Abstract
Aims:
The objective of this real-world, observational study was to evaluate change in continuing glucose monitoring (CGM) metrics for 1 year after CGM initiation in adults with noninsulin-treated type 2 diabetes (T2D).
Methods:
Data were analyzed from Dexcom G6 and G7 users who self-reported: T2D, ≥18 years, gender, no insulin use, and had a baseline percent time in range (TIR) 70–180 mg/dL of ≤70%. Outcomes were change in CGM metrics from baseline to 6 and 12 months overall and for younger (<65 years) and older (≥65 years) cohorts. Additional analyses explored the relationship between use of the high alert feature and change in TIR and time in tight range (TITR) 70–140 mg/dL.
Results:
CGM users (n = 3,840) were mean (SD) 52.5 (11.2) years, 47.9% female, mean TIR was 41.7% (21.4%), and 12.4% of participants were ≥65 years. Significant improvement in all CGM metrics not meeting target values at baseline was observed at 6 months, with continued improvement at 12 months. Mean baseline TIR increased by 17.3% (32.1%) from 41.7% (21.4%) to 59.0% (28.9%), and mean glucose management indicator decreased by 0.5% (1.2%) from 8.1% (0.9%) to 7.6% (1.1%) (both P < 0.001). Participants who maintained or customized the high alert default setting of 250 mg/dL had a greater increase in TIR and TITR compared with participants who disabled the alert. Days of CGM use over 12 months were high in 84.7% (15.9%).
Conclusion:
In this large, real-world study of adults with suboptimally controlled T2D not using insulin, Dexcom CGM use was associated with meaningful improvements in glycemic control over 12 months. Use of the high alert system feature was positively associated with glycemic outcomes. High use of CGM over 12 months suggests benefits related to consistent CGM use in this population.
Introduction
It is well established that the use of continuing glucose monitoring (CGM) significantly improves glycemic status in individuals with type 1 diabetes (T1D) 1 –3 and type 2 diabetes (T2D) who are treated with intensive insulin therapy. 4,5 Research has also demonstrated glycemic improvements in people with T2D treated with less-intensive insulin therapies, such as basal insulin. 6 –10 Evidence from these studies prompted a shift in clinical practice recommendations for CGM use to include both patients treated with intensive and nonintensive insulin therapy. 11,12
A limited number of randomized trials and real-world studies have investigated the impact of CGM use in individuals with noninsulin-treated T2D, although this group comprises the majority of the T2D population. 13 –16 While the results from these studies are promising and have demonstrated significant improvement in A1C and CGM metrics, additional study data are needed.
The objective of this real-world observational study was to evaluate changes in CGM-measured glycemic control over 1 year in a large cohort of adults with T2D not using insulin and not meeting glycemic targets.
Methods
Design and study population
This retrospective study analyzed data from Dexcom G6 and G7 (Dexcom, San Diego, CA) users who created an account in the Dexcom app and initially uploaded data between September 2021 and October 2022. CGM users were included in the analysis if they self-reported a T2D diagnosis, age ≥18 years, gender, and no insulin use, and had a baseline time in range (TIR) 70–180 mg/dL ≤70% over their initial 10 days of CGM use. CGM data sufficiency criteria of 70% of readings over 10 days at the 6- and 12-month follow-up were required. Participants were excluded if they self-reported insulin use or had connected data transfer from insulin delivery devices (multiple daily injections, connected insulin pens, insulin pumps) or any manually entered insulin use during the study period. All data were anonymized and managed in accordance with Dexcom’s privacy policies, and all users provided permission for Dexcom to store and analyze their data.
Outcome measures
CGM use and change in glycemic control
The main outcome measures were change from baseline to 6 and 12 months in CGM metrics, including mean glucose, glucose management indicator (GMI); percentage coefficient of variation (%CV); percentage: TIR 70–180 mg/dL; time in tight range (TITR) 70–140 mg/dL; time above range (TAR) >180 mg/dL and >250 mg/dL; time below range (TBR) <70 mg/dL, and days of CGM sensor use over 12 months. 17 Secondary outcomes included the proportion of participants meeting the TIR target of >70%.
Use of the high alert setting
The Dexcom high alert is a customizable setting in the Dexcom app that alerts users when their sensor reading is at or above their selected glucose level. The high alert setting appears as a yellow line on the user’s glucose graphs. The default setting is 250 mg/dL with a customizable range from 100 mg/dL to 400 mg/dL. Participant behaviors related to the use of the high alert feature were evaluated in two ways. First, the relationship between having the high alert setting enabled or disabling the default high alert setting of 250 mg/dL and the change in TIR and TITR at 12 months was evaluated. The second analysis examined the impact on change in TIR and TITR at 12 months in participants who: (1) disabled the high alert; (2) raised the setting to >250 mg/dL; (3) maintained the default 250 mg/dL setting; or (4) lowered the setting to <250 mg/dL.
Statistical analysis
Analyses were performed for the cohort overall and stratified by adults <65 years of age (younger) and ≥65 years of age (older). Paired t-tests were performed to assess overall and within-group change in all glycemic metrics and days of CGM sensor from baseline measure to 6 and 12 months. Welch’s t-test was used for between-age group comparisons of younger and older adults. Data were analyzed using Python, with significance defined as P < 0.05. A Bonferroni test with a P value threshold of 0.005 was performed to account for multiple comparisons for CGM metrics.
Results
Study population
A total of 3,840 CGM users, n = 3,364 (87.6%) <65 years and n = 476 (12.4%) ≥65 years, met the inclusion criteria and were included in the analysis. Participant baseline characteristics are presented in Table 1.
Participant Baseline Characteristics
Data are reported as mean (SD), unless otherwise indicated.
n = 1 reported gender as other.
Self-reported diabetes duration n = 3,544 (all), n = 3,122 (<65 years), and n = 422 (≥65 years).
CGM use and change in glycemic control
CGM use was high, 84.7% (15.9%) of days over 12 months. Significant improvements in all glycemic metrics not meeting target levels at baseline were observed at 6 months with continued improvement at 12 months (Fig. 1, Table 2). The improvements in TIR and TITR were attributable to reduced time in hyperglycemia. Outcomes were similar for the younger and older adult cohorts except for the change in TITR (P < 0.001), which was lower at baseline in the younger group and increased by a greater amount than the older group (Table 3). Overall, the 17.3% increase in TIR represents an additional 4.2 h spent in the target glycemic range. The 0.5% decrease in GMI from 8.1% to 7.6% at 12 months satisfies the new Health Effectiveness Data and Information Set (HEDIS) clinical metric, GMI of <8.0%, as an indicator of adequate control of diabetes. 18 The proportion of CGM users meeting the target TIR of >70% increased from 0% to 43.9% in the younger cohort and from 0% to 37.4% in the older cohort at 12 months (P = 0.008).
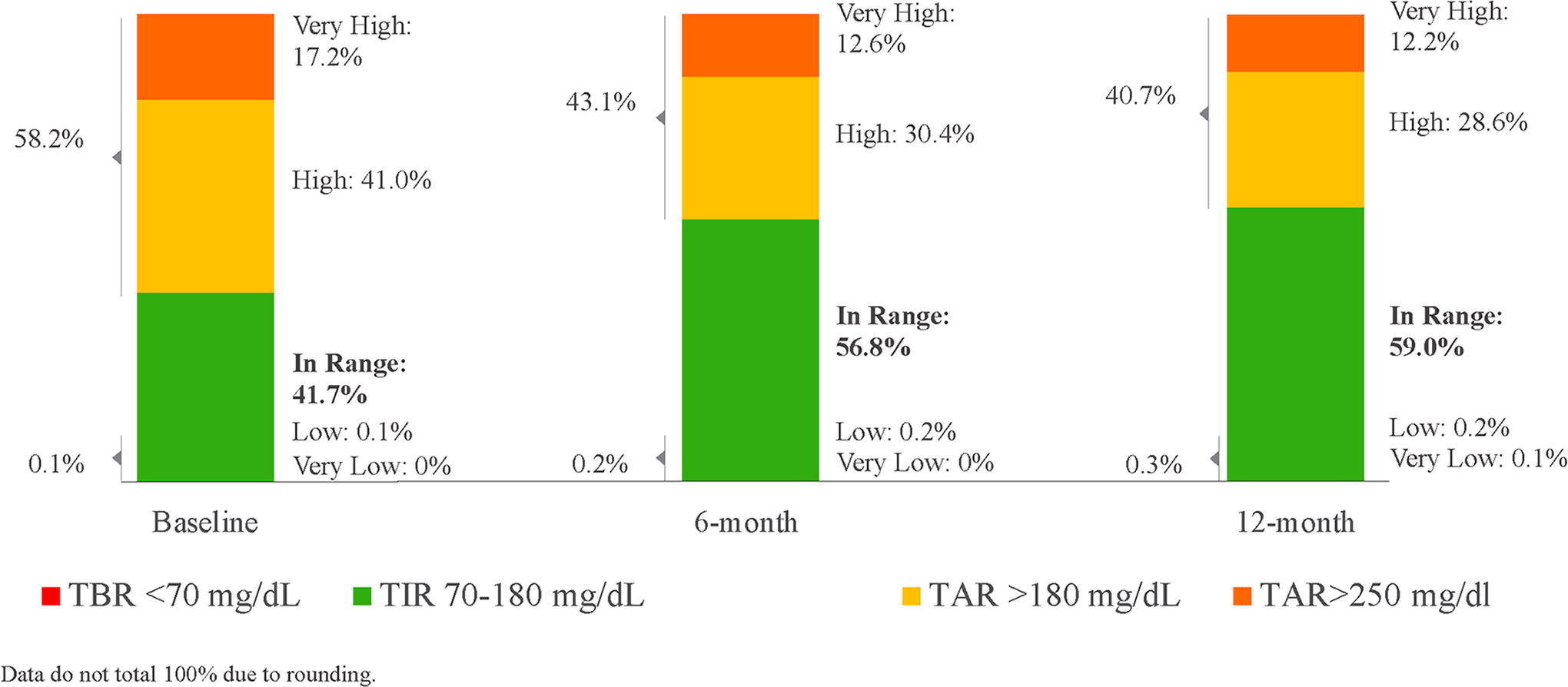
Time in ranges at baseline, 6 months, and 12 months for adults with noninsulin-treated T2D. T2D, type 2 diabetes.
Changes in Continuous Glucose Monitoring-Derived Glycemic Control from Baseline to 6 Months and 12 Months in Adults with Noninsulin-Treated T2D
Data are reported as mean (SD). A paired t-test was performed to assess changes in CGM metrics from baseline to follow-up time points. GMI, glucose management indicator; CV, percentage coefficient of variation; TIR, time in range; TITR, time in tight range; TAR, time above range; TBR, time below range.
Changes in Continuous Glucose Monitoring-Derived Glycemic Control from Baseline to 6 Months and 12 Months in Younger and Older Adults with Noninsulin-Treated T2D
Data are reported as mean (SD). A paired t-test was performed to assess changes in CGM metrics from baseline to follow-up time points.
P value for Welch’s t-test was used for between-age group comparisons of younger and older adults. BL, baseline; GMI, glucose management indicator; CV, percentage coefficient of variation; TIR, time in range; TITR, time in tight range; TAR, time above range; TBR, time below range.
Use of the high alert setting
Use of the high alert was similar between the younger and older age groups. Most participants either maintained the high alert or customized its threshold setting (n = 2,925; 76.2%), while a smaller percentage disabled the alert (n = 915; 23.8%). Participants who maintained or customized the alert had a greater increase in TIR and TITR compared with participants who disabled the alert (Fig. 2).
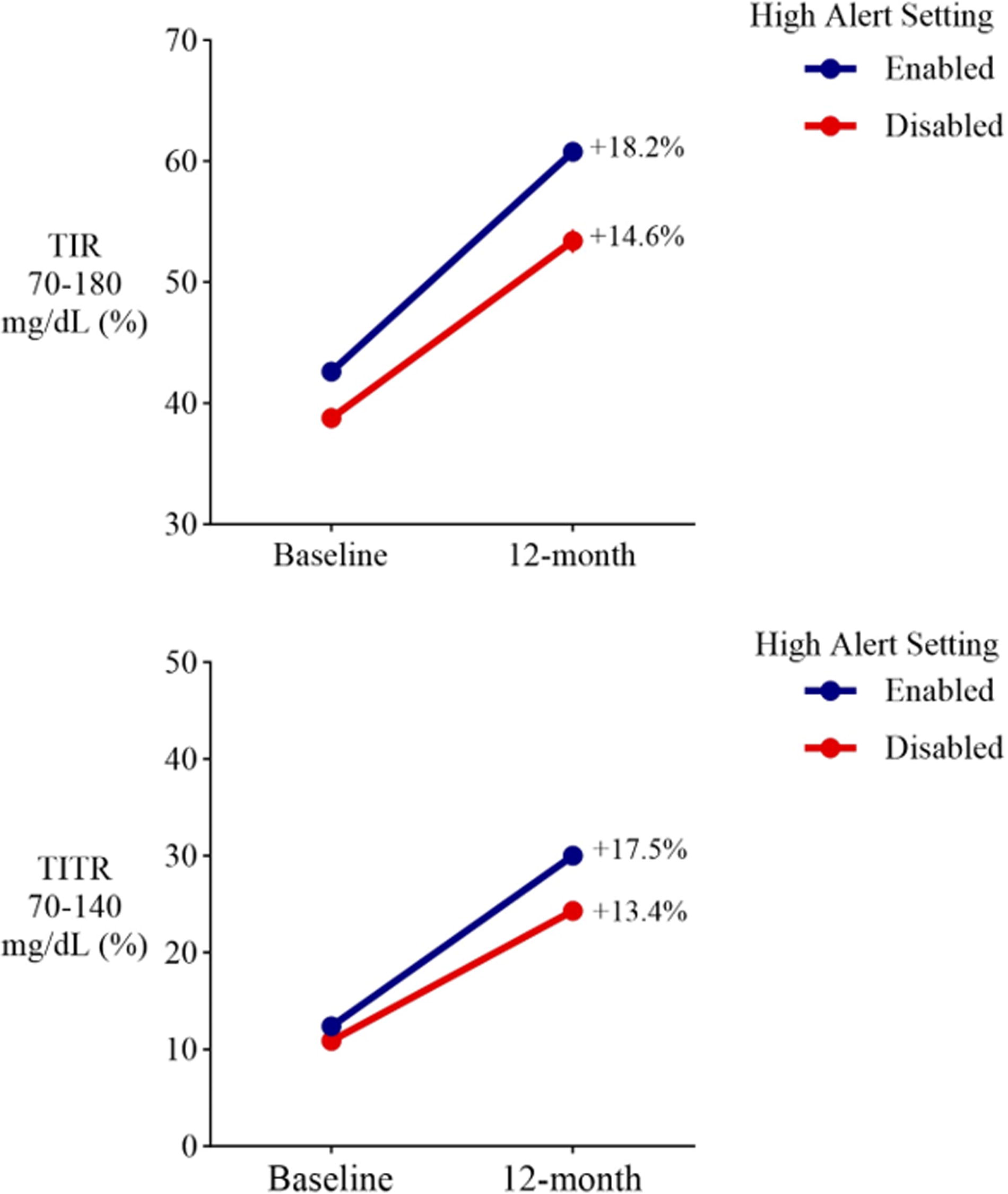
Changes in TIR and TITR at 12 months in adults with noninsulin-treated T2D with the high alert enabled or disabled. Baseline and 12-month TIR and TITR in participants who had the high alert feature enabled are shown in blue (n = 2,925 patients; 76.2%) or who had the high alert feature disabled shown in red (n = 915; 23.8%) at 12 months. TIR, time in range;TITR, time in tight range.
Changes in TIR and TITR based on customization of the high alert setting (lowered the glucose level, maintained the default, increased the glucose level, or disabled the alert) are shown in Figure 3. The most common change was to increase the high alert setting to >250 mg/dL (n = 1,483; 38.6%). A smaller percentage lowered the setting to <250 mg/dL (n = 512; 13.3%). The greatest increases in TIR were observed in participants who either lowered or maintained the default threshold of 250 mg/dL. The most notable increase in TITR (+22.8%) was observed in participants who lowered the high alert threshold.
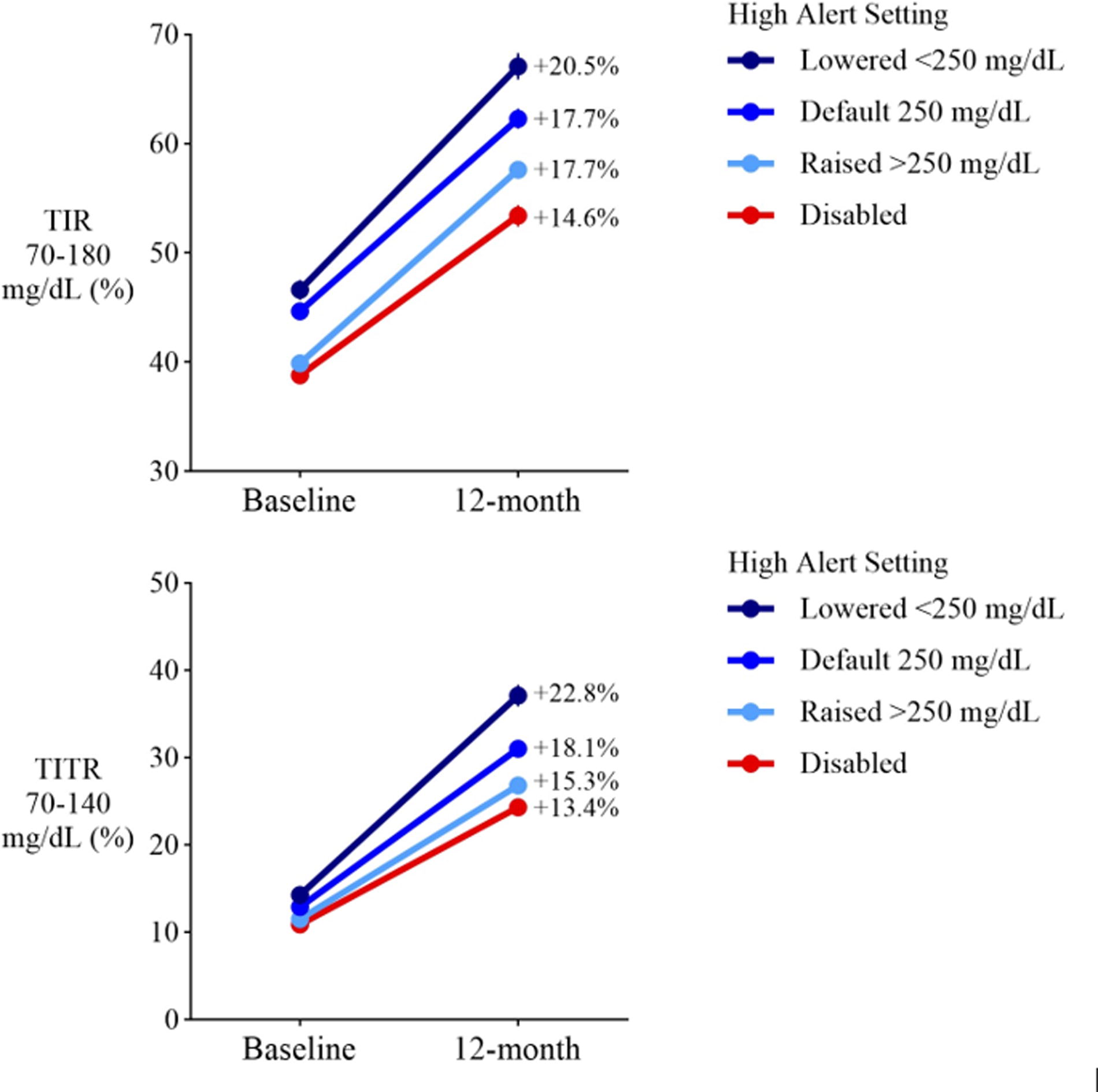
Changes in TIR and TITR at 12 months in adults with noninsulin-treated T2D who customized the high alert setting. Baseline and 12-month TIR and TITR in participants who had lowered the high alert setting to <250 mg/dL (n = 512; 13.3%; dark blue), maintained the default high alert setting of 250 mg/dL (n = 930; 24.2%; medium blue), raised the high alert setting to >250 mg/dL (n = 1,483; 38.6%; light blue), or disabled the high alert (n = 915; 23.8%; red) at 12 months.
Discussion
This large real-world, observational study of >3,800 adults with suboptimally controlled T2D not using insulin demonstrated that Dexcom CGM use was associated with clinically meaningful improvements in glycemic control at 6 months with ongoing improvement at 12 months. Similar benefits were generally observed in younger and older adults, including a 0.5% decrease in GMI, a 17% increase in TIR and a 16% increase in TITR that was attributable to a decrease in TAR. CGM use was high, with the use of CGM on approximately 85% of days, over the 12 months. Use of the Dexcom high alert feature was associated with the greatest improvements in TIR and TITR.
A limited number of studies have evaluated CGM use in noninsulin-treated T2D, with only two studies reporting CGM metrics as an outcome. 13,16 Although these studies are not directly comparable with the present investigation due to prospective designs, shorter durations, interventions, and use of a different type of CGM, they provide helpful context for the results of our study. The first study, the IMMEDIATE randomized controlled trial (RCT) evaluated the use of intermittently scanned (isCGM) in conjunction with diabetes self-management education (DSME) compared with DSME alone in 116 adults with noninsulin-treated T2D over 16 weeks. 16 TIR increased by approximately 20% from 56% to 76% in the isCGM + DSME group and by approximately 8% from 58% to 66% in the DSME-alone group for an adjusted mean difference of 9.9% TIR. Significant improvements in TITR and TAR were reported. In our retrospective study, the 17% increase in TIR occurred without the structured intervention or monitoring of a clinical trial, and participants had a lower baseline TIR of 42% compared with the IMMEDIATE study. The second previously published study in the T2D noninsulin using population was a 24-week randomized parallel-group investigation in 93 adults in Japan. Significant improvement in CGM metrics with isCGM compared with self-monitoring of blood glucose was observed at 12 weeks, including a between-group difference in TIR of 2.4 h, which was similar to the IMMEDIATE study. 13,16
In the present study, the 17% increase in TIR is equivalent to an additional 4.2 h spent in the target glycemic range, which was attributed to a decrease in TAR. This finding has important near- and long-term implications as an increase in TIR >10% has been associated with a reduced risk of microvascular and macrovascular complications in people with T2D. 19 –23 In addition, more than 40% of participants meet the target TIR of >70% at 12 months when all had a baseline TIR ≤70%.
TITR is emerging as a metric of interest in clinical practice and research, possibly because it more closely reflects glycemic levels in individuals without diabetes. 19,24,25 Further study is needed, however, to understand the applicability of TITR based upon clinical profiles and goals, and to establish targets for this metric. 25 It is also important to note the strong correlation and overlap between TIR and TITR, 25 which was observed in the present study as there was a 17% increase in TIR and a 16% increase in TITR. Notably, there was a twofold increase in TITR at 6 months from a baseline level of 12%, with continuing improvement to 29% TITR at 12 months.
Another important outcome was the decrease in GMI to 7.6%, which meets the new HEDIS target of GMI <8.0%. 18 High achievement in this HEDIS measure is important to providers and payers because it is factored into Medicare’s star ratings, which provides financial incentives for high performing Medicare Advantage plans, which, in turn, may incentivize their providers on performance in the star rating measures. 26
CGM use was high, 85% of days over 12 months. To the best of our knowledge, this is the largest long-term report of self-selected CGM use by people with noninsulin-treated T2D, outside of a clinical study with a CGM use schedule. A recent literature review suggests intermittent CGM use, at least once every three months, as standard of care for people with T2D on noninsulin therapies. 19 As with glycemic targets, many patient factors may contribute to determining optimal CGM wear cadence and further research may support different recommendations based upon these factors.
Emerging evidence points to CGM as a motivational tool to help people with suboptimally controlled T2D successfully implement lifestyle changes. 19,27 We investigated participant behaviors specific to the Dexcom high alert, which showed that approximately 75% of participants maintained or customized the high alert default setting of 250 mg/dL, while 25% disabled the alert. Any use of the high alert resulted in greater improvement in TIR and TITR compared with those who disabled the alert. These improvements were larger among users who lowered their high alert threshold. However, even those who disabled that alert had a meaningful improvement in TIR of >10%. These findings are important for clinicians and Dexcom CGM users to underscore the benefits of using and customizing the high alert feature.
The key strengths of our study are the large sample size of individuals with noninsulin-treated T2D and long-term outcomes up to 12 months. While it is well known that glycemic improvements occur soon after starting CGM use, 4,16,28 our study addresses a gap for long-term data. The significant improvements we observed at 6 months were sustained over 12 months, which suggests that shorter CGM studies may be predictive of longer term results in the noninsulin-treated T2D population.
This unique data set provides insights into self-determined, real-world CGM use patterns and the clinically meaningful benefits that can be achieved, independent of a clinical trial or intervention. In addition, similar improvements in glycemic metrics in younger and Medicare-age adults may support expanded CGM use to a larger noninsulin-treated population.
The present study has several limitations inherent in retrospective analysis and self-reported data. In addition, CGM users are not asked to provide information related to factors that may have contributed to glycemic improvements or related metabolic health improvements. For example, possible contributions of greater interactions with the health care team, including pharmacists, medication management, and potential use of other injectable therapies, including glucagon-like peptide-1 receptor agonists, and increased DSME engagements over the course of the 12-month observation period. These factors are also likely to contribute to positive outcomes independent of the type of CGM system. It should be noted that social determinates of health and insurance coverage are also important factors in CGM use and warrant further study.
Conclusions
In this large, real-world study of adults with suboptimally controlled T2D not using insulin, Dexcom CGM use was associated with meaningful improvements in glycemic control at 6 months, with continued improvement over 12 months. Self-selected CGM use was high over 12 months, suggesting benefits of ongoing CGM use in noninsulin-treated individuals with T2D not meeting treatment targets.
Footnotes
Authors’ Contributions
J.E.L.: Conceptualization, methodology, investigation, writing—original draft, writing—review and editing, supervision, project administration, and funding acquisition. L.H.J.: Methodology, investigation, and visualization. A.M.C.: Formal analysis, software, and data curation. C.G.P.: Writing—original draft and writing—review and editing. R.M.B.: Conceptualization, methodology, writing—original draft, and writing—review and editing.
Author Disclosure Statement
R.M.B. has received research support, consulted, or has been on a scientific advisory board for Abbott Diabetes Care, Ascensia, CeQur Corporation, Dexcom, Hygieia, Insulet, Johnson & Johnson, Lilly, Medtronic, Novo Nordisk, Onduo, Roche, Sanofi, United Healthcare, and Zealand. C.G.P. has received consulting fees from Abbott Diabetes Care, Biomea Fusion, Dexcom, Hager, Insulet, Roche Diabetes Care, and Tandem. J.E.L., L.H.J., and A.M.C. are employees of Dexcom.
Funding Information
Funding for the study was provided by Dexcom, Inc.
