Abstract
Aims:
This study addressed the challenge of postprandial glycemic variability in type 1 diabetes (T1D), even with AID (automated insulin delivery). We evaluated the effectiveness of a non-carbohydrate counting (non-CC) meal bolus strategy in adults with T1D utilizing open-source AID.
Methods:
A total of 32 adults with T1D, aged 18 to 50 years, participated in a randomized crossover trial utilizing open-source AID. Following a 7-day run-in period, participants were randomly assigned to one of two groups: automatic mode (closed loop) or manual mode (open loop). After 2 weeks, the participants underwent a crossover to the alternate treatment mode. Prandial boluses were administered according to a sliding scale based on preprandial glucose levels, without utilizing either the exact carbohydrate content of meals or meal announcement buttons. The study compared the differences in time in range (TIR) and insulin dosage across the different phases.
Results:
Compared with the open-loop phase, the TIR for patients during the closed-loop phase increased significantly during the night (75.45% ± 12.08% vs. 83.05% ± 7.20%, P < 0.001) and 24 h (73.40% ± 9.98% vs. 79.21% ± 4.84%, P = 0.019), with a more pronounced effect observed at night. During the closed-loop phase, the frequency of 24-h hypoglycemic events (<3.9 mmol/L) was reduced compared with the open-loop phase, with no difference in nocturnal hypoglycemic events. In addition, compared with the open-loop phase, there were no significant differences in average postprandial blood glucose and peak blood glucose levels during the closed-loop phase; however, the time to reach peak postprandial blood glucose was delayed (86.06 ± 20.80 min vs. 99.08 ± 15.05 min, P < 0.001).
Conclusions:
A non-CC meal bolus strategy based on preprandial glucose in adults with T1D utilizing open-source AID effectively prevents glycemic excursions and maintains a mean TIR over 70%.
Introduction
Carbohydrate counting (CC), a meal-planning method that focuses on the amount of carbohydrates (CHO) consumed at meals, is recommended for optimal glycemic control in type 1 diabetes (T1D). 1,2 The Diabetes Control and Complications Trial (DCCT) considers CC beneficial in assisting T1D patients in achieving glycemic outcomes and improving quality of life. 3 Subsequent evidence suggests that CC may positively affect metabolic control and reduce glycosylated hemoglobin(HbA1c) concentration. 3 –5
According to the CC algorithm, patients should identify the CHO in their food and determine the amount of insulin required to cover the CHO portion. For instance, if a patient has an insulin-to-CHO ratio (ICR) of 1 unit for every 10 g of CHO and consumes 60 g, he or she needs to administer a prandial bolus of 6 units of rapid-acting insulin. Alongside the potential benefits, however, for many patients with T1D, CC can also be a burdensome, error-prone, and complex task. The previous study reported that 63% of patients underestimated their meal content, and the average error in CC is approximately 20%. 6 The challenge is particularly evident in Asia. As we know, Asian countries usually have higher daily CHO intake (mean values about 60%) than non-Asian countries in North America and Europe (mean values generally ≤50%) and prefer the high glycemic index (GI) diet. 7,8 In many regions of Asia, where intricate culinary techniques and a tradition of communal dining prevail, evaluating the CHO in various foods can be difficult.
Randomized controlled trials (RCTs) and meta-analyses have demonstrated the efficacy and safety of automated insulin delivery (AID) systems. These systems have improved glycemic outcomes in youths and adults with T1D compared with multiple daily injections (MDI) or sensor-augmented pump (SAP) therapy. 9,10 For example, the ADAPT study (MiniMed 780G, USA) found that within a 6-month observation period, the average HbA1c in the advanced AID group decreased from 9.00% to 7.32%. In contrast, the MDI group only experienced a drop of 0.20% in HbA1c. 11 Another study similarly reported that the time in range (TIR) for the hybrid closed-loop systems (HCL) group (Control-IQ) increased from a baseline of 61% ± 17% to 71% ± 12% after 6 months, while the control group remained at 59% ± 14%. 12 Currently, most AID systems still require users to calculate and manually input the CHO content before meals to determine the appropriate bolus. However, many disturbances stand in the way of closed-loop control; postprandial glucose remains the most significant challenge. 13
Given the lack of a fully closed loop, there has recently been heightened interest in simplifying the approach to meal announcements for people using AID systems and exploring the abilities and limits of AID systems regarding unannounced meal disturbances. Simple semiqualitative meal-size estimation (in the context of single- or dual-hormone AID) has been proposed. 14 –16 For instance, in 2014, El-Khatib FH et al. reported that a prandial bolus based on body weight (0.05 U of insulin per kg) within the context of dual-hormone AID improved mean postprandial glucose (PG) without increasing the time spent with PG less than 60 mg/dL, but postprandial periods only modestly reduced in the adult subgroup. 14 In a subsequent inpatient trial, the efficacy of dual-hormone AID combined with a CHO-matched bolus was compared with dual-hormone AID accompanied by prandial boluses based on qualitative categorization (regular or large) of meal size (without CC). The AID without CC achieved overall comparable mean blood glucose compared with CC but yielded higher postmeal excursions after meals with >90 g of CHO. 15
Until now, no commercial AID has been marketed in China. Therefore, patients who desire to construct loop systems using open-source AID, also known as do-it-yourself APS (DIY-APS). Previous studies observed a significant reduction in HbA1c levels among Chinese patients with T1D after using DIY-APS for 3 months. 17 However, none has been conducted using the simple bolus strategy in patient-free living settings combined with open-source AID in Asia.
In this study, we present the results of a crossover trial comparing open-source AID combined with a simplified preprandial bolus estimation in adults with T1D over two periods of 2 weeks. The system has been used in other studies. 18,19
Research Design and Methods
Study design
This prospective, free-living, randomized, open-label crossover trial was performed at the First People’s Hospital of Yunnan Province. The First People’s Hospital Ethics Committee of Yunnan Province has approved this study (KHLL2022-KY165). This pilot trial is registered as ChiCTR2200066140. All participants signed a written informed consent form.
Participants
The recruitment began in December 2022 and ended in February 2024. Participants were 18–50 years old, with an HbA1c of 6.5%–10%, duration of T1D over a year, and a stable insulin regimen using insulin pumps or MDI within 3 months before the screen was recruited. Patients who had significant diabetes complications and/or a severe episode of diabetic ketoacidosis (DKA) or hypoglycemia in the past 6 months were excluded.
Study procedures
Before testing, all study participants completed a screening visit, including a medical examination, HbA1c testing, anthropometric measurements, and examination of insulin therapy records. After obtaining consent, the trial started with a 7-day run-in period to train participants on using the continuous glucose monitor (CGM) (SIBIONICS GS1, China), an insulin pump (DANA R/RS, Korea), and AndroidAPS (Version 3.1.0.3). During this run-in period, participants already using insulin pumps transitioned directly to the study pump while maintaining their customary parameters. Individuals on MDI transitioned to a conservative insulin regimen. An engineer and the principal investigator set up the locked Chinese version of AndroidAPS and fine-tuned the pump settings for optimal performance. This included the highest basal rate, ICR, insulin sensitivity factor, the upper limit for CHO, the maximum permissible bolus, and the maximum insulin on board. 20 The target glucose level was 5.6 mmol/L, with an insulin action duration of 3 h. At the same time, the researchers provided the participants with the bolus dosage for their regular meals.
Participants were randomly assigned to two groups: automatic mode (closed loop) and manual mode (open loop). Each phase, open loop and closed loop, lasted for 2 weeks, with a switch in mode occurring at 8:00 AM on the first day of the third week.
After randomization, a registered nutritionist reviewed the participants’ usual average CHO intake and explained the simplified meal bolus strategy. In this simplified meal bolus strategy, participants did not report the exact CHO content of their meals or use meal announcement buttons. Prandial boluses were used on a sliding scale based on preprandial glucose (Supplementary Table S1) after the training period. The recommended bolus dose is calculated based on the average mealtime insulin requirements observed during the training period for breakfast, lunch, and dinner, separately.
For example, a participant is typically recommended to administer seven units of insulin at breakfast. The premeal blood glucose level is measured at 13 mmol/L. Based on Supplementary Table S1, two additional units of insulin are necessary to adjust the blood glucose level. Therefore, the patient will receive nine units as a prandial bolus.
No restrictions on dietary intake or daily activities were advised during the study. Standard local hypoglycemia and hyperglycemia treatment guidelines were followed. Patients were instructed to give their insulin prandial boluses within 10 min before the beginning of the meal.
Outcomes and analyses
The primary outcome was the difference between open loop and closed loop in TIR (70–180 mg/dL) in the 2-week study phase. Secondary outcomes were between-group differences in other CGM-derived metrics for glycemic outcomes (including time above range, time below range, mean sensor glucose, SD, and coefficient of variation [CV]), the insulin total daily dose [TDD], and postprandial glucose. Safety endpoints included episodes of severe hypoglycemia (defined as a hypoglycemic event that requires medical assistance) and/or DKA. An overview of the primary and secondary endpoints is shown in Tables 2 and 3.
Statistics
The study hypothesis was that the closed loop is expected to show better glycemic outcomes than the open loop. In addition, we wanted to learn whether the simple semiquantitative meal bolus strategy with open-source AID still reaches international targets for glycemic control. Based on the hypothesis and an α of 0.05, a power of 80%, a TIR in the closed loop of 82.4, an SD of 7.8%, and an effect size of 5%, 18 we calculated that 26 participants would be required. To allow for dropouts, we planned to enroll 34. All analyses were performed for the entire study population. Between-group differences were analyzed using paired Student t-test or Wilcoxon test (in case of non-normality). Within-group differences were analyzed using ANOVA. A two-sided α-level of 0.05 was used, with no adjustment for multiplicity for secondary outcomes. Statistical analyses were performed using SPSS Statistics 26 (IBM, USA).
Results
Participant characteristics
Thirty-two individuals with T1D were approached to participate. Twenty-eight agreed to enroll, and all enrolled participants completed the study in full.
Table 1 presents baseline demographics and glycemic control characteristics. The mean age of participants was 35.1 ± 8.5 years. Eight participants were male (33.3%), and 20 were female (66.7%). The mean duration of diabetes was 13.2 ± 9.2 years (range 1.3–30 years). Mean HbA1c was 7.1% ± 1.0%. Eighteen patients had previously used CGM, with an average CGM usage time of 1 month (interquartile range 0.75–15.5 months).
Baseline Characteristics
Data are mean ± SD, unless otherwise indicated.
CGM, continuous glucose monitor; MDI, multiple daily injections; TDD, total daily dose.
Glycemic outcomes
Figure 1 displays the glycemic outcomes in the trial. The subjects achieved a TIR of 70% during the trial period. However, only during nighttime and the whole day, participants spent less time within the target range in the open-loop period than closed loop; TIR improvement is most evident during the night phase (75.45% ± 12.08% vs. 83.05% ± 7.20%, P < 0.001).
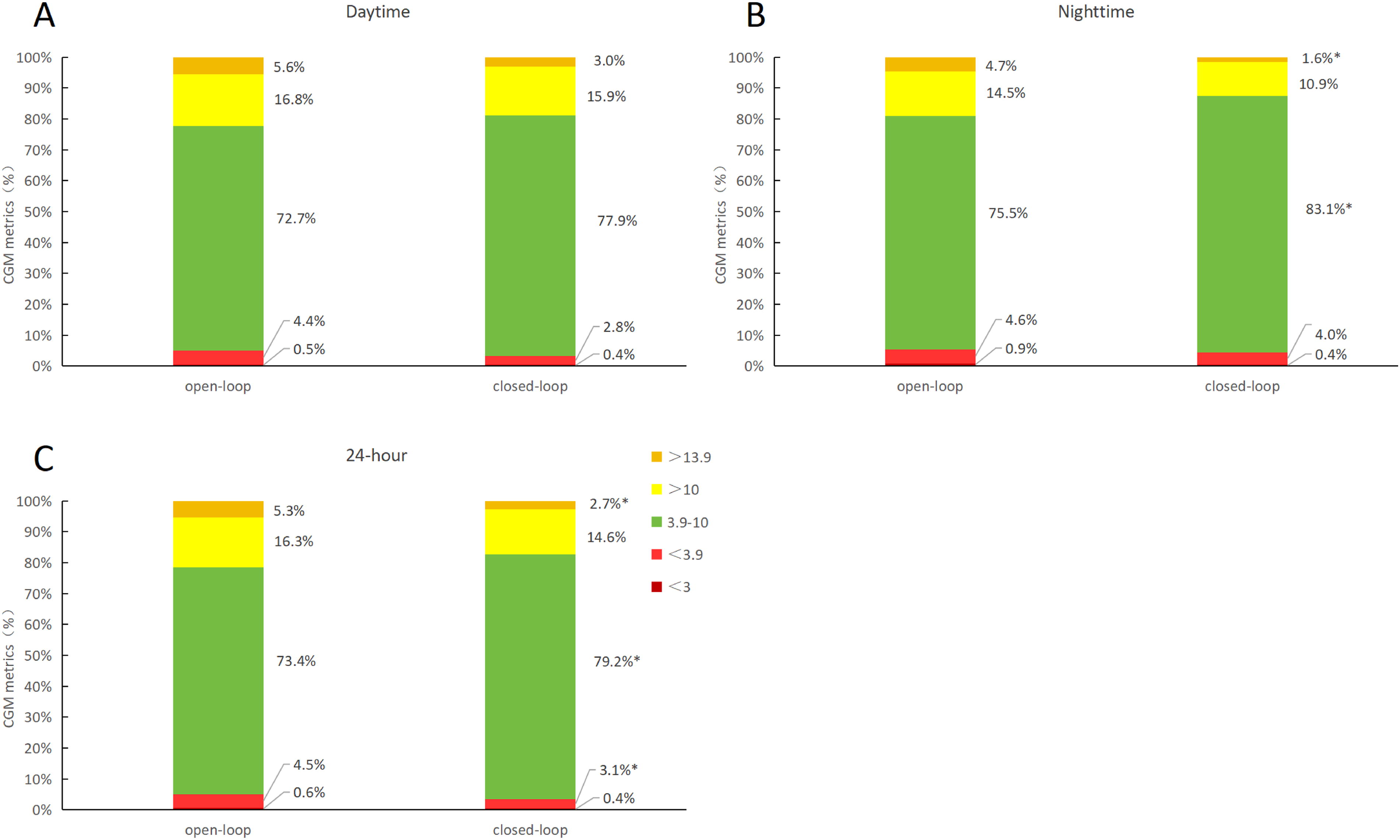
Glucose levels during open-loop and closed-loop (TIR).
In the 24 h (37.97% ± 5.50% vs. 34.57% ± 4.38%, P = 0.003), daytime (38.12% ± 6.76% vs. 34.87% ± 4.16%, P = 0.005), and nighttime (34.84% ± 8.27% vs. 30.25% ± 5.23%, P = 0.011), the coefficient of variation (CV) in the closed-loop phase was lower compared with the open-loop phase. Other CGM-derived metrics did not reveal significant between-group differences, except for the time spent above 13.9 mmol/L during the 24 h (5.34% ± 4.35% vs. 2.67% ± 1.34%, P = 0.049) and nighttime (3.56% (1.34, 7.63%) vs. 0.51% (0.00, 3.06%), P = 0.041) and the time spent below 3.9 mmol/L over 24 h (4.46% ± 2.93% vs. 3.08% ± 1.98%, P = 0.032), which were both improved in the closed-loop phase compared with the open-loop phase (Fig. 1).
Postprandial glycemic outcomes
Table 2 shows peak glucose levels and time to peak after meals. Closed loop and open loop had similar peak glucose levels 180 min after meals (10.70 ± 1.03 vs. 10.59 ± 1.49 mmol/L, P = 0.945). However, the closed loop changed the mean time to peak glucose (86.06 ± 20.80 vs. 99.08 ± 15.05 min, P < 0.001). The closed loop showed similar mean glucose levels (180 min) after meals to the open loop (Table 2).
Postprandial Glycemic Outcomes
ΔPeak, the difference between the average blood glucose level measured 10 min before a meal and the postprandial peak blood glucose level within 180 min after the meal. ΔLow, the difference between the average blood glucose level measured 10 min before a meal and the postprandial lowest blood glucose level within 180 min after the meal.
Figure 2 displays glucose excursions (mmol/L) at 30-min intervals for both the open-loop and closed-loop phases over the 180 min following meals, showing no significant differences.
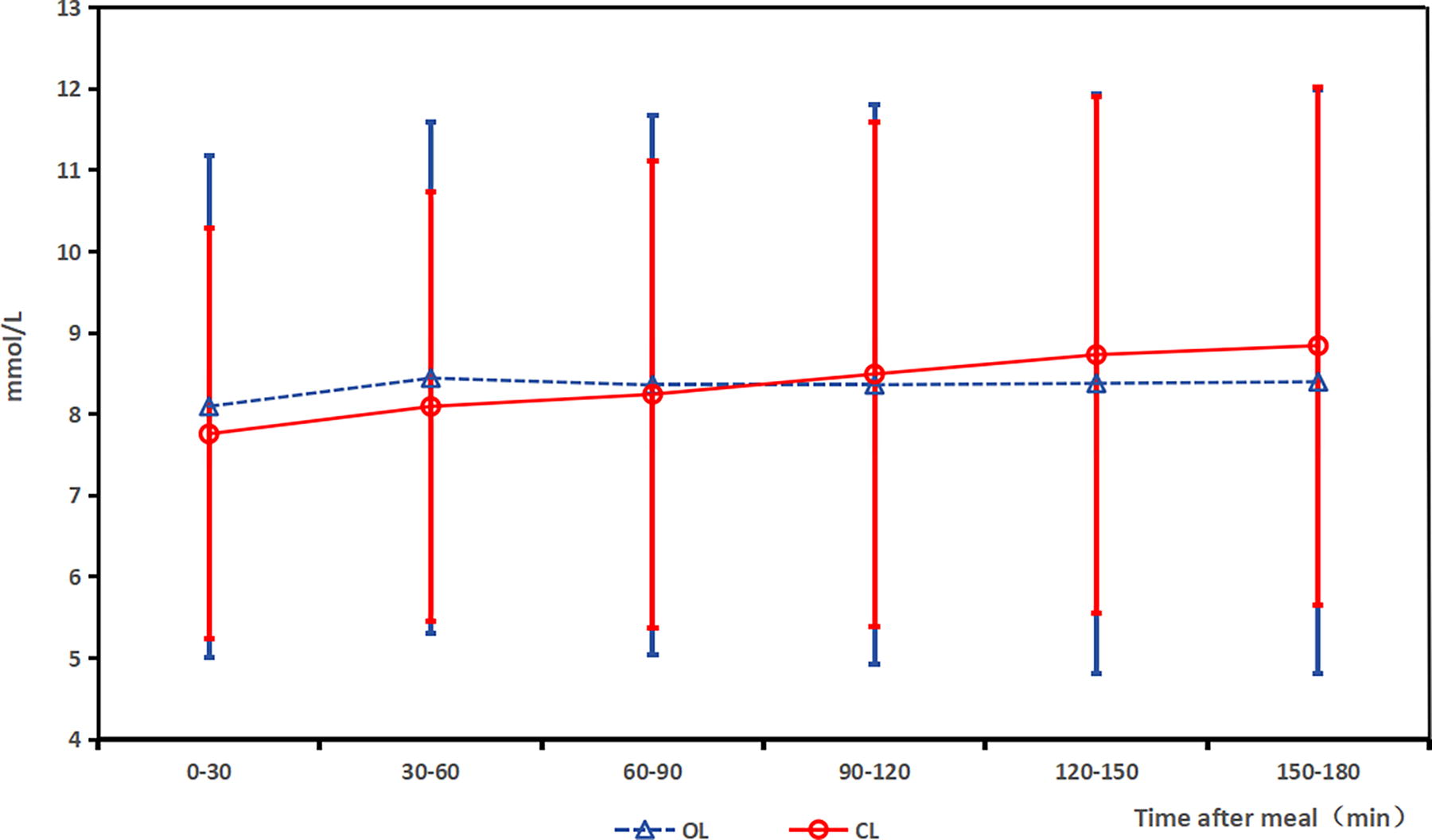
Glucose excursions (mmol/L) at 30-min intervals by continuous glucose monitoring system (CGMS) (x ± s). L, open loop; CL, closed loop.
Insulin delivery
Table 3 shows insulin delivery during the study. The TDD had no group difference (P = 0.112). Basal insulin as a percentage of TDD decreased by 16.0% in the closed-loop period (47.21% ± 13.02% to 31.22% ± 8.51%, P < 0.001). The mean bolus amount was lower in the open-loop period (17.86 ± 8.13 vs. 23.77 ± 8.13 units/day, P < 0.001).
Insulin Dosing
Safety
No serious adverse events, including episodes of severe hypoglycemia, DKA, or the need for hospitalization for any cause or serious or unanticipated adverse device effects, were reported during the research period.
Discussion
Our study is the first crossover trial to demonstrate the safety and efficacy of a meal bolus strategy without CC in adults with T1D using open-source AID. All subjects achieved a TIR of 70% during both the open-loop and closed-loop phases; however, this approach was associated with an increased TIR and reduced glycemic variability in the closed-loop period.
Postprandial glucose control has always been challenging for patients with T1D; even on the background of AID, accurately estimating CHO intake is still an essential task with current AID systems. 21 –24 Previous studies have explored a simplified preprandial dosing strategy and the limits of AID systems regarding unannounced meal disturbances. In 2023, Haidar A. et al. 23 conducted a 3-week follow-up trial in patients with T1D using the iPancreas system for AID. They demonstrated that a simplified “qualitative meal-size estimation” strategy failed to reach noninferiority versus CC. The mean TIR in the study was 74.1% with CC versus 70.5% with the simplified method.
Other trials in adolescents with T1DM using the MiniMed 780G system investigated glycemic control with CC versus simplified meal announcement. 4,6 They showed that the CC group resulted in superior TIR versus the simplified meal announcement group. However, both groups met the international consensus targets for all time points. Our study reconfirmed that the simplified approach achieved appropriate glycemic control in adult T1D open-source AID users (TIR 79.21% ± 4.84%) without increasing hypoglycemia, even in the postprandial period, which represents a viable alternative to CC, particularly in people who find the latter burdensome.
Our study also found that a 26% increased bolus dosage with a 29% basal dosage decrease caused a higher reduction of glycemic rise and provided stable and in-range glycemic values in a closed-loop period. Similar results were reported in T1D children with high GI breakfast of 50% increased prandial insulin, and the simultaneous suspension of basal insulin for 2 h can avoid postprandial hyperglycemia. 25 In another in silico model, the meal bolus amount should be increased by 50%–60% with a subsequent basal decrease. 26,27 Furthermore, in the text of closed loop, the increased bolus strategy changed the time to postprandial peak glucose, which decreased late hypoglycemia occurrence. Faster insulin delivery can explain this, as it compensates for postprandial hyperglycemia with little impact after a postprandial period.
Commercial AID systems are the new standard in diabetes care as they have been shown to improve glycemic outcomes, reduce the burden of care, and likely diminish the risk of long-term diabetic complications. 28,29 However, current commercial systems are either too expensive or unavailable in undeveloped countries. Unlike many high-income countries, CGM and insulin pumps are not funded in China. Despite a lack of regulatory approval, open-source AID is now an affordable alternative popular among people with T1D in China. The 2025 American Diabetes Association (ADA) guidelines recommend supporting and providing diabetes management suggestions for patients with diabetes who opt for open-source AID based on level B evidence. 30 Compared with commercial products, AndroidAPS can offer advanced features, such as super micro bolus (SMB) and unannounced meal (UAM). SMB is an automated bolus correction method that allows for the borrowing of future basal insulin to be included in a bolus. This approach aims to provide a safe and appropriate upfront bolus amount while utilizing reduced temporary basal rates to manage insulin peaks effectively and safely. UAM can automatically detect and handle any sudden glycemic excursion resulting from misestimating CHO intake or omitting CHO input. The CREATE trial supports the efficacy and safety of open-source AID systems; participants had a mean TIR of 12.2% higher using the AID system compared with SAP, comparable with studies examining commercially available AID systems. 31 Our analysis also supported the efficacy and safety of AndroidAPS, even in the context of a simplified prandial bolus strategy.
The study had some limitations that should be acknowledged. The limitations of our study included its relatively short duration and single-center design, as well as the exclusion of patients with high HbA1c. Furthermore, this study was unable to recruit subjects adhering to CC as a control group. This also reflects the need to simplify the current preprandial insulin dosing calculation protocols, given the complexities of managing Chinese meals in real-life situations. In addition, in this study, participants received their insulin administration 10 min before meals. This may differ from the personalized premeal insulin injection timing typically observed in real-world settings, potentially affecting postprandial glycemic outcomes. Since diabetes is a chronic disease and most titration would be done by patients at home, studies evaluating the long-term, multicenter use of a simple strategy for insulin titration are needed.
Despite the limitations, one key strength of the study was that patients with limited experience in CC were included, ideal candidates for the simplified meal announcement strategy. Another key strength of the present study was that it was conducted in the real world among Asian T1D adults, providing generalizable and valuable findings, especially for people with high CHO intake. In addition, in our trial, patients had no prior experience with open-source AID, suggesting that a range of patients with T1D can benefit from this system.
Conclusions
The present study showed a simple bolus strategy without CC in adults with T1D who use open-source AID. This strategy prevents a glycemic excursion and maintains a mean TIR >70%; the mean TIR was significantly higher in the AID period. The simplified meal bolus strategy in the context of current open-source AID could benefit patients who find CC challenging or are reluctant to estimate CHO intake.
Footnotes
Acknowledgments
The authors are grateful to all the patients who took part in the present study and the whole endocrinology and nutrition team that includes doctors, nurses, and medical staff.
Authors’ Contributions
Y.C.: Methodology and writing—original draft preparation. L.M.: Data curation. L.Z.: Project administration. Q.N.: Writing—review and editing. H.S.: Conceptualization and funding acquisition. All authors have read and agreed to the published version of the article.
Funding Information
This work was supported by the First People’s Hospital of Yunnan Province, National Clinical Key Specialty Cultivation Project Platform for Endocrinology (grant number 2024NMKFKT-09) and the Special Project for “Famous Doctor” of Yunnan Ten Thousand Talents Plan (grant number YNWR-MY-2019-020).
Institutional Review Board Statement
The First People’s Hospital Medical Ethics Committee of Yunnan Province approved the study (No: KHLL2022-KY165). Written informed consent was obtained from individuals.
Availability of Data and Materials
The data supporting this study’s findings are available from the corresponding author upon reasonable request.
Author Disclosure Statement
The authors declare no conflict of interest.
Supplementary Material
Supplementary Table S1
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
