Abstract
Objective:
To evaluate real-world outcomes in adults with type 1 diabetes initiating open-source automated insulin delivery systems (OS-AID).
Methods:
Adults with type 1 diabetes who commenced OS-AID, between May 2016 and April 2021, across 12 centers in the United Kingdom were included. Anonymized clinical data, collected during routine clinical care between December 2019 and November 2023, were submitted to a secure web-based tool within the National Health Service network. Outcomes included change in hemoglobin A1c (HbA1c), sensor glucometrics, diabetes distress score, Gold score (hypoglycemia awareness), user opinion of OS-AID, and event rates (hospital admissions, paramedic callouts, severe hypoglycemia, and adverse events) between baseline and follow-up.
Results:
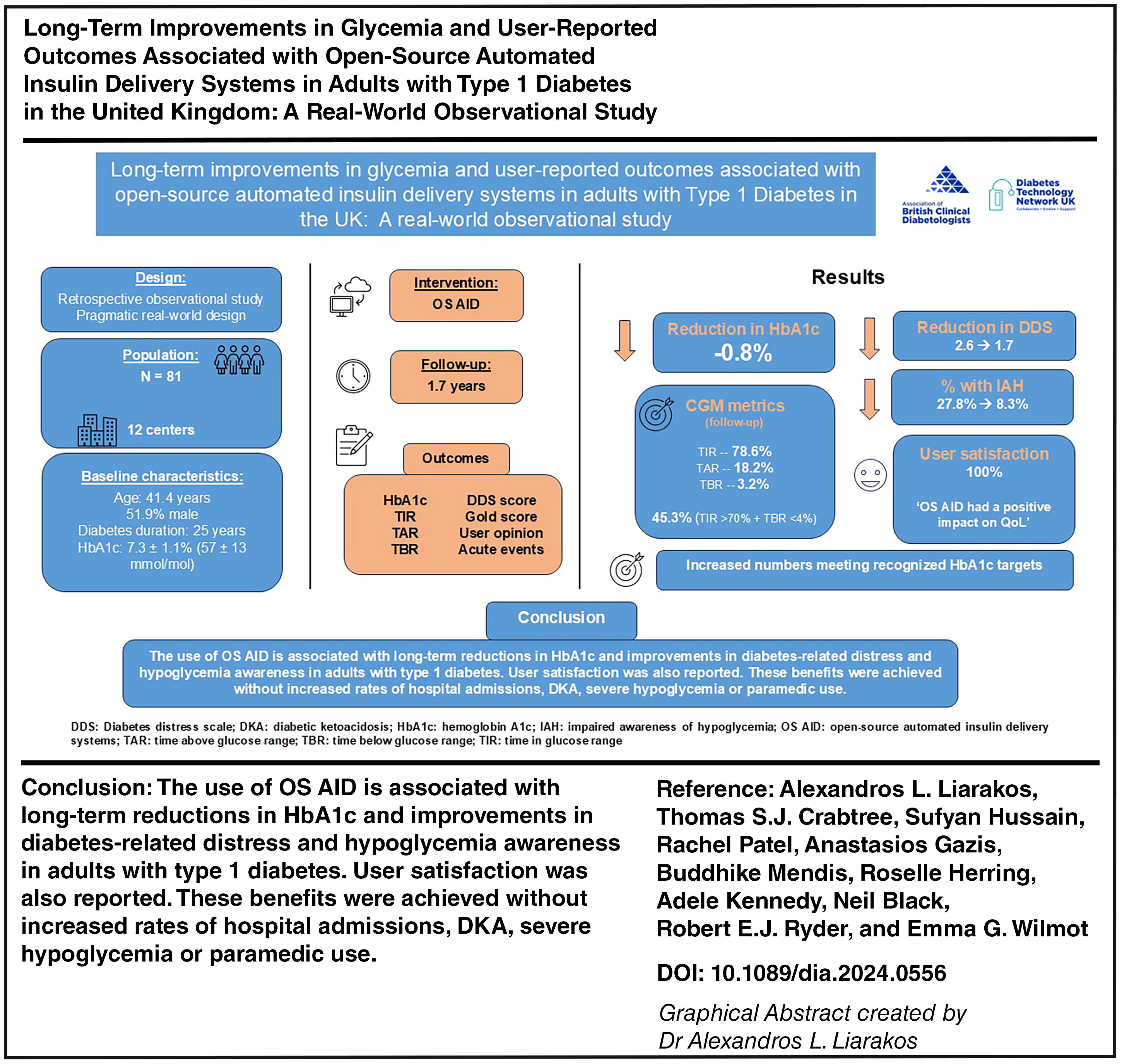
In total, 81 OS-AID users were included (51.9% male; 90.1% White British; mean age 41.4 years; median diabetes duration 25 years [IQR 17–32]). Over a mean follow-up of 1.7 years, HbA1c reduced by 0.8% (9 mmol/mol) (7.3 ± 1.1% vs. 6.5 ± 0.7%; P < 0.001), and the percentage of individuals achieving HbA1c ≤ 7.0% (53 mmol/mol) increased from 48.6% to 75.7% (P < 0.001). Diabetes-related distress score reduced by 0.9 (95% confidence interval [CI] −0.3, −1.5; P = 0.006), and Gold score reduced by 0.7 (95% CI −0.1, −1.3; P = 0.022). The percentage of individuals with impaired hypoglycemia awareness (Gold score ≥4) reduced (27.8% at baseline vs. 8.3% at follow-up; P = 0.039). Of those asked, all participants stated that OS-AID had a positive impact on quality of life. The number of hospital admissions was low.
Conclusions:
The use of OS-AID is associated with long-term improvements in HbA1c, hypoglycemia awareness, and diabetes-related distress in type 1 diabetes. These benefits were achieved without increased rates of hospital admissions, diabetic ketoacidosis, or severe hypoglycemia.
Introduction
Type 1 diabetes is a complex chronic condition that places significant demands on the individual living with the disease. Optimal glucose outcomes, while central to minimizing the future risks of micro and macrovascular events, can place a significant burden on the individual living with diabetes. Throughout the day, there is a recurrent need to check glucose, calculate carbohydrate intake, and administer insulin. Despite best attempts, and as a result of the complexity of the disease, few achieve the definition of optimal glucose outcomes. A minority of people with diabetes achieve the international time in range (TIR) recommendations of >70% time between 3.9 and 10 mmol/L (70–180 mg/dL) combined with less than 4% in hypoglycemia. 1 This is a source of frustration and impacts on long-term progression toward complications. Many people with type 1 diabetes feel that they are failing with their diabetes management and/or report feeling overwhelmed by the demands of living with diabetes. 2
Automated insulin delivery (AID) systems, also known as artificial pancreas systems (APS) or closed-loop systems, have revolutionized the care of people with type 1 diabetes. These systems consist of a real-time continuous glucose monitoring device, an insulin pump, and an algorithm that computes and regulates insulin delivery via the pump based on continuous glucose monitoring (CGM)-captured glucose levels. Evidence from randomized trials and real-world observational studies has shown that commercially available AID systems can support people to increase TIR without increasing hypoglycemia, reduce diabetes-related distress, and improve quality of life (QoL). 3 –5 As a result, commercially approved AID therapies are now recommended for the care of people with type 1 diabetes who can use the devices safely. 6,7
Before the introduction of commercially approved AID systems, online communities, consisting of people living with type 1 diabetes and their loved ones, founded the #WeAreNotWaiting movement and developed open-source (OS) AID systems (OS-AID), also known as do-it-yourself (DIY) APS. 8 These communities have produced detailed and accessible resources on how commercially available and approved medical devices, such as CGM and insulin pumps, can be connected and remotely controlled via open-source, off-label, and unregulated algorithms to automate insulin delivery.
Qualitative studies suggest that OS-AID users improve their QoL and feel empowered by the ability to customize their systems. 9,10 A randomized controlled trial (RCT) showed that the use of an OS-AID system significantly improved glycemia without increasing the rates of severe hypoglycemia or diabetic ketoacidosis (DKA) in type 1 diabetes population. 11 These findings are also supported by real-world studies, with the caveat of typically small sample sizes, short follow-up duration, and often self-reported data. 12
Historically, health care professionals (HCPs) have expressed their concern that legal or regulatory body actions could ensue if they support individuals who opt for using OS-AID. A survey of HCPs indicated the challenges experienced by both HCPs and people with type 1 diabetes when discussing about OS-AID. 13 Although an individual’s right to use OS-AID was recognized in the 2020 Diabetes UK’s position statements, the authors could not recommend the use of such therapies due to the lack of regulatory body approval and published research to support safety or effectiveness. 14 In the 2022 international consensus statement and practical guidance on OS-AID, although the use of open source over commercial AID therapies was not universally recommended, the authors suggested that the best interest of the person living with type 1 diabetes should be balanced against the risks of using OS-AID. 15
Despite the availability of commercially available AID systems in some developed countries, the use of OS-AID continues. It is estimated that over 10,000 people with type 1 diabetes manage their condition with OS-AID globally. 15 HCPs play a vital role in helping and supporting people with type 1 diabetes to achieve beneficial outcomes from AID therapies. In response to the previously highlighted concerns regarding the use of OS-AID, we designed an observational study to provide HCPs with insights into the effect of OS-AID in routine clinical practice. The aim of this study was to evaluate long-term, real-world, clinical, and user-reported outcomes associated with OS-AID in adults with type 1 diabetes in the United Kingdom.
Methods
Patient recruitment and data collection
Data for this observational study were obtained from the Association of British Clinical Diabetologists (ABCD) DIY APS audit tool (http://www.diabetologists-abcd.org.uk/APS/DIY-APS_Audit.htm), which aimed to capture real-world outcomes from nonpregnant adults, attending diabetes services with a diagnosis of type 1 diabetes managed with an insulin pump, who initiated OS-AID between May 2016 and April 2021. Anonymized clinical data were collected at baseline and follow-up during routine clinical care, and clinical systems and electronic health records were reviewed and submitted to a secure web-based tool within the National Health Service (NHS) network. Data collection was performed between December 2019 and November 2023.
Outcome measures
The primary outcome was change in laboratory-derived hemoglobin A1c (HbA1c) between baseline and follow-up. Secondary outcomes included event rates (hospital admissions, paramedic callouts, severe hypoglycemia requiring third-party assistance, and adverse events), two-item diabetes distress screening instrument (question 1 “feeling overwhelmed by the demands of living with diabetes”; question 2 “feeling that I am often failing with my diabetes regimen”), 16 Gold score to assess hypoglycemia awareness, 17 30-day sensor glucometrics (TIR [3.9–10 mmol/L or 70–180 mg/dL], time below range [TBR; <3.9 mmol/L or <70 mg/dL], time above range [TAR; >10 mmol/L or >180 mg/dL]), and user opinion of OS-AID (question 1 “would you recommend DIY APS to other people with diabetes?” and question 2 “what impact would you rate DIY APS has had on your quality of life?”; a 7-point Likert scale was used: 1 = would not recommend at all, 7 = would highly recommend [for question 1]; 1 = extremely negative impact, 7 = extremely positive impact [for question 2]). Demographic data included weight, gender, ethnicity, and education level. Data on OS-AID, insulin pumps, and CGM used as well as funding resources for these systems were reported. Follow-up frequency was determined by the responsible clinical team based on clinical need. Data were captured at baseline (for the 12 months prior to OS-AID initiation) and at follow-up (during routine clinical follow up) and can be shared on reasonable request to the corresponding author.
Statistical methods
Continuous variables were presented as mean ± standard deviation (SD) or median and interquartile range (IQR) and assessed using paired t-test or Wilcoxon signed-rank test depending on normality of distribution (determined by Shapiro–Wilk test and Kolmogorov–Smirnov test). Categorical variables were expressed as numbers and percentages and assessed using Chi-squared tests. To address loss to follow-up, the analysis of each outcome of interest included only individuals who had available data at both baseline and follow-up. A two-sided P value <0.05 was considered statistically significant. Statistical analysis was performed on SPSS v26.0 (IBM, Chicago, IL). A.L.L. and T.S.J.C. are the guarantors of this work and, as such, had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Results
Baseline data were available for 102 adults, with follow-up data reported for 84 (82.4%), of whom 96.4% (81 of 84) continued to use OS-AID across 12 centers in the United Kingdom. The mean ± SD follow-up was 1.7 ± 1.2 years. Supplementary Data S1 contains the flow diagram for this analysis.
Baseline characteristics
For the 81 individuals who had baseline and follow-up data and continued to use OS-AID at follow-up, the mean ± SD age was 41.4 ± 8.8 years; 51.9% (n = 42) were male and 90.1% (n = 73) were White British; median diabetes duration was 25 years (IQR 17–32) and insulin pump use was 7 years (IQR 4–10). Among those with available data on education level (n = 34), 76.5% (n = 26) had higher education status (degree level). The baseline characteristics of the study population are summarized in Table 1.
Baseline Characteristics of Total Population (n = 81)
APS, artificial pancreas system; HbA1c, hemoglobin A1c; IQR, interquartile range; OS-AID, open-source automated insulin delivery systems; SD, standard deviation.
The OS-AID used included AndroidAPS (54.3%, n = 44), Loop (24.7%, n = 20), and OpenAPS (12.3%, n = 10); the system was not recorded for seven (8.7%) individuals. The types of insulin pump used were recorded for 44 individuals and included Dana RS or Dana R (34.1%, n = 15); Roche Combo or Roche Insight (31.8%, n = 14); Omnipod (15.9%, n = 7); Medtronic Paradigm Veo 554 or 754 (9.1%, n = 4); and other pumps (9.1%, n = 4); 86.4% (n = 38) of these insulin pumps were funded via the NHS and 20.5% (n = 9) were out of warranty. Data on the type of CGM used and funding resources were recorded for 49 individuals (FreeStyle Libre with MiaoMiao: 44.9% [n = 22]; Dexcom G6: 34.7% [n = 17]; FreeStyle Libre 2: 20.4% [n = 10]; 71.2% funded via NHS).
Glycemic outcomes
HbA1c reduced from 7.3 ± 1.1% (57 ± 13 mmol/mol) at baseline to 6.5 ± 0.7% (48 ± 8 mmol/mol) at follow-up, a mean reduction of 0.8% (9 mmol/mol) (95% confidence interval [CI] −0.6, −1.0; P < 0.001, n = 70), over a mean follow-up of 1.7 years. These results were observed without an increase in body weight (83.9 ± 22.0 kg at baseline vs. 84.7 ± 21.2 kg at follow-up; P = 0.115) and are demonstrated in Figure 1A and Table 2. The percentage of people, with both baseline and follow-up HbA1c data available (n = 70), who achieved an HbA1c ≤ 6.5% (48 mmol/mol) was 21.4% (15/70) at baseline versus 51.4% (36/70) at follow-up (P < 0.001). An HbA1c ≤ 7.0% (53 mmol/mol) was achieved by 48.6% (34/70) of the cohort at baseline versus 75.7% (53/70) at follow-up (P < 0.001) (Fig. 2).
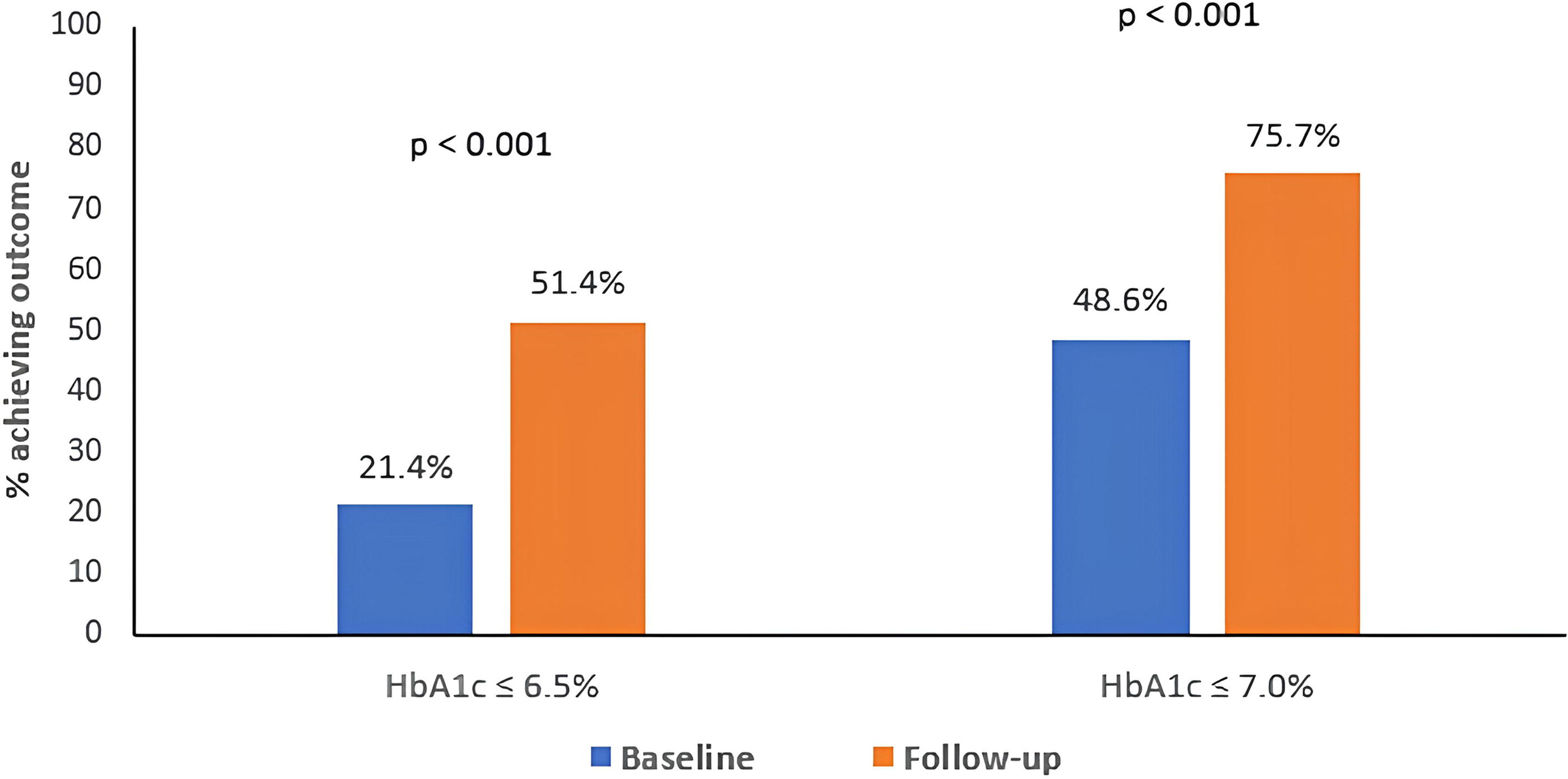
Proportion of individuals achieving targets for HbA1c at baseline and follow-up.
Baseline and Follow-up HbA1c, Weight, and Patient-Reported Outcomes
Data are mean ± SD unless otherwise indicated.
Number of individuals with available paired data at baseline and follow-up included in analysis for a given outcome; total cohort = 81.
DDS, diabetes distress scale; IAH, impaired awareness of hypoglycemia.
Stratified by baseline HbA1c, individuals with baseline HbA1c < 7% (53 mmol/mol) (n = 28) had a mean reduction of 0.4% (5 mmol/mol) in HbA1c, from 6.5 ± 0.3% at baseline to 6.1 ± 0.5% at follow-up (P < 0.001). Patients with baseline HbA1c between 7% (53 mmol/mol) and 8% (64 mmol/mol) (n = 29) experienced a mean reduction in HbA1c by 0.8% (8 mmol/mol) (7.4 ± 0.3% vs. 6.6 ± 0.6%, P < 0.001), and individuals with baseline HbA1c > 8% (64 mmol/mol) (n = 13) had a mean reduction of 1.8% (20 mmol/mol) in HbA1c (9.1 ± 1.3% vs. 7.3 ± 0.6%, P < 0.001) over the follow-up period (Fig. 1B).
Once the population with paired baseline and follow-up HbA1c data available was stratified by OS-AID system used (n = 65, missing data: 7.1%), HbA1c reduced by 0.8% (8 mmol/mol) in AndroidAPS users (P < 0.001; n = 37), 0.6% (7 mmol/mol) in Loop users (P < 0.001; n = 20), and 1.1% (12 mmol/mol) in OpenAPS users (P = 0.05; n = 8).
CGM metrics were reported in 53 individuals at follow-up. TIR was 78.6 ± 11.8%, TAR was 18.2 ± 12.5%, and TBR was 3.2 ± 2.0%. The target of TIR >70% and TBR <4% was achieved by 45.3% (24/53) of the cohort. No analysis comparing baseline versus follow-up data was performed due to the small number of CGM metrics at baseline.
Diabetes distress score, Gold score, and user satisfaction
There was improvement in mean DDS score, which decreased from 2.6 at baseline to 1.7 at follow-up, a mean reduction of 0.9 (95% CI −0.3, −1.5; P = 0.006; n = 24). Gold score reduced from 2.7 to 2.0, a mean reduction of 0.7 (95% CI −0.1, −1.3; P = 0.022; n = 36) over the follow-up period. The percentage of individuals with impaired awareness of hypoglycemia (Gold score ≥4) reduced from 27.8% at baseline to 8.3% at follow-up (P = 0.039). These results are shown in Table 2. Of those asked to rate on a 7-point Likert scale (n = 39), 100% stated that OS-AID had a positive impact on their QoL and would recommend the system to other people with diabetes (average score of 7 in Likert scale for both questions).
Hospital admissions, paramedic use, and acute and adverse events
The number of hospital admissions was low. Compared with seven hospital admissions (hypoglycemia: one; diabetes-related complications other than hyperglycemia/DKA or hypoglycemia: six) in the 12 months prior to OS-AID, a total of three admissions (hypoglycemia: two; hyperglycemia and/or DKA: one) were reported at follow-up (P = 0.48). Paramedic callouts without resulting in admission were not reported at either baseline or follow-up. Among those individuals (n = 60) with both baseline and follow-up data on severe hypoglycemic episodes, 10.0% (6/60) reported ≥1 event (10 events in total) at baseline, compared with 3.3% (2/60; 3 events in total) at follow-up (P = 0.2).
A total of three users (3.6%) discontinued OS-AID (Supplementary Data S1). Reasons for discontinuation were reported in one individual and included inappropriately increased insulin delivery due to app interference, which did not result in severe hypoglycemia, paramedic callout, or hospital admission.
Discussion
This real-world evaluation of OS-AID use in adults with type 1 diabetes demonstrates significant long-term improvements in HbA1c, diabetes-related distress, hypoglycemia awareness, and user satisfaction over 1.7 years of follow-up. Hospital admissions related to hypoglycemia, hyperglycemia, or DKA were small in number, and severe hypoglycemic episodes did not increase. Only a small proportion of OS-AID users discontinued the AID system. This analysis indicates that the use of OS-AID in the real world was associated with glucose outcomes, which were comparable to real-world data from commercially available systems. 18 –20
The reduction in HbA1c of 0.8% (9 mmol/mol) across all OS-AID systems in this analysis is consistent with the results of two other observational studies with smaller sample size and shorter follow-up duration. 21,22 Specifically, Jeyaventhan et al. (n = 30) and Patel et al. (n = 35) showed that OS-AID were associated with a significant reduction in HbA1c of 0.9% at 6 months and 1.4 years, respectively. 21,22 Hence, our study suggests sustained improvements in glycemia with OS-AID use.
Stratified by the specific system used, the AndroidAPS users in our study experienced a clinically meaningful reduction in HbA1c of 0.8% (8 mmol/mol), which was similar to the changes observed in a RCT and three observational studies with self-reported and objective outcomes in adults with type 1 diabetes who used such a system. 11,23 –25 It should be noted that AndroidAPS uses the OpenAPS algorithm and an Android phone as a controller, compared with OpenAPS, which uses a separate microcontroller. 26 OpenAPS users in our study experienced a significant reduction in HbA1c of 1.1% (12 mmol/mol); however, these results should be interpreted with caution given the relatively small number of people using this system in our analysis. Nevertheless, the benefits of OpenAPS on glycemia have been described in the real-world analyses of Choi et al. and Melmer et al., which showed a significant reduction in HbA1c ranging between 0.4% and 0.5%. 27,28 HbA1c was significantly reduced by 0.6% (7 mmol/mol) in the Loop users in our analysis, with similar results reported in previous observational studies with self-reported and objective outcomes. 29,30
Our study indicated that individuals with higher HbA1c levels at baseline achieved the greatest reduction in HbA1c after initiating OS-AID. Similar observations were described in the study of Wu et al. 23 This suggests that HbA1c levels significantly above target should not be a restrictive factor for supporting the implementation of OS-AID in people who can use the devices safely.
Our data showed that OS-AID were associated with user satisfaction and significant long-term improvements in diabetes-related distress and hypoglycemia awareness. These results are in line with the findings from previous studies that showed improvements in QoL, diabetes distress, fear of hypoglycemia, and sleep quality with OS-AID. 23,31 Also, a qualitative study including Loop users highlighted the benefits of OS-AID in reducing the behavioral or mental burden related to diabetes care, with notable improvements observed, especially in overnight glycemia and sleep quality. 10 However, the findings observed in our study should be interpreted cautiously given the low number of individuals that answered the questions related to patient-reported outcomes.
Potential safety issues for OS-AID have been an area of concern for both HCPs and regulatory bodies. 32,33 Our real-world data showed that OS-AID did not increase the number of paramedic use or admissions related to hypoglycemia, hyperglycemia, or DKA, with similar outcomes among the main OS system subtypes. Similarly, no episodes of severe hypoglycemia or DKA were reported in the 6-month RCT or previous real-world studies. 11,21,22 Also, our analysis indicated that OS-AID were associated with a nonsignificant reduction in severe hypoglycemic episodes. However, the small numbers included in the study limit the conclusions that can be drawn from these data. Hence, vigilance for OS-AID-related adverse events will need to be maintained.
The main strength of this study includes the real-world setting that enabled the collection of data from adults with type 1 diabetes managed with commonly used OS-AID subtypes in routine clinical practice across multiple diabetes centers in the United Kingdom. The real-world nature of our analysis provides observations that are more representative of the usual care for OS-AID users in a public health system, without restrictive inclusion and exclusion criteria commonly used in randomized trials (e.g., exclusion of high-risk groups). Also, our analysis involves 81 OS-AID users, which is one of the biggest samples reported in relevant observational studies in the literature. Our population was balanced in terms of gender, including similar proportion of male and female individuals. In contrast to some previous observational studies, the data reported here were objective (HbA1c, TIR, admissions, etc.) rather than self-reported. Another key strength is the long-term follow-up period of 1.7 years, which allowed for evaluation of the durability of clinical and patient-reported outcomes observed beyond the initial treatment period. To the best of our knowledge, there are no other studies evaluating the effects of all the main subtypes of OS-AID with longer follow-up than our analysis.
The study should be interpreted within the context of its limitations. The inherent limitation of its retrospective design along with a lack of control group can introduce the risk for selection bias. Other limitations include the possibility of unmeasured confounders due to data collection via an audit tool, underreporting of adverse events, and lack of information about the percentage of time that OS-AID were in closed-loop mode during follow-up. Missing data (e.g., 13.6% of total population did not have HbA1c data available at both baseline and follow-up), loss of follow up in 17.6% (n = 18) of people with baseline data available including lack of information about OS-AID continuation or discontinuation during follow-up, and lack of information about follow-up frequency of the patients are some other limitations. We also acknowledge that the study population was predominantly of White British ethnicity and high education status, which limits the generalizability of our findings to other racial and ethnic populations or individuals with lower education level. Lastly, our study would have benefited from reporting changes in CGM metrics, which was not possible due to the small number of sensor metrics available at baseline.
Conclusions
In conclusion, our analysis demonstrated that OS-AID were associated with substantial longitudinal reductions in HbA1c and improvements in diabetes-related distress in adults with type 1 diabetes in the United Kingdom. These benefits were achieved without increased rates of hospital admissions, DKA, severe hypoglycemia, or paramedic use. However, given the limitations previously described, these findings should be interpreted with caution. Nevertheless, our findings suggest long-term benefits of OS-AID and support the use of such systems in motivated individuals demonstrating a high level of self-care.
Footnotes
Acknowledgments
The authors would like to thank Dr. Rowan Hillson, MBE, and the Association of British Clinical Diabetologists Executive Committee for their enthusiasm and support to help the team develop a more detailed understanding of the impact of OS-AID on the clinical outcomes in people living with type 1 diabetes. Also, the authors would like to thank all the health care professionals and support staff who participated in the audit. Specifically, we are thankful to Melissa Louise Cull (Sandwell and West Birmingham NHS Trust, City Hospital), Linn Langeland, Peter Jennings (University Hospitals of Derby and Burton NHS Foundation Trust, Royal Derby Hospital), Dennis J. Barnes (Maidstone and Tunbridge Wells NHS Trust, Kent and Sussex Weald Diabetes Center), Lisa Goodman, Paul Lambert, Gita Modgil (Taunton and Somerset NHS Foundation Trust, Musgrove Park Hospital), Alexandra Ward (Royal United Hospitals Bath NHS Foundation Trust, Royal United Hospital), Chloe Humphries (County Durham and Darlington NHS Foundation Trust, Darlington Memorial Hospital), Rachael Bealey (York and Scarborough Teaching Hospitals NHS Foundation Trust, York Hospital site), Lisa Forrest (Hywel Dda Health Board, Glangwili Hospital), Sara Hartnell (Cambridge University Hospitals NHS Foundation Trust, Addenbrooke’s Hospital), Cintia Gonzalez, Rosa Corcoy (Hospital Santa creu i Sant Paul), Anna-Marie Jesson (University Hospitals Birmingham NHS Foundation Trust, Good Hope Hospital), Chris Kelly (NHS Forth Valley, Forth Valley Royal Hospital), Andy Pettit (Airedale NHS Foundation Trust, Diabetes Center, Airedale General Hospital), Roshell Jeyavenanthan (King’s College London), Ben Field (Surrey and Sussex Healthcare NHS Trust), Tabitha Randell (Nottingham University Hospitals NHS Trust, Nottingham Children’s Hospital, QMC Campus), and Helen Partridge (University Hospitals Dorset NHS Foundation Trust, Royal Bournemouth Hospital).
Ethical Approval
The ABCD national audit program, which includes the ABCD DIY APS audit, has Caldicott Guardian Approval and has also been approved by the Confidentiality Advisory Group. 34 The program collects anonymized and routinely available clinical data. Tests not performed routinely were not required to be performed. Hence, this study did not require specific approval by a research ethics committee.
Authors’ Contributions
E.G.W., T.S.J.C., and R.E.J.R. designed the audit and the data collection tool. S.H., T.S.J.C., E.G.W., R.P., A.G., B.M., R.H., A.K., and N.B. undertook the data collection. A.L.L. and T.S.J.C. performed the data analysis. A.L.L. wrote the first draft of the article, and E.G.W., S.H., R.E.J.R., T.S.J.C., A.G., B.M., R.H., A.K., and N.B. reviewed and revised the content.
Author Disclosure Statement
A.L.L. has received speaker fees and/or support to attend conferences from Dexcom and Novo Nordisk and research support from the Association of British Clinical Diabetologists. T.S.J.C. has received speaker fees and/or support to attend conferences from Eli Lilly, Novo Nordisk, Sanofi, Abbott Diabetes Care, Dexcom, and Insulet. S.H. has served on the advisory board for Tandem, Dexcom, and Medtronic; undertaken nonpromotional educational and/or consultancy work for Abbott UK, Insulet, Dexcom, and Roche; and received research support from Abbott and Insulet. R.P., A.G., B.M., R.H., A.K., and N.B. have no competing financial interests to disclose. R.E.J.R. has received speaker fees and/or consultancy fees and/or educational sponsorships from Abbott, Astra Zeneca, Besins, BioQuest, GI Dynamics, and Novo Nordisk. E.G.W. has received personal fees from Abbott, AstraZeneca, Dexcom, Eli Lilly, Embecta, Insulet, Medtronic, Novo Nordisk, Roche, Sanofi, Sinocare, and Ypsomed and research support from Abbott, Embecta, Insulet, Novo Nordisk, and Sanofi.
Funding Information
The Association of British Clinical Diabetologists funded the development of the online tool and clinical research fellow time to undertake the analysis. S.H. is supported by the Medical Research Council Clinical Academic Partnership award (MR/W030004/1).
Supplementary Material
Supplementary Data S1
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
