Abstract
Background and Aims:
Guidelines now recommend inpatient continuous glucose monitor (CGM) use with confirmatory blood glucose measurements. However, the Food and Drug Administration has not yet officially approved CGM for inpatient use in large part because its accuracy has not been established in this setting. We tested the accuracy of the Dexcom G6 (G6) in 28 adults on an insulin infusion in a medical–surgical intensive care unit with 1064 matched CGM and arterial point-of-care pairs.
Methods:
The participants were on average 57.29 (SD 2.39) years, of whom 13 had a prior diagnosis of diabetes and 14 were admitted for a surgical diagnosis. The first 19 participants received the G6 without calibration and had a mean absolute relative difference (MARD) of 13.19% (IQR 5.11, 19.03) across 659 matched pairs, which just meets the critical care expert recommendation of MARD <14%. We then aimed to improve accuracy for the subsequent 9 participants using a calibration protocol.
Results:
The MARD for calibrated participants was 9.65% (3.03, 13.33), significantly lower than for uncalibrated participants (P < 0.001). Calibration also demonstrated excellent safety with 100% of values within the Clarke Error Grid zones A and B compared with 99.07% without calibration. Our protocol achieved the lowest MARD and safest CEG profile in the critical care setting and well exceeds the critical care expert recommendations. Our large sample of heterogenous critically ill patients also reached comparable accuracy to the MARD of 9% for G6 in outpatients. We believe our calibration protocol will allow G6 to be used with sufficient accuracy in inpatients.
Introduction
During the COVID-19 pandemic, the Food and Drug Administration (FDA) issued a nonobjection statement for the use of continuous glucose monitors (CGMs) in inpatients to facilitate efforts to minimize contact and conserve personal protective equipment. Since then, studies in inpatients have suggested that CGM can decrease hypoglycemic events and improve the overall glycemic control. 1 –3 Given these findings, the Endocrine Society released a Clinical Practice Guideline stating that CGM use is preferred over point-of-care (POC) blood glucose (BG) testing alone in a noncritical care inpatient setting when used with confirmatory POC BG measurements. 4 As such, CGM is emerging as the standard of care for glycemic control in inpatients. However, its accuracy compared with POC testing has not been clearly established in the inpatient setting. We chose to study CGM accuracy in the intensive care unit (ICU) since critically ill patients have additional factors such as vasopressor use, large-volume interstitial fluid shifts because of resuscitation or renal replacement therapy (RRT), and high glucose variability that may affect CGM accuracy. In addition, critically ill patients are most vulnerable to the adverse effects of poor glycemic control. 5,6 As such, establishing the accuracy of CGM in the ICU is a priority as it can be extrapolated to other inpatient settings with less variables that may confound CGM accuracy.
Several studies have shown that glycemic excursions are associated with adverse clinical outcomes, including mortality, end-organ failure, infection, and length of hospital stay in critically ill patients. 7 –13 However, precise glycemic targets in the ICU remain under debate, with a recent trial by Van den Berghe et al. demonstrating similar clinical outcomes for patients with intensive versus conventional glucose targets. 14,15 Based on existing trials, the American Diabetes Association recommends initiating insulin for persistent hyperglycemia (glucose >10 mmol/L or 180 mg/dL) and a target glucose range of 7.8 mmol/L (140 mg/dL) to 10 mmol/L (180 mg/dL) for most critically ill patients. 16 The current standard of care in critical care settings attempts to meet these targets by using frequent BG measurements from arterial line or capillary glucose samples that are analyzed on a POC glucose meter. If the accuracy of CGM can be validated in critical care settings, it has the potential to become a more efficient and effective standard of care.
The objectives of our study are first, to determine the accuracy of the Dexcom G6 CGM (G6; Dexcom, San Diego, CA) in ICU patients compared with the standard of care using arterial POC BG values and second, to optimize the accuracy of the G6 using a simple calibration protocol.
Methods
Study design
This was a single-center, prospective, observational study in a large, mixed medical–surgical academic ICU at Vancouver General Hospital (VGH; British Columbia, Canada). Thirty-eight participants older than 18 years who were on an insulin infusion and anticipated to continue infusion for ≥72 h were recruited with consent between April 2021 and October 2022. The first 27 participants enrolled received a factory-calibrated G6 as per the usual manufacturer’s recommendations for outpatient use. 17 No additional calibration was performed. The subsequent 11 enrolled participants had sensor calibration at 2, 12, and then 24 h post-insertion. The calibration protocol was designed for the study following consultation with the manufacturer Dexcom Inc., which considers the sensor warm-up period of 2 h and decreased sensor accuracy within the first 12 and 24 h. 17 –19 Patients were excluded from the study if they were younger than 18 years or expected to be on the insulin infusion for <72 h. Patients on substances known to interfere with the accuracy of the G6, including hydroxyurea and acetaminophen (>1 g every 6 h), as per the manufacturer’s manual. 17 Patients receiving large-volume fluid resuscitation, on vasopressors, or RRT were included in the study to provide a representative population sample. Consent was obtained from the patient or designated substitute decision maker.
Measurement
Nursing staff continued to perform POC BG measurements from arterial blood samples using the Accu-Chek II meter every 15 min to 4 h as per the VGH ICU insulin infusion protocol. The average frequency of POC BG measurement was every 2 h while the participant was on the insulin infusion. The G6 was attached to the upper arm of the participant instead of the abdomen used in outpatient settings for ease of access and to avoid interference from surgical sites, lines, and proning for respiratory failure. The G6 measured BG values every 5 min and the values were blinded from the study staff, patients, and patient care providers. All times of arterial BG measurements were recorded to the exact minute and compared with the G6 reading performed within 5 min of the arterial POC value.
The G6 sensor remained in place until the insulin infusion was stopped or the sensor had been used for 7 days, whichever was sooner. The 7-day data collection period was chosen arbitrarily as the duration of insulin infusion for critically ill patients at our center generally does not exceed 7 days and to avoid inadvertent continuation of sensor wear beyond the approved 10-day period. In the first part of the study, there was no calibration performed after sensor insertion, according to the validated manufacturer guideline for the outpatient setting. 17 In the second part of the study, the sensor was calibrated by study personnel at 2 h, 12 h, and 24 h post-insertion using a corresponding POC BG measurement (using arterial samples on an Accu-Chek II meter), and none thereafter for the duration of the sensor use.
Statistical analysis
Mean absolute relative difference (MARD) was used to analyze differences between arterial and CGM values. MARD was calculated as the average relative difference between matched pairs of arterial POC and G6 values for each participant, shown as a percentage with interquartile range. A modified Bland–Altman plot was used to visualize the bias between POC and G6 readings. The web-based tool used can be found at https://www.diabetestechnology.org/seg/. A linear mixed-model regression analysis using the R nlme package 20,21 was used to determine CGM accuracy in subgroups based on glucose ranges, RRT, and vasopressor use, all of which can potentially affect CGM accuracy. Categorical data were described as count (%), and continuous data were described as mean and standard deviation.
Our data were compared with the FDA accuracy requirements for integrated continuous glucose monitoring systems. 22 For G6 measurements <3.9 mmol/L (<70 mg/dL), the %15/15 criteria are met when the percentage of values within 15 mg/dL (0.8 mmol/L) of the POC value is calculated and the lower one-sided 95% confidence bound exceeds 85%. For G6 values from 3.9 to 10.0 mmol/L (70–180 mg/dL), the criteria are met when the percentage of values within 15% of the POC value is calculated and the lower one-sided 95% confidence bound exceeds 70%. For G6 values >10.0 mmol/L (>180 mg/dL), the criteria are met when the percentage of values within 15% of the POC value is calculated and the lower one-sided 95% confidence bound exceeds 80%. The %40/40 criteria are defined analogously and outlined in Table 2. A one-way analysis of variance was used for differences between categorical variables. The R ega package was used for Clarke Error Grid (CEG) analysis. 23 All analyses were performed in R.
The datasets generated during and/or analyzed in the current study are available from the corresponding author upon reasonable request.
Results
We recruited a total of 38 adults who consented to study participation. After exclusion of participants who withdrew consent, did not have data because of sensor failure, had early termination from minor side effects, or were outliers with MARD >25%, a total of 28 participants with 1064 matched arterial POC and CGM pairs were available for analysis (Fig. 1). One participant with preexisting coagulopathy terminated the study early due to minor bleeding at the sensor insertion site. No other adverse events related to sensor use were noted. One patient in the uncalibrated group had no data due to a sensor connection error. In the calibrated group, the medical chart was unavailable for analysis for one patient, and calibration could not be completed as per protocol for one patient.
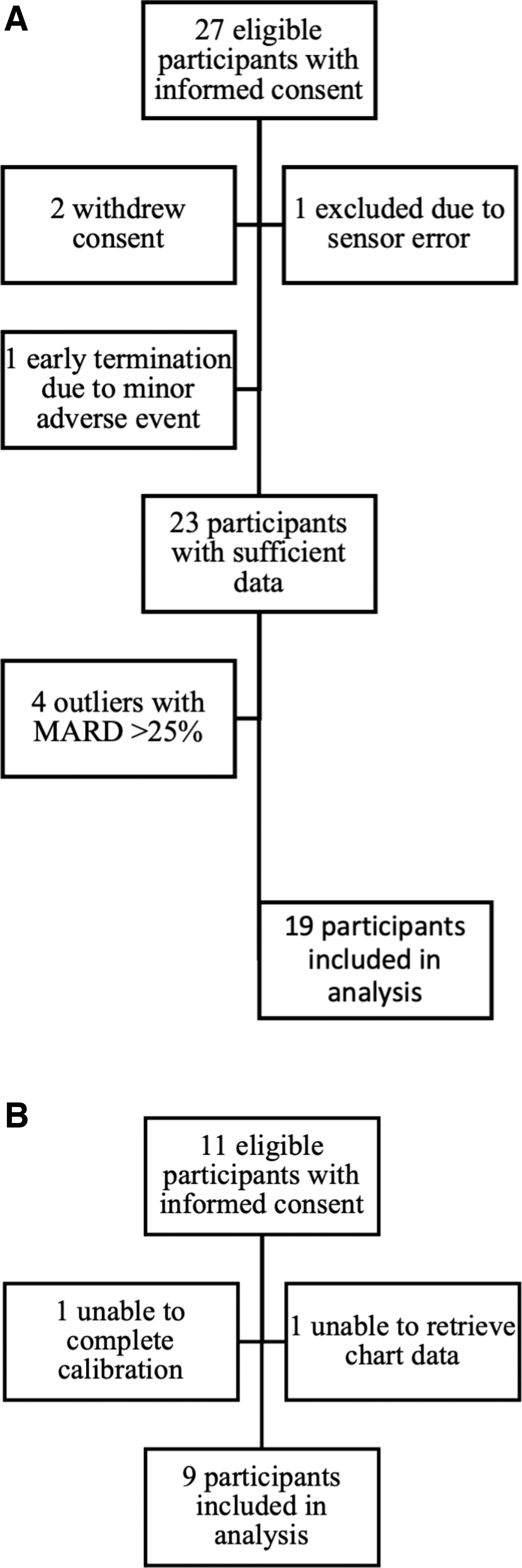
Participant flowchart for uncalibrated protocol
Participants had a mean of 99.04 ± 22.02 h (4.1 ± 0.92 days) of wear (range 2–221 h). There were 19 participants and 659 matched pairs in the uncalibrated arm, and nine participants and 405 matched pairs in the calibrated arm. There was a mean of 38 POC-CGM matched pairs per participant.
Demographic information
The participants were on average 57.29 ± 2.39 years old, with three female participants (Table 1). Thirteen participants had a history of diabetes, 13 were on vasopressors, and 11 underwent RRT (continuous RRT or hemodialysis) during the data collection period. There were more participants on vasopressors in the calibrated group compared with the uncalibrated group (eight vs. five participants), which was statistically significant (P < 0.01). There was no other statistically significant difference in participant demographic data between calibrated and uncalibrated participants.
Demographic Information of Study Participants
Statistically significant values on one-way analysis of variance test are in bold (P < 0.01).
SD, standard deviation.
There was no statistically significant difference in age, sex, diabetes history, medical versus surgical diagnosis, or vasopressor use between included and outlier participants. All outliers were uncalibrated participants. None of the outlier participants was on RRT. There was no significant difference in hemoglobin levels between included and outlier participants (P = 0.070).
Sensor accuracy
The overall MARD was 11.84% (interquartile range [IQR] 4.23, 16.59) across all 1064 matched pairs for the 28 participants. The MARD for uncalibrated participants was 13.19% (5.11, 19.03) across 659 matched pairs. The MARD for calibrated participants was 9.65% (3.03, 13.33) across 405 matched pairs, which was significantly lower than the MARD for uncalibrated participants (P < 0.01).
We analyzed all matched pairs as per the FDA %15/15 and %40/40 criteria. Overall, 100% of G6 measurements <3.9 mmol/L (<70 mg/dL) are within 15 mg/dL of the POC value, 74.24% of G6 measurements 3.9–10.0 mmol/L (70 mg/dL to 180 mg/dL) are within 15%, and 65.42% of G6 measurements >10.0 mmol/L (>180 mg/dL) are within 15%. The results in comparison with FDA criteria using lower 95% confidence bound values are summarized in Table 2.
Comparison of %15/15 and %40/40 Criteria Between Our Data and the FDA Guidelines 22
The %15/15 criteria are met when the percentage of G6 measurements are within 15 mg/dL or 15% of the corresponding POC value is calculated and the lower one-sided 95% confidence bound exceeds the FDA criteria. The %40/40 is defined analogously. The criteria are divided across glucose ranges.
FDA, Food and Drug Administration; G6, Dexcom G6; POC, point-of-care.
A modified Bland–Altman plot was done for all participants combined, which showed an overall mean relative difference (MRD) of 4% between G6 and POC measurements (Fig. 2). The uncalibrated group had an MRD of 7.6%, whereas the calibrated group had an MRD of −1.8%, indicating positive bias from the uncalibrated group.
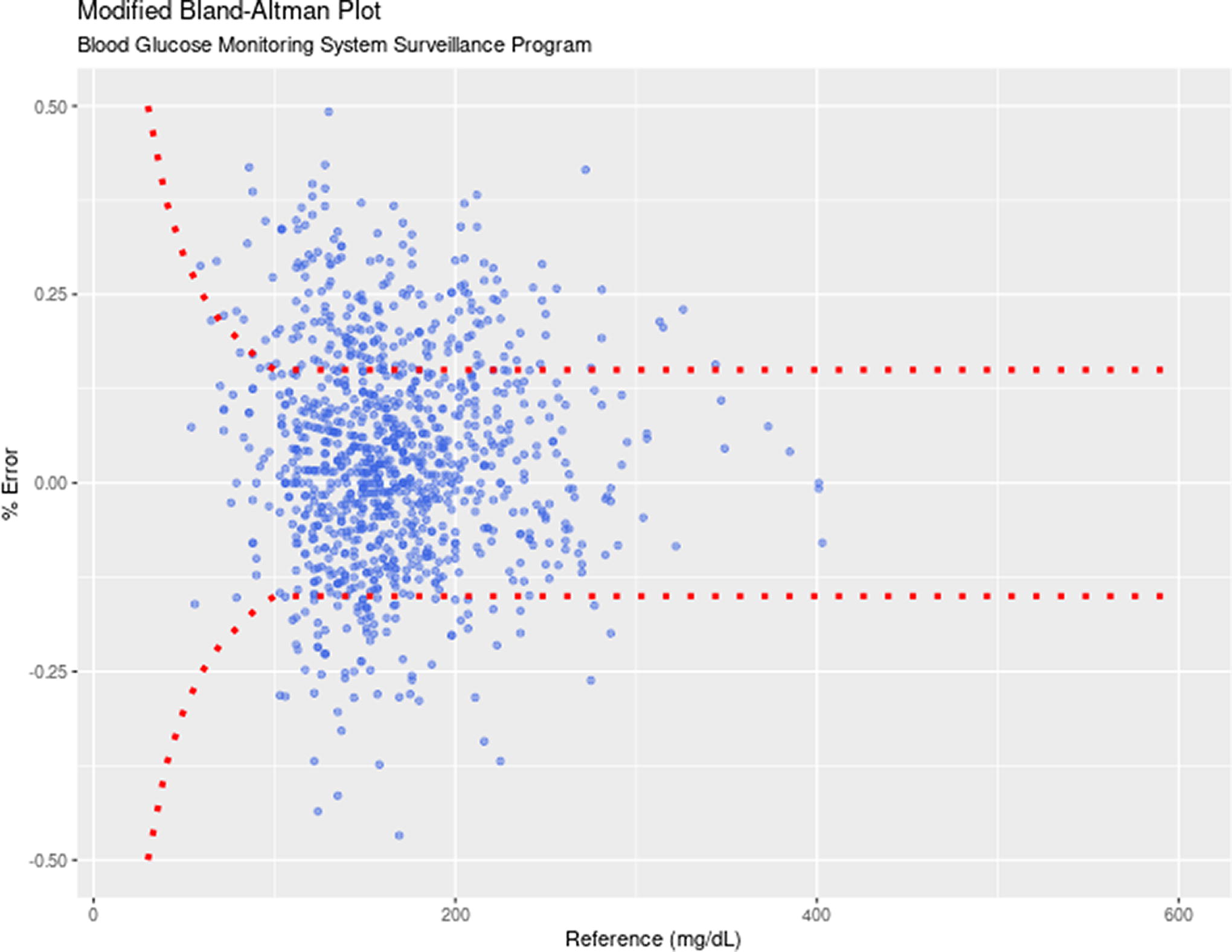
Modified bland–altman plot for POC and G6 pairs for all participants. G6, Dexcom G6; POC, point-of-care.
Sensor safety
CGM safety metrics were also calculated. As per the FDA guidelines, 22 at no point do we have G6 values of <3.9 mmol/L with corresponding BG of >10.0 mmol/L (>180 mg/dL), or G6 values of >10.0 mmol/L (>180 mg/dL) with corresponding BG of <3.9 mmol/L. Among the uncalibrated pairs, 99.1% were in CEG zones A and B, with 0.9% in zones C and D. Among the calibrated pairs, 100% were in CEG zones A and B (Fig. 3).
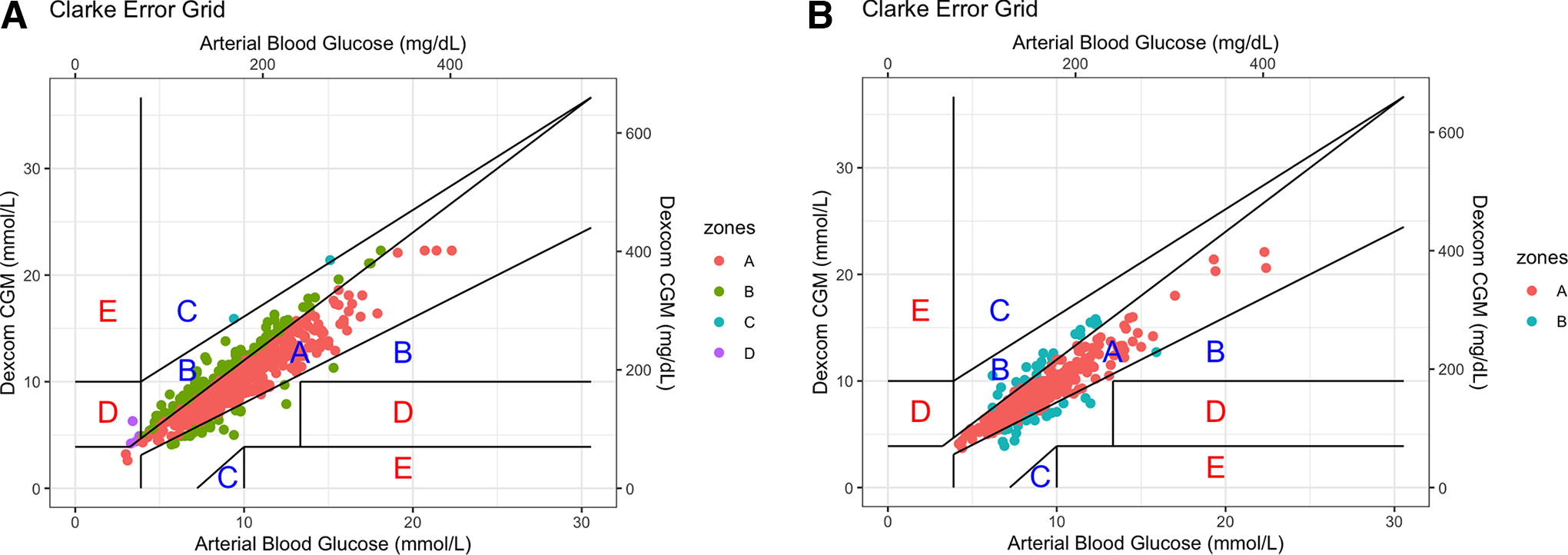
Clarke Error Grid of uncalibrated
Performance across vasopressor and RRT use
On a linear mixed-model regression analysis, there was no statistically significant difference in accuracy across participants who were on vasopressors or RRT. There were no statistically significant differences in %15/15 criteria or MARD between vasopressor and no vasopressor use (Table 3). There were no statistically significant differences in %15/15 criteria between RRT and no RRT use (Table 3). Of note, data could not be obtained on vasopressor use for one participant and for RRT use for another participant.
Accuracy Analysis Across Matched Pairs with and Without Vasopressor Use and with and Without Renal Replacement Therapy Measured in %15/15 Criteria
CI, confidence interval; IQR, interquartile range; MARD, mean absolute relative difference; RRT, renal replacement therapy.
Performance across glycemic ranges
There was no significant difference in accuracy across glycemic ranges ≥3.9 mmol/L (70 mg/dL) using %15/15 criteria (Table 4). There were insufficient glucose measurements below 3.9 mmol/L (70 mg/dL) for analysis. There was no significant difference in accuracy across glycemic ranges using a linear mixed-model regression analysis.
Accuracy Performance Across Glycemic Ranges Measured in %15/15 Criteria
There were insufficient matched pairs with glucose <3.9 mmol/L (70 mg/dL) for analysis. There was no statistically significant difference in accuracy between the glucose ranges.
Performance across hemoglobin ranges
Hemoglobin levels had an average of 89.57 (standard deviation [SD] ± 3.17) across all matched pairs. Hemoglobin levels were stratified as Hb <70, Hb 70–100, and Hb 100–140 mmol/L. There were 59 matched pairs with Hb <70 mmol/L, 783 with Hb 70–100 mmol/L, and 222 matched pairs with Hb 100–140 mmol/L. There was no significant difference in accuracy using MARD or %15/15 criteria across hemoglobin subgroups (Table 5).
Accuracy Performance Across Hemoglobin Ranges Measured in %15/15 Criteria
There was no statistically significant difference in accuracy between the hemoglobin subgroups.
Performance across duration of wear
For all participants, there was no significant difference between MARD at <24 h after insertion (13.32%) and at
Discussion
CGMs are emerging as the new standard of care for inpatients due to its potential benefit in reducing hypoglycemia and improving overall glycemic control. However, the accuracy standard for CGMs in the inpatient setting has not yet clearly been established. Critically ill patients are especially vulnerable to the effects of glycemic excursions and have a higher likelihood of hypoglycemia unawareness; therefore, it is of priority to study the accuracy of CGMs in this population. 24 Critical care settings pose a unique challenge as theoretically the use of vasopressors, large-volume shifts induced by fluid resuscitation or RRT, or low hemoglobin levels may affect CGM accuracy. 25 A recent single-center study comparing CGM accuracy in critically ill versus noncritically ill patients found a higher MARD for critically ill patients (16.3% vs. 13.8%). 26 Therefore, if CGM accuracy can be established in the critically ill patient setting, it can be extrapolated for accuracy in the general inpatient setting.
There is currently a lack of expert consensus on acceptable accuracy for CGMs in inpatient settings. The current FDA accuracy standards for integrated CGM devices require 85% of CGM measurements <3.9 mmol/L (<70 mg/dL) to be within 0.8 mmol/L (15 mg/dL) of a laboratory comparator and for 70% of measurements between 3.9 and 10 mmol/L (70 to 180 mg/dL) to be within 15%, and for 80% of measurements >10 mmol/L (180 mg/dL) to be within 15% of the comparator. 22 This differs from the criteria set by the International Organization for Standardization (ISO) in 2013, which suggests 95% of CGM values to be within 15% of comparator values for BG ≥100 mg/dL (5.6 mmol/L), 95% of values to be within 15 mg/dL (0.8 mmol/L) of comparator values for BG <100 mg/dL (5.6 mmol/L), and 99% of values to be within zones A and B of the consensus error grid. 27 The Critical Care journal expert panel suggests 98% of values within 12.5% of a reference standard for BG values <5.5 mmol/l (99.0 mg/dL) and an MARD of <14%. 28,29 The FDA and ISO guidelines do not differentiate between inpatient and outpatient criteria. Several studies on inpatient CGM accuracy have been published since the development of these guidelines; however, no single study, especially in the critical care setting, has been able to meet these criteria.
Given these uncertainties, current expert guidelines necessitate the use of confirmatory POC BG measurements when CGMs are used in inpatients. 4 However, there is no guideline on the indication or frequency of the confirmatory measurements. Many existing studies using CGM in inpatient settings have different protocols for confirmation of CGM values, which are often complex and necessitate several POC measurements per day to verify CGM accuracy. As such, for CGM to be accepted in practice as the standard of care for inpatients, there needs to be a consensus on accuracy standards and a calibration protocol for widely adapted CGM models.
Our data from a heterogenous mixed medical–surgical adult ICU population on IV insulin infusion showed that among 1064 matched CGM and arterial BG pairs for more than 28 participants, the G6 had a MARD of 11.84% (IQR 4.23, 16.59) overall, which was retained across glycemic ranges. Our overall MARD is comparable with other studies in critical care settings in participants with COVID-19 using the G6, which have reported an MARD of 11–21%. 30 –35
We demonstrated that by using a simple calibration protocol with sensor validation at 2, 12, and 24 h post-insertion, the accuracy of the G6 was significantly improved from an MARD of 13.19% (5.11, 19.03) for uncalibrated participants to an MARD of 9.42% (3.03, 13.33) for calibrated participants. As a comparison, the MARD for G6 in outpatients for adults is 9%. 18 This protocol also allowed for excellent safety with 100% of CGM values in CEG zones A and B with calibration. To our knowledge, our calibration protocol was able to achieve the composite lowest MARD and highest safety profile using the G6 in a critical care setting. Other studies have used frequent validation (e.g., every 2 h) or a hybrid protocol, which allows providers to decrease the frequency of confirmatory POC testing if accuracy criteria were met. 28,31,36,37 We believe that our calibration protocol can be a proven standardized and simple solution to using G6 with sensor validation for inpatient use.
Our data met the %15/15 FDA criteria for G6 measurements <3.9 mmol/L (<70 mg/dL) and for measurements from 3.9 mmol/L to 10.0 mmol/L (70 mg/dL to 180 mg/dL). The %15/15 criteria for values >10 mmol/L (180 mg/dL) and %40/40 criteria were not met (Table 2). These FDA criteria are not specified for validation of CGMs in the inpatient or critical care setting. In addition, these criteria are intended for validation of CGMs against a laboratory-based glucose measurement method, whereas our patients continued the standard of care with a glucose meter used in clinical practice. Therefore, although our results did not fulfill these FDA criteria, we believe our low calibrated MARD still demonstrates excellent accuracy as it is comparable with outpatient MARD and may be a more clinically interpretable measure of accuracy.
We found no identifiable clinical reason for sensor inaccuracy among the outliers. There were no outliers in our calibrated group, which suggests that the calibration protocol may be effective for early identification and correction of discrepancies. Further studies are needed to determine if outliers are observed with calibrated sensors and whether there are clinical factors contributing to the inaccuracy of the outliers. We did not find a statistically significant difference in accuracy for participants on vasopressors or RRT. These results are comparable with a retrospective analysis by Faulds et al. 32 using G6 in 18 COVID-19 participants, where there was no difference in MARD for participants on dialysis or vasopressors. Studies using other sensor models have also demonstrated no difference in accuracy with vasopressor or dialysis use, or demonstrated good accuracy in such heterogenous ICU populations. 34,37,38 This suggests that despite the hypotheses that the effects of vasopressors and RRT on microcirculation and interstitial fluid dynamics may decrease the accuracy of CGMs, they can be used in critically ill populations with retained accuracy. Of note, there were more participants on vasopressors in the calibrated group compared with the uncalibrated group. However, the significance of this difference on sensor accuracy is unclear as there was no statistically significant difference in accuracy between patients with and without vasopressor use. Our study showed retained accuracy across hemoglobin ranges. In comparison, a study by Davis et al. in adult participants in a general inpatient ward using G6 showed decreased accuracy with hemoglobin values <70 mmol/L. 19 This may be due to the small number of matched pairs with Hb <70mmol/L in our cohort.
Overall, with our findings in a heterogenous mixed medical–surgical ICU population, we suggest the following accuracy standards for G6 in critical care: (1) MARD ≤10% as the accuracy standard and (2) ≥99% of values in the CEG zones A and B. We also suggest sensor validation to be done 2, 12, and 24 h post-insertion to meet these accuracy standards. Given that CGM accuracy is higher in the general inpatient setting, these standards may be extrapolated for general inpatient use.
The strengths of our study are the heterogeneity of our study population with mixed medical and surgical diagnoses, vasopressor, and RRT use. We are the first to demonstrate the effectiveness of a pre-designed calibration protocol on CGM accuracy in an inpatient setting. Our simple and effective calibration protocol was able to achieve the lowest MARD and highest safety profile using the G6 in inpatient settings in comparison with other protocols using more frequent sensor validation. As such, it provides a practical solution to a major barrier to CGM adoption in inpatients.
There are some limitations of our study. Our calibration protocol may not be applicable to other CGM models or different inpatient settings. However, we do not consider this a major limitation to our results as our study is the first to demonstrate that by using a simple calibration protocol, a CGM can achieve excellent accuracy in an inpatient setting comparable with that of the outpatient setting. Other studies have shown decreased accuracy for patients with extracorporeal membrane oxygenation, induced hypothermia, and disorders related to hypoglycemia. 26 We did not have sufficient sample sizes in these patient populations to determine accuracy in these settings. Also, there is a MARD inherent to our comparator Accu-Chek II meter, as with any glucose monitoring device. Lastly, there were few women in our study, although the clinical relevance of biological sex on CGM accuracy is unclear.
Our study highlights the need for further research in inpatient CGM use. This is a single-center study of 28 participants. Future studies with larger sample sizes across multiple centers would be helpful to further test our protocol. In addition, our data collection period was seven days, and studies are needed to determine sensor accuracy toward the end of sensor life. We also observed outliers for sensor accuracy. Further studies are needed to determine if there is an acceptable threshold of discrepancy at the time of sensor validation, as this can potentially identify outliers that require sensor replacement or removal.
Conclusions
The Dexcom G6 CGM demonstrated excellent accuracy in a heterogenous critically ill adult population. Our calibration protocol using sensor validation on the first day post-insertion achieved the highest accuracy and safety profile of any study using the same device in inpatients.
Footnotes
Acknowledgments
The authors would like to acknowledge Barbara Allan for her help in ethics submission, participant enrollment, and data collection. Dexcom Inc. supplied the sensor equipment at no cost.
Authors’ Contributions
S.A.B. wrote the first draft of the article. J.C.H., S.A.B., and J.P.L. performed data collection and analysis. D.J.T., J.C.H., and S.A.B. contributed to and edited the article. S.A.B., P.A., and A.A. designed the study, enrolled participants, and collected data. M.S. assisted with the study design and data collection. All authors approved the final version of the article. D.J.T. is the guarantor of the study.
Author Disclosure Statement
The authors have no conflicts of interests to disclose.
Ethics approval
This study received ethical approval from the University of British Columbia Clinical Research Ethics Board (REB number H19-02392).
Funding Information
Dexcom Inc. (F20-01590) partially provided funding and supplied the sensor equipment at no cost but had no involvement in the data analysis or article preparation.
