Abstract
Background:
Scuba diving was previously excluded because of hypoglycemic risks for patients with type 1 diabetes mellitus(T1DM). Specific eligibility criteria and a safety protocol have been defined, whereas continuous glucose monitoring (CGM) systems have enhanced diabetes management. This study aims to assess the feasibility and accuracy of CGM Dexcom G7 and Free Style Libre 3 in a setting of repetitive scuba diving in T1DM, exploring the possibility of nonadjunctive use.
Material and Methods:
The study was conducted during an event of Diabete Sommerso® association in 2023. Participants followed a safety protocol, with capillary glucose as reference standard (Beurer GL50Evo). Sensors’ accuracy was evaluated through median and mean absolute relative difference (MeARD, MARD) and surveillance error grid (SEG). Data distribution and correlation were estimated by Spearman test and Bland–Altman plots. The ability of sensors to identify hypoglycemia was assessed by contingency tables.
Results:
Data from 202 dives of 13 patients were collected. The overall MARD was 31% (Dexcom G7) and 14.2% (Free Style Libre 3) and MeARD was 19.7% and 11.6%, respectively. Free Style Libre 3 exhibited better accuracy in normoglycemic and hyperglycemic ranges. SEG analysis showed 82.1% (Dexcom G7) and 97.4% (Free Style Libre 3) data on no-risk zone. Free Style Libre 3 better performed on hypoglycemia identification (diagnostic odds ratio of 254.10 vs. 58.95). Neither of the sensors reached the MARD for nonadjunctive use.
Conclusions:
The study reveals Free Style Libre 3 superior accuracy compared with Dexcom G7 in a setting of repetitive scuba diving in T1DM, except for hypoglycemic range. Both sensors fail to achieve accuracy for nonadjunctive use. Capillary tests remain crucial for safe dive planning, and sensor data should be interpreted cautiously. We suggest exploring additional factors potentially influencing sensor performance.
Background
Recreational diving has attracted more and more people around the world, amateurs and “lovers” alike. Among them, there is also an increasing number of people with type 1 diabetes mellitus (T1DM) who were once excluded from this activity. In fact, until the 90s, diving was forbidden for people with diabetes because of the risk of hypoglycemia and its potentially lethal consequences while underwater. In the following years, retrospective observational data collected in the United States by the Divers Alert Network led to a slow opening of the scientific world and worldwide medical diabetes community to this activity. A protocol was developed 1 based on serial capillary blood glucose checks before and after each dive, and specific criteria were established for defining eligibility of people with T1DM for diving activity. Those criteria were centered on glycemic control, presence or absence of complications, and hypoglycemia awareness. 2 –6
Remarkably, in the last 10 years, while recreational diving was opening toward people with diabetes, we have experienced the widespread diffusion of flash and continuous subcutaneous glucose monitoring systems (FGM and CGM). Their high reliability and the progressive improvement in analytical and clinical accuracy have allowed their use to become nonadjunctive to self-monitoring of blood glucose (SMBG), so much as to be essential in routine clinical practice, paving the way for artificial pancreas systems. The excellent patient acceptance rate has also contributed significantly to make them an almost indispensable tool for glycemic monitoring and thus optimization of therapy and glucose control in patients with T1DM. One of the most appreciated aspects is the possibility of reducing capillary measurements, which can be limited to particular situations and contexts. These certainly include physical activities, such as scuba diving, where safety is essential and CGM data must be still interpreted with caution. 7,8
Indeed, the assessment of the accuracy of these devices in the context of scuba diving remains to be investigated at present, with only a few data in the literature having evaluated this performance in comparison to capillary checks before/after diving and even fewer under pressure conditions comparable with a real dive.
Aim
In this context, the purpose of the present study is (1) to evaluate the feasibility of use and the analytical and clinical accuracy of two widely commercialized CGM systems (Dexcom G7 9 and Free Style Libre 3 10 ) in divers with T1DM participating in a campus of repetitive recreational scuba diving and (2) to consider the possibility of a nonadjunctive use of the CGM in the particular setting of recreational scuba diving in patients with T1DM.
Material and Methods
Setting
Diabete Sommerso® (diabetes underwater) is a nonprofit association founded in Italy in 2011, in the wake of a research project started in 2004 at the Diabetes Center of Niguarda Hospital, Milan (https://www.diabetesommerso.org). Its goal is to coordinate and organize activities merging people with T1DM and recreational diving. It is a guidance for all patients with diabetes interested in this sport, and from a scientific–technical standpoint, it brings together various professionals involved (i.e., diabetologist, hyperbarist, sports physician) stimulating mutual discussions.
The association’s activities include educational school camps, during which patients with T1DM are trained in scuba diving and glycemic management, obtaining an Open Water Diver and more advanced licenses, and other activities lasting from 1 to 7 days addressed to people already certified and trained. Among them, the “blue week” takes place every year in different locations, as conclusion of the association’s annual activities. During this week, therapeutic education activities and data collection are carried out.
Case selection and study design
In 2023, the “blue week” was held from October 28 to November 4 in the Maldives. Twenty-four people, including patients with T1DM, from Italy and Switzerland participated. Participants who signed informed consent from the nonprofit organization were included in the study. All people applied a Free Style Libre 3 sensor on the arm and a Dexcom G7 on the same arm or abdomen, covered with TegadermTM and tape reinforcement patches approximately 30 h before the first dive. All participants also received a Beurer GL50 Evo glucometer and consumables to perform all necessary capillary blood glucose tests during the week. Participants were asked not to calibrate the Dexcom G7 sensor, to minimize the chance of bias against the Free Style Libre 3. In case of failure or detaching before the normal deadline, the sensor was replaced (subject to material availability). Acquisition of sensor data took place via dedicated apps (Dexcom G7 app and Free Style Libre 3 app) and glucometer data via the Beurer Health Manager Pro app. All participants with diabetes used capillary blood glucose readings, performed according to a safety protocol, to make therapeutic decisions. The protocol includes serial capillary blood glucose check at −60, −30, and −10 min before the dive and a check immediately after the dive. The condition for dive is to keep the blood glucose before the dive stable (changes < 15%) in the range of 150–250 mg/dL (8.3–13.9 mmol/L). Diving is not allowed in case of blood glucose >250 mg/dL (13.9 mmol/L) with ketonemia, >300 mg/dL (16.7 mmol/L) rising, <150 mg/dL (8.3 mmol/L) not rising, or <120 mg/dL (6.7 mmol/L). Participants were also provided with a small towel to keep in the dry bag and to use specifically to dry their hands before capillary tests. It is indeed well known as the wet environment in which the tests are performed (in particular at −10 min and after dive) can represent a source of inaccuracy of the capillary data. Information about diabetes duration, routine diabetes therapy, glucose control, complications, and comorbidities were collected through questionnaires and medical records obtained in the 6 months before the event. Divers wore wet suits during every dive.
Reference standard for glucose data
Capillary blood glucose was used as reference standard to assess the accuracy of Free Style Libre 3 and Dexcom G7. Sensor glucose data were collected 1 min before or after the SMBG data.
Measures
Regarding analytical accuracy, considering the non-Gaussian distribution of our data, we first calculated median absolute relative difference (MeARD). However, it is known that using MeARD reduces the impact of outliers compared with mean absolute relative difference (MARD), and, from a clinical point of view, outliers are of relevance when the patient is adapting his therapy on such a value. 11 On this basis, we decided to perform both MeARD and MARD on overall data, within the glycemic ranges <70 mg/dL (<4 mmol/L), 70–180 mg/dL (4–10 mmol/L), >180 mg/dL (>10 mmol/L), 180–250 mg/dL (10–13.9 mmol/L), >250 mg/dL (>13.9 mmol/L), and in the safety protocol suggested range between 150 and 250 mg/dL (8.3–13.9 mmol/L), according to gender (assuming a potential difference linked to body composition), the time of the day (fasting and >8 h after last dive vs. rest of day), during the days of the week, and per single patient. For the Dexcom G7, we also considered and analyzed separately according to the site of application (arm vs. abdomen).
In addition to considering the MARD threshold of 10%, 12 to assess the possibility of using the two sensors in nonadjunctive mode, we performed an analysis according to the criteria established by the Food and Drug Administration (FDA) in 2017 for integrated continuous glucose monitoring (iCGM). 13 The conditions listed in points (v, A-I) have been verified. It was not possible to carry out the assessments for items J and K concerning the rate of glucose change because of the frequency of the capillary blood glucose sampling we performed.
The clinical accuracy was analyzed via Surveillance Error Grid Analysis using a web-based tool available at https://www.diabetestechnology.org/seg/.
Satisfaction and perceived accuracy of the two sensors were evaluated by a questionnaire completed within 7 days from the end of the week.
Statistical analysis
The two-tailed Spearman test was performed to estimate the correlation between capillary and sensor data. Bland–Altman plots were used to depict the distribution of the data and the bias between sensor data and capillary data. Wilcoxon and t-test were performed to assess the significance of MeARD and MARD results and significance of analyses according to FDA criteria for iCGMs. 13 The ability of the two sensors to correctly identify hypoglycemia was assessed by evaluating sensitivity, specificity, and predictive values, used to calculate the diagnostic odds ratio (DOR). The analysis was conducted on 3 levels of hypoglycemia: level 1 or mild (54–69 mg/dL; 3–3.8 mmol/L), level 2 or moderate (<54 mg/dL; < 3 mmol/L), and level 3 or severe (requiring assistance).
Statistical analysis and figures were performed using GraphPad Prism v7 (GraphPad Software, Boston, MA).
Results
Participants’ characteristics
Thirteen patients with T1DM, 9 females and 4 males, were included in the study. The median age was 37 years (interquartile range [IQR] 17 years), and duration of diabetes was 23 years (IQR 16 years). Diabetes treatment consisted of an advanced hybrid closed loop (AHCL) system (Medtronic MiniMedTM 780G, CamAPS® FX, Tandem Control-IQTM or Loop) in 9/13 patients, patch pump + sensor monitoring in 2/13 patients, and multiple daily injections (MDI) + sensor monitoring in 2/13 patients. During the camp, patients managed their therapy independently and with different approaches: patients usually on Omnipod and one of those on Medtronic 780G switched to MDI therapy. The other patients in closed loop (with a third sensor—Dexcom G6) maintained this therapy, disconnecting the pump in the period immediately preceding the dive and reconnecting it at the end of the same, freely choosing from day to day whether to keep the device in closed or open loop based on the alignment of Dexcom G6 data with capillary data. Glycemic control was good to optimal in the quarter preceding the study week (median HbA1c 6.7%, IQR 0.9%), with a median insulin requirement of 35 IU/day (IQR 7 IU/day) and 0.54 IU/kg/day (IQR 0.11 IU/kg/day). No patient had a history or regular paracetamol intake >1 g/day or vitamin C >500 mg/day, nor they did during the “blue week.” The characteristics of participants are summarized in Table 1.
Patients Characteristics
AHCL, advanced hybrid closed loop; AOWD, advanced open water diver; BMI, body mass index; CGM, continuous glucose monitoring; CSII, continuous subcutaneous insulin infusion FGM, flash glucose monitoring; IQR, interquartile range; MDI, multiple daily injections; NPDR, nonproliferative diabetic retinopathy; PADI, Professional Association of Diving Instructors; TDD, total daily dose; yrs, years.
Seven Dexcom G7 sensors (of which 6 were replaced) and 5 Free Style Libre 3 sensors (all replaced) stopped working sooner than expected, and an accidental overnight detachment of a Dexcom G7 sensor occurred (not replaced).
Glycemic data
Data were collected between one dive and the next, both at the timing established by the safety protocol (−60, −30, and −10 min before and immediately after each dive) and during the day/night regardless of the diving schedule. The total number of dives considered was 202, at an average depth of 27.1 meters and constant water temperature of 28–29°C without thermocline, with an average duration of ∼45–60 min each. Overall, 625 capillary/Dexcom G7 data pairs and 696 capillary/Free Style Libre 3 data pairs were available for statistical analysis.
Glucose profile from predive to postdive will be the subject of a separate publication in which the trends of capillary glycemia will be analyzed, while in this publication, we focused on evaluating the accuracy of the two sensors.
Data distribution was not normal, and global correlation analysis between CGM and SMBG data showed a Spearman’s r of 0.58 (P < 0.0001) for Dexcom G7 (n = 625) and 0.94 (P < 0.0001) for Free Style Libre 3 (n = 696).
The average blood glucose value measured by the glucometer was 195 ± 74 mg/dL (10.8 ± 4.1 mmol/L), and the mean bias versus capillary data was 54 mg/dL (3 mmol/L) for Dexcom G7 and 25 mg/dL (1.4 mmol/L) for Free Style Libre 3.
MARD and MeARD data
The overall MARD was 31% for Dexcom G7 and 14.2% for Free Style Libre 3, whereas MeARD was 19.7% and 11.6%, respectively. Figure 1 shows data correlation, distribution, and overall MARD and MeARD.
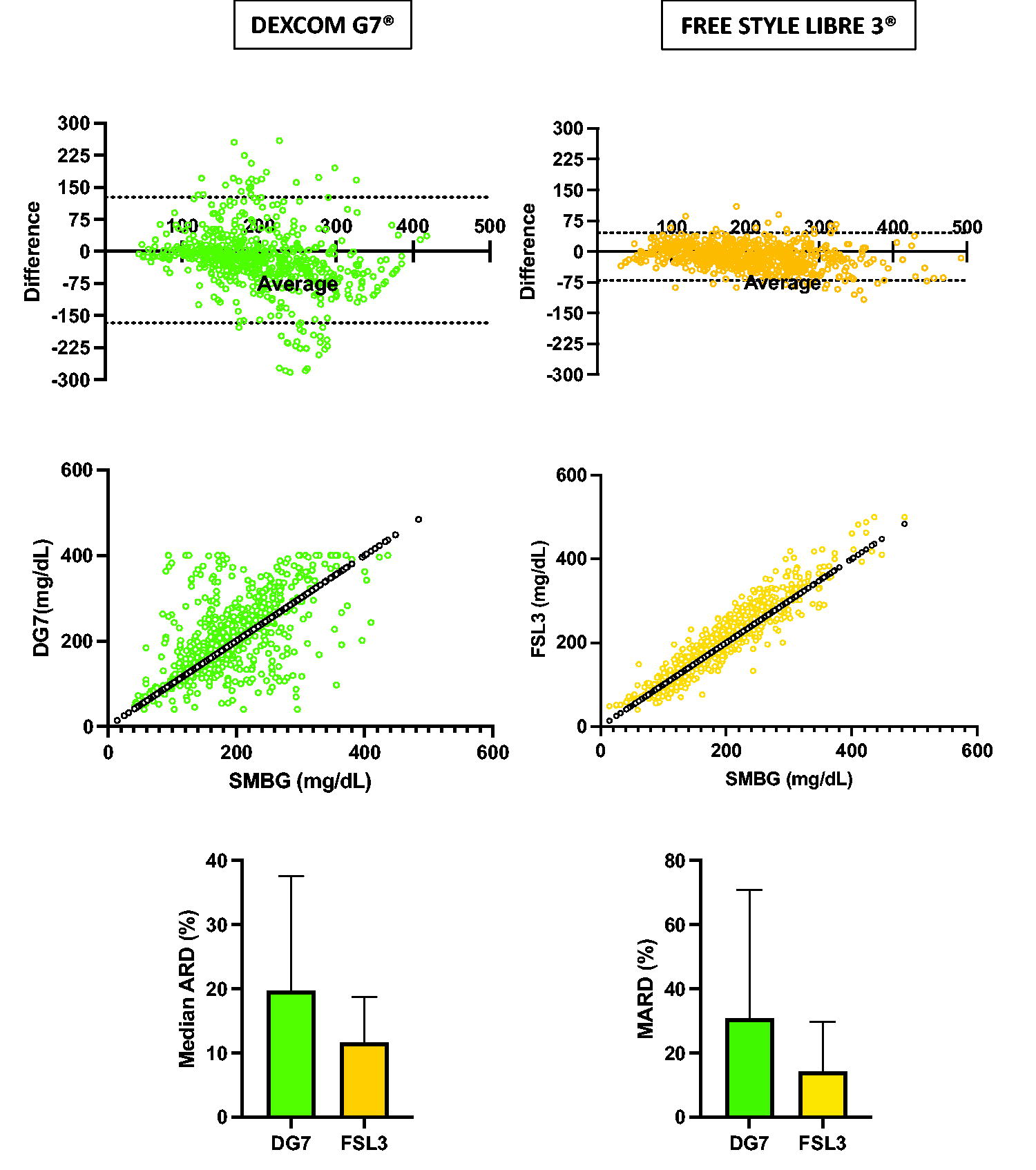
Data distribution (Bland–Altman plots), linear correlation (two-tailed Spearman), MeARD, and MARD of DexcomG7®/Free Style Libre 3® versus SMBG Beurer GL50 Evo®. MARD, mean absolute relative difference; MeARD, median absolute relative difference; SMBG, self-monitoring of blood glucose.
In both normoglycemic (70–180 mg/dL; 4–10 mmol/L) and hyperglycemic (>180 mg/dL or >10 mmol/L) ranges, Free Style Libre 3 showed better analytical accuracy than Dexcom G7, with a MARD of 14.3% (n = 291) versus 38.6% (n = 286) in the former and 12.2% (n = 383) versus 23.7% (n = 323) in the latter (P < 0.0001). The analysis of the MARD through different days of consecutive dives was performed: regarding Dexcom G7, we observed a clear deterioration in accuracy on day 2 (MARD 38%), with a partial recovery on day 3 and eventual stability (MARD 31.1–31.7%). In contrast, the Free Style Libre 3 maintained a stable accuracy (MARD 12.4%–13.7%) apart from a transient worsening on day 5 (MARD 17.6%).
We analyzed data according to gender, but we did not find clinically significant differences: MARDs of Dexcom G7 and Free Style Libre 3 were 34.6% and 14.6% in males and 29.8% and 14% in females, respectively. The single-patient analysis showed a homogeneous result in most of patients for Free Style Libre 3, whereas we observed a wide heterogeneity for Dexcom G7, not correlated with age, gender, and type of therapy (MDI vs. CSII) but with a significant impact of the sensor placement site in favor of the abdomen versus the arm (MARD 22.4% vs. 15.7%, P < 0.0001; MeARD 41.6% vs. 24.1%, P < 0.0001).
Sixteen episodes of hypoglycemia were identified at capillary control during the “blue week” (0.18 episodes/person/day). They were not severe, and they never occurred while diving or required the interruption of a dive. Within this glycemic range, the MARD was 41.2% for Dexcom G7 (n = 16) and 48.6% for Free Style Libre 3 (n = 22).
Table 2 shows the MARDs and MeARDs of the two systems in overall data and in the different subcategories of glycemic range, gender, daytime, different days of the study week, per single patient, and according to placement site (for Dexcom G7).
Dexcom G7 and Free Style Libre 3 MARD and MeARD (Overall and Subgroup Analysis)
MARD, mean absolute relative difference; MeARD, median absolute relative difference.
Dexcom G7 and Free Style Libre 3 Analysis According to iCGM FDA 2017 Criteria (Point V, A-I)
FDA, Food and Drug Administration; iCGM, integrated continuous glucose monitoring; SMBG, self-monitoring of blood glucose.
Other accuracy analysis
Considering only the glycemic data relating to the protocol (−60, −30, −10 min, and postdive, n = 407), we performed an analysis of the glycemic trend detected by SMBG and the two sensors between the timing −60 and −10 min and between −10 min and postdive: in the first case (−60 vs. −10 min), the SMBG variation was +4.9%, whereas Dexcom G7 identified a −3% and Free Style Libre 3 a +11.2%. In the second case (−10 min vs. postdive), the change was −3.7%, −5.3%, and −1.7%, respectively. We collected no data regarding the trend arrow of the sensors at the time of capillary glycemic controls.
Both sensors failed to meet the required thresholds at points (v, A-I) listed in the FDA criteria for iCGM, with the exception of Free Style Libre 3 for points H (no sensor value < 70 mg/dL corresponding to an SMBG data > 180 mg/dL) and I (no sensor value > 180 mg/dL corresponding to an SMBG data < 70 mg/dL). This analysis is reported in Table 3.
We also performed a clinical accuracy analysis according to the surveillance error grid (SEG) used for glucometers, which divides the differences between the analyzed tool and the gold standard into risk zones, requiring 97% of the data to be in the green “SEG no risk” zones to confirm an accuracy of <5% of data pairs outside the 15 mg/dL (0.83 mmol/L)/15% standard limits.
This analysis showed a percentage of data in the green A + B “SEG no risk” zones of 82.1% for the Dexcom G7 and of 97.4% for the Free Style Libre 3, with 17.9% and 2.6% of data in the remaining risk zones, respectively (Fig. 2).
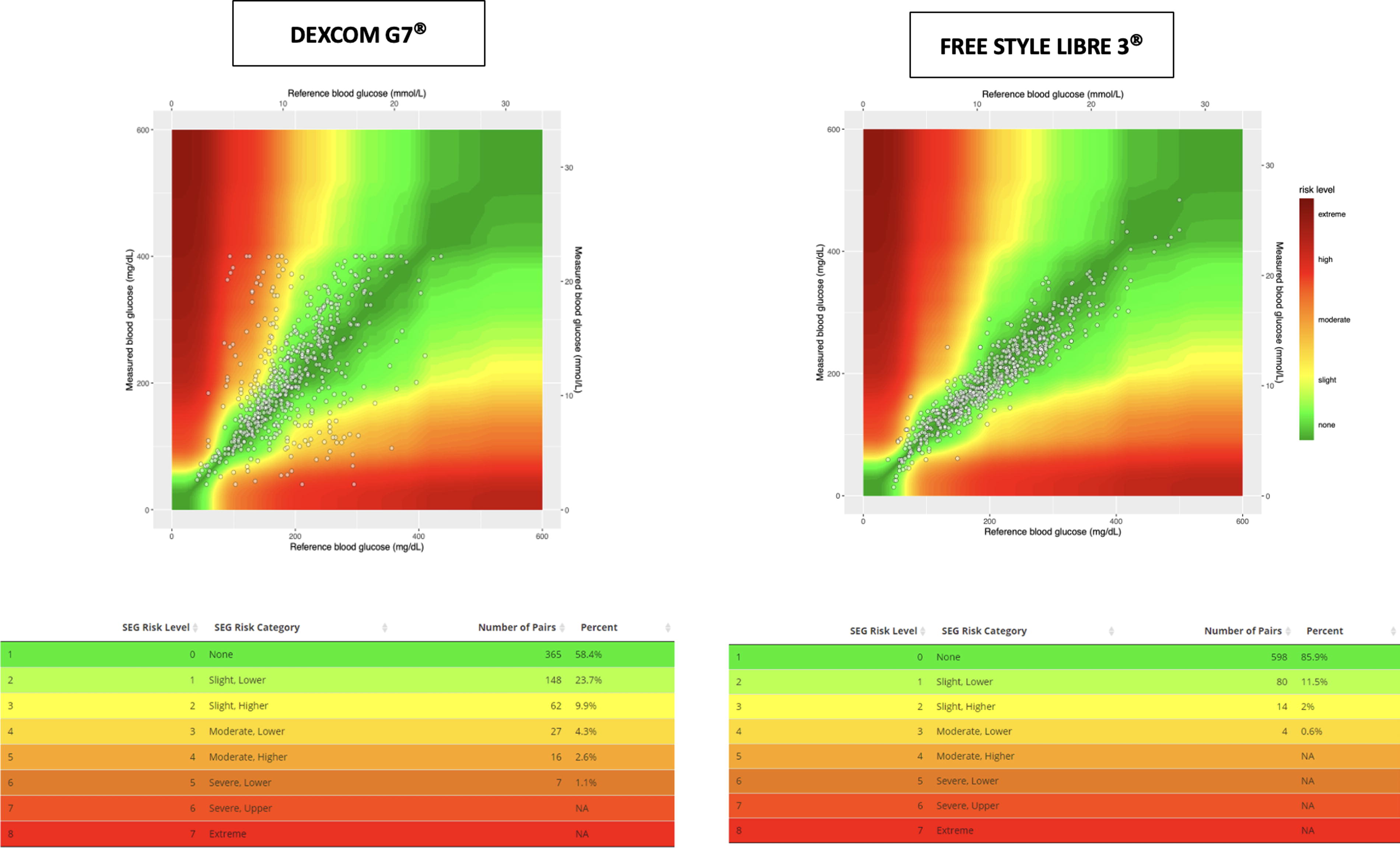
Surveillance error grid for DexcomG7 and Free Style Libre 3.
The ability of the two sensors to correctly identify hypoglycemia over the course of 7 days, before/after the dives, was assessed by contingency tables: as shown in Table 4, in a context of low prevalence of hypoglycemia, specificity and negative predictive value of Dexcom G7 and Free Style Libre 3 were quite similar. Sensitivity and positive predictive values of Free Style Libre 3 were higher than that of Dexcom G7 in the overall analysis and in level 2 hypoglycemia, with better performance of the Abbott sensor in terms of positive and negative likelihood ratio, and thus of DOR. In level 1 hypoglycemia, we find the opposite situation, with sensitivity, positive predictive value, positive and negative likelihood ratio, and DOR slightly in favor of Dexcom G7. This last parameter measures the effectiveness of the two sensors in identifying hypoglycemia in terms of the ratio between the probability that the sensor detects hypoglycemia if the subject is actually hypoglycemic and the probability that the sensor detects hypoglycemia when the subject is not. Thus, it allows us to comment on the performance of the two sensors independently of the low prevalence of hypoglycemia.
Results of Contingency Tables on the Identification of Hypoglycemia by the Two Sensors
Patient satisfaction analysis
The satisfaction questionnaire completed at the end of the week by participants revealed that 5/13 participants found problematic the application of the Free Style Libre 3 on the arm, but the minimal size and bulking of this sensor were appreciated by all of them. In total, 11/13 had a good subjectively perceived accuracy, and the overall assessment was positive for 12/13 users.
On the contrary, Dexcom G7 size and the possibility to apply the sensor in several body areas were appreciated by all subjects. Although the accuracy was subjectively rated as good only by 5/13 patients, the overall rating was positive for 9/13 users. No one reported skin reactions during the week or at sensor removal, except one case of skin inflammation in the area of application of the Tegaderm reinforcement patch.
Discussion
The convenience CGM offers is certainly a strength of these devices, especially in a situation such as dive preparation. However, it is essential to know how the device will perform in such a peculiar setting to avoid making decisions based on inaccurate and, therefore, potentially dangerous data.
Considering that the symptoms of hypoglycemia during diving can be easily confused with those derived from other problems, such as nitrogen narcosis or alternobaric vertigo, it is essential that divers with diabetes are properly trained and follow the safety protocols developed over the years, with the main goal of avoiding life-threatening situations such as hypoglycemia and ketoacidosis. Technology, now serving as a therapeutic tool especially in insulin-treated diabetes, can indeed enhance diving safety for individuals with diabetes. However, regarding the use of CGM, caution is needed: to date, no device has statistically demonstrated to meet the criteria for nonadjunctive use while diving or in contexts of multiple days of repetitive dives. Therefore, extreme caution must be taken in offering these devices in this particular high-risk setting, making sure that the person with diabetes is aware of this current limitation and knows how to correctly interpret values obtained from the sensor.
This study allowed us to collect additional data and clarify the accuracy of two of the newest subcutaneous glucose sensor in the context of scuba diving activity in people with T1DM.
First, it shows that CGM use is feasible and can be particularly useful for monitoring blood glucose during several days of repetitive recreational scuba diving in patients with T1DM.
Second, the level of accuracy required in terms of MARD 12 and according to more recent FDA iCGM criteria 13 to make nonadjunctive use of these data is not met for either sensor model.
In view of the “extreme setting,” the expectation of finding accuracy completely in accordance with data obtained in iCGM trials was certainly not reasonable. However, considering the peculiarity of scuba diving and the potentially fatal risk associated with an error in this context, it is desirable to achieve the highest possible accuracy, similar to what is required under standard conditions. On these premises, the objective of the present study is exploratory and descriptive to provide potentially useful information to diabetes professionals and patients and to reduce the possibility of potentially inappropriate and risky use of this technology.
Third, Free Style Libre 3 shows significantly better performance than Dexcom G7 in terms of MARD in all categories analyzed, with the exception of hypoglycemia, day −1 (day before the start of diving), and the day after the last dive, where data sample is very limited, and the tests do not reach the significance. In addition, analysis through the SEG showed that in the case of Free Style Libre 3, the requirement of 97% of the data in the “no risk” zone is met, whereas only 82.1% of the Dexcom G7 data falls within that zone. Finally, Free Style Libre 3 demonstrates a better diagnostic odds ratio for identifying hypoglycemia.
Regarding the analysis of the trend detected for blood glucose measurements conducted in accordance with the diving protocol, capillary data showed an overall predive trend (−60 vs. −10 min) increasing by +4.9%, whereas the Dexcom G7 exhibited an opposite trend (−3%) and the Free Style Libre 3, a consistent trend but of approximately double magnitude (+11.2%). These discrepancies are clinically of limited relevance as they translate into a maximum difference from SMBG of 0.8 mmol/L (14 mg/dL). Regarding the pre−post dive trend (−10 min vs. postdive), the trend of SMBG, Dexcom G7, and Free Style Libre 3 showed a consistent trend with a reduction of −3.7%, −5.3%, and −1.7%, respectively.
To date, we do not have an explanation to the large difference in accuracy between the Dexcom G7 and the Free Style Libre 3. On the basis of data previously collected by our group on 2019 on the Dexcom G6, 14 in which we found an overall MARD of 13.6%, such a marked difference was not expected. It should be underlined that in the previous data collection, patients were allowed to calibrate Dexcom G6, whereas in this case, they were expressly requested not to do so with G7. We therefore hypothesize that the Dexcom G7, although factory-calibrated, may need, in this particular context, one or more calibrations. The only factor we found to significantly affect the accuracy of the Dexcom G7was the site of application, with a significantly better performance if the sensor was placed on the abdomen.
Many potential factors could underly the deterioration of sensors performance: repeated changes in pressure and humidity over the course of the day and the wider glycemic range with greater glycemic variability than during standard living conditions could have stressed the sensors and contributed.
The literature in this field exists, but data collected are still limited. Multiple factors are mentioned and assumed as potentially affecting the accuracy of CGM systems during/between scuba diving, including pressure, temperature, and humidity. In addition, local variations at the sensor insertion site of parameters, such as vascularization and pH, can hold a part.
Researchers have investigated the performance of previous versions of FGM/CGMs under both hyperbaric chamber pressure 15,16 and real-life conditions during 17 or in the period of repetitive dives 18 –21 in healthy individuals or patients with T1DM. Tables 5 and 6 illustrate the main features of previously published studies in this field. Overall, evidence suggests that FGM/CGM use is preferable in patients with T1DM during recreational diving. In particular, the potential usefulness in identifying hypoglycemia, even unawareness, is emphasized to develop strategies to increase the safety for this activity in this patient category. These conclusions were drawn despite the confirmation, as in nondive conditions, of a worse performance in terms of accuracy in the hypoglycemic range, although a progressive improvement was observed over days (postulating a beneficial effect of calibrations and stabilization of the environment around the sensor). It should be specified that these findings in favor of CGM never mention a nonadjunctive use. It is also necessary to consider the scientific−cultural context of previous evidences, which was certainly different from today in several respects: (1) the accuracy cutoff needed for a nonadjunctive use of CGMs was not yet clearly defined; (2) AHCL systems, for which sensor accuracy is very important as the decision-making algorithms use the CGM as data source, did not yet exist; and (3) there was still some mistrust and concerns from both physicians and patients toward the nonadjunctive use of CGMs, tools that were relatively new in clinical practice. The recent technological evolution has significantly changed the perspective on all these aspects: First, the nonadjunctive use threshold of the MARD has been defined on 2015 (<10%), 12 and specific FDA criteria for iCGM systems exist since 2017. 13 Second, AHCL systems were developed on this basis, with an awareness of the significance and reliability of the MARD and its limitations. 11,22,23 Third, the technological push and the convenience of using sensors versus the greater invasiveness and complexity of the capillary glucose meter have led to more (sometimes excessive) confidence in CGM data. This leads to less awareness of the potential risks of neglecting the capillary test, which remains necessary in some contexts. The idea to collect data for this study arose in this context and from the observation of how in real life, many patients with T1DM, especially in a hostile environment such as a dinghy, are understandably inclined to use sensor data rather than carrying out many capillary tests, as required by safety protocol (minimum 3 before and 1 after diving). However, convenience comes with price: the use of CGM data could lead to errors, with potentially fatal consequences. Even several hours after the dive (e.g., the night following a dive), CGM data remain unreliable and could represent a danger in case of AHCL.
Characteristic and Major Results of Articles Evaluating the Performance of FGM/CGMs Between Subsequent Repetitive Dives in Healthy Subjects or T1DM
NPV, negative predictive values; PPV, positive predictive values; Se, sensibility; Sp, specificity; T1DM, type 1 diabetes mellitus.
Characteristic and Major Results of Articles Evaluating the Performance of CGMs Under Hyperbaric Condition or While Diving in Diabetic Subjects
CEG, Clarke Error Grid; ICC, intraclass correlation coefficient; ISO-15197, International Organization for Standardization guideline 15197; MRD, mean relative difference.
It should also be mentioned that the accuracy of capillary glucose meters can also be affected in extreme situations by various environmental factors such as temperature and humidity. 24 To our knowledge, there are no studies that have validated capillary versus venous data in the context of scuba diving. The only data available are those obtained by Baines et al., 15 in the context of a hyperbaric chamber and then inserting the variable pressure. However, it is known that exposure to extreme temperatures and rapid temperature changes seems to have the greatest impact on the reliability of capillary data. It is essential to choose a glucometer that meets ISO 15197:2015 standards and to use it according to the approved conditions declared by the manufacturer. In our case, ambient temperatures were consistently around 28°C and humidity between 70% and 80%, with rare increases at around 90%, thus meeting the recommended conditions for the Beurer GL50Evo (temperature 10°C–40°C and humidity <90%).
One of the missions of Diabete Sommerso is to empower people with diabetes practicing diving activity, as part of a therapeutic education project. The safe practice of this activity can be achieved through good glycemic management: nowadays, it requires the conscious use of all available technological tools.
Data presented in this article constitute, to our knowledge, the largest published report to date about the use of CGM systems in a setting of repetitive recreational diving in people with diabetes. This is also the first and only dataset available for the Dexcom G7 and Free Style Libre 3 system (with the exception of the case report by Herold for the latter 17 ).
Other strengths of this study are also the head-to-head use of the two sensors without potential confounding factors in favor of one or the other. Finally, the context where the study was carried out represents well real-life conditions of divers with T1DM, thus including all those variables that cannot be reproduced in the context of hyperbaric chamber studies. One limitation of the present study is certainly the absence of the best gold standard reference method (venous sampling). At the same time, however, it must be considered that in real life, the reference method for patients is represented by capillary sampling. In addition, an evaluation of the performance of the sensors >24 h after the end of the dives is lacking, which could have shown accuracy changes. As stated, we chose not to calibrate the Dexcom G7 to reduce the risk for potential bias against the Free Style Libre 3, which does not offer this possibility. We wonder whether the performance of the Dexcom G7 would have been better if it had been calibrated (at start-up, one day after start-up, or repeatedly over the course of the week).
Possible future studies should aim at increasing the sample size, testing new sensors that are becoming available in clinical practice, and investigate the impact on sensor performance of other variables, such as the use of wetsuits versus dry suits, mixtures with different oxygen percentages (e.g., Nitrox, of particular interest in people with diabetes because of the possibility of reducing the risk of decompression sickness), different environmental conditions such as temperature and salinity (e.g., lake vs. sea diving), or of the subject such as the gender, body mass index, and heart rate.
Conclusions
This study shows, in a peculiar real-life setting of recurrent diving in people with T1DM, a significant superiority of the Free Style Libre 3 over the Dexcom G7 in terms of analytical and clinical accuracy, except for the hypoglycemic range and on the day after the diving cycle. Both sensors fail to achieve the standard of accuracy required for nonadjunctive use.
Therefore, capillary tests remain an essential tool for a safe dive planning, as recommended by international protocols.
The use of sensors is feasible and can represent an additional tool and support, especially in early identification of hypoglycemia, but data must be interpreted with great caution.
At present, why sensor performance would or would not be affected by one or more variables remains to be fully elucidated. We advise for further investigation considering calibration, insertion site, and other potentially influencing variables/factors.
Furthermore, based on our data, patients on AHCL systems using DexcomG7 or Free Style Libre 3 should be very cautious when using the system in a closed loop between subsequent dives or in the hours immediately following a dive. Additional studies are needed to further investigate this crucial aspect.
Footnotes
Acknowledgments
This project would not have been possible without Prof. Matteo Bonomo: thank you for starting and strongly believing in Diabete Sommerso project, which began 20 years ago and continues to enable many people with T1DM to safely discover the wonders of the underwater world and to increase scientific knowledge on the topic.
Many thanks to Prof. Matteo Bonomo and Dr. Alessandro Bisio for their support in writing and revising the article.
In addition, the authors would like to acknowledge the study participants who generously shared their time and insights.
Author Disclosure Statement
The authors have no relevant financial or nonfinancial interests to disclose.
Funding Information
This study did not receive any fund.
