Abstract
This article offers a systematic literature review (SLR) on the use of the MiniMed 780G automated insulin delivery system (MM780G) in people with type 1 diabetes (PwT1D) during Ramadan intermittent fasting. It also presents consensus recommendations on the use of MM780G during the Ramadan period. The SLR was performed following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses methodology. The recommendations resulted from a consensus-forming process involving a panel of experts. The process considered evidence found in the SLR as well as the expert opinions. In total, six studies were included in the SLR. The evidence and expert opinions led to recommendations related to (a) pre-Ramadan counseling of MM780G users who plan to fast; (b) suggested MM780G settings, meal announcement strategy, and safety aspects during Ramadan (including a contingency plan); and (c) post-Ramadan transition into and out of Eid-al-Fitr festivities. The SLR findings showed that the MM780G maintains glycemic control at target in PwT1D during Ramadan (meeting continuous glucose monitoring–based clinical targets proposed by the International Consensus on Time-in-Range) while ensuring low rates of hypoglycemia and diabetic ketoacidosis. Automated insulin delivery also helps PwT1D fast more days of Ramadan compared with users of other less advanced modalities of treatment. Pre-Ramadan guidance on specific aspects of the MM780G along with the International Diabetes Federation and Diabetes and Ramadan International Alliance counseling guidelines is recommended. There is still a challenge with post-Iftar hyperglycemia, which could potentially be mitigated by following the recommendations outlined in this article.
Introduction
The month of Ramadan is an important period for Muslims worldwide, occurring in the ninth month of the Islamic calendar. During Ramadan, it is mandatory for healthy Muslims who have reached puberty to perform fasting. Ramadan fasting is a form of intermittent fasting that involves abstaining from eating and drinking from dawn to sunset for 29–30 consecutive days. Typically, two main meals are consumed during a day of Ramadan, being Suhur (predawn meal to prepare for fasting) and Iftar (evening meal after sunset to break the fast). The nonfasting period between Iftar and Suhur is characterized by frequent overnight snacking for most people. The Ramadan period leads to significant changes in daily routines (e.g., diet, activity, sleep pattern), which, in turn, have effects on physiological aspects such as energy supply, hormonal balance, and mental health. 1
Muslims currently make up almost a quarter of the world’s population, and this proportion is expected to increase in the coming decades. 2 Despite the Quran exempting those with illnesses from fasting and medical recommendations advising against fasting for many Muslims with type 1 diabetes (T1D), a considerable number still choose to fast. The 2019 Diabetes and Ramadan global survey, conducted in 13 countries with predominant Muslim population, revealed that 71.1% of people with T1D (PwT1D) observed fasting during Ramadan, even amid the COVID-19 pandemic. 3 Children have shown a positive attitude and willingness to participate in Ramadan fasting as well. 4 According to recent worldwide data, more than 148 million Muslims are diagnosed with (any type of) diabetes, and ∼116 million of them opt to observe the fast. 5,6
PwT1D who choose to fast during Ramadan face risks, including but not limited to hypoglycemia (especially during fasting hours), hyperglycemia (especially after Iftar meal and during daytime for some PwT1D who may intentionally reduce or skip insulin doses to minimize hypoglycemia risk and avoid breaking the fast), dehydration, and increased variability in blood sugar levels. 1 To ensure safe fasting, various organizations such as the International Diabetes Federation (IDF) and Diabetes and Ramadan International Alliance (DAR), 1 and the International Society for Pediatric and Adolescent Diabetes (ISPAD) 7 have developed guidelines for health care professionals (HCPs) and PwT1D.
The rapid progress of automated insulin delivery (AID) systems has significantly improved glycemic control for PwT1D. These systems, now integrated into global guidelines for treating T1D, 8 –10 have shown remarkable effectiveness and safety in both adults and children. The MiniMed 780G AID system (MM780G), introduced for clinical use in 2020, features the advanced hybrid closed loop algorithm. This algorithm automatically adjusts basal insulin and delivers correction doses every 5 min (as needed), with the goal of maintaining a predetermined glucose target (GT). 11 Through clinical trials and real-world evaluations, the MM780G has been proven to safely surpass the international continuous glucose monitoring (CGM) targets and to be adaptable to homeostatic changes. 12 –19 Recently, the first studies on MM780G during Ramadan were undertaken. As many HCPs begin using this system, there is a need for education on its use during Ramadan. In addition, the current versions of the guidelines mostly predate the AID era and do not yet include any recommendations for these advanced systems. 20
This article serves a dual purpose. First, it offers a systematic review of existing literature on the use of MM780G in PwT1D during Ramadan intermittent fasting. Second, it presents recommendations for PwT1D on the use of MM780G during the Ramadan period. These recommendations result from a consensus-forming process involving a panel of experts. The process considers the evidence found in the initial literature review as well as the expert opinions.
Methods
Part 1: Systematic literature review on MM780G use during Ramadan
A systematic literature review (SLR) was performed, focusing on original publications on insulin pump therapy in PwT1D during Ramadan. After that, the search was narrowed down to publications on MM780G. The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) principles were followed. 21
The search began in July 2023, exploring Medline, Embase, and PubMed for full manuscripts in the English language. A detailed overview of keywords and the search strategy is available in Supplementary Table S1. Identified records were screened by title and abstract, using the inclusion and exclusion criteria from the Population, Interventions, Comparison, Outcomes, and Study (PICOS) framework in Table 1. The eligibility of the remaining records was then confirmed by reviewing the full text. In November 2023, a manual search, including scrutinizing references and a fresh engine search, was conducted as a last step and led to the final list of included publications. Details of all included records were listed, and those focusing on MM780G only were summarized separately. For each record, the Oxford evidence level was provided. 22
PICOS Overview
PICOS Overview
RCT, randomized controlled trial.
Part 2: Recommendations on the use of MM780G during Ramadan
Medtronic extended invitations to 13 adult and pediatric endocrinologists and diabetologists to participate in the expert panel. Selection criteria for experts encompassed substantial clinical expertise in managing T1D during Ramadan, either in individuals using the MM780G or in individuals using other treatment modalities, and participation in previous Ramadan guidelines. An equitable geographical distribution was maintained (experts from eight countries spread across four continents). From this panel of experts, a two-person working group was formed, comprising individuals who recently published studies on the use of the MM780G during Ramadan (N.E. and M.A.). This working group identified clinical topics that need recommendations, across three distinct periods: pre-Ramadan, during Ramadan, and post-Ramadan. Subsequently, the two experts collaboratively formulated the recommendations based on the evidence found in the SLR (part 1 of this article) and clearly stated if this recommendation was based on scientific evidence, expert opinion, or both. A rationale was provided for each recommendation. Following this, the recommendations underwent a comprehensive review by the broader expert panel, leading to deliberations and consensus.
Results
Part 1: SLR on MM780G use during Ramadan
The PRISMA chart, including included and excluded records for each step, is provided in Figure 1. The engine search led to 34 records, which was reduced to 18 based on the PICOS eligibility criteria, and then extended to a final 38 records after reference scrutiny and the fresh search. An overview of those 6 records confined to MM780G can be found in Table 2. They are also summarized below, in order of the Oxford evidence level. A detailed overview of all records, including those not addressing MM780G, can be found in Supplementary Table S2.
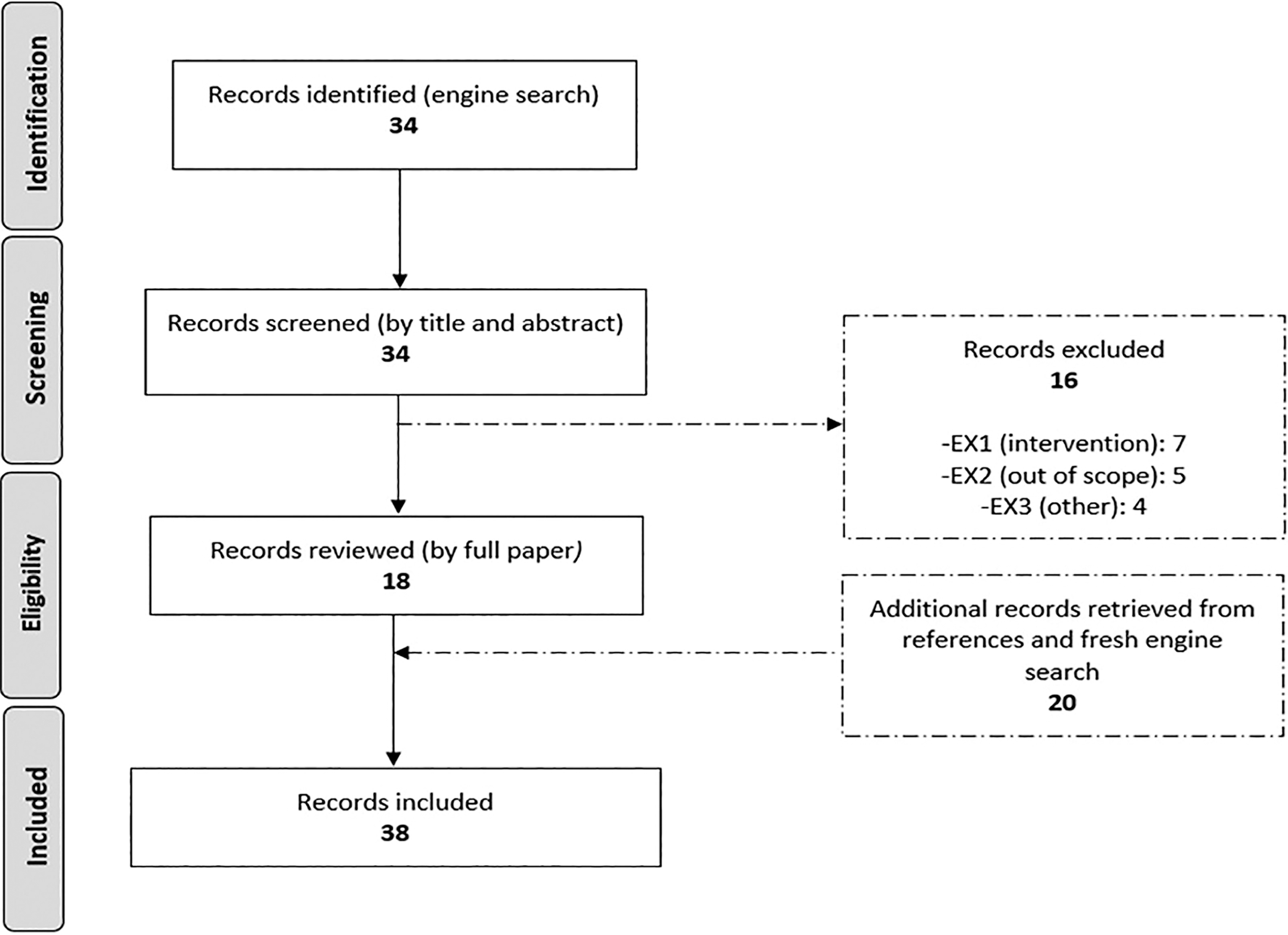
PRISMA chart. PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
Studies Derived from Systematic Literature Review Involving MM780G Use During Ramadan
AHCL, advanced hybrid closed-loop; AID, automated insulin delivery; AIT, active insulin time; CGM, continuous glucose monitoring; GMI, Glucose Management Indicator; GT, glucose target; ICR, insulin-to-carbohydrate ratio; IQR, interquartile range; MDI, Multiple daily injections; MM780G, MiniMed 780G; NR, not reported; SAP-PLGM, sensor-augmented pump therapy with predictive low-glucose management; SMBG, Self-monitoring blood glucose; TA250, time above range (>250 mg/dL); T1DM, type 1 diabetes mellitus; TA180, time above range (>180 mg/dL); TB70, time below range (<70 mg/dL); TIR, time in range.
Elbarbary et al. 23 conducted a randomized clinical trial (RCT) to assess the safety and effectiveness of MM780G in T1D during Ramadan. The trial involved 42 adolescents and young adults in Egypt, evenly split between an intervention group and a control group. The intervention group used lenient settings during Ramadan with a GT of 120 mg/dL and active insulin time (AIT) of 3 h, while the control group used a GT of 100 mg/dL and AIT of 2 h (i.e., recommended or optimal settings). 12,24 The latter group with the optimal MM780G settings demonstrated excellent sensor glucose control; time in range (TIR) reached 82.0%, time above range (>180 mg/dL) (TA180) 14.8%, and it was not at the expense of time below range (<70 mg/dL) (TB70) that reached only 3.0%. However, these metrics did not significantly differ from the glycemic control in the intervention group with the lenient settings. Users in both groups fasted for more than 28 days, with over 95% completing the entire month. Only one person in each group broke their fast in the afternoon during the first week of Ramadan due to mild hypoglycemia. There were no severe hypoglycemic or diabetic ketoacidosis (DKA) events. This study is classified as Oxford level 1 evidence.
In another RCT, Elbarbary et al. 25 investigated the efficacy and safety of oral vildagliptin as an add-on therapy to MM780G use during Ramadan. Among 50 adolescents and young adults with T1D, the use of adjunctive vildagliptin treatment mitigated postprandial hyperglycemia and increased TIR without compromising safety. However, MM780G therapy alone (with optimal settings) already reached a substantial TIR of 79.1%, a TA180 of 17.5%, and a TB70 of 3.1%. No severe hypoglycemia or DKA was reported, and the number of days the fasting was completed reached to almost 28. This study is classified as Oxford level 1 evidence.
Al-Sofiani et al. 26 showed the safety and efficacy of the MM780G in PwT1D during Ramadan in a large retrospective real-world study. In this study, 449 MM780G users from the Gulf region were monitored in the month before, during, and after Ramadan. Throughout Ramadan, TIR averaged 70.7%, TA180 was 27.1%, and TB70 was 2.3%. These values were comparable with the month before and after Ramadan, meeting CGM-based clinical targets from the International Consensus on Time-in-Range. A limitation of this study was the inability to confirm who actually fasted during Ramadan. However, glucose profiles showed clear spikes for Suhur and Iftar meals, and users who manually bolused the least during daytime (based on quartiles) had similar results. Users consistently using a GT of 100 mg/dL had a numerically higher TIR of 74.0% (not statistically tested against the overall group), and those also consistently using an AIT of 2 h even reached to 78.1%, without increasing hypoglycemia risk (TB70 = 2.7%). Other relevant conclusions were the lower nighttime TIR during Ramadan (60.4%) compared with daytime TIR (80.0%), in parallel with a slightly elevated TA180 during nighttime (37.3%). In addition, there was absence of an increased hypoglycemia risk during Ramadan daytime. By breaking down the data per day, authors additionally found that the adaptation of the MM780G to lifestyle changes was very fast, starting as soon as users transitioned from Shaban (the month prior to Ramadan) to Ramadan and from Ramadan to Shawwal (the month after Ramadan). Safety events and data on days the fasting was completed were not available. This study was rated as Oxford level 2 evidence.
In another prospective study, Al-Sofiani et al. 27 directly compared fasting experience and glycemic control during Ramadan in PwT1D using AID systems (n = 62) versus other treatment modalities (conventional open-loop pump + CGM, n = 37; pump + self-monitoring blood glucose (SMBG), n = 8; MDI + CGM, n = 155; multiple daily injections (MDI) + SMBG, n = 32) in Saudi Arabia. Use of AID, including MM780G, MiniMed 670G system (MM670G), and Tandem Control IQ, demonstrated superiority over other treatment modalities in various endpoints. The AID group achieved a TIR of 73.0%, TA180 of 25.0%, and TB70 of 2.0% statistically outperforming the two CGM groups (conventional pump: 51.0%, 46.0%, and 3.0%, respectively; MDI: 52.0%, 45.0%, and 3.0%, respectively). The median number of days the fast was broken was only 2 for AID users, whereas this was 5 for conventional pump + CGM, 3 for MDI + CGM, 3.5 for pump + SMBG, and 2.5 for MDI + SMBG users. Furthermore, 53% of AID users achieved the “double target” of breaking the fast due to diabetes ≤2 days and maintaining TIR ≥70%, which was significantly higher than the 3% of conventional pump users and 44% of MDI + CGM users. This study was rated as Oxford level 2 evidence.
In a pilot study, 28 Wannes et al. followed 19 children with T1D using either MM670G or MM780G. The study covered the month before Ramadan and the month of Ramadan. Among these children, 11 observed Ramadan fasting. For those fasting, the average glycemic metrics during Ramadan were a TIR of 71.7%, TA180 of 26.8%, and TB70 of 1.5%, which were the same as the month before. Comparing the two AID systems, both showed similar glycemic control, but MM780G significantly reduced the burden with fewer exits from automation (0.36 vs. 1.67 exits/day). No episodes of DKA, admissions, or emergency room consultations for severe hypoglycemia occurred during Ramadan. This study is classified as Oxford level 4 evidence.
In another publication by Wannes et al., 29 they presented a case report of an 11-year-old child from Saudi Arabia who underwent two successive Ramadan seasons under two different treatment modalities: sensor-augmented pump therapy with predictive low-glucose management (SAP-PLGM using the MiniMed 640G system) and MM780G. The user broke the fast six times with the SAP-PLGM system and three times with MM780G, all due to mild hypoglycemia. Glycemic control was significantly better with MM780G, achieving a TIR of 75%, a TA180 of 23%, and a TB70 of 2%. This case report is rated as Oxford level 5 evidence.
Part 2: Recommendations on the use of MM780G during Ramadan
Recommendation 1.1: MM780G users who plan to fast during Ramadan are encouraged to continue using the system during Ramadan for a better and safer fasting experience
Although only six studies were found in the SLR, their notable consistency provided valuable insights on the efficacy and safety of MM780G during Ramadan. First, all studies indicated that individuals using MM780G can achieve good glucose control during Ramadan, with average study TIRs ranging between 70.7% and 84.7%. These TIRs fall within the CGM-based clinical targets proposed by the International Consensus on Time-in-Range. 30 Second, glycemic control was achieved safely, with the average study TB70 ranging from 1.8% to 3.1% and no severe safety events such as DKA or severe hypoglycemia being detected in any of the trials. Third, the trials showed a high proportion of full fasting days. The minimal average reported the number of fasting days was almost 28, and across all studies only a few breaks were observed (mainly for mild hypoglycemia). Fourth, glycemic control during Ramadan was comparable with that in the month before and after. Finally, one study broke down the CGM data into Ramadan fasting and nonfasting hours, 26 and it became apparent that daytime hypoglycemia risk was low (only 2.3%). The post-Iftar TA180 was slightly above international targets (37.3%) and may be addressed by closely adhering to the recommendations.
The SLR revealed that PwT1D using MM780G during Ramadan on average achieved high levels of glycemic control when compared with less advanced treatment modalities. In a real-world study by Al-Sofiani et al., the use of AID during Ramadan was associated with better glycemic control (TIR of 73%) compared with the use of other T1D treatment modalities, including conventional pump (TIR of 51%) and MDI (TIR of 52%). 27 AID users also had the lowest number of days during which fasting was broken with a median of 2 days compared with 5 days for conventional pump + CGM group, 3 days for MDI + CGM group, 3.5 days for the pump + SMBG group, and 2.5 days for MDI + SMBG group. Other large studies directly comparing AID with less advanced modalities are lacking.
Recommendation 1.2: Pre-Ramadan counseling on specific aspects of the MM780G during Ramadan is advised. This guidance should complement the pre-Ramadan counseling guidelines specified by IDF-DAR
Pre-Ramadan consultation ideally occurs at least 2 weeks before the start of Ramadan. The exact timing can be individualized; high-risk MM780G users or new users may benefit from earlier consultations (up to 2–3 months prior to Ramadan), while those who are already experienced or well-controlled can be consulted closer to Ramadan.
Pre-Ramadan consultation ideally involves a team comprising an endocrinologist, a diabetes educator, and a dietitian. If feasible, at least one HCP in the team should have experience and proficiency in MM780G use.
Table 3 provides an outline of topics suitable for discussion during pre-Ramadan consultation, and a summary is presented below. The consultation team should explain the (dis)advantages of using this system during Ramadan. Advantages include improved glycemic control, less hypoglycemia and risk, and less efforts from PwT1D compared with other modalities; disadvantages involve acceptance of relinquishing control. The team can perform a competency assessment (if needed), discuss user versus HCP responsibilities, and provide MM780G-specific guidance on insulin-to-carbohydrate ratio (ICR), AIT, GT, and temporary target (TT), as well as on meal announcement strategy, consistent SmartGuard use (>85% of the time), manual mode basal rate and correction factor update, and safety. CareLink System reports can be used to review settings. In addition, preparing the MM780G user for fasting should include a contingency plan in case SmartGuard is deactivated. It is also crucial to provide post-Ramadan advice, including considerations for Eid-al-Fitr.
Topics to Discuss During the Pre-Ramadan Counseling
Topics to Discuss During the Pre-Ramadan Counseling
HCP, health care professional; TT, temporary target.
Recommendation 1.3: The health care team is encouraged to confirm that the CareLink Personal accounts of the MM780G users who plan to fast are linked to the accounts of their treating HCPs
CareLink Personal is a software application designed to collect data directly from MiniMed systems. 11 When configured correctly and connected to the treating HCP’s account, this allows the HCP to track progress. Users should ensure that their data are available and updated prior to the pre-Ramadan visit. Users should ensure automatic uploads, if possible, rather than manual uploads, to furnish HCPs with up-to-date information during Ramadan. HCPs have the advantage to conduct regular virtual check-ins with users and intervene as required. The availability of 24-h technical support by the manufacturer is another safety measure to put in place.
Recommendation 2.1
For the Iftar meal, postprandial hyperglycemia can be mitigated by intensifying the ICR (e.g., intensify ICR by 10%–20% for the first Ramadan Iftar meal and further personalize the ICR in the following days until a safe and effective ICR is established) and/or by augmenting the meal announcement.
For the Suhur meal, we suggest maintaining the same pre-Ramadan ICR setting, although adjustments can be made based on individual needs. Close monitoring of SG levels after Suhur and further adjustment of the ICR as needed is advised.
In most parts of the world, the nutritional composition of the Iftar meal is often characterized by both high glycemic index carbohydrates and high fatty components, and post-Iftar hyperglycemia is a common phenomenon. There are multiple strategies to mitigate post-Iftar hyperglycemia.
One commonly used strategy is to intensify the ICR for the Iftar meal. While only some studies touched the role of modifying the ICR during Ramadan, 23,25,31 it is established that an intensified ICR results in increased insulin for an equivalent amount of carbohydrates and can mitigate post-Iftar hyperglycemia. A frequently used approach is to intensify ICR by 10%–20% for the first Ramadan Iftar meal. This adjustment should be closely monitored by the HCP and further personalized in the following days, if necessary, until a safe and an effective ICR has been established.
Another strategy is to announce additional carbohydrates to what the user has actually consumed and divide the announcement. This approach can be applicable to Iftar meals that are rich in carbohydrates and fat. Empirical evidence regarding the optimal strategy for announcing Iftar meals with the MM780G is, however, limited. 23,25 An often used approach is to initiate the announcement based on the actual counted carbohydrates for the first Ramadan Iftar meal, followed by a subsequent announcement of 20%–30% thereof after 1.5–2 h. This adjustment should be closely monitored by the HCP and further personalized in the following days, if necessary. Caution should be used, however, as splitting the announced meal can be associated with increase in time below range. It must be noted that this strategy contradicts the instruction for use as provided by the manufacturer, but the expert panel deems it justified due to the high glycemic index carbohydrates and high fatty components in many Iftar meals.
The Suhur meal, at dawn, is often delayed to shortly before the fasting begins. Not intensifying the ICR for this meal aims at minimizing the risk of breaking the fast because of early daytime hypoglycemia. The decision to maintain or relax the ICR depends on individual preferences and needs. For the Suhur meal, the split meal announcement approach is not recommended.
Recommendation 2.2: Users of the MM780G who plan to fast are encouraged to count carbohydrates and announce meals using the Bolus Wizard feature for all snacks and meals that they consume during the nonfasting hours
Strategies for announcing meals can contribute to preventing severe hyperglycemia after Iftar. One meal announcement strategy has already been explained in recommendation 2.1.
Although it is typically advised for rapid-acting insulin in MM780G and open-loop systems to announce meals 15 min before the first bite (to address early postprandial hyperglycemia), this can be modified for Iftar for some MM780G. Announcing the meal right before Iftar might alleviate concerns about reaching a hypoglycemic state right before breaking Iftar, particularly if the pre-Iftar glucose level is approaching a low level. Nonetheless, it is the experience shared by some authors that a 15-min premeal announcement might be necessary in many MM780G users. For users of ultrarapid acting insulin, meal announcement is recommended at the start of Iftar in almost all individuals.
Overnight snacking is a common practice for many people during the nonfasting hours in Ramadan, especially for those living in regions with longer nonfasting hours such as the Middle East and North Africa. MM780G users should be encouraged to announce all meals and snacks using the Bolus Wizard feature. For snacks with high fat and protein content, the 20%–30% additional announcement strategy (as mentioned before) can be applied. It is important to highlight that the MM780G incorporates a safety feature known as the “safe meal bolus,” 11 which automatically reduces the bolus amount if the SmartGuard predicts a potential hypoglycemia risk after food consumption.
Recommendation 2.3
No adjustments to GT and AIT are needed at the start of Ramadan for most users of the MM780G who plan to fast, unless there is an increased hypoglycemia risk in certain individuals or in countries with prolonged fasting hours. The optimal settings of GT at 100 mg/dL and AIT at 2 h are also suitable during Ramadan for most users if good glycemic control had already been achieved with these settings before Ramadan.
The GT represents the desired glucose value used by the algorithm in its calculations, while AIT signifies the duration during which bolus insulin is considered active (with a shorter time indicating a more proactive algorithm). 11 It is important to emphasize that the MM780G has demonstrated optimal efficacy while still being safe with a GT of 100 mg/dL and an AIT of 2 h, as evidenced by both trials 13,32 and real-world studies. 16,24 In addition, one RCT 23 and one real-world study 26 from the SLR have also highlighted that these optimal settings result in the highest TIR during Ramadan while maintaining low TB70 levels. The supporting bar charts are shown in Figure 2, but it must be noted that a favorable outcome of optimal over lenient settings was either not proven (RCT) or not tested (real-world study).
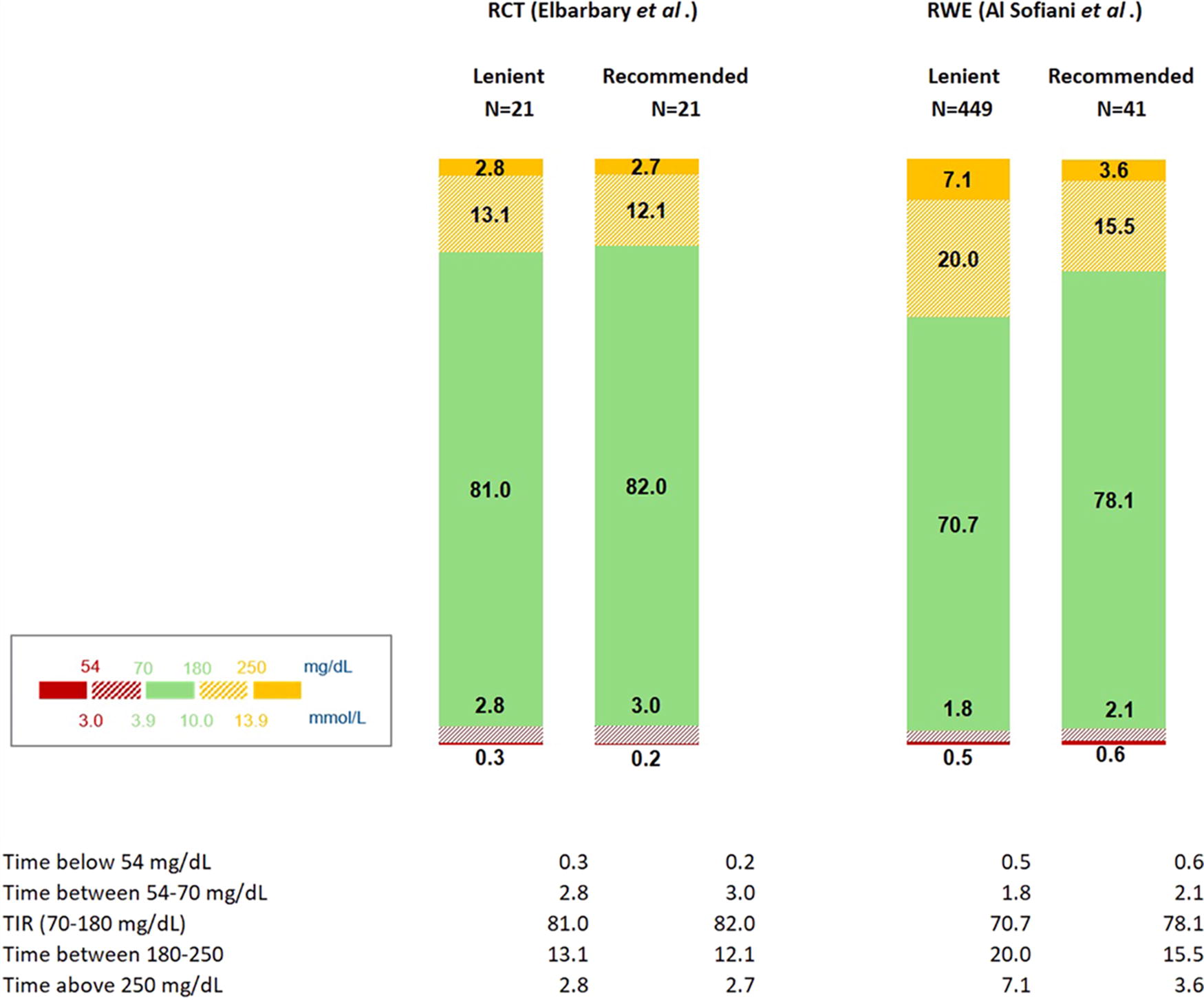
Performance of the MM780G in PwT1D during Ramadan—optimal settings versus lenient settings. MM780G, MiniMed 780G; PwT1D, people with type 1 diabetes.
The studies on optimal settings during Ramadan have been used in North Africa and in the Gulf countries, where shorter fasting hours are seen, and the option of less working hours is offered during Ramadan. However, in countries farther from the equator, where fasting hours can extend beyond 18 h, some experts have suggested to start Ramadan with more lenient settings to avoid increase in hypoglycemia during fasting hours. In countries close to the arctic circle where daytime hours are longer than average, PwT1D who wish to fast should consult with their local religious authorities to decide on duration of fasting and with their HCPs to ensure safe fast.
Recommendation 3.1: For guidance on breaking the fast, we suggest reviewing the IDF-DAR guidelines
According to the IDF-DAR guidelines, PwT1D should break the fast promptly if glucose levels drop below 70 mg/dL (even if asymptomatic), rise above 300 mg/dL, or if symptoms of hypoglycemia, hyperglycemia, dehydration, or acute illness occur. It is best to rely on actual blood glucose levels by fingerpicking rather than CGM measures, but if blood glucose measurements are not available, PwT1D should break the fast when sensor glucose levels drop below 70 mg/dL. The experts experienced that the upper level of 300 mg/dL is rarely reached with the MM780G during fasting hours, and they therefore recommend that any sudden sustained hyperglycemia may be a reason to break the fast. Together with immediate checking of the infusion set and site, the contingency plan (recommendation 3.4) should be used. Setting alarms can aid in detecting exceeding sensor glucose values. It can be beneficial to keep a glucagon kit (either injection kit or nasal spray) easily accessible, especially for situations involving severe hypoglycemia and loss of consciousness. In addition, it is important to ensure that an acquaintance is trained on how to use the kit.
Recommendation 3.2: Optimizing the utilization of the CGM sensor and SmartGuard feature at all times during Ramadan is advisable
Findings from studies outside of Ramadan have shown that both high sensor use and high SmartGuard use play a crucial and independent role in achieving a high TIR and a low TB70. 24 Ideally, disengaging from the sensor and SmartGuard should be limited to occasions when sensor and infusion set changes are necessary.
Recommendation 3.3: The TT feature can be utilized during the daytime to minimize the risk of hypoglycemia when needed
The TT feature temporarily sets the GT to 150 mg/dL and inactivates autocorrections. This aims at reducing hypoglycemia risk when rapid decrease in insulin requirements is anticipated (e.g., during exercise). Using TT in the last few hours before breaking the fast at Iftar is a frequently used strategy when sensor glucose is approaching low and expected to further drop. However, it is essential to deactivate the TT feature and return back to SmartGuard shortly before Iftar to minimize the risk of post-Iftar hyperglycemia. The TT can also be used in physical activity during Ramadan or during Tarawih prayer.
It is important to recognize that the suggestion to utilize TT applies to situations when glucose level is still above 70–80 mg/dL but trending down and the user is concerned about hypoglycemia risk. Whenever blood glucose drops below 70 mg/dL, PwT1D should break the fast promptly (even if asymptomatic) as per recommendation 3.1. The utility of TT during the last few hours of fasting remains subject to debate. Since glucose levels are low during prolonged fasting, the anticipated minimal basal insulin rate can diminish the potential impact of TT. Therefore, some authors suggest that it might be safer to encourage users to break the fast when glucose is approaching 70 mg/dL than to rely on TT.
Recommendation 3.4: Establishing a contingency plan for users of the MM780G who plan to fast during Ramadan is essential
While SmartGuard exits during Ramadan are very low (one per week on average, similar to the month before Ramadan 26 and comparable with other regions outside Ramadan 16,18 ), it is important to recognize that situations may arise where a user cannot or chooses not to use SmartGuard. A contingency plan should therefore be in place. In case the contingency plan includes using the MM780G in manual mode, it is essential for the HCP to check the manual basal rates and correction factor during the pre-Ramadan counseling. The user can apply the specific Ramadan manual basal settings, 33 and users can switch to Ramadan settings during the night before starting the fast. If the contingency plan includes other treatment modalities as backup, it is recommended to adhere to IDF-DAR guidelines. The contingency plan should also include troubleshooting of common pump and sensor issues and educating users on how to switch to alternative modalities.
Recommendations regarding the post-Ramadan period
Recommendation 4.1
In countries where Eid-al-Fitr involves consuming high glycemic index meals with high fat components in the evening hours, MM780G users may continue to use the intensified ICR as described for the Ramadan Iftar meals. MM780G users who consume this type of meals throughout the entire day of Eid-al-Fitr and who know how to change the ICR setting may extend the intensified ICR to the entire day of Eid-al-Fitr. In addition, counting and announcing carbohydrates using the Bolus Wizard feature for all snacks and meals consumed are encouraged.
The end of Ramadan is celebrated with Eid-al-Fitr, a multiday festive event. Similar to the Ramadan Iftar meal, Eid-al-Fitr is often characterized by plentiful food and the presence of high glycemic index carbohydrates and fatty foods. In some countries, the consumption of these meals is more common in the evening hours of Eid-al-Fitr, while in other countries, this practice may extend throughout the entire day. It is recommended to use a similar intensified ICR and meal announcement strategies as described for Ramadan Iftar meals. Whether the intensified ICR should be limited to evening hours or be extended throughout the entire day depends on the user’s eating pattern and type of food as well as the user’s ability to independently change the ICR. In many Muslim-majority countries, outpatient clinics are closed during Eid-al-Fitr days, and accessibility to HCPs who can help with ICR change is limited. Therefore, it may be more practical and safer to use the same Ramadan ICRs during Eid-al-Fitr.
In certain countries, Eid-al-Fitr is not recognized as an official public holiday, and the celebrations do not typically involve consuming high glycemic index meals throughout the day. For those individuals who do not indulge excessively during Eid-al-Fitr, it is recommended to revert to pre-Ramadan strategies for ICR and meal announcements to avoid hypoglycemia. This adjustment is justified because continuing with an intensified ICR during evening hours may lead to hypoglycemia after dinner.
MM780G users are encouraged to use the Bolus Wizard feature and bolus for all the traditional Eid-al-Fitr meals, high glycemic index sweets, and snacks even if they are consumed within a short period of time. The risk of insulin stacking with MM780G is reduced compared with MDI and other less advanced treatment modalities, as mentioned before. 11
Recommendation 4.2: After Eid-al-Fitr festivities are over, users of the MM780G are advised to go back to the pre-Ramadan ICR setting
When an MM780G user returns to the regular non-Ramadan lifestyle, system settings should be adjusted accordingly. Updating the ICR and timing thereof is essential. If the contingency plan involved utilizing MM780G in manual mode, necessary updates need to be made here as well. MM780G settings and CareLink System reports can be reviewed after Ramadan and discussed with users in anticipation of future fasting.
Discussion
This article presents an evidence-based systematic exploration on the efficacy and safety of MM780G during Ramadan. The SLR findings showed that MM780G maintains glycemic control within target in PwT1D during Ramadan while ensuring low rates of hypoglycemia and DKA. In addition, comprehensive recommendations were offered for the optimal utilization of MM780G in PwT1D who plan to fast during Ramadan. The recommendations are summarized in Table 4.
Recommendations for MM780G Use During Intermittent Ramadan Fasting
IDF-DAR, International Diabetes Federation–Diabetes and Ramadan International Alliance; SG, Sensor Glucose.
The currently available IDF-DAR and ISPAD guidelines for treating PwT1D during Ramadan do not yet offer specific guidance on using AID systems. 1,7,34 Since the effectiveness of these systems, particularly MM780G, is evident and given that a substantial proportion of Muslims with T1D already use AID systems, we think it is opportune to include these AID systems in the guidelines. However, the question of whether AID use should allow for a different score in the IDF-DAR risk calculator is a topic that goes beyond the scope of this article and requires a broader discussion within the IDF-DAR community.
As different AID systems have different algorithms and differ in the adjustable settings, some of the recommendations provided are specific to MM780G (e.g., those on AIT and GT). Recommendations on ICR and meal announcement strategy may also apply to other AID systems, although there is a lack of confirmatory studies at this time. It must be noted that at present, most AID studies during Ramadan concentrate on assessing MM780G, while some case reports also describe MM670G (which does not have the autocorrection). 28,35 To the best of our knowledge, two articles have delved into other AID systems. 31,36 Whether other AID systems display similar efficacy and safety during Ramadan requires further research.
The recommendations from this article can be applied to other (religious) situations that involve intermittent fasting such as fasting for the 6 days of the month of Shawwal, adult females making up for the missed days of fasting during Ramadan because of the menstrual cycle, the first 9 days of Dhul-Hijjah, fasting during the month of Muharram, and fasting during the month of Shaban for Muslims. Likewise, our recommendations may apply to fasting during Yom Kippur for Jews, Lent for Christians, and other religious forms of fasting. Moreover, intermittent fasting for health-related reasons (e.g., weight loss) would be another situation where our recommendations can be applicable.
The recommendations from this article are by no means intended to pressure PwT1D who are at moderate to high risk or uncertain about fasting. The exemption for those who are ill not to fast (as mentioned in the Quran), as well as the medical advice against fasting for most individuals with T1D, still stands and should be respected.
Making small yet significant adjustments to the algorithm could potentially enhance the MM780G performance and further improve glycemic control and safety during Ramadan. For instance, the performance of MM780G during Ramadan can be further improved by offering users with an option of time-specific SmartGuard GT, where the SmartGuard GT can be set slightly higher during the last hours of fasting. Moreover, designing a Ramadan-specific algorithm warrants consideration by AID manufacturers. Such an algorithm designed through in silico modeling of Ramadan-specific changes in lifestyle, behaviors, and human physiology will likely help optimize glycemic control during Ramadan and may address the current issue with post-Iftar hyperglycemia.
The current of MM780G adoption rate in many predominant Muslim countries remains relatively low. However, the use of AID systems is increasing in some of those countries, as well as in Western countries, where significant Muslim communities observing Ramadan may reside. Therefore, this guidance on MM780G during Ramadan is timely.
The limitation of this article is the scarcity of research specifically focusing on MM780G during Ramadan. This prevented us from using GRADE (Grading of Recommendations, Assessment, Development, and Evaluations) methodology, 37 which is the standard for creating recommendations. However, we were able to utilize the Oxford methodology on grading evidence. 22 The strength lies in the PRISMA process used, 21 the robust process used to formulate the recommendations and reach alignment among the expert panel, and the participation of key experts in the field of diabetes, technology, and Ramadan. Because of the limited data about AID use during Ramadan, some of the recommendations are based on clinical experience rather than being evidence-based recommendations. These specific recommendations would have been more robust should the empirical evidence be available.
For future, it would be beneficial to explore topics such as (1) the optimal ICR strategy to minimize post-Iftar hyperglycemia in MM780G users during Ramadan, (2) the optimal meal announcement approach to reduce post-Iftar hyperglycemia, (3) the optimal use of TT to prevent daytime hypoglycemia, and (4) consideration of multinational studies to understand the impact of social and cultural differences on fasting in T1D. Another area that warrants further investigation is the potential use of adjunctive therapy (e.g., Glucagon-like peptide-1 receptor antagonists, dual Glucagon‐like peptide‐1/ Gastric inhibitory polypeptide receptor agonists, or other medications) in reducing post-Iftar hypoglycemia. One initial study on Dipeptidyl peptidase 4 inhibitor vildagliptin has already demonstrated a significant reduction in post-Iftar TA180 during Ramadan in MM780G users. 25 For more tailored recommendation on pregnant women, the elderly, and patients with impaired renal function, future studies are needed.
In conclusion, this is the first SLR on the efficacy and safety of MM780G during Ramadan intermittent fasting. The SLR showed that the MM780G maintains glycemic control at target in PwT1D during Ramadan (meeting international targets) while ensuring low rates of hypoglycemia and DKA. The MM780G also helps PwT1D fast more days of Ramadan compared with users of MDI and other less advanced modalities. Pre-Ramadan guidance on specific aspects of MM780G along with IDF-DAR counseling guidelines is recommended. There is still a challenge with post-Iftar glucose levels staying above 180 mg/dL, which could potentially be mitigated by following the recommendations outlined in this article. Further studies related to the post-Iftar challenge are required and will be planned.
Footnotes
Acknowledgments
The authors would like to thank the following people: Ghassan Nabulsi for project management, Teodora Bellone and Paola Cardano for SLR, and Ohad Cohen for medical input. All are employees of Medtronic.
Authors’ Contributions
N.E., M.A., W.C., and T.H. designed the study and wrote the article. N.E. and M.A. formulated the recommendations. All authors interpreted and discussed the data/recommendations and reviewed the article. Medtronic employees (not part of the author list) performed the SLR.
Guarantor Statement
N.E. and M.A. are the guarantors of this work and, as such, had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Author Disclosure Statement
N.E. served as speaker and served in the Advisory Board for Medtronic. A.A. takes part in the advisory panel for Medtronic, Novo Nordisk, Eli Lilly, Vital Air, and Sanofi and has received honoraria for speaking from AstraZeneca, Eli Lilly, Medtronic, Novo Nordisk, and Sanofi; received research support from AstraZeneca and Novo Nordisk. H.Z. served on the advisory panel for Abbott, Medtronic, and Novo Nordisk and received honoraria for speaking/consultancy from Abbott, Medtronic, and Novo Nordisk. M.H., A.D., R.A.D., and R.A. take part in the Medtronic advisory board. G.P. served as a speaker for Abbott, Novo Nordisk, Sanofi, Dexcom, and Medtronic events and served on Medtronic and Sanofi advisory Boards. S.H. served on the advisory panel for Tandem, Dexcom, and Medtronic and received honoraria for nonpromotional educational and/or consultancy work from Abbott, Insulet, Dexcom, Roche, and Sanofi. M.I. had consultancy agreement and received speaker honoraria from Novo Nordisk, Boehringer Ingelheim, Medtronic, and Pfizer. S.S. served on advisory boards conducted by Novo Nordisk, Lupin pharma, Astra Zeneca, and USV pharma and on the advisory panel for Medtronic and had spoken at meetings organized by Zydus pharma, Cipla pharma, Alkem pharma, Biocon Pharma, and Novo Nordisk. S.B.Z. takes part in the advisory panel for Medtronic. W.C. and T.v.d.H. are employees of Medtronic. M.A. served on an advisory panel for Abbott, Medtronic, Insulet, VitalAire, and Sanofi and received honoraria for speaking/consultancy from Abbott, Eli Lilly, Insulet, Medtronic, Novo Nordisk, Sanofi, and VitalAire.
Funding Information
S.H. is supported by the Medical Research Council Clinical Academic Partnership award (MR/W030004/1). This work was sponsored by Medtronic.
Supplementary Material
Supplementary Table S1
Supplementary Table S2
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
