Abstract
Background:
Early feasibility studies have demonstrated safe and effective automated insulin delivery use in individuals with suboptimally controlled type 2 diabetes (T2D). The present study investigated MiniMed™ 780G advanced hybrid closed-loop (AHCL) therapy safety and effectiveness in adults with insulin-requiring T2D.
Materials and Methods:
This 13-site, single-arm, open-label study included 95 adults (mean ± standard deviation [SD] age of 60.3 ± 10.8 years and T2D duration of 18.6 ± 8.6 years) using basal-bolus insulin therapy. Participants underwent a run-in period (∼21 days) of open-loop or HCL followed by a study period (∼90 days) of AHCL. The primary safety end point was mean change in glycosylated hemoglobin (HbA1c) from baseline to the end of the 3-month study period. The primary and secondary effectiveness end points were noninferiority and superiority in the percentage of time in range (%TIR 70–180 mg/dL) during the last 6 weeks of the study period (computed by the Hodges–Lehmann method). Safety metrics, including the rates of severe hypoglycemia, diabetic ketoacidosis (DKA), and hyperosmolar hyperglycemic state (HHS), were summarized.
Results:
HbA1c was reduced from 7.9% ± 1.0% (62.4 ± 10.4 mmol/mol) at baseline to 7.2 ± 0.7% (54.7 ± 8.0 mmol/mol) (P < 0.001). The %TIR estimate was 80.9% (95% confidence interval: 78.4%, 83.1%), and the significance criteria for both the primary and secondary effectiveness end points were met (P < 0.001). While total daily insulin dose was increased from run-in to the end of the study (77.4 ± 38.5 U vs. 91.8 ± 49.3 U, P < 0.0001), announced carbohydrates were unchanged, and the number of daily user-initiated boluses was reduced (3.9 ± 1.9 vs. 3.2 ± 1.8, P < 0.0001). There was no significant change in participant weight or body mass index, no severe hypoglycemia, DKA, or HHS, and no serious or unanticipated adverse device effects.
Conclusions:
These findings show that MiniMed 780G AHCL use provides safe insulin intensification in type 2 diabetes and significantly improves mean HbA1c and %TIR.
Introduction
The incidence and burden of type 2 diabetes (T2D) is increasing globally, and it is estimated that approximately 500 million people are living with T2D worldwide. 1 Within the United States, there are at least 38 million people with T2D, 2 and its clinical management involves considerable attention to not only dysglycemia but also comorbidities requiring adherence to multiple and, often, complex pharmacotherapy regimens. Due to the progressive nature of T2D and the side effects or perceived lack of efficacy of some second-line noninsulin glucose-lowering drugs (GLDs), 3 maintaining a recommended glycosylated hemoglobin (HbA1c) goal (<7.0% [53 mmol/mol]) with basal insulin can be challenging. Delaying the initiation of bolus insulin and/or insufficient insulin intensification (i.e., therapeutic inertia) can increase the risk of heart failure, myocardial infarction, stroke, and retinopathy. 4 To prevent or reduce these complications, the American Diabetes Association and European Association for the Study of Diabetes strongly recommend a “person-centered” approach to T2D care that takes into account interventions that work in tandem to address dysglycemia, comorbidities, and behavioral and psychosocial factors that maintain quality of life. 5
Early randomized controlled trials of continuous subcutaneous insulin infusion (CSII) in T2D dysglycemia, like the OpT2mise study, 6,7 were the first to enlist diabetes technology-enabled insulin intensification. The OpT2mise investigators reported CSII intervention reductions in HbA1c with lower total daily doses of insulin along with increased treatment satisfaction 8 compared with basal-bolus multiple daily insulin injection (MDI) therapy. Small feasibility studies, 9 –12 retrospective analysis, 13 and the recent SECURE-T2D pivotal trial 14 provide evidence of safe automated insulin delivery (AID) use in adults with suboptimally controlled T2D and draw similarities with AID therapy improvements in glycemia and quality of life observed 15 –17 and recognized 18 –21 in type 1 diabetes (T1D).
Therapeutic inertia and the concerns of increased burden with basal-bolus therapy and/or additional noninsulin medications often prevent many with T2D from achieving glycemic goals. AID that titrates to dysglycemia has become a sensible “patient-centered” approach, and the MiniMed™ 780G advanced hybrid closed-loop (AHCL) system has been shown to provide glycemic need-dependent insulin intensification (i.e., increased automated correction insulin and increased basal insulin) while lowering the number of daily user-initiated insulin boluses in T1D. 22 –24 As such, the present study evaluated MiniMed 780G system safety and effectiveness in individuals with insulin-requiring T2D, a majority of whom did not meet target HbA1c.
Methods
Study design and participants
The In-Home Study with MiniMed 780G Pump Automated Control in Type 2—Evaluation of the AHCL System in Adults with Insulin-Requiring Type 2 Diabetes (IMPACT2D) pivotal trial began enrollment of adults 18–80 years of age at 13 investigational centers in the United States on February 2, 2022. This open-label, single-arm observational study assessed safety and effectiveness of the MiniMed 780G system (Medtronic, Northridge, CA, USA) with the Guardian™ 4 sensor/Guardian 4 transmitter (Medtronic) and the Accu-Chek® Guide Link blood glucose (BG) meter (Roche Diabetes Care, Inc., Indianapolis, Indiana, USA) and followed the EQUATOR checklist for STrengthening the Reporting of OBservational studies in Epidemiology.
Internal review board approval for each investigational center, in addition to study participant informed consent, was obtained in accordance with the Code of Federal Regulations Title 21, Part 50 and the California Experimental Subject’s Bill of Rights and Health Insurance Portability and Accountability Act, before study start. Investigators and their respective site personnel were trained on the study devices and the study protocol. Training included instructions regarding the transition from MDI therapy to insulin pump therapy and risks associated with hypoglycemia, hyperglycemia, diabetic ketoacidosis (DKA), and hyperosmolar hyperglycemic state (HHS). In addition, the study aligned with the ethical principles of the Declaration of Helsinki.
Study inclusion required a clinical diagnosis of T2D for ≥2 years and use of either MDI therapy (i.e., ≥2 manual daily injections consisting of a basal long-acting insulin and bolus rapid-acting insulin analog) or CSII pump therapy, with or without continuous glucose monitoring (CGM), for ≥3 months before screening. In addition, study participants had to be willing to perform finger-stick BG measurements as needed, wear the investigational system continuously throughout the study, and have a HbA1c of <10%. Exclusion criteria were a history of one of the following within the 6 months before screening: ≥2 episodes of severe hypoglycemia that resulted in medical assistance, coma, or seizure; hospitalization or an emergency room visit related to uncontrolled diabetes diagnosis; DKA; or HHS. Individuals with a positive glutamic acid decarboxylase (GAD) antibody test were excluded. The complete list of inclusion and exclusion criteria is provided (Supplementary Data S1).
Study visits and procedures
There was a run-in period of ∼21 days (Visits 2 through 8) and a study period of 90 days (Visits 9 through 18), and the trial lasted ∼20 months from the initiation of the first investigational center to phase completion (Supplementary Data S2). Briefly, visit 1 included informed consent signing, screening for eligibility, weight measurement, and blood sample collection for HbA1c and other testing. At visit 2, participants completed psychometric questionnaires and were trained on diabetes management principles and the use of study devices and insulin pump features, while using their own insulin lispro (Eli Lilly and Company, Indianapolis, Indiana, USA or Sanofi, Bridgewater, New Jersey, USA) or insulin aspart (Novo Nordisk Inc., Plainsboro, New Jersey, USA). If they previously used CSII (with or without CGM) or a non-Medtronic AID system before study start, the study pump was set to Manual Mode for the run-in period. Participants had the option to use the “suspend before low” or “suspend on low” features. If participants previously used a Medtronic AID system, the study pump was set to closed loop with automated basal function only. Per protocol, noninsulin glucose-lowering drug (GLD) regimens were not titrated before study start and were required to remain stable.
The study period began at visit 9, where automated basal and automated correction were turned on and the glucose target (GT) was set to 120 mg/dL during the first three weeks (Visits 9–14). The active insulin time (AIT) was initially set at 4 h and titrated toward 2–3 h. Between visits 14 and 16, the GT was to be set to 100 mg/dL. For the remaining six weeks of the study period (or any time after visit 16), the GT and AIT were set as per the discretion of the investigator. End-of-study psychometric questionnaires and blood sample collection for HbA1c occurred during the last study period visit (i.e., visit 18).
Safety and effectiveness end points
The primary safety end point was the overall change in mean HbA1c from baseline to the end of the study period (see Statistical analyses). Other safety end points were summarized and included the incidence of adverse events (AEs), serious AEs (i.e., severe hypoglycemia, severe hyperglycemia, DKA, and HHS), serious adverse device effects, and unanticipated adverse device effects.
Primary and secondary effectiveness end points were analyzed for the last 6 weeks of the study period. The primary effectiveness end point was the mean percentage of time in range (%TIR 70–180 mg/dL) compared with a predefined threshold using a noninferiority test, and the secondary effectiveness end point compared the mean %TIR with a predefined threshold using a simple superiority test (see Statistical analyses). Additional end points were analyzed for mean sensor glucose (SG), glucose variability (i.e., standard deviation of mean SG and coefficient of variation [CV] of mean SG), the percentage of time below range (%TBR, <54 mg/dL and <70 mg/dL), time in tight range (%TITR, 70 mg/dL–140 mg/dL), and time above range (%TAR, >180 mg/dL and >250 mg/dL) from baseline/run-in to the end of the study period for the 24-h day, day time (6:00 AM to 11:59 AM) and nighttime (12:00 AM to 5:59 AM). Change in the mean ± SD of daily user-announced carbohydrate, number of user-initiated insulin boluses, total daily insulin dose [TDD] (including total basal and total bolus insulin), and automated correction insulin were also analyzed.
Participant responses to validated psychometric questionnaires completed during run-in and at the end of the study period were summarized. The questionnaires included the Diabetes-39 (D-39) with five dimensions regarding energy and mobility, diabetes control, anxiety and worry, social burden, and sexual function, with items ranked on a 7-point scale (1 “not affected at all” to 7 “extremely affected”); the Diabetes Distress Scale (DDS) with four dimensions regarding emotional burden, physical distress, regimen-related distress, and diabetes-related interpersonal distress composed of 17 items ranked on a 6-point scale (1 “not a problem” to 6 “a very serious problem”); and the hypoglycemia fear survey-II (HFS-II) with two subscales (worry and behavior [i.e., hypoglycemia avoidance or maintaining high glucose]) comprised of 15 and 18 items, respectively, and ranked on a 5-point scale (0 “never” to 4 “almost always”). In addition, the change version of the Diabetes Treatment Satisfaction Questionnaire (DTSQc) was completed at the end of the study and provided a metric of overall treatment satisfaction (from −18 to +18) based on the sum of six items (current treatment satisfaction, convenience, flexibility, understanding of diabetes, treatment recommendation to others, and willingness to continue treatment) ranked on a 7-point scale (“much less […] now” to “much more […] now”). The overall DTSQc and the perceived frequency of hypoglycemia and perceived frequency of hyperglycemia items of the DTSQc (ranging from −3 to +3) asked participants to compare their current treatment experience with that of their previous treatment before beginning the study.
Exploratory analyses
The mean ± SD of CGM-derived metrics, delivered insulin, and system interactions during the run-in and the last six weeks of the study period were stratified by baseline HbA1c (i.e., <7.0% [53 mmol/mol], 7.0%–8.0% [64 mmol/mol], and >8.0%) and subgroups of participants who used and did not use a glucagon-like peptide-1 receptor agonist (GLP-1 RA) or sodium-glucose cotransporter-2 (SGLT-2) inhibitor. The former was based on established literature informing that glycemic outcomes may be baseline glycemic control dependent. The latter investigated the effect of AHCL insulin intensification during concurrent use of specific second-line noninsulin GLDs. The proportion of participants meeting an HbA1c of <7.0%, %TIR of >70%, %TITR of >50%, %TBR 70 mg/dL of <4%, and %TAR 180 mg/dL of <25% were summarized individually and as grouped composites.
Statistical analyses
The primary, secondary, and descriptive end points were evaluated for an intention-to-treat (ITT) population that included all subjects who entered the study period. Adverse events data were evaluated for all subjects who were enrolled. A sample size calculator with one-sample t test showed that a total of 80 subjects would provide >80% power to detect noninferiority and superiority with a significance level of 0.025 (one sided).
For primary and secondary end points, noninferiority and superiority margins were tested against predefined thresholds (e.g., for primary safety end points, the noninferiority margin was set at 0.4% compared with a threshold of 0%). For the %TIR effectiveness end points, the Hodges–Lehmann method was used to compute an estimate and its accompanying 95% confidence interval (CI). Additional end points (including percentages of time spent at sensor glucose ranges) and exploratory analyses were tested for equivalence between the run-in and the study period with a significance level of 0.05 (two sided). For all tests of statistical significance of the difference between baseline or run-in and the end of study period, a one-sample t test was conducted if the normality assumption was met; otherwise, a Wilcoxon signed-rank test was used. Data entry error or nonreasonable values were resolved before data analysis, and no imputation was applied; analysis was conducted on all available data. Descriptive statistics, including questionnaires, were summarized as mean ± SD or median (interquartile range). Statistical analyses were performed using SAS™ 9.4 (SAS Institute, Cary, North Carolina, USA).
Results
The study screened 165 individuals, and there were 58 screen failures primarily due to positive GAD antibody (N = 19) and low hemoglobin (N = 18) diagnoses (Supplementary Data S3). Of the 104 participants who entered the run-in period, there were 9 early exits, 95 who entered the study period, and 89 who completed the study. The baseline demographics and characteristics of the ITT population (N = 95, 60.3 ± 10.8 years of age with T2D duration of 18.6 ± 8.6 years) are listed in Table 1. At baseline, approximately 37% had prior experience with an insulin pump or CGM system.
Participant Demographics and Baseline Characteristics
Data are shown as mean ± SD or number (percentage).
HbA1c, glycosylated hemoglobin; BMI, body mass index; SD, standard deviation; MDI, multiple daily insulin injection; CSII, continuous subcutaneous insulin infusion; CGM, continuous glucose monitoring.
Safety end points
Throughout the study, the overall group spent 91.2% ± 16.8% of the time in AHCL, and there was no severe hypoglycemia, severe hyperglycemia, DKA, or HHS events and no device-related serious or unanticipated adverse device effects (Supplementary Data S4). There were nine serious adverse events, none of which was related to the study procedure or device. Five of the events occurred in the run-in period and four were during the study period. Compared with baseline, AHCL significantly reduced HbA1c from 7.9% ± 1.0% (62.4 ± 10.4 mmol/mol, N = 95) to 7.2% ± 0.7% (54.7 ± 8.0 mmol/mol, N = 88), resulting in a mean change of −0.71% (−7.7 mmol/mol, P < 0.0001).
Effectiveness and additional end points
Overall day glycemic metrics, daily carbohydrate entries, number of insulin boluses, in addition to system-delivered insulin, and weight and body mass index (BMI) for the run-in and study periods are shown in Table 2. For purposes of testing the primary and secondary effectiveness end points, the Hodges–Lehmann method %TIR (70–180 mg/dL) during the study period was 80.9% (95% CI: 78.4%, 83.1%), and significance criteria were met for both the noninferiority test (P < 0.0001) and simple superiority test (P < 0.0001). Compared with run-in, AHCL use during the study period significantly increased mean %TIR from 72.2% to 79.8% (Δ7.2%) and mean %TITR from 41.5% to 50.9% (Δ9.2%) with significant reductions in mean SG (-Δ 9.1 mg/dL) and %TAR (>180 mg/dL [−Δ 7.1%] and >250 mg/dL [−Δ 1.7%]). There was no significant change in %TBR (<70 mg/dL or <54 mg/dL) or CV of SG. The day and nighttime CGM metrics of the overall cohort during the run-in and study periods are also shown (Supplementary Data S5).
Glycemia, Insulin, and Weight during the Run-in and Study Periods
Data are shown as mean ± SD.
Sensor-augmented pump with or without predictive low glucose management or HCL was used. AHCL was inadvertently (temporarily) enabled on 5 systems.
N = 88.
N = 92.
Paired two-sided t-test; otherwise, Wilcoxon signed-rank test.
AHCL, advanced hybrid closed loop; CHO, carbohydrate; CV, coefficient of variation; SG, sensor glucose; SD, Standard deviation; TAR, time above range; TBR, time below range; TIR, time in range; TITR, time in tight range; TDD, total daily insulin dose.
From run-in to the end of the study, TDD was significantly increased, announced carbohydrates did not change, and the number of daily user-initiated insulin boluses were significantly reduced (Table 2). Auto correction boluses accounted for a mean of 13.4% and 34.4% of TDD and bolused insulin, respectively, and there were no significant changes in participant weight or BMI.
Responses to the D-39, DDS, and HFS-II psychometric questionnaires are summarized in Supplementary Data S6. Compared with baseline, end-of-study scores during AHCL use were slightly reduced for all three questionnaires. The end-of-study overall DTSQc and perceived frequencies of hyperglycemia and hypoglycemia scores were reduced and are also shown (Supplementary Data S6).
Exploratory analyses
A comparison of CGM-derived glycemic metrics and delivered insulin stratified by baseline HbA1c is shown in Figure 1. Each subgroup had increased %TIR and %TITR with reductions in mean SG and %TAR during AHCL use, and the extent of automated correction insulin from the system ranged from a mean of 10.5 units to 15.2 units (>30% of total bolus). Study participants with the highest baseline HbA1c (>8.0%) received the greatest amount of auto correction and basal insulin and achieved a %TIR of 76.6%. A majority of this subgroup (77%, 27/35) completed the study with an HbA1c <8.0%.
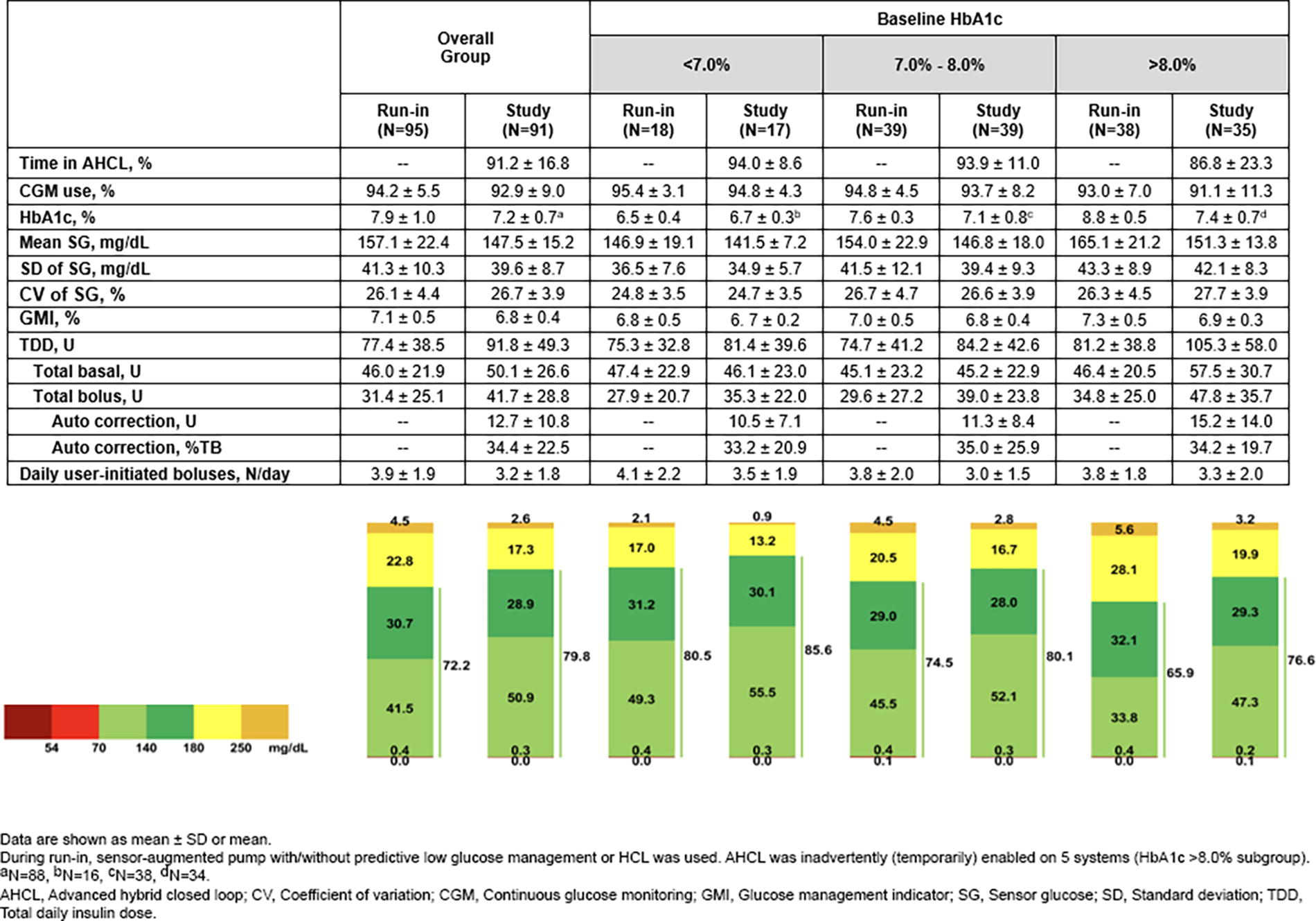
Glycemia and insulin delivery during the run-in and study periods. HbA1c, continuous glucose monitoring-derived metrics, and insulin delivery for the overall group and stratified by baseline HbA1c (from <7.0% [53 mmol/mol] to >8.0% [64 mmol/mol]). The lowest n-tile had within-target glycemic outcomes at run-in (N = 18) that were maintained at study (N = 17). For the highest n-tile, above-target glycemic outcomes during run-in (N = 38) were within target with AHCL use during the study period (N = 35). AHCL, advanced hybrid closed loop; HbA1c, glycosylated hemoglobin.
There was a separate assessment of participants who used GLP-1 RAs or SGLT-2 inhibitors versus those who did not use either (Supplementary Data S7). The mean HbA1c of participants using a GLP-1 RA (N = 17) or SGLT-2 inhibitor (N = 20) was reduced from 7.6% to 7.1% (60.1 mmol/mol to 53.8 mmol/mol) and from 8.1% to 7.3% (64.9 mmol/mol to 56.3 mmol/mol), respectively (Supplementary Data S8). The HbA1c of nonusers (N = 40) was also reduced (from 7.7% to 7.2% [60.8 mmol/mol to 55.0 mmol/mol]). Similar to the overall group, the trend in glycemic control was associated with increased TDD and with no significant change observed in daily carbohydrate, weight, BMI, or %TBR (both <54 mg/dL and <70 mg/dL).
Participants who achieved targets for HbA1c and CGM-derived metrics during the study period versus run-in are shown in Figure 2. During study-period AHCL use, the percentage meeting the HbA1c <7.0% [53 mmol/mol] target was doubled, and there was a greater proportion who met %TIR of >70%, %TITR of >50%, %TAR 180 mg/dL of <25%, and the combined target composites, compared with the run-in period.
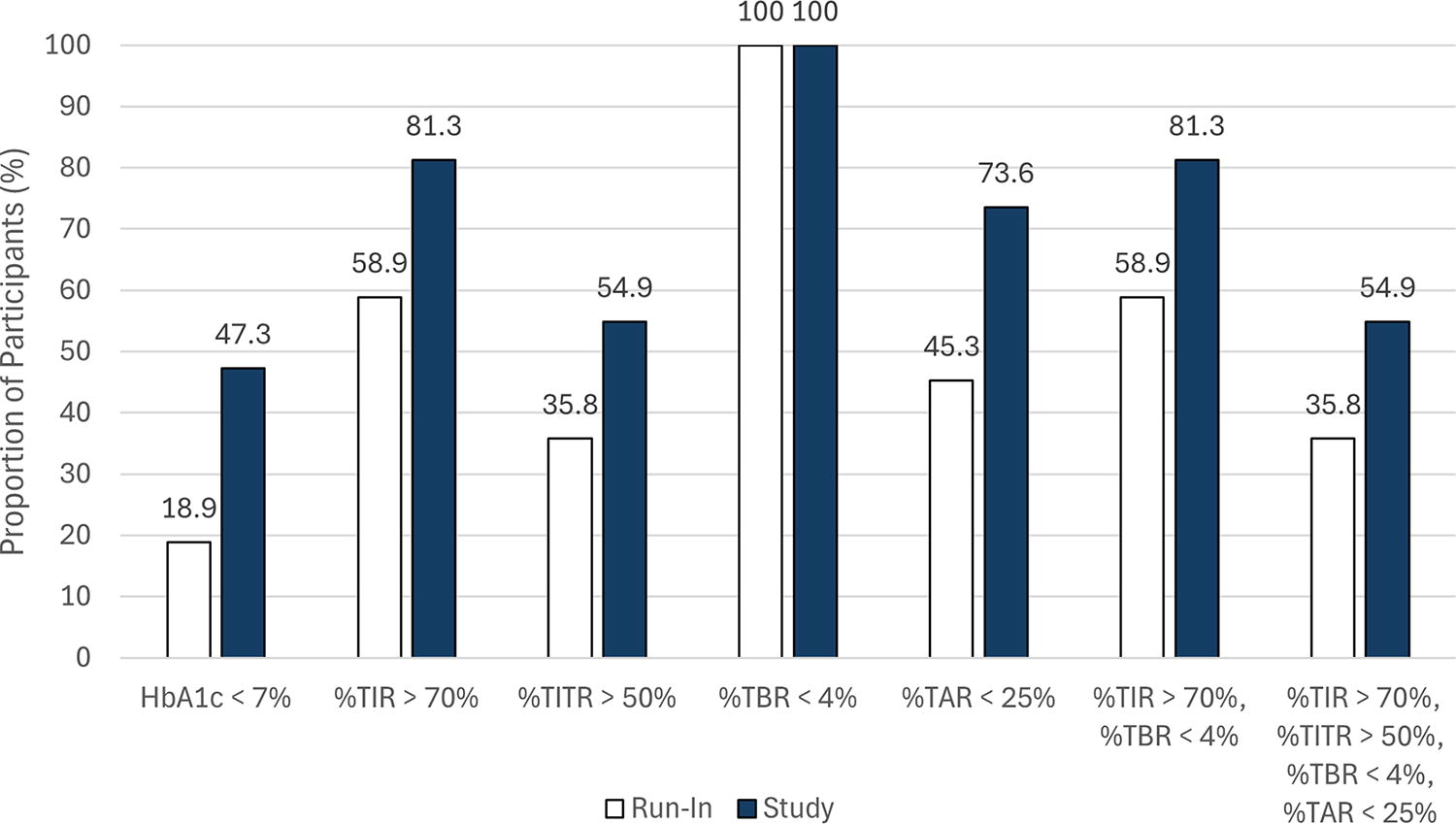
Participant glycemic targets during the run-in and study periods. The proportion of participants at HbA1c and/or continuous glucose monitoring-derived targets is shown for the run-in period (white bars) and the study period (blue bars). N = 95 and N = 88 for HbA1c at run-in and study, respectively. TAR, time above range; TBR, time below range; TIR, time in range; TITR, time in tight range.
Discussion
This pivotal trial of MiniMed 780G AHCL use by more than 90 adults with insulin-requiring T2D demonstrates clinically significant improvement in HbA1c, %TIR, and %TAR with no increase in hypoglycemia exposure or weight and without any episodes of DKA, severe hypoglycemia, or HHS. Improved glycemic control was also observed for the day and night periods. Compared with run-in, 3-month AHCL use allowed more participants to achieve an HbA1c of <7.0% (53 mmol/mol), %TIR >70%, and %TBR 70 mg/dL of <4%. Increased insulin delivery was one important factor in the improved glycemic control, as participants who began the study with a mean HbA1c >8.0% (64 mmol/mol) had a mean TDD that increased by 24.1 units and >75% completed the study with a mean HbA1c <8.0%.
In a subgroup using GLP-1 RAs and SGLT-2 inhibitors, AHCL insulin intensification afforded a reduction in mean HbA1c from 7.6% (60.0 mmol/mol) to 7.1% (54 mmol/mol) and 8.1% (65 mmol/mol) to 7.3% (56 mmol/mol), respectively. A similar reduction in HbA1c and trends on weight and time in hypoglycemia were observed in nonusers (N = 40), demonstrating that AID therapy is additive to the benefits of GLD use. The present work is the largest AID pivotal study in T2D to demonstrate glycemic need-dependent insulin intensification (with auto correction insulin) across participant baseline HbA1c levels.
The Levy et al. multicenter, single-arm feasibility study investigated 6-week use of an AID system, providing both automated basal and automated bolus insulin to adults with T2D (N = 17, aged 54 ± 12 years, median diabetes duration of 13 years, and baseline HbA1c of 8.9 ± 1.3% [74 mmol/mol]) who also used a basal-bolus insulin regimen and continued noninsulin GLDs. 12 Compared to an “open-loop optimization” run-in (2–4 weeks), mean %TIR increased from 61% to 68% and %TITR increased from 31% to 40% with significant reductions in %TAR >180 mg/dL, %TAR >250 mg/dL, and mean SG from 39% to 31%, 6.3% to 2.9%, and 174 mg/dL to 166 mg/dL, respectively. Similar to the present study, TDD was significantly increased, weight did not increase, and there were no episodes of severe hypoglycemia, DKA, or HHS.
The present study findings and Levy et al. show that AID therapy initialization can play a substantial role in managing progressive insulin needs in T2D. This role is more pronounced for systems that can correct for late meal boluses or miscalculated meal carbohydrate. For example, the extent of increased TDD and auto correction insulin delivered across baseline HbA1c level in the present study revealed that insulin insufficiency contributed, in part, to the above-target HbA1c, mean SG, and %TAR observed with open-loop or automated basal only therapy during the run-in. This aligned with the increased TDD and automated boluses comprising more than 50% of total daily boluses in the Levy et al. study participants using either basal only or basal-bolus therapy with or without GLP-1 RA or SGLT-2 inhibitor use. 12 Those authors also observed improvements in dysglycemia that appeared dependent on baseline glycemic control similar to other clinical trials 17,25,26 and real-world data analyses 13,27,28 of AID system use by individuals with T1D.
Another multicenter, single-arm feasibility study investigated outcomes between 2-week standard therapy (ST, with masked or unmasked CGM) and 8-week use of a tubeless AID system with a 26-week extension phase. 29 The population was racially and ethnically diverse, and a subgroup (N = 12, aged 61.8 ± 8.9 years, mean diabetes duration of 20 years, and baseline mean HbA1c 9.4% ± 1.0% [79 ± 11 mmol/mol]) used basal-bolus insulin with or without second-line noninsulin GLDs. Primary end points were change in mean %TAR 250 mg/dL and %TBR 54 mg/dL. By the end of the 26-week extension, improvements in %TAR 250 mg/dL (12.4% vs. 21.5% for ST), %TBR 54 mg/dL (0.02% vs. 0.19% for ST), and HbA1c (8.0% [64.0 mmol/mol] vs. 9.4% [79 mmol/mol] for ST) were maintained. 29 TDD was significantly reduced, weight was unchanged, device satisfaction scores were improved, and there was no DKA or severe hypoglycemia.
A recent multicenter but randomized and controlled feasibility study in 17 adults (aged 63 ± 9 years, mean diabetes duration of 24 ± 9 years, and baseline HbA1c 7.9% ± 0.9% [63 mmol/mol]) investigated 6-week use of an AID system comprising an application (Regulation v2017.04.20; Diabeloop, Paris, France) installed on an Android smartphone that is only available outside of the United States. 30 Compared with open-loop (CSII + CGM) control, AID system intervention resulted in a significantly increased mean %TIR (76.0%) versus 61.0% in control. Similar to the present study, participants had a mean HbA1c of 7.9% (63 mmol/mol) at study start, and most were using either a GLP-1 RA or SGLT-2 inhibitor. Although some found the application difficult to use, diabetes treatment satisfaction between treatments did not differ and there was no severe hypoglycemia or DKA.
While caution is warranted for comparisons drawn between the present study and the AID feasibility studies in T2D given the different study designs, baseline HbA1c levels, and durations of AID intervention, they show that AID in T2D can be safe. It can significantly reduce dysglycemia, as well as psychosocial factors relating to diabetes impact, distress, and fear of hypoglycemia during multiple pharmacotherapy use. In the present study, participants using AHCL also experienced less diabetes management burden as indicated by their reduced number of daily insulin boluses in the wake of improved glycemia. These findings are similar to those seen in T1D compared with MDI therapy 31 –34 or insulin delivery with open- or closed-loop therapy. 22,31,35 –37
As more than half of individuals with T2D have suboptimal adherence to insulin therapy, AID systems may reduce this burden and result in fewer long-term diabetes complications and health care costs. 38,39 There is, also, report of a significant percentage of health care partners (85%) who wish there was an insulin treatment that covered missed doses, yet, hesitancy to intensify insulin treatment given concerns about hypoglycemia. 40 It will be critically important, however, to identify and address the institutional, financial, social, and cultural barriers that limit access to beneficial advanced diabetes technologies like AID in insulin-requiring T2D.
Strengths of this clinical trial are that it demonstrates safe MiniMed 780G AHCL use by more than 90 individuals with insulin-requiring T2D as there was no severe hypoglycemia, DKA, or HHS and no increase in weight or time in hypoglycemia. It includes analyses of automated insulin intensification across ranges of baseline HbA1c and exploratory investigation of AHCL therapy use in GLP-1 RA and SGLT-2 inhibitor users and nonusers that revealed the important contribution of automated correction insulin to these regimens. Limitations of the present study include its nonrandomized design without a control group and the comparison of 3-month AID intervention to a run-in period of shorter duration. That comparison, in which run-in included automated basal insulin delivery and sensor-augmented pump use (with or without a “suspend before low” or “suspend on low” feature), may have resulted in an underestimation of the AHCL effect when factored alongside the study inclusion criterion for baseline HbA1c (<10% [86 mmol/mol]). It is, also, important to note that 11.5% of potential study participants were excluded because they were GAD positive. We recommend that future studies of AID use in people with T2D conduct similar screening to preclude study population dilution with latent autoimmune diabetes of adults (or LADA) who may respond more like individuals with T1D. If future trials include GAD-positive individuals, secondary analyses comparing AHCL outcomes of that cohort versus those of a non-GAD cohort may be very informing.
Conclusions
Safe AID therapy use in T2D is growing, and improved glycemic and psychosocial outcomes are mirroring those observed in people living with T1D. Use of AID systems, such as the MiniMed 780G system, may mitigate clinical inertia in T2D and safely facilitate insulin intensification when glycemic targets are not achieved with insulin or insulin with noninsulin GLD use.
Footnotes
Acknowledgments
The authors gratefully thank the study participants and their families for their time and contributions and the investigational staff and personnel for their support. Medtronic Diabetes obtained the license (HPR 462) for use of the Diabetes Treatment Satisfaction Questionnaire (change) form, which is owned by Professor Clare Bradley and licensed by Health Psychology Research Ltd. (![]() ). At the time of article submission, the MiniMed 780G system is investigational and not approved for use in T2D by the Food and Drug Administration or other regulatory bodies. The MiniMed 780G system includes technology developed by DreaMed Diabetes (Petah Tikvah, Israel).
). At the time of article submission, the MiniMed 780G system is investigational and not approved for use in T2D by the Food and Drug Administration or other regulatory bodies. The MiniMed 780G system includes technology developed by DreaMed Diabetes (Petah Tikvah, Israel).
Authors’ Contributions
The principal investigator authors made substantial contributions to the acquisition and interpretation of data. The
IMPACT2D Study Group
Atlanta Diabetes Associates (Atlanta, Georgia, USA): Bruce W. Bode, MD, FACE. East Coast Institute for Research (Canton, Georgia, USA): Jason Berner, MD. East Coast Institute for Research (Jacksonville, Florida, USA): David Sutton, MD. East Coast Institute for Research (Macon, Georgia, USA): Thomas C. Jones, MD, FACE. Endocrine & Metabolic Consultants (Rockville, Maryland, USA): Michael A. Dempsey, MD. Endocrine Research Solutions, Inc. (Roswell, Georgia, USA): John “Chip” Reed, MD. International Diabetes Center, HealthPartners Institute (Minneapolis, Minnesota, USA): Richard M. Bergenstal, MD; Anders L. Carlson, MD; Molly J. Carlson, MD; and Thomas W. Martens, MD, FACP. Iowa Diabetes (West Des Moines, Iowa, USA): Anuj Bhargava, MD, MBA, CDCES, FACP, FACE. Arkansas Diabetes & Endocrinology Center (Little Rock, Arkansas, USA): Katherine R. Cameron, PA-C; Loran Seib, RN; Lesa Thrasher, RRT, CCRP; and James R. Thrasher, MD, FACE.
Prior Presentation or Publication
Portions of data were presented at the 84th Scientific Sessions of the American Diabetes Association (June 21–24, 2024 in Orlando, Florida, USA), the 60th European Association for the Study of Diabetes Annual Meeting (September 10–13, 2024, Madrid, Spain), and the Advanced Technologies & Treatments for Diabetes: 1st Asian Conference on Innovative Therapies for Diabetes Management (November 18–20, 2024, Singapore).
Author Disclosure Statement
A.B. has received grants or research support, via Iowa Diabetes Research, from Abbott Diabetes Care, AbbVie, Inc., Akero Therapeutics, Barbara Davis Center for Diabetes, Boehringer Ingelheim Pharmaceuticals, Inc., Carmot Therapeutics, Covance, Inc., Dexcom, Inc., Eli Lilly and Company, Gasherbrum Bio, Inc., Insulet Corporation, IQVIA, Inc., Kowa Pharmaceuticals America, Inc., Madrigal Pharmaceuticals, Inc., MannKind Corporation, Medtronic plc, Novo Nordisk A/S, Thermo Fisher Scientific, Inc., Viking Therapeutics, vTv Therapeutics LLC, Zydus Pharmaceuticals, and 89Bio, Inc. R.M.B. has received research support, acted as a consultant, or been on the scientific advisory board for Abbott Diabetes Care, Ascensia Diabetes, CeQur Corporation, Dexcom Inc., Eli Lilly and Company, Embecta Corp., Hygieia, Insulet Corporation, Medtronic plc, Novo Nordisk A/S, Verily, Roche Diabetes Care, Sanofi, Tandem Diabetes Care, United Healthcare, Vertex Pharmaceuticals, and Zealand Pharma. R.M.B.’s employer (the nonprofit HealthPartners Institute) contracts for his services, and he receives no direct personal income from these activities. M.L.W. has served on the advisory board of Eli Lilly and Company and on the speaker’s bureau of Abbott Diabetes Care, Amgen Inc., and Eli Lilly and Company and received research support from Amgen Inc., Amolyt Pharma, AstraZeneca, Bayer AG, Biomea Fusion Inc., Calcilytix Therapeutics, Inc., Medtronic plc, Mineralys Therapeutics, Inc., Novo Nordisk A/S, and Novartis AG. J.R.T. has received grants, research support, or speaking fees from AstraZeneca, Eli Lilly and Company, Inversargo Pharma, Medtronic plc, Merck & Co., and Novo Nordisk A/S and has served as an advisory board member and consultant for Medtronic plc. M.A.D. has received research support from Eli Lilly and Company, Medtronic plc, and Novo Nordisk A/S. B.W.B. has stock in Aseko LLC; served as a speaker for Abbott Diabetes Care, Eli Lilly and Company, MannKind Corporation, Medtronic plc, Novo Nordisk A/S, Sanofi, and Xeris Pharmaceuticals, Inc.; consulted for Beta Bionics, MannKind Corporation, Medtronic plc, and Novo Nordisk A/S; and received grant or research support via his employer (Atlanta Diabetes Associates), from Abvance Therapeutics, Abbott Diabetes Care, Bayer AG, Carmot Therapeutics, Dexcom Inc., Diasome Pharmaceuticals, Inc., Dompé Farmaceutici S.p.A., Eli Lilly and Company, Insulet Corporation, MannKind Corporation, Medtronic plc, Novo Nordisk A/S, REMD Biotherapeutics, Sanofi, and vTv Therapeutics LLC. J.L. has received research support from Medtronic plc. A.L.C. has served as a consultant or advisory board member for Insulet Corporation, MannKind Corporation, Novo Nordisk A/S, and Zealand Pharma and served as a clinical research investigator for Abbott Diabetes Care, Companion Medical, Dexcom Inc., Eli Lilly and Company, Insulet Corporation, Medtronic plc, Novo Nordisk A/S, Sanofi, Tandem Diabetes Care, and United Health Inc. A.L.C.’s employer (the nonprofit HealthPartners Institute) contracts for his services, and he receives no direct personal income from these activities.
Data Availability Statement
The data generated or analyzed for the present study are included in the published article and its online supplementary files.
Funding Information
This study (Clinicaltrials.gov ID NCT05238142) was funded by Medtronic Diabetes (Northridge, California, USA). The investigator authors received research support from Medtronic to conduct the study at each of their centers.
Supplementary Material
Supplementary Data S1
Supplementary Data S2
Supplementary Data S3
Supplementary Data S4
Supplementary Data S5
Supplementary Data S6
Supplementary Data S7
Supplementary Data S8
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
