Abstract
Objective:
Since the introduction of continuous glucose monitoring (CGM) technology, developers have rigorously researched the feasibility of creating a noninvasive glucose monitoring device. In a recent pilot study, investigators reported a strong correlation between glucose values obtained from novel noninvasive monitoring device (GWave) values to venous and capillary glucose measurements.
Research Design and Methods:
We investigated whether the level of accuracy observed in the pilot study could be reproduced in a larger cohort, using a smaller third-generation manufacturable device (Gen III GWave) containing a standardized sensor chip that can be mass produced for commercial use. The evaluation assessed concordance with capillary blood glucose, reproducibility between two Gen III devices, and accuracy during insulin-induced hypoglycemia.
Results:
Assessment of samples from 75 subjects (type 2 diabetes, n = 6; type 1 diabetes, n = 28; nondiabetic pregnant subjects, n = 10; and nondiabetic, n = 31) showed that 97% of values were in Zone A with 3% in Zone B of the Clarke Error Grid, with a mean absolute relative difference of 6.7% from reference blood glucose. Comparison between two independent Gen III GWave devices demonstrated reproducibility between the sensors (R 2 = 0.95), with 100% of values within Zone A. In the hypoglycemia assessment, measurements from the Gen III sensor tightly followed the capillary glucose measurements down to 42 mg/dL (2.3 mmol/L), whereas the CGM measurements from two different CGM only converged with the GWave and capillary glucose readings after 90 min of decreasing glucose levels.
Conclusion:
Our results show promise as potentially the first noninvasive technology. Future studies will focus on larger number of people in all glucose ranges. Real-time noninvasive blood glucose monitoring is possible using GWave technology.
Introduction
Frequent measurement of glucose levels is an essential component of effective diabetes management. Although the majority of individuals with diabetes use fingerstick blood glucose monitoring (BGM) for their daily self-management, adherence to prescribed monitoring regimens remains suboptimal. 1 –3 In addition to the pain and inconvenience of fingerstick testing, it is susceptible to user errors, 4 which can result in erroneous high or low readings.
The introduction of continuous glucose monitoring (CGM) addressed many of the barriers associated with fingerstick testing. Ongoing innovations in CGM technology have led to smaller, more accurate, and less invasive devices that are factory calibrated. Given these improvements, increasing number of people with diabetes are adopting CGM as their preferred method for glucose monitoring. 5
However, CGM devices are not without their own limitations. Because the CGM sensor measures glucose levels in subcutaneous interstitial fluid instead of blood, the sensor reaction time to rapidly changing glucose can be delayed up to 15–20 min. 6,7 This delay, referred to as “lag time”, may cause patients to doubt the accuracy of their CGM results and discontinue using their devices. 8,9 Moreover, current and past CGM users have reported concerns about body image, insertion pain, discomfort, and skin irritation. 8
Although previous attempts to develop noninvasive glucose monitoring systems have been unsuccessful, 10 Hagar (Haifa, Israel) has developed a novel noninvasive CGM device (GWave) that uses real-time high-frequency radiofrequency (RF) to accurately measure capillary/venous blood glucose levels covering the physiological range (40–400 mg/dL). The Hagar device does not require per patient calibration or “learning period” and does not rely on predictive algorithm but rather on real-time glucose monitoring.
In a small pilot study by Schwarz et al., investigators assessed the performance of a GWave prototype in five adults with type 2 diabetes (T2D) and HbA1c of 6.0%–8.0% (42–64 mmol/mol). 11 Investigators reported a strong correlation between the GWave values and venous glucose measurements (R 2 = 0.924 [95% confidence interval [CI] 0.929–0.970, P < 0.0001) and the capillary glucose measurements (R 2 = 0.975 [95% CI 0.975–0.994, P < 0.0001).
This study was conducted to determine whether this level of accuracy could be reproduced in a larger cohort, using a smaller second-generation device containing a standardized sensor chip that can be mass produced for commercial use.
Research Design and Methods
Study design and population
This analysis assessed the accuracy of the GWave device compared with capillary blood glucose values, using a commercial BGM system that has been previously shown to provide 96.9% of test results within the International Organization for Standardization (ISO) accuracy limits. 12 The primary objective was to demonstrate correlations between GWave glucose measurements and capillary glucose values and to evaluate the accuracy of the G7 system when used in adult participants with diabetes. The study was conducted at the Weizmann Institute of Science (Rehovot, Israel) and approved by an independent institutional review board. Participants included individuals with diabetes and without diabetes. There were no exclusion criteria. All participants provided written informed consent.
Devices
The noninvasive GWave device technology is based on the electrical reaction of biological tissue in the dermis to the broadcasted RF wave and the dielectric change in the tissue in correlation with the change of blood glucose levels. The study device utilized an on-chip radio frequency generator. The reaction is measured on a dielectric material when the electromagnetic field flux is generated in a unique range. The device is powered by standard commercially available batteries. The device is equipped with Bluetooth capability and can transfer the data to an application for further use.
The FreeStyle Lite BGM system (Abbott Diabetes Care, Alameda, CA) is used for the quantitative measurement of glucose in fresh capillary whole blood drawn from the fingertips or palm. In an early evaluation of the system, linearity assessments demonstrated exceptional accuracy with 98.8% of test results in Zone A and 1.2% of the results in Zone B of the Parke Error Grid. 12
Procedures
Participants reported to the clinic at 8:00 a.m. after an 8 h fast (no caloric intake, water permitted). The device was placed on the anterior portion of the distal wrist for all analyses.
Concordance and correlation
Comparisons between GWave and capillary glucose values were performed across two glucose ranges using the Gen II GWave (prototype) and Gen III GWave (manufacturable) devices:
(1) Gen II GWave Prototype Chip: Samples from 20 subjects were tested (T2D, n = 12; type 1 diabetes [T1D], n = 2, nondiabetic, n = 6). To obtain readings in the high end of the physiological range, up to 500 mg/dL (27.8 mmol/L), five participants drank sweetened apple juice and were subjected to another cycle of GWave and FreeStyle readings. Each measurement entailed capillary glucose blood determination by a single GWave measurement, and four different blood glucose values obtained with the FreeStyle blood glucose meter.
(2) Gen III GWave Manufacturable Chip: The manufacturable chip, which contains the same RF circuitry (sensor component of the device), was integrated into the Gen III GWave device. Using the same testing methodology used with the Gen II device, samples from 75 subjects (T2D, n = 6; T1D, n = 28; pregnant subjects, n = 10; and nondiabetic, n = 31) were analyzed across a glucose range of 0–300 mg/dL (0–16.7 mmol/L). Among the 31 nondiabetic subjects, 5 were younger than 15 years old and 5 were dark skinned. Each subject was tested once or twice by a GWave device and then by three FreeStyle meters. The average of three capillary measurements was the comparator for the average of GWave values.
Reproducibility
To demonstrate device-to-device reproducibility of the 75 test subjects, a subset of 20 test subjects (with T1D or T2D diabetes, n = 13; nondiabetic, n = 7) was measured by two independent GWave devices.
Accuracy during insulin-induced hypoglycemia
To test Gen II GWave ability to detect capillary blood glucose in real time and how it compares with the ability of different CGMs, we performed a supervised insulin-induced hypoglycemia (≤54 mg/dL). Owing to the risks associated with this procedure, it was limited to one individual. Fast acting insulin was administered, and the glucose levels of test subject were measured over 95 min. At each measuring point, capillary blood was recorded by a FreeStyle blood glucose. The Gen III GWave sensor and two different commercial CGM sensors (Dexcom G6 system [Dexcom, Inc., San Diego, CA]; FreeStyle Libre 3 system [Abbott Diabetes Care, Alameda, CA]) were attached to the test subject 2 days before the measurement.
Statistical analysis
A Bland Altman plot was used to plot the mean absolute relative difference (MARD) between measurements derived from the GWave device and BG meter values. Pearson correlation coefficients were computed to determine the degree of correlation of the GWave measurements with capillary glucose (BG meter) and independent GWave values using GraphPad Prism V.9. The correlations between the GWave and capillary glucose values and independent GWave values were overlayed on a Clarke Error Grid Analysis (EGA), which illustrates the clinical accuracy of glucose monitoring systems over the entire range of blood glucose values. 13
The EGA considers the absolute value of the device-generated glucose value, the absolute value of the reference blood glucose value, the relative difference between these two values, and the clinical significance of this difference. The EGA is divided into five zones that represent the clinical acceptability and risk associated with the accuracy findings. Values in the A and B zones indicate accuracy and acceptability of the results. Values in the C zone may provoke unnecessary corrections in therapy that could result in poor outcomes. Values in the D zone denote a hazardous failure to detect and treat problematic glycemia. Response to values in the E zone constitutes incorrect treatment.
Results
Concordance, correlations, and reproducibility
The correlations between the GWave measurements and capillary values were overlayed on a Clarke Error Grid for both the prototype and manufacturable chip analyses. The prototype analysis showed that 100% of values fell within Zone A (Fig. 1A), with near perfect correlation between the GWave and capillary glucose measures across the physiological glucose range (R 2 = 0.9911 [95% CI 0.9893–0.9981, P < 0.0001). The analysis of the manufacturable sensor chip showed that 97% of values were in Zone A with 3% in Zone B (Fig. 1B).
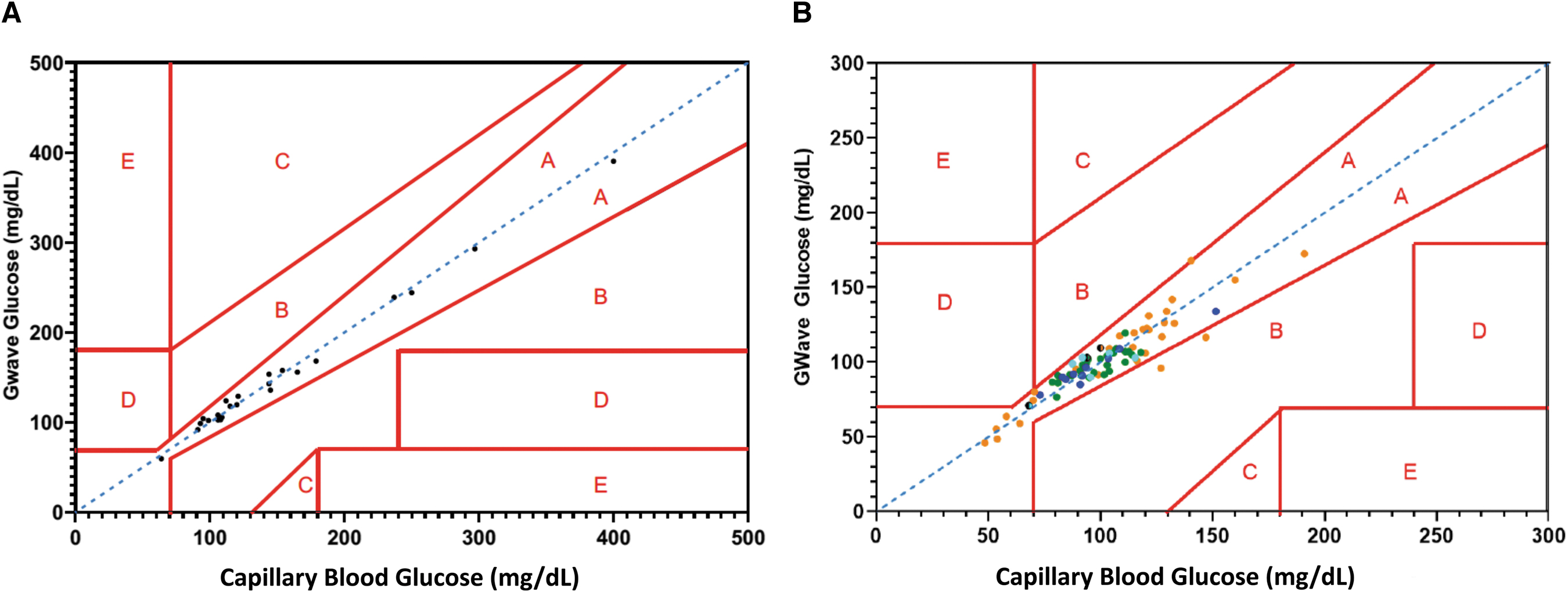
GWave Gen II capillary blood glucose correlations.
The Gen III GWave sensor performed impeccably and persisted in hypoglycemic range. A mean absolute relative difference (MARD) of 6.7% was calculated using the mean capillary blood measured by the FreeStyle meter glucose values as the comparator. Comparison between two independent Gen III GWave devices demonstrated reproducibility between the sensors (R 2 = 0.95), with 100% of values within Zone A.
Accuracy during insulin-induced hypoglycemia
As shown in Figure 2, the GWave Gen III measurements tightly followed the capillary glucose measurements down to 42 mg/dL (2.3 mmol/L), whereas the CGM measurements from the other devices only converged with the GWave and capillary glucose readings after 90 min of decreasing glucose levels.
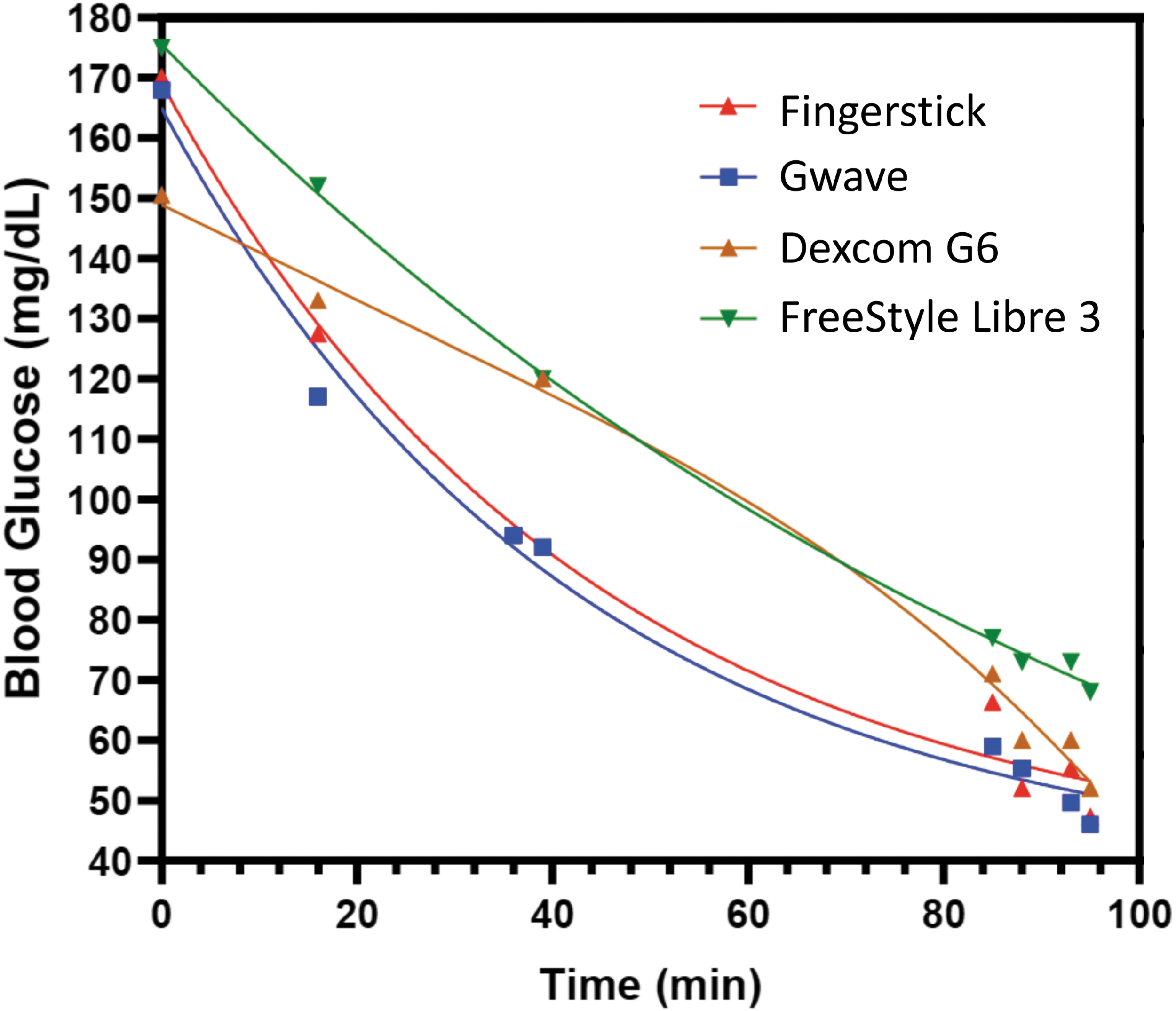
GWave ability to follow blood glucose in real time. Insulin was administered to induce blood glucose decrease from 170 mg/dL to ∼45 mg/dL over a period of ∼95 min. Blood glucose levels were measured by fingersticks (red triangle), GWave (blue square), or two types of CGMs (brown triangle) and (green triangle). Although GWave tightly followed the fingerstick pattern, the CGMs readout converged with the capillary blood values only after 90 min (n = 1).
Discussion
In this analysis, glucose measurements from the novel noninvasive GWave device were compared with a commercially available BGM system using MARD and correlations as outcomes. Our results showed that the GWave technology provides glucose measurements that are accurate (MARD 6.7%), strongly correlated with capillary glucose across the full physiological glucose range, and reproducible between devices. Our exploratory analysis also demonstrated near-perfect agreement between GWave values and capillary glucose measurements during insulin-induced hypoglycemia. Importantly, our findings closely aligned with the accuracy of the FreeStyle Lite monitoring system, which showed an R 2 > 0.99, with 98.8% of test results in Zone A, and 1.2% in Zone B of the Parke Error Grid. 12
Prior efforts to utilize RF technology for glucose measurement have been challenged by the inability to separate out the glucose from the other components in the dermis, which consists primarily of dense irregular connective tissues such as blood vessels, hair follicles, and sweat glands. The GWave device eliminates these interferences. Importantly, the GWave device emits less radiation than a smartphone and, unlike current BGM and CGM systems, the GWave device is durable in that it never needs to be replaced; the device is powered by commercially available rechargeable batteries. The commercial version will be integrated into a wristwatch. Additional information about the GWave technology is provided in Supplementary Figure S1. 14
This real-time glucose sensing technology can be incorporated into commercial CGM systems, combining the accuracy of fingerstick blood glucose devices with the demonstrated clinical benefits and convenience of CGM, utilizing noninvasive RF glucose sensing technology. The clinical utility and benefits of incorporating the GWave technology into CGM devices are evidently obvious for individuals treated with intensive insulin therapy who rely on the accuracy of their glucose test results for accurate insulin dosing as well as real-time detection and remediation of acute glycemic events. However, the potential benefits are even greater for individuals with T2D, a population wherein adherence to BGM is suboptimal. 1 –3,15,16
Integrating GWave technology into real-time CGM systems could effectively address all these barriers, which would improve clinical outcomes and quality of life. Such devices may be beneficial in managing women with gestational diabetes who are often poorly adherent to their prescribed blood glucose testing regimens, which has been shown to be associated with a greater incidence of preeclampsia and elevated HbA1c levels. 17
Moreover, a noninvasive CGM system may also be beneficial in screening for diabetes and prediabetes. Studies have shown that even short-term CGM use can more accurately detect early dysglycemia compared with the other short-term methods such as HbA1c testing and 2-h OGTT. 18 Another potential use is in early screening for insulinoma, which is currently assessed using the insulin release index based on the 72-h fasting test. As recently reported by Ma et al., CGM can be useful in identifying the etiology of insulinoma. 19 In addition, use in hospitalized patients, including those in the intensive care unit, may be possible if future studies demonstrate accuracy. The convenience of a noninvasive CGM system may prompt greater use of this technology, resulting in earlier intervention and improved outcomes.
Our study has notable limitations that affect the generalizability of our results. The correlation assessments will require confirmatory testing with yellow springs instrument or equivalent comparison to verify MARD values because Food and Drug Administration (FDA) comparator standards were not used in our evaluation. Although our cohort was somewhat diverse regarding the age of participants, it is unknown whether other factors such as racial characteristics related to skin color or body mass index may impact accuracy. In addition, because the study was conducted under a controlled environment, we were unable to assess the accuracy of the GWave system under real-world environmental conditions.
Conclusions
Current results from the GWave glucose monitoring system show promise and could potentially be the first noninvasive technology. Future studies will focus on larger number and diversity of people in all glucose ranges. Real-time noninvasive BGM is possible using GWave technology.
Footnotes
Acknowledgment
The authors thank Christopher G. Parkin, MS, CGParkin Communications, Inc., for his editorial assistance in developing this article.
Authors' Contributions
I.B.H. designed the study and edited the article. A.N. researched the data, contributed to the discussion, and wrote/edited the article. A.T. conducted the study and obtained the data.
Author Disclosure Statement
I.B.H. reports grant support from Dexcom, Tandem, and MannKind, and consulting fees from Abbott Diabetes Care, Roche, and Hagar. A.N. has received consulting fees from Hagar. A.T. has received research funding from Hagar.
Funding Information
Hager funded the research study.
Supplementary Material
Supplementary Figure S1
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
