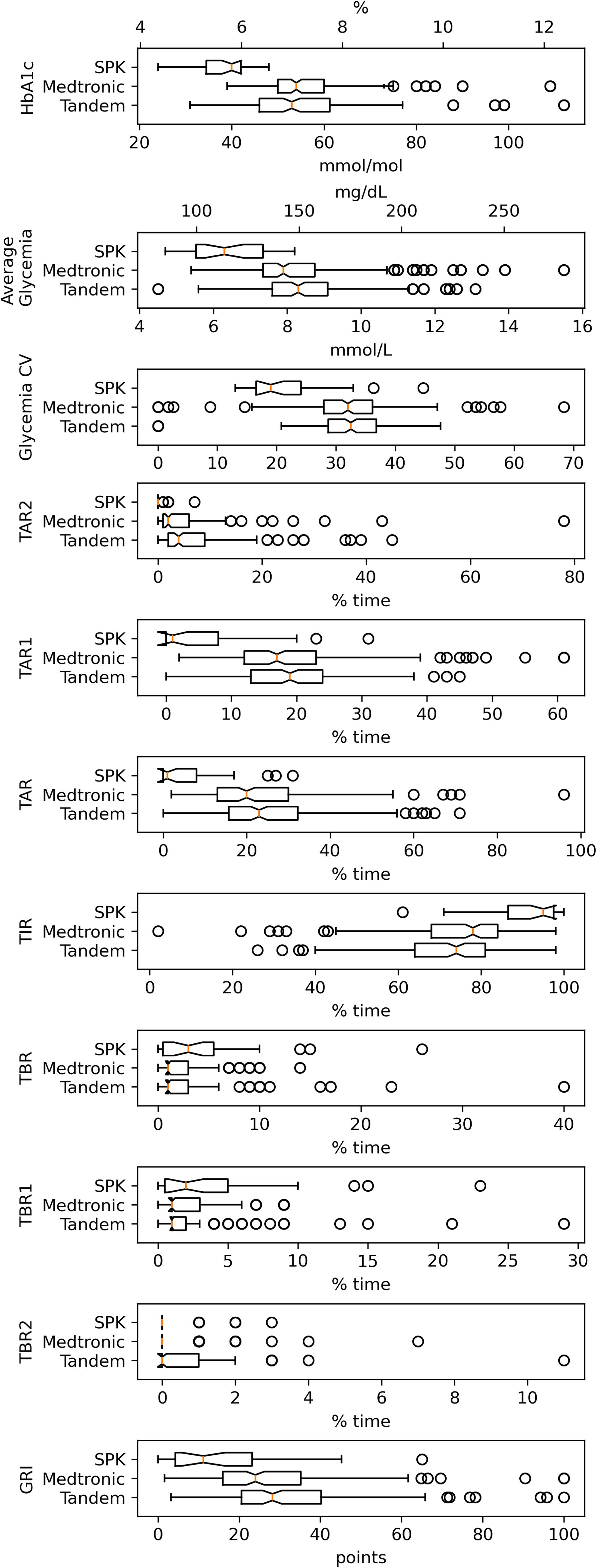
Distribution of measured results for the entire AID and SPK groups. Center lines in the notch show the medians. Box limits indicate the 25th and 75th percentiles, as determined by R software. Whiskers extend 1.5 times the interquartile range from the 25th and 75th percentiles. Outliers are represented by dots. Data points are plotted as open circles. Tandem group, n = 177; MiniMed group, n = 200; SPK group, n = 31 subjects. AID, automated insulin delivery; SPK, simultaneous pancreas and kidney; CV, coefficient of variation; TBR 1, time in <3.0 mmol/L (54 mg/dL); TBR 2, time in 3.0–3.8 mmol/L (54–69 mg/dL); TBR, TBR1, and TBR2 together; TIR, time in range; TAR 1, time above range, 10.1–13.9 mmol/L (181–250 mg/dL); TAR 2, time above range, >13.9 mmol/L (>250 mg/dL); TAR, TAR 1, and TAR 2 together; GRI, glycemia risk index.
Statistical analysis
The study findings are presented preferably with 95% confidence intervals (CIs) to provide estimates of effect sizes and their precision. This approach aligns with recommendations to report more than only P values, as CIs offer a more informative assessment of both the statistical and clinical significance of results, by indicating the range of plausible effect sizes. 18 To obtain the 95% CIs for the means of glycemia control parameters, we used bootstrap resampling with 10,000 iterations. This method was selected to avoid relying on any specific distribution of the respective variables, which was highly skewed in some instances.
Because raw data were not available from the sensors, we calculated the TITR for each group based on the mean glycemia and SD reported from each device, using normal cumulative density function. To ascertain the validity of this calculation, we calculated the TIR in the same way for each participant, and confirmed that it corresponded in aggregate to the device-reported TIR values. The group matched to the SPK cohort was selected from all AID patients, using propensity score matching with age, duration of diabetes, and eGFR. All analyses were performed using the Python ecosystem. The GRI was calculated using the formula described by Klonoff et al.
17
HbA1c levels are expressed in mmol/mol, in accordance with recommendations of the International Federation of Clinical Chemistry. Results can be converted to percentage values using the following formula:
Blood glucose levels are expressed in mmol/L, as the international standard unit for measuring the glucose concentration in blood. To convert from mmol/L to mg/dL, the blood glucose value is multiplied by 18.0182.
Results
Tandem Control IQ and Medtronic 780G users significantly differed in TIR (which slightly favored the Medtronic 780G), and time in glycemia <3.0 mmol/L and >13.9 mmol/L (Tables 2 and 3, and Fig. 2). As the two different AID user groups were identical overall, we combined them into a single cohort for comparison with the SPK group. Compared with AID users, SPK recipients exhibited significantly lower HbA1c levels, mean glycemia, glycemia variability values (SD, %CV), and GRI, and spent much less time in glycemia above range (>10.1 mmol/L). TBR (<3.8 mmol/L) did not significantly differ between groups (Tables 2 and 4, Figs. 2 and 3), and was below 1% in all groups for glycemia below 3.0 mmol/L. Pancreatic graft function remained stable, with HbA1c of 39 ± 4.2 mmol/mol at 3 months post-transplantation, and 39 ± 3.6 mmol/mol at 1 year after SPK (n = 20). The calculated TITR was 39.7% [38.3, 41.3] for patients treated using an AID, and 73.3% [66.4, 79.7] for patients after SPK. We compared the SPK cohort with a subgroup of AID users matched for age, number of subjects (n = 31), diabetes duration, and eGFR, and this analysis yielded the same results as observed for the entire AID group (Figs. 2 and 3).
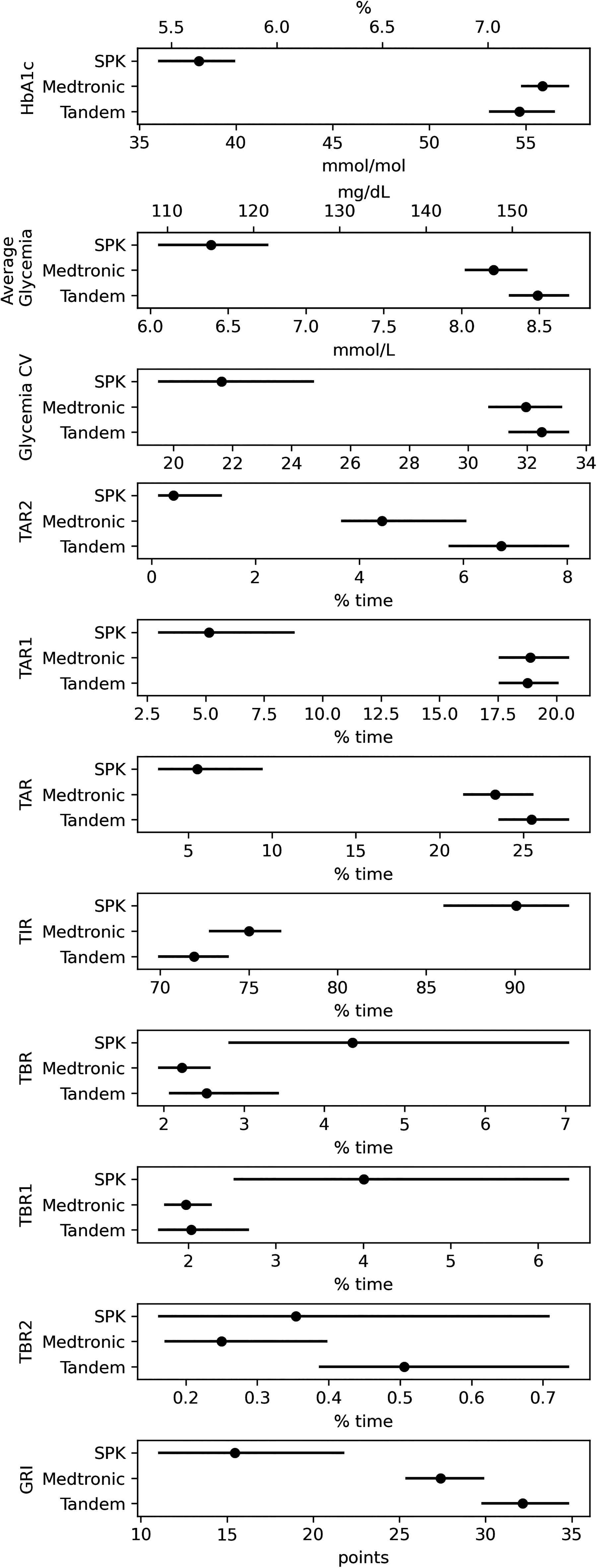
Comparison of measured parameters between the entire AID and SPK groups. Data are presented as mean and 95% confidence intervals [95% CI]. TBR 1, time in <3.0 mmol/L (54 mg/dL); TBR 2, time in 3.0–3.8 mmol/L (54–69 mg/dL); TBR, TBR1, and TBR2 together; TIR, time in range; TAR 1, time above range, 10.1–13.9 mmol/L (181–250 mg/dL); TAR 2, time above range, >13.9 mmol/L (>250 mg/dL); TAR, TAR 1, and TAR 2 together; GRI, glycemia risk index.
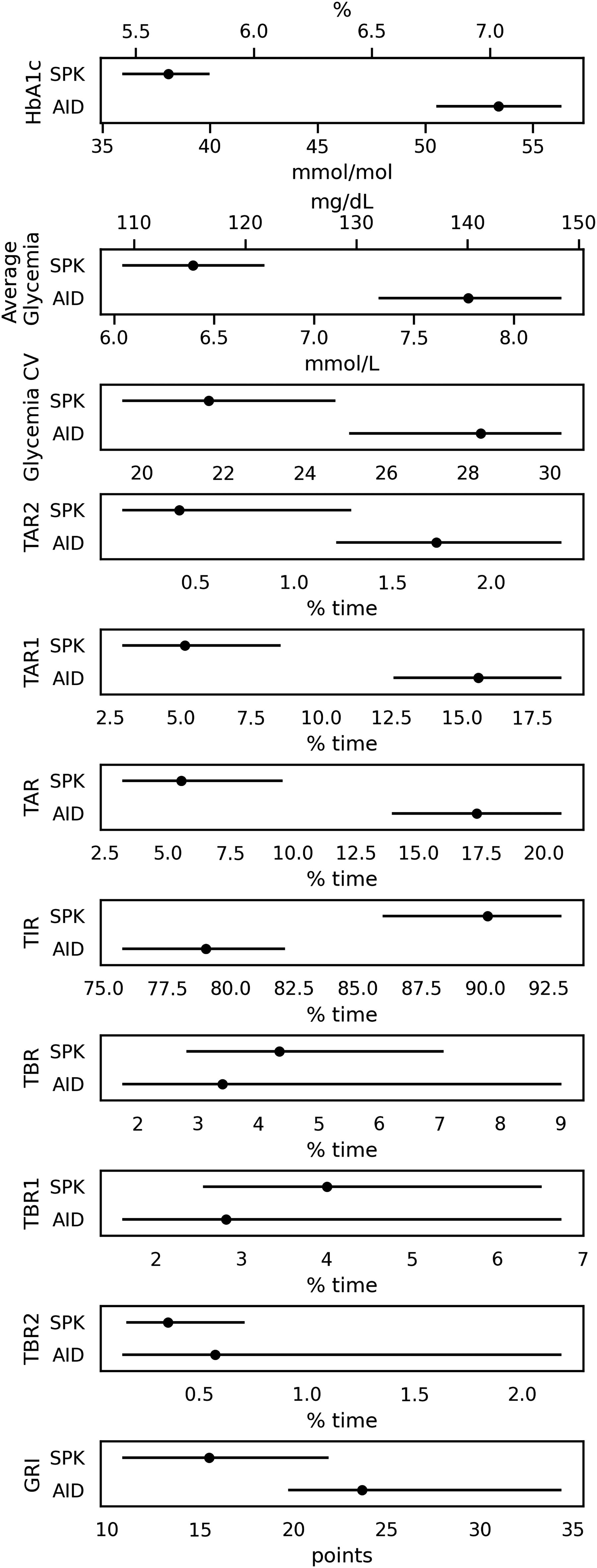
Comparison of measured parameters between the SPK group and the AID group matched to the SPK. Data are presented as mean and 95% CI. TBR 1, time in <3.0 mmol/L (54 mg/dL); TBR 2, time in 3.0–3.8 mmol/L (54–69 mg/dL); TBR, TBR1, and TBR2 together; TIR, time in range; TAR 1, time above range, 10.1–13.9 mmol/L (181–250 mg/dL); TAR 2, time above range, >13.9 mmol/L (>250 mg/dL); TAR, TAR 1, and TAR 2 together; GRI, glycemia risk index.
Glycemic Control Outcomes of Automated Insulin Delivery Group and Simultaneous Pancreas and Kidney Group at 1 Month Post-Transplant
Data are presented as mean [95% CI].
SD, standard deviation; CV, coefficient of variation; GRI, glycemia risk index; TIR, time in range.
Comparison of Metabolic Outcomes Between the Two Automated Insulin Delivery Systems
Data are presented as mean ± SD.
P values were obtained using Mann–Whitney and Student’s t test.
Comparison of Metabolic Outcomes Between the Entire Automated Insulin Delivery and Simultaneous Pancreas and Kidney Groups
Data are presented as mean ± SD.
% CV, percentage coefficient of variance.
AID systems, Tandem + Medtronic.
Discussion
The present results indicate that compared with AID systems, pancreas transplantation provides fundamentally better results in terms of controlling glucose metabolism. To our knowledge, this is the first prospective study of patients after pancreas and kidney transplantation with CGM, with analysis of outcomes compared with those of patients treated with AID systems. Our previous study focused on long-term complications and graft survival showed that SPK transplantation had the 5- and 10-year death-censored pancreatic graft survival up to 83% and 78%, respectively. 7 These results are in accord with other mainly U.S. transplant centers. 5,6 This study aimed at detailed aspects of glucose metabolic control for both types of treatment.
In cases of type 1 diabetes, insulin therapy is provided to reach the following goals: HbA1c < 53 mmol/mol, TIR > 70%, readings of <3.9 mmol/L < 4% of the time, readings of <3.0 mmol/L < 1% of the time, readings of 10.0–13.9 mmol/L < 25% of the time, and readings of >13.9 mmol/L < 5% of the time, with a CV ≤ 36%. 19 The HbA1c is a basic indicator of achieving these goals, although it does not include all aspects of glucose tolerance, especially glycemic fluctuations and hypoglycemia. To fill this gap, it has been increasing considered important to assess CGM-derived metrics as a complement to HbA1c for glycemic management, as such metrics provide a more comprehensive understanding of the glucose profile. De Meulemeester et al. 20 revealed that CGM-derived TITR and TIR were inversely related to the occurrence of microvascular complications and cerebrovascular accidents, in a large population of people living with type 1 diabetes.
Mittal et al. 21 tested the suitability of using sensors for early post-transplant monitoring of graft function, and recommended early post-transplant CGM for all pancreas transplant recipients, to identify patients likely to benefit from closer monitoring. Dmitriev et al. 22 analyzed a group of SPK patients, with a median follow-up of 8.8 years, and consistently found CGM TIR > 97% throughout follow-up, in the setting of insulin independence. For most patients with type 1 diabetes, AID systems are recommended because this therapy increases TIR and TITR, with no increase or even with a reduction of hypoglycemia, compared with other therapies. 13
In the present study, we aimed to compare metabolic parameters between patients using state-of-the-art AID technology, versus those who had undergone SPK transplantation. A glucose sensor was used after transplantation, as a part of the post-transplant follow-up. For comparison, we used data from a hospital database for all patients with type 1 diabetes who were receiving treatment with AID systems.
We found that the median TIR of 74% in the AID group was quite good with respect to recommendations 19 ; however, TIR did not reach 70% in 32% of these patients. On the contrary, the mean TIR among transplanted patients was significantly higher (90%), and only 6.4% of patients did not reach the recommended 70% TIR threshold. The between-group difference was even greater for the calculated TITR values (39.7% vs. 73.3%, favoring the SPK group). In our study, the TITR values were calculated from the mean glycemia and SD reported by each device using normal cumulative density function; however, Beck et al. showed that TIR and TITR are highly correlated despite their nonlinear relationship. 23
AID systems (both Medtronic and Tandem) have proven to be effective at reducing hypoglycemia exposure. In our study, the subjects using AID systems achieved a 2% TBR level 1 and a 0.37% TBR level 2, which is in line with the recommended goals of therapy. 19 Patients who received pancreas transplantation spent even more time in TBR (4.36% compared with 2.37% among those with AID systems) but never experienced any episodes of severe hypoglycemia. TBR > 2% was found in seven SPK patients. In four of these patients, hypoglycemia during mixed-meal test between the 40th and the 120th minute was observed. This finding shows reactive hypoglycemia on carbohydrate absorption. According to a previous study of Redmon et al., postprandial hypoglycemia may occur in some pancreas transplant recipients and it does not appear to be a highly significant clinical problem. 24 In three patients, short lasting episodes of hypoglycemia came randomly during night or day and were independent of a meal or physical activity. The majority of these episodes were recorded only and had no adequate clinical symptoms. On the contrary, in the AID-treated group, 1% experienced at least one episode of severe hypoglycemia (level 3). Most of these incidents were caused by inadequate patient reaction to physical activity, and inadequate estimation of carbohydrate content in a meal.
Although SD and CV are commonly available in CGM reports, the Advanced Technologies & Treatments for Diabetes Congress has decided on CV as the consensus marker of glycemic variability. 25 Patients treated with AID systems met the criterion of CV ≤ 36%, 19 but pancreas transplant recipients had significantly lower mean CV values (32.2% vs. 21.6%).
HbA1c is undoubtedly the most important parameter to consider. On average, the recommended target was not reached for patients treated using AID, 19 with only 49.3% reaching HbA1c ≤ 53 mmol/mol. In contrast, all or most participants in the SPK group exhibited HbA1c ≤ 53 mmol/mol at 1 month and 3 months, and even at a year (n = 27) after transplant surgery.
The GRI is a single-number summary of the glycemic quality. It comprises both hypoglycemia and hyperglycemia components, providing actionable scores. Clinicians and researchers can use its graphical display (the GRI grid) to determine the glycemic effects of prescribed and investigational treatments. 17 Our GRI results showed the majority of pancreas transplant recipients in “Zone A,” whereas AID-treated patients exhibited scores in “Zone B.”
This study had several limitations. The group of nontransplanted AID-treated patients was quite heterogeneous, and included younger people with normal renal function who did not meet the criteria for SPK. In addition, different CGM systems were used, and comparisons in the SPK group were restricted to the early post-transplant period, without any long-term assessment of CGM metrics in this group. Longer follow-up would provide more data on graft survival and complications, but this was addressed in detail in our previous study. 7 The AID-treated patients were a relatively large group, but they were systematically monitored and educated. On the contrary, the group of SPK recipients was relatively small but more homogeneous.
A more clinically relevant comparison would have been between patients who received only pancreas transplantation and patients treated with AID systems. However, pancreas transplant alone is performed much less frequently, partly due to the shortage of suitable organs for transplantation. SPK transplantation is more frequent, has very good outcomes, and is usually the option of choice offered to patients with diabetes and end-stage renal disease. 7 Indications for pancreas transplant alone are limited by a wider use of modern approaches (e.g., islet transplantation and AID systems), and are thus mainly reserved for patients with hypoglycemia unawareness, in whom all proven technological means have failed. 26 We can speculate that results identical to our present findings might also be achieved with the transplantation of isolated islets, 3,4,15,27 or in the future by using insulin-producing tissues grown in vitro. 28
In conclusion, our study demonstrates that at 1 month post-transplant, SPK recipients exhibit better metabolic control, compared with patients treated with commonly used AID systems. The exception was hypoglycemia, for which the AID systems showed efficacy comparable with that of SPK.
Footnotes
Acknowledgments
The authors thank all study participants for their time and efforts. They thank the Educational Nurse Team (Department of Diabetes, IKEM) for their assistance with patient care and for being able to handle all of the everyday challenges of life with technology. They also thank the IT team for supporting the institutional registry of patient’s data.
Authors’ Contributions
S.F. conceptualized the study and contributed to the protocol draft and study planning. H.M. and G.P. head the Department of Diabetes, thereby enabling patient recruitment, and run the institutional registry. Z.M. and N.L. contributed to participant recruitment, screening, and enrollment. Z.M. conducted the study visits, collected and analyzed data, and finally wrote this article. V.D. collected and analyzed data from the registry. B.R. and H.M. coordinate the institutional registry. K.M. performed the statistical analyses.
Data Sharing
The dataset generated and analyzed in this work is available from the corresponding author on reasonable request.
Author Disclosure Statement
The authors of this article have no conflicts of interest to disclose as described by the Diabetes Technology & Therapeutics journal.
Funding Information
This trial was an independent investigator-initiated study funded by the: AZV MZ ČR (NW24-01–00138), Czech Ministry of Health; supported by the project National Institute for Research of Metabolic and Cardiovascular Diseases (Program EXCELES, ID Project No. LX22NPO5104), funded by the European Union, Next Generation EU; and supported by MH CZ — DRO (IKEM, IN 00023001).
Supplementary Material
Supplementary Data
