Abstract
Aims/hypothesis:
To compare glycemic metrics during pregnancy between women with type 1 diabetes (T1D) delivering large-for-gestational-age (LGA) and appropriate-for-gestational-age (AGA) infants, and to identify predictors of LGA infants.
Materials and Methods:
A cohort study including 111 women with T1D using intermittently scanned continuous glucose monitoring from conception until delivery. Average sensor-derived metrics: mean glucose, time in range in pregnancy (TIRp), time above range in pregnancy, time below range in pregnancy, and coefficient of variation throughout pregnancy and in pregnancy intervals of 0–10, 11–21, 22–33, and 34–37 weeks were compared between women delivering LGA and AGA infants. Predictors of LGA infants were sought for. Infant growth was followed until 3 months postdelivery.
Results:
In total, 53% (n = 59) delivered LGA infants. Mean glucose decreased during pregnancy in both groups, with women delivering LGA infants having a 0.4 mmol/L higher mean glucose from 11–33 weeks (P = 0.01) compared with women delivering AGA infants. Mean TIRp >70% was obtained from 34 weeks in women delivering LGA infants and from 22–33 weeks in women delivering AGA infants. Independent predictors for delivering LGA infants were mean glucose throughout pregnancy and gestational weight gain. At 3 months postdelivery, infant weight was higher in infants born LGA compared with infants born AGA (6360 g ± 784 and 5988 ± 894, P = 0.04).
Conclusions/interpretations:
Women with T1D delivering LGA infants achieved glycemic targets later than women delivering AGA infants. Mean glucose and gestational weight gain were independent predictors for delivering LGA infants. Infants born LGA remained larger postdelivery compared with infants born AGA.
Introduction
Over the past 20
Maternal hyperglycemia is considered to be a major determinant of fetal growth, predicting fetal overgrowth and neonatal adverse outcomes. 6 In women with T1D, there is a positive independent association between HbA1c during pregnancy and fetal growth. 9 However, continuous glucose monitoring (CGM) probably reflects the actual fetal glycemic exposure better than HbA1c. 10 Excessive gestational weight gain is emerging as an important risk factor for fetal overgrowth and up to 65% of pregnant women with T1D have excessive gestational weight gain irrespective of prepregnancy body mass index (BMI). 11,12
Recently, the use of CGM during pregnancy has become part of routine care for T1D in many places. Continuous use of CGM during pregnancy in women with T1D has been associated with lower HbA1c, reduced fetal overgrowth, and fewer neonatal complications compared with routine care using blood glucose monitoring alone. 13
In our center, all women with T1D, who are not already using CGM, are offered intermittently scanned CGM (isCGM) during pregnancy planning and pregnancy. 14 The isCGM measures subcutaneous interstitial glucose concentrations every 60 s and reports a glucose value every 15 min to the raw data set. 15 The use of isCGM offers easily accessible sensor-derived glycemic metrics across 24 h via smartphone and thereby a unique insight to daily glycemic patterns by the women themselves and their caregivers. 16
Higher mean sensor glucose levels in second and third trimester and higher time above range in pregnancy (TARp) have been associated with LGA in women with T1D using CGM. 9,17,18 However, these studies did not include the potential role of gestational weight gain on fetal growth and it remains unknown which sensor-derived glycemic metrics are best for prediction of pregnancy outcomes such as fetal overgrowth, and at which time during pregnancy. 9,17
The aim of this study was to compare sensor-derived glycemic metrics during pregnancy and to follow infant growth until 3 months postdelivery between those who delivered LGA and those who delivered appropriate-for-gestational-age (AGA) infants. We also investigated sensor-derived glycemic metrics as predictors of LGA infants when taking gestational weight gain and other potential predictors into account.
Materials and Methods
Study design and population
This study was a preplanned secondary analysis of sensor-derived glycemic metrics based on prospectively collected data as part of the CopenFast trial, a randomized controlled open-label, single-center trial evaluating the effect of faster acting insulin aspart compared with insulin aspart from early pregnancy until 3 months postdelivery in women with T1D and type 2 diabetes during 2019–2022. The primary outcome was birthweight standard deviation score. Secondary outcomes included gestational weight gain and infant weight 3 months postdelivery. The cohort has previously been described in detail. 1,16
In the original CopenFast trial, 150 women with T1D used isCGM or real-time CGM (rtCGM) during pregnancy. 1 Twenty-six (17%) women using rtCGM were excluded. At randomization, 122 women used isCGM initiated before pregnancy and two women initiated isCGM shortly after randomization in early pregnancy. Out of these 124 (83%) women using isCGM, two women with small-for-gestational-age infants (birthweight <10th percentile), five with early fetal loss, and six without available isCGM data were excluded. This secondary analysis included 111 women with T1D (including one woman with maturity-onset diabetes of the young, who in the original study was stratified as T1D based on her clinical presentation) 1 using isCGM, 93% (n = 103) Freestyle Libre and 7% (n = 8) Freestyle Libre 2 in early pregnancy. During pregnancy, six women changed from Freestyle Libre to Freestyle Libre 2.
Sensor-derived glycemic metrics
All isCGM data were recorded and uploaded using LibreView (Abbott Diabetes Care, Alameda, CA). For each woman, the raw spreadsheets with isCGM data were downloaded from periconception (defined as date of first day of last menstrual period) or day of isCGM application in early pregnancy, as relevant, until 37 completed gestational weeks. In case of preterm delivery (before 37 weeks), isCGM data were downloaded until delivery. Based on trial visits in the original study at 10, 21, and 33 weeks, the isCGM data were analyzed in four pregnancy intervals: 0–10, 11–21, 22–33, and 34–37 weeks. 1
Based on a standard range of summary glycemic metrics, the following were calculated from each raw isCGM spreadsheet; mean sensor glucose, percentage of time in range in pregnancy (3.5–7.8 mmol/L, TIRp), percentage of TARp (>7.8 mmol/L), percentage of time below range in pregnancy (<3.5 mmol/L, TBRp) and mean sensor glucose coefficient of variation (CV). Mean sensor glucose CV (%) was calculated by dividing glucose SD by the mean of the corresponding glucose reading. The mean number of glucose readings was calculated per woman per week for each of the four pregnancy intervals.
Routine diabetes and pregnancy care
All women followed the routine antenatal care at our center, with consultations by a diabetes specialist approximately every 2 weeks throughout pregnancy. 16
The women were recommended to use sensor-derived glycemic metrics to adjust meal-time insulin dose every 3–5 days between routine visits to obtain isCGM targets 4–5.5 mmol/L preprandially and 4–7 mmol/L postprandially aiming for the following targets: mean sensor glucose value of 5–6 mmol/L, TIRp >70%, TARp <25%, and TBRp <4%. 15,16 HbA1c targets were <48 mmol/mol (6.5%) before 20 weeks and <38 mmol/mol (5.6%) after 20 weeks. 1
All women received the same recommendations on medical nutritional therapy, carbohydrate counting, and physical activity as part of routine care. 1,14,16
According to local recommendations, women were recommended to aim for gestational weight gain according to the prepregnancy BMI as follows: 10–15 kg if BMI <25 kg/m2, 5–8 kg if BMI 25–29.9 kg/m2, and 0–5 kg if BMI ≥30 kg/m2. 14
The women attended routine obstetric visits at ∼10, 12, 21, 27, 33, and 36 weeks. Fetal growth was routinely assessed by ultrasound at 27, 33, and 36 weeks, with more frequent assessments if fetal growth deviations were detected.
Data collection and definitions
At trial visits in the original study at 10, 21, 33, and 35 weeks, the following data were recorded: gestational age, HbA1c, weight, blood pressure (BP), insulin dose, and presence of proteinuria. 1 Gestational weight gain was defined as the difference between the last weight measured before delivery and the self-reported prepregnancy weight. 19 For this analysis, women were categorized as having excessive gestational weight gain if exceeding the recommendations from the Institute of Medicine (IOM), that is, ≥16.0 kg if prepregnancy BMI <25 kg/m2, ≥11.5 kg if prepregnancy BMI 25–29.9 kg/m2, and ≥9.0 kg if prepregnancy BMI of ≥30 kg/m2. 14
Gestational age at delivery given as weeks (+0 days), birthweight, and offspring sex were recorded. LGA was defined as birthweight >90th percentile adjusted for gestational age and sex. 20 AGA was defined as birthweight between the 10th and 90th percentile.
Preeclampsia was defined as hypertension (office BP ≥140/90 mmHg) developed after 20 weeks with coexistence of proteinuria, defined as ≥+1 on a sterile urine dipstick, or symptoms from other organs. 21 Gestational hypertension was defined as hypertension diagnosed after 20 weeks without fulfilling the criteria for preeclampsia. 21 Early preterm delivery was delivery before 34 completed weeks and preterm delivery was delivery before 37 completed weeks.
At 1 and 3 months postdelivery, infant weight and data on breastfeeding were recorded. Infant weight SD-score at 1 and 3 months and maternal weight retention (>5.0 kg compared with prepregnancy weight) at 3 months were calculated. 1
Statistical analysis
Data are presented as numbers (%) for categorical variables and mean (SD) or median (IQR) for continuous data. Categorical variables were compared with X 2 test. Continuous variables were compared using Student's t test. If data did not follow normal distribution, logarithmic transformation was applied, and parametric statistics was used when normal distribution was obtained. One variable (duration of diabetes) did not obtain normal distribution and was analyzed with the Mann–Whitney U test.
Mean sensor-derived glycemic metrics, mean sensor glucose, mean sensor glucose CV, TARp, TIRp, and TBRp, were calculated for each participant for each gestational week and plotted on curves for women who delivered LGA infants versus women who delivered AGA infants. All glycemic metrics were calculated for both 24 h, during nighttime (00:00 to 05:59) and during daytime (06:00 to 23:59), 15 for each woman and each week. The glycemic metrics were analyzed in each pregnancy interval: 0–10, 11–21, 22–33, and 34–37 weeks and compared between women delivering LGA and AGA infants.
A difference of ≥5% in TIRp between groups was considered a clinically meaningful difference for an individual. 15
Univariate linear regression analysis was used to examine the differences in sensor-derived glycemic metrics between women delivering LGA and AGA infants at the different pregnancy intervals. The assumptions for linear regression were assessed by model diagnostics using quantile-quantile plots and histograms of residuals. Sensor-derived glycemic metrics were analyzed for 24 h, daytime and nighttime. Univariate logistic regression analysis was conducted with LGA as the dependent variable. To identify independent factors associated with LGA, multivariate logistic regression analyses were performed including variables with an a priori known clinical significance or based on P-values of <0.10 in the univariate regression analysis. All analyses were adjusted for trial treatment allocation (faster acting insulin aspart vs. insulin aspart) to address the randomization of the original randomized controlled trial. 1
Mean sensor glucose, TARp, TIRp, and HbA1c were calculated for each participant for the entire pregnancy and entered, as relevant, in the multiple regression analyses as independent variables in four different models. Model 1: mean sensor glucose for the entire pregnancy (mmol/L), nulliparity (yes/no), trial treatment allocation [faster acting insulin aspart (yes/no), gestational weight gain (kg), and baseline HbA1c (mmol/mol)]. Model 2: TIRp for the entire pregnancy (per 5%), nulliparity (yes/no), trial treatment allocation [faster acting insulin aspart (yes/no), gestational weight gain (kg), and baseline HbA1c (mmol/mol)]. Model 3: TARp for the entire pregnancy (per 5%), nulliparity (yes/no), trial treatment allocation [faster acting insulin aspart (yes/no), gestational weight gain (kg), and baseline HbA1c (mmol/mol)]. Model 4: HbA1c for the entire pregnancy (mmol/mol), nulliparity (yes/no), trial treatment allocation [faster acting insulin aspart (yes/no), and gestational weight gain (kg)]. Correction for multiple testing was not performed.
R version 4.1.0 (R Core Team, 2021, R Foundation for Statistical Computing, Vienna, Austria) was used for all the data analyses. 22 A two-sided P-value <0.05 was regarded as statistically significant.
Ethics
Written consent was obtained from all participants. The trial protocol was approved by The Danish Medicines Agency (2018-004680-31) and the Regional Ethics Committee (H-19029966).
Results
Baseline characteristics were similar between women who delivered LGA (53% (n = 59)) and AGA (47% (n = 52)) infants (Table 1).
Baseline Characteristics in 111 Women with Type 1 Diabetes Using Intermittently Scanned Continuous Glucose Monitoring According to Infant Size at Delivery (Large-for-Gestational-Age vs. Appropriate-for-Gestational-Age)
Data are presented as median (IQR), mean (SD), or n (%).
One woman with maturity-onset diabetes of the young was treated with meal-time insulin from conception until delivery and did not require long-acting insulin during pregnancy.
BMI, body mass index.
Sensor-derived glycemic metrics
Mean glucose readings per woman per week were 590 (495–650) and 580 (490–640) in the pregnancy interval 0–10 weeks (P = 0.28), 635 (585–659) and 632 (603–650) in the pregnancy interval 11–21 weeks (P = 0.33), 623 (618–629) and 642 (591–660) in the pregnancy interval 22–33 weeks (P = 0.25), and 622 (570–658) and 644 (602–662) in the pregnancy interval 34–37 weeks (P = 0.26) in women delivering LGA and AGA infants, respectively.
Mean sensor glucose decreased initially rapidly with no differences between women who ultimately delivered LGA and AGA infants. From 11 weeks, the curves diverged, and mean sensor glucose was 0.4 mmol/L higher both in the pregnancy intervals 11–21 weeks (P = 0.01, [95% CI 0.1–0.7]) and 22–33 weeks (P = 0.01, [95% CI 0.1–0.6]) in women delivering LGA compared with women delivering AGA infants (Fig. 1A).
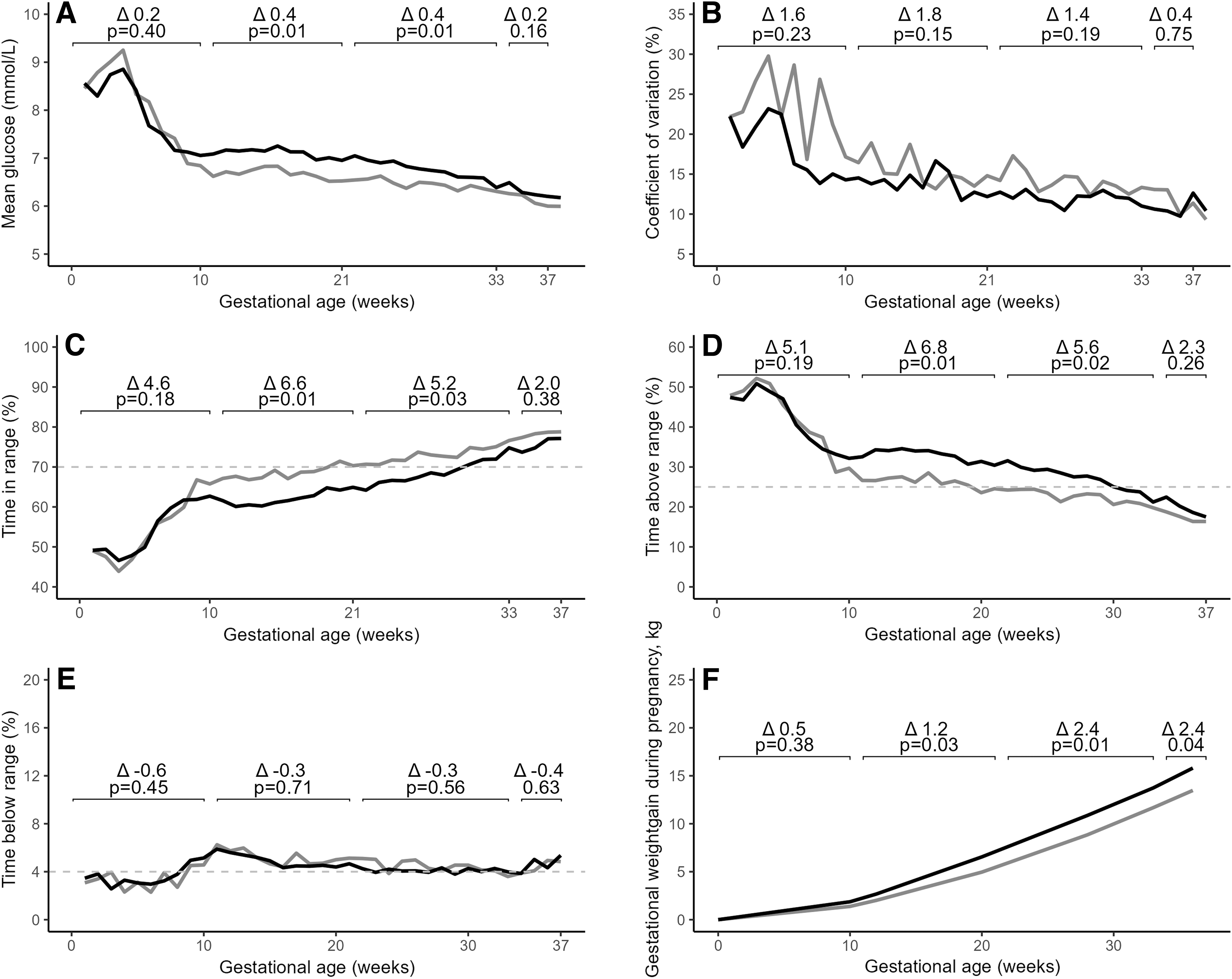
Sensor-derived glycemic metrics throughout pregnancy in 111 women with type 1 diabetes using intermittently scanned continuous glucose monitoring. The sensor-derived glycemic metrics were calculated for each participant for each gestational week and plotted according to infant size at delivery as either LGA infants (birthweight >90th percentile) (black) or AGA (birthweight between the 10th and 90th percentile) (gray). The sensor-derived glycemic metrics were analyzed in four pregnancy intervals: 0–10, 11–21, 22–33, and 34–37 weeks and compared between women delivering LGA and AGA infants. Data were available for 88% (n = 95), 100% (n = 111), 100% (n = 111), and 95% (n = 105) of women in the periods 0–10, 11–21, 22–33, and 34–37 weeks.
No differences in mean sensor glucose CV throughout pregnancy were found between the two groups (Fig. 1B).
TIRp increased throughout pregnancy in both groups. The estimated mean difference between groups was 6.6% in the pregnancy interval 11–21 weeks (P = 0.01) and 5.2% at 22–33 weeks (P = 0.03). Women delivering LGA infants obtained mean TIRp ≥70% in the pregnancy interval 34–37 weeks, while this was obtained from 22–33 weeks onward in women delivering AGA infants (Fig. 1C).
TARp decreased throughout pregnancy in both groups. The estimated mean difference between groups was 6.8% in the pregnancy interval 11–21 weeks (P = 0.01) and 5.6% at 22–33 weeks (P = 0.02). Women delivering LGA infants obtained mean TARp <25% in the pregnancy interval 34–37 weeks, while this was obtained from 22–33 weeks onward in women delivering AGA infants (Fig. 1D).
TBRp remained low, stable, and comparable between the two groups throughout pregnancy (Fig. 1E).
During daytime, mean sensor glucose was 0.4 mmol/L higher in the pregnancy interval 11–21 weeks (P = 0.01) and 0.3 mmol higher at 22–33 weeks (P = 0.02) in women delivering LGA infants compared with women delivering AGA infants. The estimated mean difference between groups in TIRp was 7.6% at 11–21 weeks (P = 0.005) and 6.3% at 22–33 weeks (P = 0.02) (Table 2).
Sensor-Derived Glycemic Metrics According to Daytime (06:00–23:59) and Nighttime (00:00–05:59) in 111 Women with Type 1 Diabetes Using Intermittently Scanned Continuous Glucose Monitoring According to Infant Size at Delivery (Large-for-Gestational-Age vs. Appropriate-for-Gestational-Age)
Results are presented as mean ± SD, number (%). Data are available for >95% of the women unless otherwise stated.
Data are available for 88% (n = 95), 100% (n = 111), 100% (n = 111), and 95% (n = 105) of women in the pregnancy intervals 0–10, 11–21, 22–33, and 34–37 weeks.
TARp, time above range in pregnancy; TBRp, time below range in pregnancy; TIRp, time in range in pregnancy.
During nighttime, mean sensor glucose was 0.5 mmol/L higher (P = 0.01) at 11–21 weeks in women delivering LGA infants compared with women delivering AGA infants, while other glycemic metrics did not differ between the groups (Table 2).
Insulin doses were similar between the two groups during pregnancy except for higher meal-time insulin dose at 35 weeks in women delivering LGA infants compared with women delivering AGA infants (Table 3).
Insulin Dose Throughout Pregnancy in 111 Women with Type 1 Diabetes Using Intermittently Scanned Continuous Glucose Monitoring According to Infant Size at Delivery (Large-for-Gestational-Age vs. Appropriate-for-Gestational-Age)
Data are presented as mean ± SD. Data are available for 95% (n = 106), 100% (n = 111), 100% (n = 111), and 91% (n = 101) of women at 10, 21, 33, and 35 weeks. Data on basal insulin dose are available in 85 women using multiple daily injections. Data on meal-time insulin dose are available in 86 women using multiple daily injections. Data on total daily insulin dose were available in 111 women.
Pregnancy outcome and possible predictors of delivering LGA infants
Women delivering LGA infants had higher gestational weight gain (16.4 ± 6 kg vs. 13.9 ± 6 kg, P = 0.04) and were more likely to have excessive gestational weight gain compared with women delivering AGA infants (Table 4). Pregnancy outcomes were similar in both groups except that women delivering LGA infants more often had an elective cesarean section and delivered infants with lower blood glucose levels 2 h postdelivery compared with women delivering AGA infants (Table 4).
Maternal, Pregnancy, and Postdelivery Outcomes in 111 Women with Type 1 Diabetes Using Intermittently Scanned Continuous Glucose Monitoring According to Infant Size at Delivery (Large-for-Gestational-Age vs. Appropriate-for-Gestational-Age)
Data are presented as median (IQR), mean (SD), or n (%). Data are available for >95% of the women. Excessive gestational weight gain: Gestational weight gain exceeding the recommendation by the U.S. Institute of Medicine (prepregnancy BMI <25 kg/m2: >16 kg, prepregnancy BMI 25–29.9 kg/m2: >11.5 kg and prepregnancy BMI of ≥30 kg/m2: >9.0 kg).
Data availability: a87% (n = 97), b50% (n = 52), c81% (n = 90), d60% (n = 67), and e79% (n = 88).
Univariate logistic regression analysis identified mean sensor glucose, TIRp, TARp, and gestational weight gain as being associated with delivery of an LGA infant (Table 5).
Risk Factors for Delivering a Large-for-Gestational-Age Infant in 111 Women with Type 1 Diabetes Using Intermittently Scanned Continuous Glucose Monitoring by Univariate and Multivariate Logistic Regression Analysis
Multivariate regression analysis identified mean sensor glucose and gestational weight gain as being independently and positively associated with delivering an LGA infant. Entering TIRp, TARp, or HbA1c, respectively, instead of mean sensor glucose in the multivariate regression analysis, showed comparable results, but these variables did not reach statistical significance (P = 0.08, P = 0.07, and P = 0.08, respectively).
Postdelivery
One and 3 months postdelivery, infant weight and infant weight SD-score were higher in infants born LGA compared with infants born AGA, while fewer infants born LGA were breastfed at 3 months. Maternal weight retention and percentage of women with postdelivery weight retention >5 kg were similar in the two groups (Table 4).
Discussion
In this prospective cohort study involving 111 women with T1D who used isCGM from conception to delivery, women delivering LGA infants achieved recommended sensor-derived glycemic targets later than women delivering AGA infants. It is novel that mean sensor glucose and gestational weight gain were independent and potentially modifiable predictors for delivering an LGA infant. Infants born LGA remained larger 1 and 3 months postdelivery compared with infants born AGA.
Regardless of initial glucose levels, mean sensor glucose decreased markedly in the first 10 weeks and diverged from 11 weeks onward, being higher in women who went on to deliver LGA infants compared with women delivering AGA infants. Interestingly, this pattern resembles findings previously described by Scott et al. where 60% of infants were born LGA, however, mean sensor glucose obtained in our study was lower. 23 The women in the present study entered pregnancy with lower mean sensor glucose, which may to some extent explain this discrepancy between the current study and the study by Scott et al. 23 Differences in the definitions of LGA may also play a role for the difference in sensor mean glucose, as we used a growth curve for Scandinavian neonates, whereas GROW curves were used in the study by Scott et al. 20,23
Nevertheless, the findings in our study suggest that obtaining lower mean sensor glucose already by 11 weeks is important to reduce the risk of LGA as higher mean sensor glucose in the second and third trimesters has also previously been associated with LGA in studies by Kristensen et al. and Scott et al. 17,23
The current study found the biggest difference in mean sensor glucose between women delivering LGA and AGA infants in the pregnancy intervals 11–21 and 22–33 weeks, indicating that glucose levels in early and midpregnancy may have a pronounced influence on fetal growth in this population. Achieving a mean sensor glucose ≤7.0 mmol/L after 10 weeks has been associated with appropriate fetal growth, 23 however, in the current study women who delivered LGA obtained mean sensor glucose around 7.0 mmol/L or lower from 11 weeks, suggesting that the target for mean sensor glucose in pregnancy should be lower than 7.0 mmol/L, that is, a target for mean sensor glucose of 5–6 mmol/L may be appropriate.
Women who delivered LGA infants did not achieve the recommended target of TIRp >70% until the pregnancy interval 34–37 weeks, ∼12 weeks later than women delivering AGA infants. This is in agreement with previous studies in which women delivering LGA infants achieved TIRp >70% only shortly before delivery or not at all. 17,23 Likewise, women delivering LGA infants obtained TARp <25% in late pregnancy, which is in line with the study by Scott et al. where women delivering LGA never achieved TARp <25% during pregnancy. 23 Insulin doses were similar between women delivering LGA and AGA infants until 35 weeks where women delivering LGA infants received higher meal-time insulin doses. This suggests that it is important to adequately increase insulin doses earlier in pregnancy to obtain sensor-derived metrics in target in the pursuit to achieve more AGA infants.
To our knowledge, this is the first study to show that both mean sensor glucose and gestational weight gain were independent predictors of delivering LGA infants. This highlights the impact of gestational weight gain on offspring birthweight, independent of glycemic control, in women with T1D. 12 Overweight and obesity before pregnancy are increasing in women with T1D and the recommendations for gestational weight gain in these women are tighter than for women with normal weight. 12,14 Strikingly, nearly 70% of women delivering an LGA infant exceeded the IOM recommendations for gestational weight gain despite being recommended to follow the stricter local recommendations for gestational weight gain, thus emphasizing the importance of increased clinical focus on gestational weight gain in addition to glycemic control during pregnancy.
Outside of pregnancy, the sensor-derived metrics mean sensor glucose, time in range, and time above range are highly correlated among each other but are relatively weakly correlated with HbA1c. 24 In pregnant women with T1D, HbA1c has been shown to correlate with both mean sensor glucose and TIRp and TARp, and a TIRp of >78% is necessary to achieve an HbA1c of less than 42 mmol/mol (6.0%) during pregnancy. 25 In the present study, women who delivered LGA infants achieved a mean HbA1c of 45 mmol/mol (6.3%) in late pregnancy, thus emphasizing the importance of achieving tight glycemic control to reduce the prevalence of LGA infants. Nonetheless, HbA1c was not significantly associated with LGA, and HbA1c has several gestational limitations. 10
We found associations between mean sensor glucose, TIRp, and TARp, respectively, and delivery of LGA infants in the univariate analysis and an independent association between mean sensor glucose and delivery of LGA infants in the multivariate analysis, while an association with HbA1c was not present. These findings are in accordance with the CONCEPTT trial, where both mean sensor glucose and TIRp and TARp were associated with the delivery of LGA infants, 9 and other studies 17,23 suggesting that CGM is at least as good as HbA1c for monitoring glycemic control during pregnancy, and that CGM might even be better at predicting adverse pregnancy outcomes such as fetal overgrowth. There is a need for more studies to establish whether sensor-derived glycemic metrics can replace HbA1c or need to be used instead of or as a supplement to HbA1c during pregnancy for monitoring the risk of poor pregnancy outcome.
Use of faster acting insulin aspart or insulin aspart was similarly distributed between women delivering LGA and AGA infants, and despite a numerically lower OR when using faster acting insulin aspart, it was not significantly related to the delivery of LGA infants. This is in line with the original report where treatment with faster acting insulin aspart during pregnancy resulted in similar fetal growth compared with treatment with insulin aspart, both in the total cohort of women with T1D and type 2 diabetes and in a subanalysis of all women with T1D. 1
Previous data from the CONCEPTT trial showed that differences in glucose levels between women delivering LGA and AGA infants were higher during daytime compared with nighttime. 9 The current study also identified the biggest difference in glycemic metrics during daytime, which may imply that postprandial hyperglycemia is still a challenging issue in this population.
CGM is recommended to women during pregnancy and pregnancy planning in Denmark. Since isCGM is easy to use, data from the previous week are readily available on a smartphone, and it is the cheapest CGM device on the market, the majority of women in our center use isCGM. The precision of isCGM has improved over time and rtCGM often needs an additional indication such as hypoglycemia unawareness. Previous studies comparing isCGM and rtCGM found no difference in mean sensor glucose between the devices. 17,26 Although the use of isCGM in pregnant women with T1D has been associated with higher TBRp, especially at nighttime, the prevalence of TBRp in the current study remained low and stable throughout pregnancy. 26 Only 17% of women with T1D in the original CopenFast cohort used different types of CGM and our analysis was restricted to isCGM to ensure data availability and homogeneity.
More than half of the women delivered LGA infants. Infants born LGA had significantly higher weight at 1 and 3 months postdelivery, implying a potential increased risk of childhood obesity starting already at birth. 3,4 Fewer infants born LGA were breastfed at 3 months, which may be an additional risk for future overweight and obesity in the offspring. 27,28
Achieving sensor-derived glycemic metrics in target already in early pregnancy might be of paramount importance for appropriate fetal growth. Automated insulin delivery (AID) systems are leading to a paradigm shift in diabetes management for persons with T1D. 29 In women with T1D and HbA1c 48–86 mmol/mol (6.5–10.0%) in early pregnancy, AID is associated with achieving more TIRp without increasing hypoglycemia and a lower prevalence of LGA infants. 30 However, no study on AID in pregnancy has been powered for pregnancy outcomes, and studies on AID in unselected pregnant women with T1D and powered for LGA and other pregnancy outcomes are warranted. 30 –32
Strengths of the current study include the prospectively collected cohort of women with T1D, treated by the same diabetes team and with large amounts of isCGM data from conception until delivery. The evaluation of glycemic metrics was based on similarly high numbers of glucose measurements in the two groups in all pregnancy intervals. Data on gestational weight gain were included in the analysis to assess its predictive significance. Both women and infants were followed until 3 months postdelivery. The cohort originated from a well-defined geographical area, and the women were from a single center with expertise in managing pregnant women with diabetes.
This may limit the generalizability of the results to other populations where glycemic control and weight management might be different. The women were predominantly of Nordic origin, which may limit applicability to other ethnic groups. We did not correct for multiple testing and therefore there is a risk of a type 1 statistical error.
In conclusion, women who delivered LGA infants achieved recommended sensor-derived glycemic targets later in pregnancy compared with women who delivered AGA infants. It is novel that higher mean sensor glucose in pregnancy and higher gestational weight gain were potentially modifiable predictors for delivering LGA infants. This may have an impact later in life since infants born LGA remained larger at 3 months of age. Future studies in larger cohorts of women with T1D are warranted to determine whether early achievement of sensor-derived glycemic metrics in target during pregnancy can reduce the prevalence of LGA.
Footnotes
Acknowledgments
The authors would like to kindly thank all the participants in the trial and the nurses at the Center for Pregnant Women with Diabetes, Ann-Sofi Lunde, Birgitta Ellingsgaard, Charlotte Barfred, Olga M. Green, Signe Lou-Møller, and Vibeke Ladefoged. The authors also acknowledge Julie Lyng Forman from the Statistical Advisory Section of Biostatistics at the University of Copenhagen and Rigshospitalet and Mads Albrecht Andersen.
Data Availability
The data set analyzed during the study is available from the corresponding author on reasonable request.
Authors' Contributions
The original trial was initiated by the principal investigator L.R. in collaboration with senior investigators E.R.M. and P.D. J.C.S., S.K.N., E.R.M., and L.R. collected the data. J.C.S. performed all the statistical analyses. J.C.S. wrote the first draft of the article. S.K.N., K.N., T.D.C., P.D., E.R.M., and L.R. critically read and revised the article. All the authors have approved the article and have consented to the publication of the article before submission.
Author Disclosure Statement
E.R.M. has contracts with Novo Nordisk for the Expect trial and the Evolve trial investigating newer insulin analogues and insulin pump treatment in pregnant women with diabetes, has received fees from Novo Nordisk for lectures, received financial support from Novo Nordisk for traveling to one international scientific meeting in the field of diabetes, and is participating on an advisory board for Novo Nordisk. P.D. has participated in clinical studies on the use of insulin in pregnant women with preexisting diabetes in collaboration with Novo Nordisk, but no personal honorarium was involved. K.N. has received funding from Novo Nordisk for an investigator-initiated trial (grant number U1111-1209-6358) with faster acting insulin aspart in insulin pump-treated adults with T1D, and is an advisory board member, for which an honorarium was given to her institution Steno Diabetes Center Copenhagen. K.N. owns stocks in Novo Nordisk. T.D. declares no conflict of interests. L.R. participated in the European Association for the Study of Diabetes annual meeting 2023 as an invite by Novo Nordisk.
Funding Information
This trial was an Investigator Sponsered Study funded by Novo Nordisk, grant number U1111-1209-6358. The grant covered full-time salaries for J.C.S. and S.K.N., as well as part-time salaries for E.R.M. and L.R.
