Abstract
Aims:
We evaluated attainment of the hemoglobin A1c (HbA1c) target of ≤7.0%, its temporal trends, and associated factors among adults with type 1 diabetes in Ontario, Canada, using administrative data.
Methods:
We conducted a retrospective cohort study, including Ontarians with type 1 diabetes ≥18 years old with ≥1 HbA1c test between April 1, 2012 (fiscal year 2013), and March 31, 2023. Generalized estimating equations were used to determine probabilities of meeting the HbA1c target, as well as associations between fiscal year and individual-, physician-, and system-level factors on odds of meeting the target.
Results:
Among 28,827 adults with type 1 diabetes [14,385 (49.9%) female, 17,998 (62.4%) pump users], with median age at index of 25 years [interquartile range (IQR) 18–37] and median diabetes duration of 12 years [6–18], there were 474,714 HbA1c tests [median 2/individual/year (IQR: 1–3)]. The model-estimated probability of meeting the HbA1c target of ≤7.0% was 22.1% (95% confidence interval, CI: 21.6 to 22.5) in 2013, remained stable until 2020, and increased to 34.7% (95% CI: 34.3 to 35.2) in 2023. The age- and sex-adjusted odds ratio for meeting the target in 2023 versus 2013 was 1.87 (95% CI: 1.79 to 1.96). Young adults (18–25 years), diabetic ketoacidosis, greater comorbidity, and receiving diabetes care from a nonspecialist physician were associated with reduced odds of meeting the HbA1c target.
Conclusions:
One-third of adults with type 1 diabetes in Ontario met the recommended HbA1c target of ≤7.0% in 2023, with improvement noted since 2021, which may be due to advanced technologies or effects of the COVID-19 pandemic.
Introduction
Over 30
Despite 30 years passing since the publication of the DCCT, attainment of recommended glycemic targets appears to be consistently low in all regions or countries. 6,7 Hypoglycemia, lack of truly physiologic insulin, burden associated with intense management, and social determinants of health such as socioeconomic status contribute to difficulties in meeting recommended HbA1c targets. 1,6,8 –10 However, these findings come from studies that predominantly use clinic- or registry-based data sources, which are at risk of selection bias and may systematically affect results.
Few studies have evaluated temporal trends in attainment of HbA1c targets, especially among adults with type 1 diabetes, to determine if recent advances in therapeutics and technologies have had meaningful impact. 7,9 Furthermore, there has been no comprehensive assessment of patient-, physician-, and health care system-level factors associated with the attainment of glycemic targets. Evaluation of these factors may identify those that are potentially modifiable and could be targeted in future quality-of-care improvement strategies.
Using a large administrative data-based cohort of adults with type 1 diabetes, our objectives were to estimate the annual probability of meeting the HbA1c target of ≤7.0% among adults with type 1 diabetes and assess whether this probability has changed over time. In addition, we aimed to identify patient-, provider-, and system-level predictors associated with attainment of the recommended HbA1c target.
Materials and Methods
The use of data in this project was authorized under section 45 of Ontario's Personal Health Information Protection Act, which does not require review by a Research Ethics Board
Study design and setting
We conducted a retrospective cohort study in Ontario, Canada, using administrative health data available at ICES (formerly the Institute for Clinical Evaluative Sciences) for the time period from April 1, 2012 (fiscal year 2013), to March 31, 2023. The study was designed and reported according to the “Strengthening the Reporting of Observational Studies in Epidemiology” (STROBE) and REporting of studies Conducted using Observational Routinely-collected health Data (RECORD) guidelines. 11,12
Data sources
Individuals were linked across multiple health administrative databases using a unique encrypted version of the Ontario health card number. Databases used in this study were Ontario Health Insurance Plan (OHIP) for physician service codes, Ontario Laboratory Information System (OLIS) for laboratory test results, Registered Persons Database for demographics, Discharge Abstract Database and National Ambulatory Care Reporting System for hospitalizations and emergency department visits, the Canadian Census for derivation of neighborhood income quintile, Canadian Classification of Health Interventions for hospital procedural codes, ICES Physician Database for physician characteristics, and the Assistive Devices Program (ADP) for insulin pump application records (Supplementary Table S1).
Participants
Criteria for inclusion were residence in Ontario and age ≥18 years at the time of cohort entry between April 1, 2012 (fiscal year 2013), and March 31, 2022, meeting criteria for the type 1 diabetes algorithm, and having at least one HbA1c value between cohort entry and March 31, 2023 (end of follow-up). The type 1 diabetes algorithm has been previously validated and consists of meeting criteria for pediatric diabetes (four physician service codes for diabetes within 1 year before age 19) or having applied to ADP for an insulin pump, 13 which is a public funding program for insulin pumps in Ontario, exclusively for individuals with type 1 diabetes (thus identified pump users would not include individuals with type 2 diabetes using an insulin pump).
The algorithm has a positive predictive value of 79% and a negative predictive value of 98%. The cohort entry date was April 1, 2012, if all inclusion criteria were met by that date, or on the earliest subsequent date when all inclusion criteria were met. Exclusion criteria were invalid birth date or sex, or incomplete eligibility for OHIP in the 5 years before cohort entry.
Exposures and covariates
The primary exposure of interest was fiscal year (defined as April 1 to March 31) for the time period of April 1, 2012 (fiscal year 2013), to March 31, 2023. Baseline covariates were defined using 5 years of data before the cohort entry date, unless otherwise indicated, and classified as factors pertaining to the individual (individual level), the physician providing diabetes care (physician level), or the health care system (system level).
Baseline individual-level characteristics consisted of sex, diabetes duration, estimated glomerular filtration rate, prior treatment for proliferative retinopathy, and level of comorbidity using a previously validated weighted sum of Aggregated Diagnosis Groups (ADGs) score. 14,15 The Johns Hopkins ACG system (v10.0) was used to derive ADGs based on 2 years of data before cohort entry. Time-varying individual-level characteristics, which were updated on the date of each HbA1c test, included age category, neighborhood-based income quintile, insulin pump use, hospitalization or emergency department visit for diabetic ketoacidosis or hypoglycemia in the prior year, number of diabetes appointments with the diabetes physician per year, and transfer to a new diabetes physician.
Use of continuous glucose monitors (CGM) is not well captured in administrative health data since there is limited public funding for these devices; thus, the use of CGM and corresponding metrics such as time in range were not available. Proliferative retinopathy was defined based on procedural codes for treatment of retinopathy (laser or injections). Neighborhood income quintile was assigned to individuals based on their postal code of residence using a postal code conversion file (PCCF) created by Statistics Canada. 16
Physician-level covariates included physician specialty, sex, age, and years in practice, with all being updated at the start of each fiscal year. To identify physicians providing diabetes care, all outpatient physician service codes during the study period with a diagnostic code for diabetes (ICD-9 250) were identified. We used a hierarchical approach to assign one diabetes physician to each patient per fiscal year with the order of assignment being endocrinologist, general internist, pediatrician, family physician, or no physician (if there was no diabetes physician services code). With this method, if any code was billed by an endocrinologist within the year, then the endocrinologist was identified as the diabetes physician. If more than one physician within the same specialty was identified, then the physician with the latest date of billing within the year was used.
System-level characteristics included distance of the patient to the diabetes physician's practice, rural or urban location of the patient, and association of the patient's diabetes physician with a registered ADP center. All system-level covariates were updated at the start of each fiscal year. Distance to physician was calculated using the patient's postal code and the postal code of the physician's office location and categorized by tertiles (<6 km, 6–20 km, and >20 km). In Ontario, public funding is provided for insulin pumps for individuals with type 1 diabetes through the ADP program. To apply, patients must attend a Diabetes Education Program that is registered with ADP. However, not all patients followed at an ADP-registered Diabetes Education Program necessarily use insulin pumps.
Outcomes
The primary outcome was HbA1c, categorized as meeting target (≤7.0%) or not meeting target (>7.0%). All HbA1c values between cohort entry and end of follow-up (March 31, 2023) were included. HbA1c values were obtained from the Ontario Laboratory Information System (OLIS), which contains laboratory tests and results for routine clinical care and includes >99% of all HbA1c test results.
Statistical methods
For the primary analyses, we used generalized estimating equations (GEE) to estimate the association between fiscal year and HbA1c being ≤7.0%, while accounting for the within-individual correlation of repeated HbA1c results over time. Exchangeable covariance matrices were used for GEE models. Results are reported as odds ratios (ORs) and 95% confidence intervals (CIs). Models were performed unadjusted, adjusted for age and sex, and adjusted sequentially for individual-level, provider-level, and system-level predictors.
Variables included in multivariable models were chosen a priori based on clinical relevance. Age was categorized (as 18–24, 25–34, 35- and ≥50) based on preliminary analyses, which found a linear term for age to be inadequate. Results from GEE models were used to compute the marginal predicted probability for HbA1c being ≤7.0% for each fiscal year. In secondary analyses, the same sequences of GEE models were fitted, but with HbA1c as a continuous outcome, and marginal mean HbA1c in each fiscal year was computed. Missing data were uncommon, other than for system-level characteristics. For multivariable models that included system-level predictors, we performed complete case analysis. SAS Version 9.4 (Cary, North Carolina) was used for all analyses. A P-value <0.05 was used to assign statistical significance threshold for all analyses.
Sensitivity analyses
To address misclassification of type 1 diabetes by the algorithm (in particular, inclusion in the cohort of individuals without type 1 diabetes), fully adjusted models were repeated in the subset who met cohort entry criteria based on application to ADP for an insulin pump, since this criterion has 100% specificity for type 1 diabetes.
Results
Cohort description
There were 28,827 adults with type 1 diabetes in Ontario, who met the cohort inclusion (Supplementary Fig. S1). Of these, 10,829 (37.6%) were identified as having type 1 diabetes based on the pediatric diabetes criterion only, and 17,998 (62.4%) were identified as having type 1 diabetes based on the insulin pump criterion (with or without, also meeting the pediatric diabetes criterion). Characteristics of the cohort are presented in Table 1. Median age at index was 25 [interquartile range (IQR): 18–37], median duration of diabetes was 12 years (IQR: 6–18), and 14,385 (49.9%) were female.
Baseline Characteristics of Adults with Type 1 Diabetes in Ontario Between Fiscal Year 2013–2023 by Reason for Inclusion (N = 28,827)
Missing data: 149 for income quintile, 35 for rural versus urban, 10,106 for eGFR, and 3788 for distance to diabetes care provider.
ADG, Aggregated Diagnosis Group; ADP, Assistive Devices Program; CAD, coronary artery disease; CHF, congestive heart failure; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; DKA, diabetic ketoacidosis; ED, emergency department; eGFR, estimated glomerular filtration rate; ESRD, end-stage renal disease.
Most [23,776 (82.5%)] individuals were assigned a diabetes physician in the first year following cohort entry, with the majority of these [14,733 (62.0%)] assigned to an endocrinologist. Among individuals assigned a diabetes physician, 19,208 (80.8%) physicians were affiliated with a registered ADP center. In total, there were 474,714 HbA1c test results, with individuals having a median of 2 (IQR: 1–3) HbA1c tests per year. Median follow-up time was 11.7 years (IQR: 8.2–11.7).
HbA1c target of ≤7.0%
The marginal model-predicted probability for HbA1c being ≤7.0% was 22.1% (95% CI: 21.6 to 22.5) in 2013 and 34.7% (95% CI: 34.3 to 35.2) in 2023. Probabilities for HbA1c being ≤7.0% over time by age category are shown in Figure 1A. While the probability for HbA1c being ≤7.0% increased minimally between 2013 and 2020 (increase in probability of meeting HbA1c target of 0.2%/year), probabilities increased for 2020 through 2023 among all age groups, with no plateau observed (increase in probability of meeting HbA1c target of 3.8%/year). Unadjusted and age- and sex-adjusted odds of meeting the HbA1c target for each fiscal year relative to 2013 are shown in Figure 2. Adjusting for age and sex, the odds of meeting the HbA1c target in 2023 were 87% higher than in 2013 (OR: 1.87, 95% CI: 1.79 to 1.96).
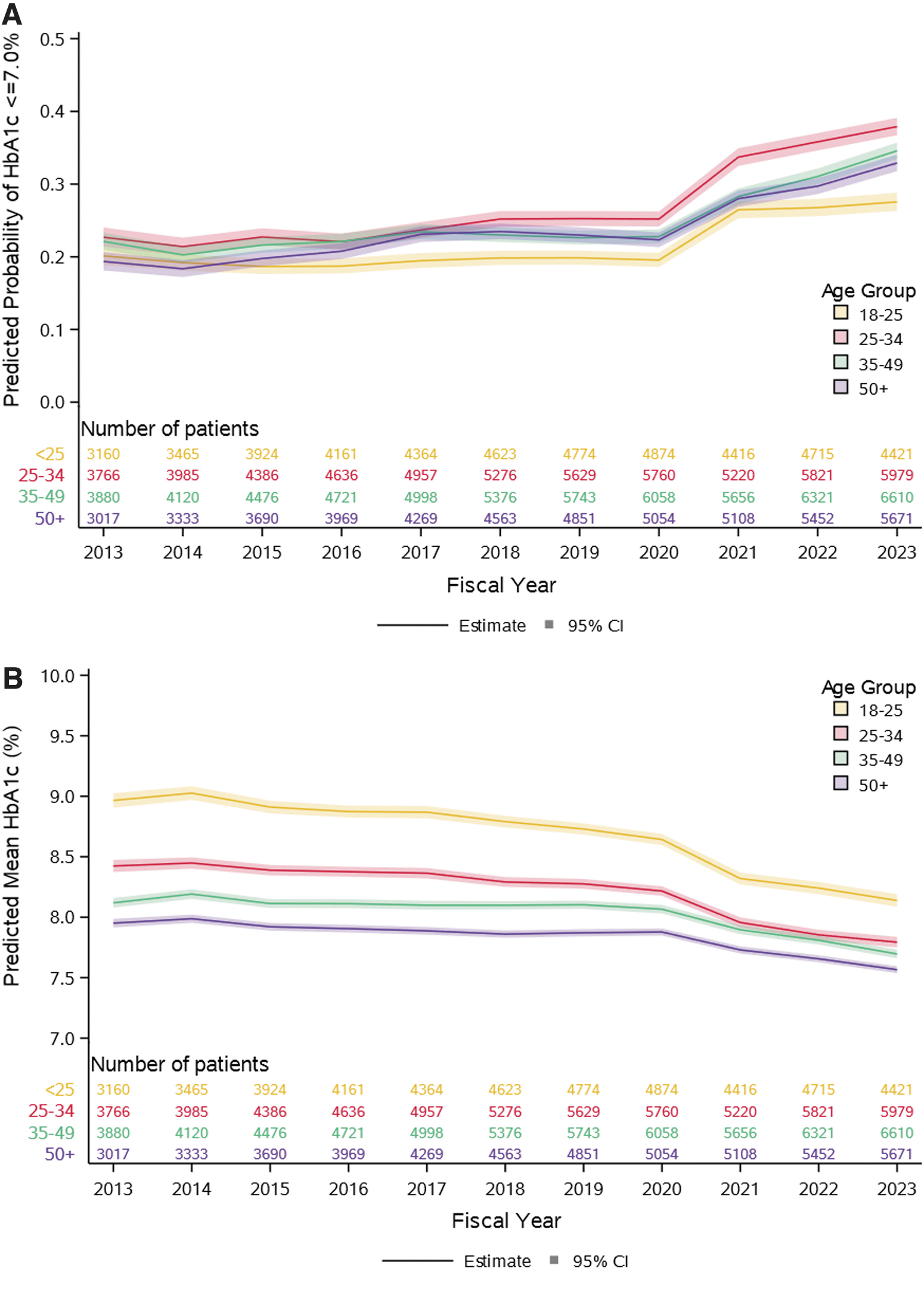
Predicted probability of HbA1c ≤7.0%
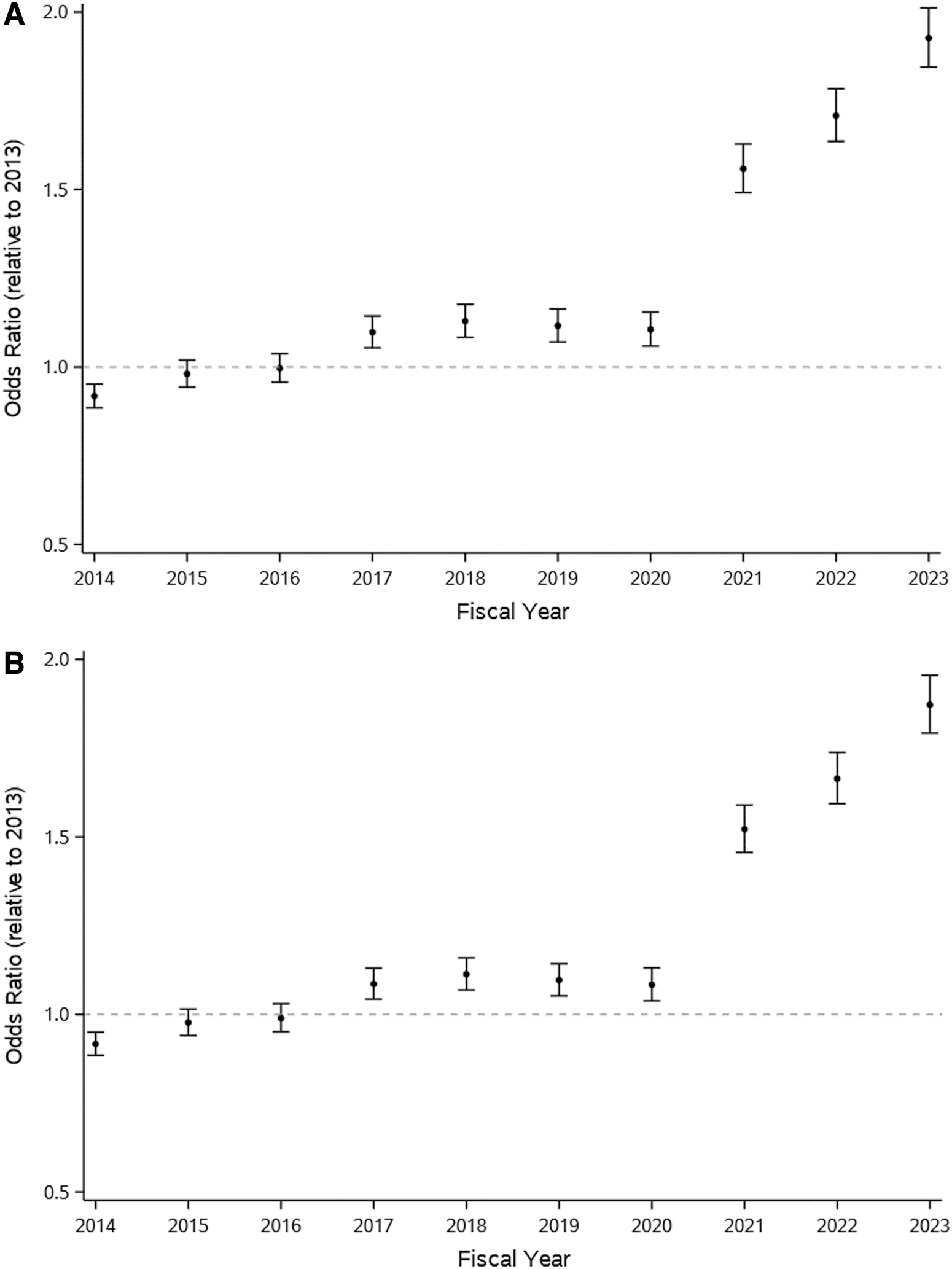
Forest plots demonstrating
GEE models sequentially adjusting for individual-level, physician-level, and system-level predictors are shown in Table 2. For the model with individual-level characteristics, age 18–24 years was associated with lower odds of meeting the HbA1c target relative to the reference age group 35–49 years [OR: 0.87 (95% CI: 0.82 to 0.92)]. Hospitalization or emergency department visit for diabetic ketoacidosis (DKA) in the prior year was associated with lower odds of meeting the target [OR: 0.69 (95% CI: 0.66 to 0.72)], while hospitalization of emergency department visit for hypoglycemia was associated with higher odds of meeting the target [OR: 1.14 (95% CI: 1.07 to 1.20)].
Generalized Estimating Equation Results for Estimating Odds Ratio of Hemoglobin A1c ≤7% Over Time Compared to Fiscal Year 2013
Sample sizes for models: Individual-level predictors n = 28,800; individual- and physician-level predictors n = 25,839; individual-, physician-, and system-level predictors (n = 21,706).
Time-varying covariate (patient-level covariates updated at the time of HbA1c test and provider-level covariates updated at the beginning of each fiscal year).
CI, confidence interval; HbA1c, hemoglobin A1c; OR, odds ratio; SD, standard deviation.
A higher weighted-ADG score, indicating greater comorbidity, was associated with reduced odds of meeting the HbA1c target [OR: 0.87 (95% CI: 0.85 to 0.89) per standard deviation increase]. Longer diabetes duration was also associated with reduced odds of meeting the target [(OR: 0.96 (95% CI: 0.95 to 0.98) per 5-year increase]. The number of outpatient diabetes visits per year was not associated with attainment of HbA1c ≤7.0% (Table 2).
In the model adding physician-level characteristics, only physician specialty was associated with the outcome; compared to individuals with an endocrinologist as their diabetes provider, the odds of meeting the HbA1c target were lower in those whose provider was a family physician [OR: 0.90 (95% CI: 0.86 to 0.94)] and higher in those whose provider was a pediatrician or other specialist [OR: 1.14 (95% CI: 1.00 to 1.31) and OR: 1.23 (95% CI: 1.14 to 1.32), respectively]. Physician age, sex, and years in practice were not associated with the outcome.
In the fully adjusted model, rural residence and having a diabetes physician who is affiliated with an ADP clinic were associated with reduced odds of meeting the HbA1c target [OR: 0.90 (95% CI: 0.84 to 0.97) and 0.88 (0.80 to 0.98), respectively]. Distance from the patient to the physician's office was not associated with attainment of the HbA1c target.
Mean HbA1c
Predicted mean HbA1c was 8.36% (95% CI: 8.33 to 8.38) in 2013 and 7.87% (95% CI: 7.85 to 7.89) in 2023. HbA1c was higher with each successively younger age category. Adjusting for age and sex, HbA1c was 0.41% (95% CI: −0.43 to −0.38) lower in 2023 compared to 2013 (Fig. 1B). Predicted Mean HbA1c declined within all age categories, with the greatest decline observed for young adults 18–25 years of age [−0.83% (95% CI: −0.91 to −0.75) between 2013 and 2023].
Estimates from GEE models with HbA1c as a continuous outcome are shown in Supplementary Table S2. In the model adjusting for individual-level predictors, age 18–24 years was associated with an HbA1c 0.34% higher (95% CI: 0.30 to 0.38) than age 35–49 years, insulin pump use was associated with an HbA1c 0.41% lower (95% CI: −0.44 to −0.39), and a hospitalization or emergency department visit for DKA in the prior year was associated with an HbA1c 0.63% higher (95% CI: 0.59 to 0.69). In the model adding physician-level characteristics, mean HbA1c was higher when a family physician [0.20% (95% CI: 0.17 to 0.23)] or pediatrician [0.42% (95% CI: 0.32 to 0.51)] was the assigned diabetes provider in comparison to an endocrinologist. In the fully adjusted model, none of the system-level characteristics had clinically meaningful associations with HbA1c.
Sensitivity analyses
Fully adjusted models were repeated in the subsample of insulin pump users (100% specificity for having type 1 diabetes), and results were consistent with the primary analyses (Supplementary Table S3). To permit comparison with reports of glycemic outcomes from other regions, unadjusted GEE models for attainment of HbA1c <7.0%, <7.5%, and >9.0% were performed for age categories 18–24, 25–39, and ≥40 years of age (Supplementary Table S4). Additional plots demonstrating HbA1c distribution for 2013 and 2023 by age category and diabetes duration are shown in Supplementary Figures S2 to S4.
Discussion
In a retrospective cohort study of 28,827 adults with type 1 diabetes in Ontario, one-third met the recommended HbA1c target of ≤7.0% in 2023. While attainment of HbA1c ≤7.0% was relatively stable between 2013 and 2020, an increase has occurred since 2021, with no plateau yet observed. Young adults (18 to 25 years of age) were least likely to have an HbA1c ≤7.0% yet, encouragingly, had the greatest reduction in HbA1c over the observed time period. Patients receiving diabetes care from a family physician, living in rural areas, and followed at clinics registered with ADP were less likely to meet the recommended HbA1c target.
Attainment of the HbA1c target was similar in Ontario to estimates in other regions, although the most recent attainment in 2023 is higher than reported in other regions. 2,4,6,17 –19 In the T1D Exchange Registry in the United States, 26% had an HbA1c ≤7.0% in 2021/2022. 20 Higher attainment in Ontario may be due to the universal health care system with universal access to insulin pumps. However, despite differences in health care systems, it is remarkably consistent that globally, fewer than one-half of individuals with type 1 meet the HbA1c target of ≤7.0%, which is recommended by most international societies. 2,4,6,17 –19 In an international comparison of glycemic control for over 500,000 individuals with type 1 diabetes, nearly all regions or countries had fewer than one-quarter to one-third of adults attaining an even higher HbA1c threshold of <7.5%. 7
Temporal trends in HbA1c have been more comprehensively evaluated in children and adolescents than adults, where improvements in HbA1c over time have been noted internationally, 21 –23 with the United States being potentially an exception. 20,24 Among individuals with type 1 diabetes of all ages, including adults, an international comparison of 500,000 individuals from 17 countries or regions using population- or clinic-based data sources demonstrated an improvement in the proportion with an HbA1c <7.5% in 2020 compared to 2013, although even in 2020, HbA1c <7.5% was attained by fewer than 50% of individuals. 7
In Scotland, median HbA1c improved from 8.7 to 8.4% between 2004 and 2016 for individuals with type 1 diabetes of all ages, but the proportion with an HbA1c at target (<7.0% for adults and <7.5% for children and adolescents) increased from 11% to only 15% during the same time frame. 25 Among nearly 100,000 individuals with type 1 diabetes in Germany and Austria, the proportion with an HbA1c >9.0% declined from 28% in 1995 to 17% in 2017, although the proportion with an HbA1c ≤7.0% was not reported. 26 Our findings that young adults (ages 18–25) had the highest HbA1c are consistent with other studies such as the T1D Exchange in the United States. 20 In Ontario, transition from pediatric to adult care occurs at the age of 18 and is associated with insufficient care (e.g., loss to follow-up) and a higher risk of adverse outcomes. 27
Our study is the first to demonstrate recent improvement in HbA1c target attainment. There are a number of potential explanations for improvement in glycemic outcomes beginning in 2021. First, the COVID-19 pandemic may have led to improved self-management behaviors. 28,29 Second, CGM and hybrid-closed loop systems became increasingly available in Ontario during this time (two commercially available hybrid-closed loop systems are available: Medtronic 670G became available late 2018 with 780G currently available, and Control IQ became available April 2021). Although the use of CGM or AID was not available in our cohort, recent estimates in studies conducted in Ontario, as well as a diabetes registry in Quebec suggest 20%–40% of individuals with type 1 diabetes use insulin pumps and 50%–80% use CGM. 30 –32 Rates of AID use in Ontario have not been reported to our knowledge. Third, access to CGM expanded, with the introduction of public funding programs for flash glucose monitoring in September 2019 and CGM in March 2022, although these programs have restrictions based on age and clinical criteria. It is not clear whether the improvements are entirely driven by increased adoption of diabetes technologies, as improvements in glycemic outcomes were similar in the analyses restricted to pump users as for the full sample, which includes those not using pumps or hybrid-closed loop systems. To robustly evaluate determinants of temporal trends in glycemic outcomes, systematic collection of CGM and AID data should be prioritized by health systems.
In our study, most physician- and system-level factors did not appear to have substantial associations with glycemic control. This is consistent with findings among adults with type 1 and type 2 diabetes in the Australian National Diabetes Audit, in which patient-level factors contributed more to variation in HbA1c than center-level factors. 33 The only physician-level factor associated with attainment of glycemic targets in our study was physician specialty, with specialist management being associated with greater likelihood of meeting the HbA1c target. This finding is consistent with previous studies and reinforces the importance of specialty-based care for type 1 diabetes and ensuring access to specialists for all individuals living with type 1 diabetes. 34
We are not aware of studies evaluating whether characteristics among endocrinologists are associated with glycemic outcomes and were not able to assess this in our study due to limitations of administrative data. It is reassuring we observed no difference in glycemic control by socioeconomic status, which may reflect the universal health care system and universal insulin pump program in Ontario.
Having a hospitalization or emergency department visit for hypoglycemia (i.e., severe hypoglycemia) was associated with a greater likelihood of having an HbA1c ≤7.0%. However, HbA1c targets should be individualized based on history of hypoglycemia, and among individuals with a history of severe hypoglycemia, an HbA1 target of ≤7.0% may not be appropriate. 2,3 With increasing adoption of AID systems, which reduce hypoglycemia, future consideration should be given to whether HbA1c targets should also differ according to use of nonuse of CGM and AID. 35,36
Strengths of our study include the use of administrative data, which comprehensively capture health care services and temporal HbA1c values in a sample not affected by volunteer bias. As far as we know, this is the first study to assess for temporal changes in glycemic control and associated characteristics in a large representative adult population.
There are some weaknesses to our study. First, the algorithm used to identify type 1 diabetes has a positive predictive value of 79% and negative predictive value of 98%, which could mean ∼ 20% of the cohort in our primary analysis do not have type 1 diabetes. The pediatric diabetes criterion also biases toward a younger cohort. To mitigate this, we repeated the analyses in pump users who have 100% certainty of having type 1 diabetes, and the results were unchanged. In addition, the algorithm is biased toward detecting younger individuals with type 1 diabetes. Since young adults typically have the highest HbA1c values, it is possible that HbA1c target attainment is underestimated in our sample, although the probability was still <40% within the oldest age category.
Second, we were unable to capture some important characteristics such as the use of CGM or hybrid-closed loop systems, race, or ethnicity (which is not currently available in administrative data in Ontario), individual measures of socioeconomic status, and health-care interactions with providers other than physicians (such as diabetes nurses and dietitians), which could affect HbA1c results. Since CGM was not available, we were not able to report on time in range. Third, we restricted cohort entry to April 1, 2012, or later as this is when HbA1c results were comprehensively captured in administrative data. We are therefore unable to assess earlier time periods. Finally, we were not able to analyze individualized targets for HbA1c, although with a younger cohort the majority would be expected to have a target of ≤7.0%.
Using administrative health data in Ontario, Canada, we demonstrated that only one-third of adults with type 1 diabetes met the HbA1c target of ≤7.0% in 2023, although improvement was noted since 2021, which could potentially be due to effects of the pandemic or increasing utilization of diabetes technologies. Ongoing surveillance is required to determine if recent improvements in HbA1c target attainment are sustained.
Footnotes
Acknowledgments
We thank ServiceOntario for use of Office of the Registrar General (ORG) information on deaths and IQVIA Solutions Canada, Inc., for use of their Drug Information File. The views expressed herein do not necessarily reflect those of ORG or the Ministry of Government Services. A.W. is the guarantor and as such takes full responsibility for the work, including the study design, access to data, and the decision to submit and publish the article.
Authors' Contributions
A.W. conceptualized the study, conducted the literature search, acquired funding, planned the data analysis, interpreted the data, and wrote the first draft of the article. G.A.T. conceptualized the study, planned the data analysis, interpreted the data, and provided critical revision of the article. K.E. performed data analysis, interpreted the data, and provided critical revision of the article. G.L.B. planned the data analysis, interpreted the data, and provided critical revision of the article.
Data Availability
The dataset from this study is held securely in coded form at ICES. While data sharing agreements prohibit ICES from making the dataset publicly available, access may be granted to those who meet prespecified criteria for confidential access, available at ![]() . The full dataset creation plan and underlying analytic code are available from the authors upon request, understanding that the computer programs may rely upon coding templates or macros that are unique to ICES and are therefore either inaccessible or may require modification.
. The full dataset creation plan and underlying analytic code are available from the authors upon request, understanding that the computer programs may rely upon coding templates or macros that are unique to ICES and are therefore either inaccessible or may require modification.
Statement of Ethics
The use of data in this project was authorized under section 45 of Ontario's Personal Health Information Protection Act, which does not require review by a Research Ethics Board.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
Alanna Weisman is supported by a Sunlife Financial New Investigator Award from the Banting & Best Diabetes Centre. This study was supported by ICES, which is funded by an annual grant from the Ontario Ministry of Health (MOH) and the Ministry of Long-Term Care (MLTC). This document used data adapted from the Statistics Canada Postal CodeOM Conversion File, which is based on data licensed from Canada Post Corporation, and/or data adapted from the Ontario Ministry of Health Postal Code Conversion File, which contains data copied under license from © Canada Post Corporation and Statistics Canada. Parts of this material are based on data and/or information compiled and provided by CIHI and the Ontario Ministry of Health. The analyses, conclusions, opinions and statements expressed in the material are those of the author(s), and not necessarily those of the funding or data sources; no endorsement is intended or should be inferred. We thank IQVIA Solutions Canada Inc. for use of their Drug Information File. Parts of this report are based on Ontario which is ServiceOntario. The views expressed therein are those of the author and do not necessarily reflect those of ORG or the Ministry of Public and Business Service Delivery.
Supplementary Material
Supplementary Figure S1
Supplementary Figure S2
Supplementary Figure S3
Supplementary Figure S4
Supplementary Table S1
Supplementary Table S2
Supplementary Table S3
Supplementary Table S4
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
