Abstract
Background:
The Omnipod® 5 Automated Insulin Delivery System was associated with favorable glycemic outcomes for people with type 1 diabetes (T1D) in two pivotal clinical trials. Real-world evidence is needed to explore effectiveness in nonstudy conditions.
Methods:
A retrospective analysis of the United States Omnipod 5 System users (aged ≥2 years) with T1D and sufficient data (≥90 days of data; ≥75% of days with ≥220 continuous glucose monitor readings/day) available in Insulet Corporation's device and person-reported datasets as of July 2023 was performed. Target glucose setting usage (i.e., 110–150 mg/dL in 10 mg/dL increments) was summarized and glycemic outcomes were examined. Subgroup analyses of those using the lowest average glucose target (110 mg/dL) and stratification by baseline characteristics (e.g., age, prior therapy, health insurance coverage) were conducted.
Results:
In total, 69,902 users were included. Multiple and higher glucose targets were more commonly used in younger age groups. Median percentage of time in range (TIR; 70–180 mg/dL) was 68.8%, 61.3%, and 53.6% for users with average glucose targets of 110, 120, and 130–150 mg/dL, respectively, with minimal time <70 mg/dL (all median <1.13%). Among those with an average glucose target of 110 mg/dL (n = 37,640), median TIR was 65.0% in children and adolescents (2–17 years) and 69.9% in adults (≥18 years). Subgroup analyses of users transitioning from Omnipod DASH or multiple daily injections and of Medicaid/Medicare users demonstrated favorable glycemic outcomes among these groups.
Conclusion:
These glycemic outcomes from a large and diverse sample of nearly 70,000 children and adults demonstrate effective use of the Omnipod 5 System under real-world conditions.
Introduction
Advances in diabetes technology have revolutionized the treatment paradigm for type 1 diabetes (T1D), with recent standards of care guidance emphasizing the importance of access to diabetes technology and use of these devices early in the disease. 1 Yet, many people with T1D in the United States (US) are not meeting recommended glycemic targets across all ages and demographic populations. 2 –7 Automated insulin delivery (AID) systems, which combine an insulin pump, continuous glucose monitor (CGM), and a dosing algorithm to automatically adjust insulin delivery, have been shown to be safe and effective for people with T1D in clinical trials. 8 –14
Beyond their performance in a controlled study environment, several AID systems have demonstrated successful use in a real-world setting, 15 –29 with glycemic outcomes from large-scale clinical trial and real-world studies across systems compiled in a recent review article. 24 Further, two recent real-world studies demonstrated improvements in time in range (TIR, 70–180 mg/dL) and time spent in hypoglycemia (<70 mg/dL) for both children and adults with T1D initiating different AID systems in a clinic setting. 30,31 Real-world studies of AID systems of various populations and use cases have reported anywhere from 60% to 82% TIR, and 1% to 3.6% time in hypoglycemia, 15 –29 emphasizing the myriad of factors (country, age group, insurance status, device settings, bolus frequency, baseline glycemia, and more) that can affect outcomes and the importance of evaluating these factors to inform clinical care.
Currently, there are four AID systems commercially available in the US for people with T1D, including the MiniMed 780G (and the earlier 670G/770G), the Tandem t:slim X2 with Control-IQ technology, the iLet Bionic Pancreas, and the Omnipod® 5 AID System. 32 –36 Each system differs by the specific combination of devices used, the system settings and features, and the proprietary dosing algorithm. The Omnipod 5 System, the only available system to use a tubeless insulin pump, was shown to be safe and effective through two pivotal trials in people with T1D aged 2–70 years, 33,34 with 3 months of use associated with an increase in TIR and either a reduction in time spent in hypoglycemia (<70 mg/dL), or maintenance of low levels of hypoglycemia. 33,34 These improvements were found to persist for up to 2 years of use during the pivotal trial extension phases. 37,38
The system was cleared by the US Food and Drug Administration (FDA) on January 27, 2022, for people with T1D aged 6 and up, with full market release for this age group taking place on August 1, 2022, and clearance for ages 2–<6 years was subsequently added on August 19, 2022. Thus, the system has been available commercially for most users in the US for 1 year at the time of the present study.
A unique feature of the Omnipod 5 System is that all US user data are automatically uploaded to the Insulet Cloud without any additional action by the user, removing the need for manual uploads and facilitating unprecedented access to evaluate real-world outcomes. While the clinical trial results are promising, real-world data are needed to determine if the effectiveness of the Omnipod 5 System persists in larger, more diverse groups of people under nonstudy conditions. This study aimed to evaluate glycemic outcomes and device use patterns of people with T1D using the Omnipod 5 System in a real-world setting 1 year after full market release.
Methods
Study design
This study was a retrospective analysis of Omnipod 5 System data from users aged ≥2 years in the US with self-reported T1D and data available in Insulet's electronic datasets. The Omnipod 5 System is comprised of a tubeless insulin pump (“Pod”) with an integrated AID algorithm and the Omnipod 5 App that controls the Pod's operations via Bluetooth wireless technology. The system works with an interoperable CGM, currently the Dexcom G6. 39 Detailed information on the Omnipod 5 System device function has been previously published. 39
The dataset consisted of automatically uploaded device data from anyone using the Omnipod 5 System in the US. Data are automatically and securely uploaded from the Omnipod 5 Controller or compatible personal smartphone with the Omnipod 5 App via SIM card or wireless internet.
For users who uploaded CGM data to the Insulet Provided Glooko cloud-based data management system while using another Insulet product before the Omnipod 5 System (i.e., the Omnipod DASH® Insulin Management System), that information was also included in the analysis. Device datasets were linked to a dataset of person-reported information (type of diabetes, age, etc.) collected as part of the onboarding process.
Study population
Upon initiating the Omnipod 5 System, all US users, or a parent or guardian for users aged <18 years, agreed to the required device use terms, which allow for the automated upload of their device data and for their data to be used for research purposes. The Western Institutional Review Board—Copernicus Group (WCG) Institutional Review Board (IRB) approved a request for a waiver of authorization for use and disclosure of protected health information for this study and found the protocol to be exempt under 45 CFR § 46.104(d)(4) on January 4, 2022.
The primary analysis included all users in the datasets aged ≥2 years in the US with self-reported T1D with at least 90 days of CGM data and using ≥5 units of insulin per day as of July 31, 2023. A day of CGM data was defined as a calendar day with any glucose sensor readings available. The 90-day threshold was selected to reflect typical device use and minimize any effects of the initial transition to the Omnipod 5 System. To ensure that adequate glucose data were available for accurate and meaningful interpretation of CGM metrics, we required that at least 75% of the days where CGM data were available had ≥220 readings per day (76% of possible daily readings). 40 Any users not meeting these criteria were excluded.
For the analysis comparing outcomes before and after initiation of Omnipod 5, those who transitioned from Omnipod DASH with CGM data available in Insulet Provided Glooko were included if they had a minimum of 30 days with ≥220 readings/day in the 45 days before transition to Omnipod 5, in addition to meeting the other above-listed criteria.
Statistical analysis
Device use patterns of interest included glucose target settings (i.e., the maximum number of different targets used per person, average glucose target setting) and time spent in Automated Mode. 39 The average glucose target setting was calculated as a time-weighted average glucose target per user. This value was calculated by taking the average of the active target glucose setting at every 5 min system cycle across the entire duration of use. Average glucose target values were grouped by rounding to the nearest of the five available target glucose settings. For some analyses, users with an average glucose target of 130–150 mg/dL were grouped due to low sample sizes at higher targets (3.3% and 1.9% of users at 140 and 150 mg/dL targets, respectively).
Glycemic outcomes evaluated included the percentage of sensor glucose values in the following ranges: 70–180 mg/dL (TIR), <54 mg/dL, <70 mg/dL, and >180 mg/dL, as well as the glucose management indicator (GMI). In addition, total daily dose of insulin was assessed. As glucose target setting is the foremost factor that most directly impacts performance of the system algorithm, subgroup analyses were conducted across glucose target settings as well as for those primarily using the lowest glucose target setting (time-weighted average glucose target 110–115 mg/dL), and stratified by baseline characteristics (e.g., age, prior therapy) and health insurance coverage. Outcomes pre- and post-Omnipod 5 System initiation for those with available data were compared.
Descriptive statistics were used to summarize the data. Wilcoxon signed-rank tests were used to compare outcomes pre- and post-Omnipod 5 System initiation. Analyses were only conducted if the sample size for a particular characteristic was sufficient (n ≥ 10) and were conducted using the Python programming language and two packages, pandas and SciPy, for descriptive statistics and Wilcoxon signed-rank tests, respectively. P-values with a two-sided significance level of 0.05 were considered significant.
Results
There were 69,902 users aged ≥2 years with sufficient data available at the time of analysis. Users were from 7,696 clinics across all 50 states in the US, with 12,031 health care providers prescribing the Omnipod 5 System for their patient's diabetes management. Of those aged ≥2 years, 22,162 (31.7%) were children and adolescents (2–17 years) with 6.0 million person-days of data and 47,740 (68.3%) were adults (≥18 years) with 11.7 million person-days of data. The overall median age was 30 years (Table 1).
Characteristics of the Study Population Aged ≥2 Years (n = 69,902) a
Data are mean ± SD, median [IQR], unless otherwise specified.
Number of users aged ≥2 years old in the United States with self-reported T1D with at least 90 days of sufficient CGM data (≥75% of days with ≥220 readings) and using ≥5 units of insulin per day at time of analysis (July 31, 2023).
Prior pump type was not available for 24.1% of users.
Analysis of Medicare users was conducted on users aged ≥18 years.
CGM, continuous glucose monitor; IQR, interquartile range; MDI, multiple daily injections; SD, standard deviation; T1D, type 1 diabetes.
Device use patterns
Overall, users aged ≥2 years had a median of 263 days of Omnipod 5 System use and spent a median 93.7% of time using the system in Automated Mode. The use of more than one glucose target was most often seen in younger age groups, with 82% of 2–5 years, 70% of 6–12 years, 57% of 13–17 years, 44% of 18–25 years, 38% of 26–49 years, 40% of 50–64 years, and 45% of 65 years and older using two or more glucose targets.
The time-weighted average glucose targets used most often were 110 mg/dL (53.8% of users) and 120 mg/dL (31.5% of users), but there were differences by age, with the younger age groups opting for higher targets (Fig. 1). Notably, while only 14.7% of users overall had an average target of 130–150 mg/dL, this number was much higher at 54.3% among the 2–5-year age group, with only 16.9% of this age group having an average target of 110 mg/dL. The proportion of users with an average target of 110 mg/dL increased with age, up to the group of adults aged 26–49 years, of whom 64.2% had an average target of 110 mg/dL.
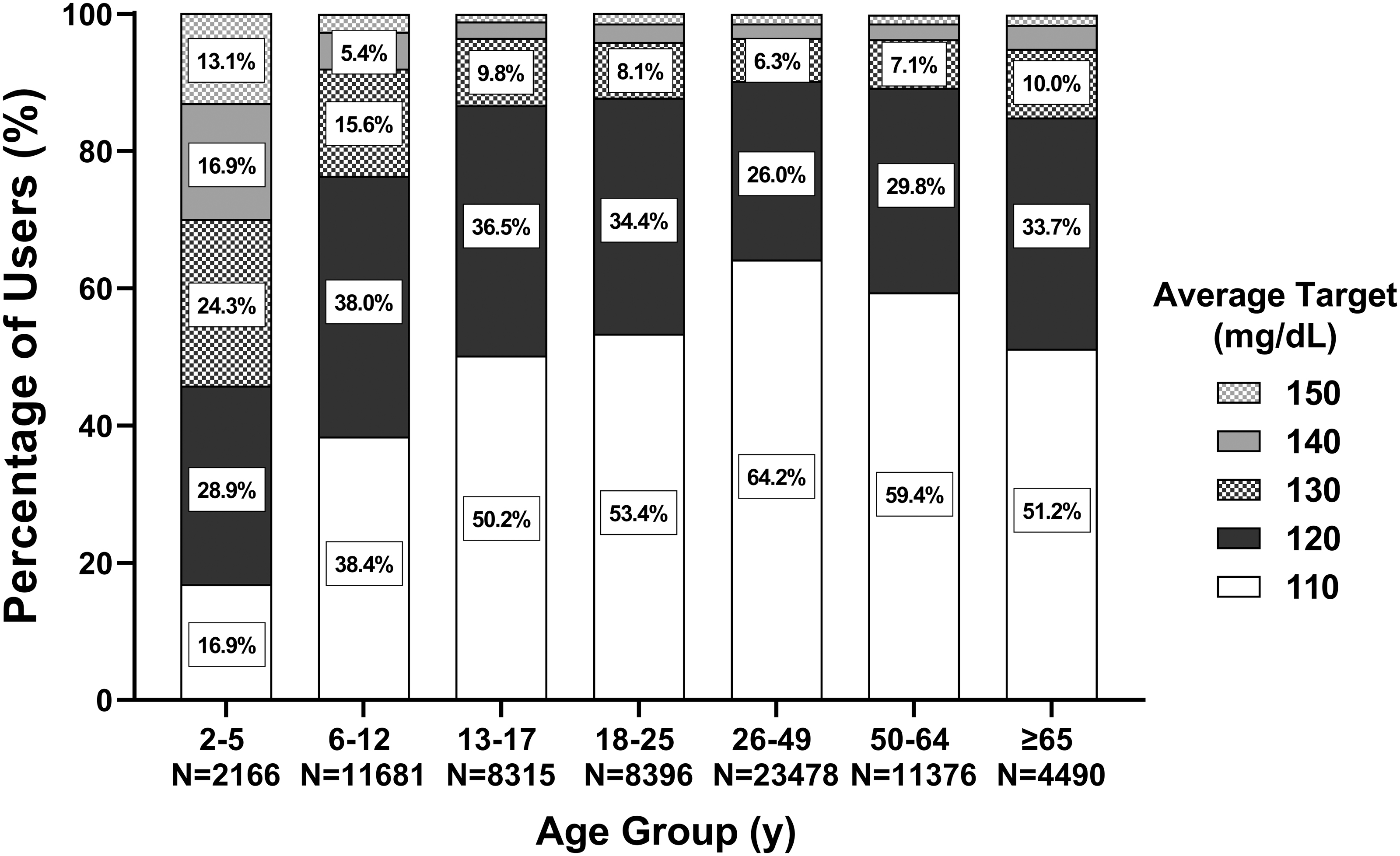
Glucose target use patterns stratified by age group. Percentage of users selecting each time-weighted average glucose target rounded to the nearest setting value by age group (n = 69,902). The stacked bar graph displays targets in descending order from top to bottom. Percentages <5% not shown for readability. Conversion factor to convert glucose from mg/dL to mmol/L, multiply by 0.0555.
Users transitioning from multiple daily injections (MDI) tended to use higher target settings than users overall, particularly among the youngest age group, while a higher percentage of users transitioning from Omnipod DASH tended to use the 110 and 120 mg/dL targets more often, similar to users overall (Supplementary Fig. S1).
Glycemic outcomes
The median percentage of TIR for the total study population was 64.2%, with children and adolescents (2–17 years) and adults (≥18 years) achieving a median TIR of 60.6% and 66.0%, respectively. The median percentages of TIR were 68.8%, 61.3%, and 53.6% for users aged ≥2 years with an average glucose target of 110, 120, and 130–150 mg/dL, respectively, with 46.1%, 24.1%, and 9.8% of users with TIR >70% (Table 2, Supplementary Fig. S2). The median percentage of time in hypoglycemia (<70 mg/dL) was 0.97% for the total study population and was 1.21% and 0.86% for children and adolescents (2–17 years) and adults (≥18 years), respectively. The median percentage of time in hypoglycemia was low for all average glucose targets (110, 120, and 130–150 mg/dL), at 0.19%, 0.15%, and 0.13% for <54 mg/dL and 1.12%, 0.87%, and 0.71% for <70 mg/dL, respectively.
Omnipod 5 Users Aged ≥2 Years Stratified by Average Glucose Target Setting (n = 69,902) a : Real-World Glycemic Outcomes and Device-Use Data
Data are mean ± SD, median [IQR], unless otherwise specified.
Number of users aged ≥2 years old in the United States with self-reported T1D with at least 90 days of sufficient CGM data (≥75% of days with ≥220 readings) and using ≥5 units of insulin per day at time of analysis (July 31, 2023).
Users with an average glucose target of 130–150 mg/dL were grouped due to low sample sizes at higher targets.
CGM, continuous glucose monitor; GMI, glucose management indicator; IQR, interquartile range; SD, standard deviation.
The outcomes at each target from the subset of users selecting only a single target throughout their entire duration of use (110, 120, or 130 mg/dL) (Supplementary Table S1) were similar to those calculated using time-weighted average glucose targets (Table 2). Glycemic outcomes for children and adolescents (2–17 years) and adults (≥18 years) stratified by glucose target setting are reported in Supplementary Table S2 and Supplementary Table S3, respectively.
Glycemic outcomes were examined in more detail for users with an average glucose target of 110 mg/dL (n = 37,640). Summaries of the glycemic outcomes for children and adolescents (2–17 years) and adults (≥18 years) (Table 3) show expected age-related variability, with a median TIR of 65.0% in children and adolescents and 69.9% in adults. The percentage of users with >70% TIR was 34.7% for children and adolescents and 49.8% for adults, with 28.3% and 44.3% achieving both >70% TIR and <4% time >70 mg/dL, respectively. Glycemic outcomes for users with an average target glucose target of 110 mg/dL stratified into more granular age groups are shown in Figure 2 and described in Supplementary Table S4.
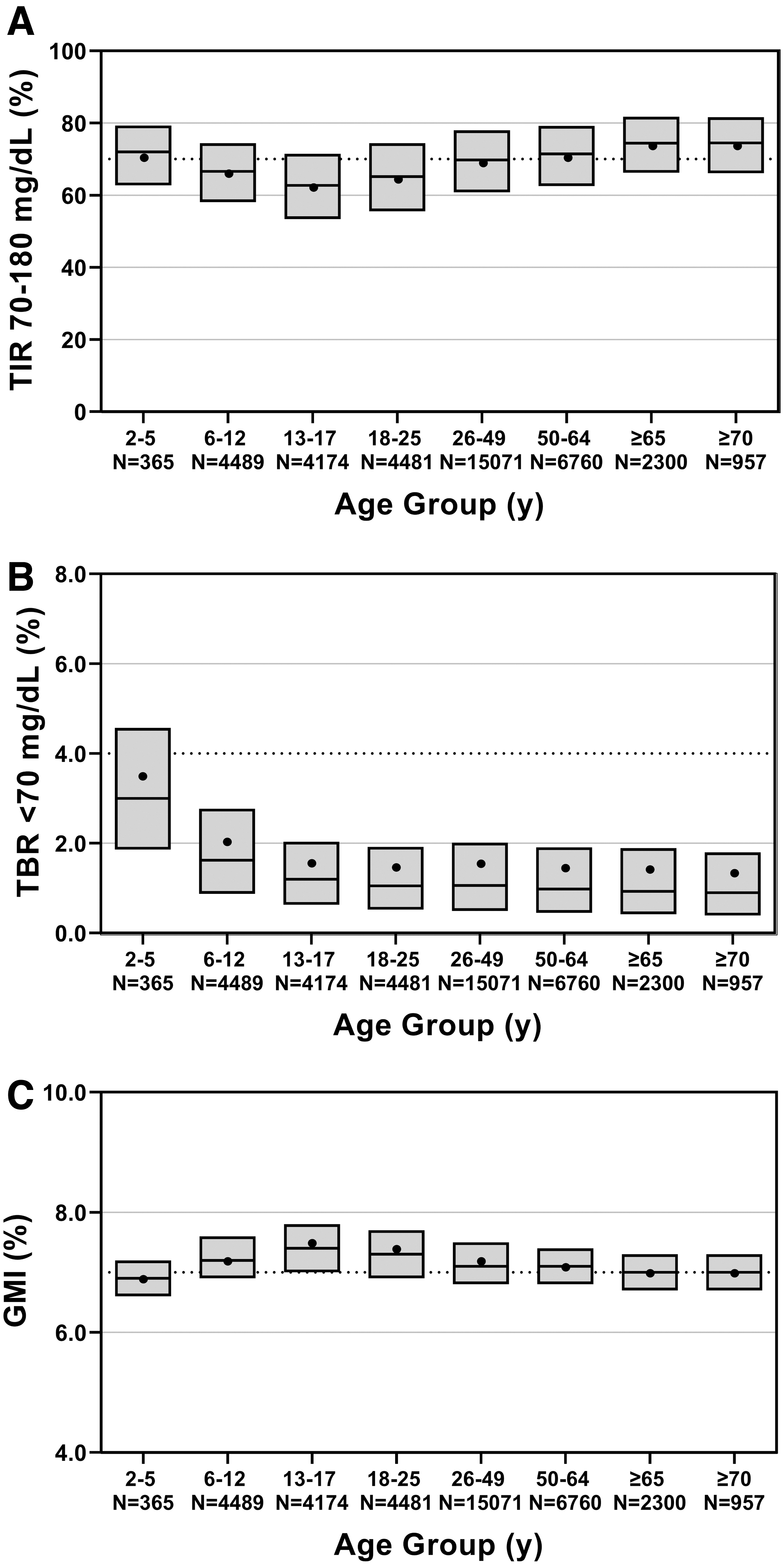
Glycemic outcomes for those using the time-weighted average target of 110 mg/dL (average target setting of 110–115 mg/dL), stratified by age group (n = 37,640).
Omnipod 5 Users Aged 2–17 Years (Children and Adolescents) and ≥18 Years (Adults) Using the Time-Weighted Average Target of 110 mg/dL (n = 37,640) a : Real-World Glycemic Outcomes and Device-Use Data
Data are mean ± SD, median [IQR], unless otherwise specified.
Number of users aged ≥2 years old in the United States with self-reported T1D with at least 90 days of sufficient CGM data (≥75% of days with ≥220 readings) and using ≥5 units of insulin per day at time of analysis (July 31, 2023) and mean target over time of 110–115 mg/dL (6.1–6.4 mmol/L).
CGM, continuous glucose monitor; GMI, glucose management indicator; IQR, interquartile range; SD, standard deviation.
Glycemic outcomes for the overall cohort with an average glucose target of 110 mg/dL stratified by the average number of daily boluses are detailed in Supplementary Table S5. Median TIR was 59.9% among users bolusing fewer than four times per day and was 72.4% among those bolusing more frequently (≥4 × per day), with a low percentage of time in hypoglycemia (<70 mg/dL) in both groups (0.85% and 1.28%, respectively).
Among users with an average glucose target of 110 mg/dL and who had pre-Omnipod 5 data available (n = 3582 previous Omnipod DASH users), the percentage of TIR and percentage of time in hypoglycemia (<70 mg/dL) improved significantly after initiating the Omnipod 5 System. For children and adolescents (2–17 years), TIR increased from a median 60.5% with Omnipod DASH to 69.3% with Omnipod 5. In addition, time <70 mg/dL decreased from 2.12% to 1.54%, and time <54 mg/dL declined from 0.32% to 0.27%. In adults (≥18 years), TIR increased from a median 65.1% to 73.3%, time <70 mg/dL decreased from 2.02% to 1.06%, and time <54 mg/dL declined from 0.27% to 0.18% (all P < 0.05) (Fig. 3).
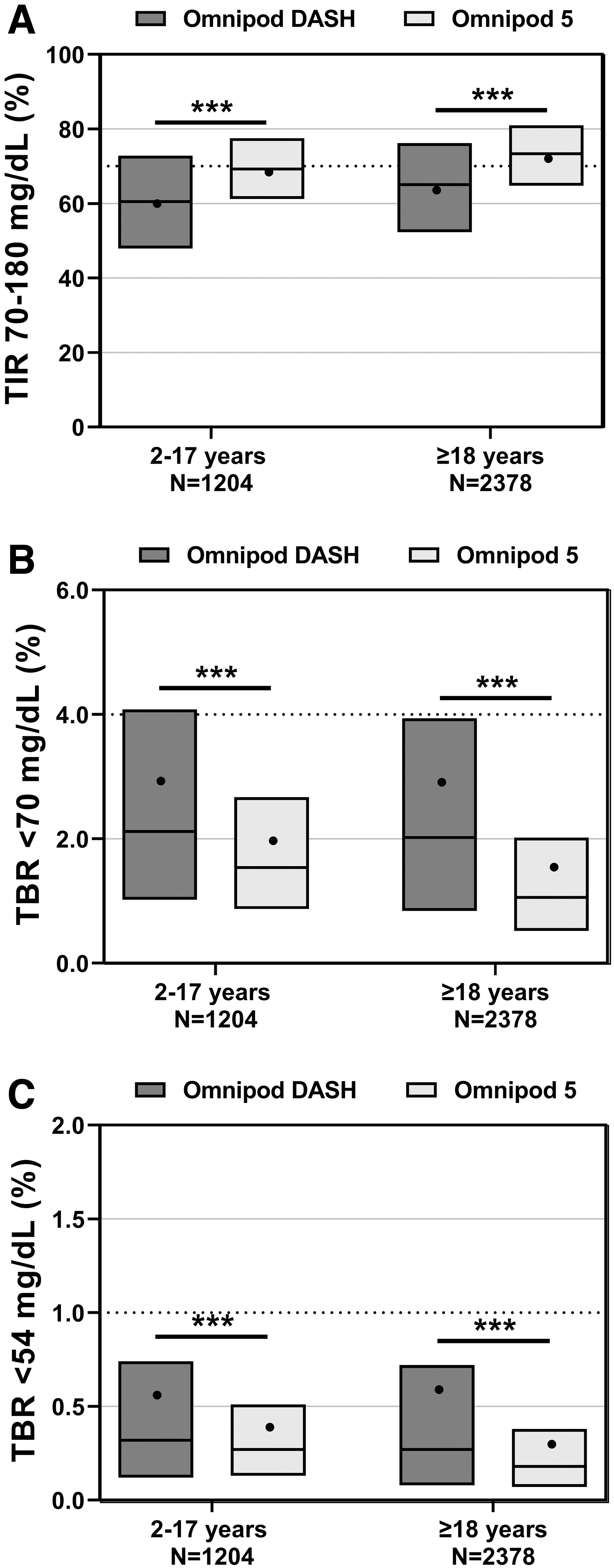
Glycemic outcomes for children and adolescents (2–17 years) and adults (≥18 years) using the time-weighted average target of 110 mg/dL (average target setting of 110–115 mg/dL) and transitioning from the Omnipod DASH to the Omnipod 5 System (n = 3,582).
Self-reported prior MDI users with an average glucose target of 110 mg/dL (n = 6525) also had favorable outcomes with Omnipod 5 use, showing a median of 70.8% TIR and 0.96% time <70 mg/dL. Results were similar among children and adolescents (2–17 years) and adults (≥18 years) previously using MDI, with a median TIR of 68.9% and 71.3%, respectively (Table 4). Median time <70 mg/dL for prior MDI users was 1.15% for children and adolescents and 0.90% for adults. Detailed outcomes among prior MDI users stratified by glucose target setting are reported in Supplementary Table S6.
Omnipod 5 Users Aged 2–17 Years (Children and Adolescents) and ≥18 Years (Adults) Using the Time-Weighted Average Target of 110 mg/dL who Self-Reported Transitioning from Multiple Daily Injections (n = 6525) a : Real-World Glycemic Outcomes and Device-Use Data
Data are mean ± SD, median [IQR], unless otherwise specified.
Number of users aged ≥2 years old in the United States with self-reported T1D with at least 90 days of sufficient CGM data (≥75% of days with ≥220 readings) and using ≥5 units of insulin per day at time of analysis (July 31, 2023) and mean target over time of 110–115 mg/dL (6.1–6.4 mmol/L).
CGM, continuous glucose monitor; GMI, glucose management indicator; IQR, interquartile range; MDI, multiple daily injections; SD, standard deviation.
Users with Medicaid insurance using an average target glucose of 110 mg/dL (n = 1064; median age 21 years) had a median TIR of 62.2% and time <70 mg/dL of 1.12% (Table 5). Users with Medicare insurance using an average target glucose of 110 mg/dL (n = 1503; median age 67 years) had a median TIR of 72.3% and time <70 mg/dL of 0.91% (Table 5). Detailed outcomes among Medicaid and Medicare users stratified by glucose target setting are reported in Supplementary Table S7 and Supplementary Table S8, respectively.
Omnipod 5 Medicaid (Aged ≥2 Years) and Medicare (Aged ≥18 Years) Users Using the Time-Weighted Average Target of 110 mg/dL (n = 2567) a : Real-World Glycemic Outcomes and Device-Use Data
Data are mean ± SD, median [IQR], unless otherwise specified.
Number of users aged ≥2 years old in the United States with self-reported T1D with at least 90 days of sufficient CGM data (≥75% of days with ≥220 readings) and using ≥5 units of insulin per day at time of analysis (July 31, 2023) and mean target over time of 110–115 mg/dL (6.1–6.4 mmol/L).
CGM, continuous glucose monitor; GMI, glucose management indicator; IQR, interquartile range; SD, standard deviation.
Discussion
Real-world evidence of Omnipod 5 System use among a diverse sample of almost 70,000 children, adolescents, and adults in the US with T1D indicates effective use of the system in a real-world setting, and it reaffirms findings from studies of this system in prospective clinical trials. 33,34 Use of the lowest glucose target (110 mg/dL) was associated with a higher TIR, with minimal time spent <70 mg/dL for users across all glucose target settings. For those using an average glucose target of 110 mg/dL, approximately one-third of children and adolescents and half of adults achieved >70% TIR, with 28.3% and 44.3% achieving both >70% TIR and <4% time >70 mg/dL, respectively, and there was a high percentage of time in Automated Mode across all age groups.
Subgroup analyses of glycemic outcomes for users transitioning to Omnipod 5 from the Omnipod DASH System or MDI and users with Medicaid or Medicare demonstrate successful system utilization across each group. These results not only demonstrate effectiveness in a large group sample under real-world conditions but are also consistent with real-world data from other AID systems showing positive glycemic outcomes and minimal risk of hypoglycemia across diverse populations. 15 –29
The customizable glucose target setting is an essential factor to investigate with the Omnipod 5 System, as this setting allows for personalized care and has the most direct impact on the behavior of the algorithm. Glucose target selection was variable with age—younger children, whose care likely reflects parental comfort, took advantage of the customizable targets more frequently, often using more than one glucose target and were also more likely to utilize the higher target settings. Although use of multiple targets was less common in older age groups compared to the youngest children, at least 40% of adult users opted to use more than 1 target setting.
These real-world results corroborate findings from clinical trials of the system, where the lowest target was the most popular among adults (aged 14–70 years, 80% of person-days) and older children (aged 6–13.9 years, 61% of person-days), 33 but was less commonly used among very young children (aged 2–6 years, 33% of person-days). 34 Similarly, real-world results from another AID system with customizable glucose targets found that the lowest setting was used most frequently (48% of time), while the three incrementally higher targets were selected for 23%, 22%, and 1.9% of time, respectively. 15 As such, this work adds to the body of evidence highlighting the importance of personalization of insulin delivery to conform to user preferences.
By design, use of the lowest average glucose target setting of 110 mg/dL was associated with highest TIR on the population level, which agrees with clinical trial findings that showed TIR incrementally increased with progressively lower Omnipod 5 glucose targets, 33,34 and is consistent with reports of other AID systems that demonstrate higher TIR with use of lower glucose target settings. 15,25 Still, 24.1% and 9.8% of users achieved TIR >70% even with the higher 120 and 130–150 mg/dL average targets, respectively, emphasizing that having the option to customize glucose targets can be beneficial to optimize outcomes and personalize care. Although time in hypoglycemia (<70 mg/dL) remained low at the population level at median 1.12% even at the lowest target setting, individual results may vary. For some, the higher targets may result in the most satisfactory outcomes when balancing several factors, including fear or unawareness of hypoglycemia, occurrence of hypo- and hyperglycemia with a given target, physical activity patterns, meal, and bolusing habits, other health risk factors, and comfort level with technology. However, users who are seeking to increase their TIR should consider decreasing their target glucose setting toward the 110 mg/dL target whenever possible.
The large sample included in this study enabled stratification of results into unique subgroups, such as by prior therapies and insurance coverage. The Omnipod DASH System is a tubeless continuous subcutaneous insulin infusion (CSII) system and the predecessor to the Omnipod 5 System. Real-world evidence on the use of the Omnipod DASH System in 4,738 children and adults with T1D demonstrated significant improvements in glycated hemoglobin (HbA1c) levels and reductions in hypoglycemia after 3 months of use when switching from MDI or other CSII systems. 41 The use of the lowest average glucose target (110 mg/dL) with the Omnipod 5 System led to further glycemic improvements for prior Omnipod DASH and CGM users under real-world conditions. Separate reports have demonstrated similar improvements in TIR for children and adults following real-world use of other AID systems after transitioning from CSII. 15,18
Conversely, there are little data available on MDI users transitioning directly to an AID system in the real world. The data available in the present study provide insights on the largest group of MDI users transitioning to an AID system in the real-world to date. Of particular note, children and adolescents in this subgroup achieved a median GMI of 7.1%, which is lower than HbA1c levels reported for MDI with CGM users of a similar age reported in the T1D Exchange Registry (8.0% and 8.8% for ages <13 years and 13- < 26 years, respectively), 4 highlighting the benefit of transitioning to AID among this population.
Medicare and Medicaid users in this sample also came close to or met consensus guidelines for TIR and time below range in both groups when using an average glucose target of 110 mg/dL. 40 These results are consistent with glycemic outcomes reported in a separate study of a similar size under real-world conditions for Medicare (n = 4,243) and Medicaid (n = 1,332) users following at least 30 days of use of another AID system. 20 Notably, in the present study, Medicare users (median age 67 years) achieved a similar TIR to all adults using the average glucose target of 110 mg/dL. While older adults with T1D are often overlooked as potential candidates for diabetes technology, 42,43 these results indicate effective utilization of Omnipod 5 among this group.
The lower TIR in the Medicaid group (median age 21 years) compared to the general population is expected given previous findings that privately insured people with T1D often have better clinical outcomes and lower HbA1c levels relative to those with public insurance. 7,44 Moreover, a high proportion of Medicaid beneficiaries are children, adolescents, and young adults who face significant barriers to access diabetes technology, 45 –47 contributing to the higher HbA1c levels in this population. 7,44
Encouragingly, Medicaid users in this study achieved a median GMI of ≤8.0% across all glucose targets, which is lower than HbA1c levels reported for publicly insured people with T1D of a similar age group in the T1D Exchange Registry (8.9%, 9.1%, and 8.4% among those aged 1–15, 16–25, and 26–40 years, respectively). 7 Thus, when provided access to advanced diabetes technologies such as AID systems, this group that has historically had the greatest deviation from recommended glycemic targets can improve their glycemic outcomes with reduced burden. 48,49
There is a growing body of evidence that reports safe and effective use of other AID systems in a real-world setting in large groups of people with T1D across a broad range of baseline characteristics. 15 –29 A recent postmarket surveillance study of Control-IQ system use under real-world conditions in the US (n = 3,061) reported TIR values of 61.2%, 60.9%, and 70.1% among those aged 6–13, 14–17, and ≥18 years, respectively. 23 These results from Graham et al. report a higher TIR compared to the adult population in the present study 23 ; however, adults using the lowest target with the Omnipod 5 System achieved a comparable TIR of 69.9%. Another larger study of the same system (n = 19,354) in the US limited to users who transitioned from a predictive low glucose suspend system and had uploaded their data revealed an overall TIR of 70.5% (lowest at 63.3% for adolescents and highest at 75.3% for ages ≥65 years). 18
A study of the MiniMed 780G System (n = 12,870 in 24 countries outside the US, primarily Europe) also reported higher TIR outcomes than the present study (mean value of 75.8%, up to 78.9% and 81.3% TIR for the ∼10% of pediatric and adult users with the recommended settings) 15 ; however, these data include outcomes for select users who took additional actions to enable the upload of their device data rather than outcomes across all users. Finally, a real-world study of the CamAPS FX system reported a mean TIR of 72.6% for 1,805 users in Europe and Australia who agreed for their data to be included. 26
Notably, the time spent in hypoglycemia (<70 mg/dL) reported herein with the Omnipod 5 System even at the lowest target (mean 1.62%, median 1.12%) was lower than that with use of the MiniMed 780G System (mean 2.5% for all users, or 3.6% and 2.8% for pediatric and adult users with the recommended settings) 15 and CamAPS FX system (median 2.3% overall), 26 and was comparable to the Control-IQ system (median 1.1% in the smaller study and mean 1.46% in the larger study). 18,23
While it is useful to examine the present results in context of the current field of real-world evidence of AID systems, the above direct comparison across real-world datasets must be taken with caution. First, it has been demonstrated that glycemic outcomes, such as TIR, are highly dependent on the baseline TIR or HbA1c of the study participants. 25,50 –52 Thus, glycemic outcomes between studies are likely to differ as a result of the baseline glycemic management of users. In contrast to the studies described above, 15,18,23,26 a recent outpatient observational study of adults with HbA1c ≥8.5% initiating a range of AID systems (largely the MiniMed 780G, Control-IQ, and CamAPS FX systems) reported a lower TIR of 61.9% for participants, 30 highlighting the importance of considering each study population as some real-world datasets may not be representative of a typical clinic population.
In addition, national registry data have demonstrated people with T1D in the US have higher HbA1c levels on average compared to people with T1D across Europe, 53 –55 and this difference may contribute to the variability seen across real-world data on AID system use between countries. Further, study inclusion criteria (e.g., time on device, sufficient CGM data threshold), device-specific features (e.g., customizable glucose targets or other adjustable settings), and device data upload processes, which could introduce selection bias, vary between studies. Collectively, these differences in both the study populations and data collection make direct comparison of real-world data across AID systems challenging, and results from each individual system are best considered only within the context of the population from which they were collected.
A major strength of this work is the large sample size of participants, enabling conclusions on a population-based level. With almost 70,000 US users, this is, to our knowledge, the largest study of AID system use under real-world conditions published to date in the current AID therapeutic landscape. 15 –29 Such a sizeable sample provides evidence of Omnipod 5 System use in a more diverse population, including nearly 6,000 users with public health insurance and users that may not be considered typical candidates for AID. Further, the large number of prescribers across a broad range of more than 7,000 clinics, including larger academic institutions and primary care clinics, is reflective of the varied clinical practice settings in the real-world.
Another important aspect of this study is the automatic upload of data, which eliminates the need for manual uploads, and thus minimizes selection bias that would result from more engaged users who upload more often tending to have better glycemic outcomes. 56 In addition, the retrospective nature of this work removes any potential participant bias arising from research participation. Finally, although the requirements for inclusion were 90 days of CGM data, the actual median days of use was ∼3 times this amount, which may indicate promise for continued success with the Omnipod 5 System over time. This is further supported by recent data demonstrating that the initial improvements seen in the Omnipod 5 pivotal trials are durable for up to 2 years of at-home use of the system. 37,38
This study is not without limitations. The exclusion of users that discontinued use (before 90 days) or had limited CGM availability (<75% of days with ≥220 readings/day) of the Omnipod 5 System limits the generalizability of the results to only those with adequate data for reliable computation. Further, while the inclusion criteria were selected to exclude off-label use of the system, the self-reported nature of users' age and demographic data is a limitation. Finally, the ability to report only associations rather than causal effects is a potential limitation of the observational nature of the study without a control group. Future investigations can help understand the challenges faced by users who are not achieving consensus targets, the underlying differences between those with and without sufficient data, and reasons for attrition.
Conclusion
These data from almost 70,000 users in the US demonstrate effective use of the Omnipod 5 System in a large and diverse sample of children and adults with T1D under real-world conditions.
Footnotes
Acknowledgments
We extend our sincere thanks to the Omnipod 5 users whose data were included in this study. The authors thank Jodi Bernstein, PhD, MPH, RD, of Jodi Bernstein Medical Writing (Toronto, Canada) who received payment from Insulet Corporation for assisting with drafting and formatting of the article. We are grateful to Steve Lowen, PhD, of Insulet Corporation for providing advice on the data analysis.
Authors' Contributions
L.M.H., I.H., and T.T.L. created the conception of the study. L.M.H., I.H., and T.T.L. designed the study. I.H. and J.J.M. acquired data for the study. G.P.F., D.J.D., L.M.H., J.J.M., and L.R.C. analyzed the data for the study. G.P.F., D.J.D., G.A., E.G.W., C.B., L.M.H., I.H., T.T.L., and J.L.S. interpreted the data for the study. G.P.F., L.M.H., and L.R.C. contributed to drafting of the article. G.P.F., D.J.D., G.A., E.G.W., C.B., L.M.H., I.H., J.J.M., L.R.C., T.T.L., and J.L.S. critically revised the work for important intellectual content. T.T.L. had full access to the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. All authors made the decision to submit this article for publication.
Author Disclosure Statement
G.P.F. reports grants and consulting fees from Insulet, during the conduct of the study; consulting fees and grants from Medtronic, Dexcom, Tandem, Lilly, and Beta Bionics; consulting fees from Abbott, outside of the submitted work. D.J.D. reports research support from Insulet during the conduct of the study and personal honoraria from Dexcom and Insulet outside the submitted work. G.A. has received consulting fees from Dexcom, Insulet, and Medscape; and grants from Fractyl Health, Insulet, MannKind Tandem Diabetes, and Welldoc. E.G.W. reports speaker and other fees from Abbott Diabetes Care, AstraZeneca, Dexcom, Eli Lilly, Embecta, Insulet, Medtronic, Novo Nordisk, Roche, Sanofi, and Ypsomed; and research support from Abbott Diabetes Care, Embecta, Insulet, Novo Nordisk, Sanofi. C.B. reports consulting and speaker fees from Insulet, Dexcom, and Embecta and educational grants from Insulet and Tandem. J.L.S. reports research support from Insulet, during the conduct of the study. J.L.S. serves, or has served, on advisory panels for Bigfoot Biomedical, Cecelia Health, Insulet Corporation, Mannkind, Medtronic Diabetes, StartUp Health Diabetes Moonshot, and Vertex. J.L.S. has served as a consultant to Abbott Diabetes, Bigfoot Biomedical, Insulet, Medtronic Diabetes, and Zealand. Yale School of Medicine has received research support for J.L.S. from Abbott Diabetes, JAEB Center for Health Research, JDRF, Insulet, Medtronic, NIH, and Provention Bio. L.M.H., I.H., J.J.M., L.R.C., and T.T.L. are full-time employees of and own stock in Insulet Corporation.
Funding Information
This study was funded by Insulet Corporation.
Supplementary Material
Supplementary Table S1
Supplementary Table S2
Supplementary Table S3
Supplementary Table S4
Supplementary Table S5
Supplementary Table S6
Supplementary Table S7
Supplementary Table S8
Supplementary Figure S1
Supplementary Figure S2
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
