Abstract
Background:
Despite advances in technology, glycemic outcomes in people with type 1 diabetes (T1D) remain suboptimal. The MiniMed 780G (MM780G) advanced hybrid closed-loop (AHCL) system is the latest technology for T1D management with established safety and efficacy. This study explores the cost-effectiveness of MM780G AHCL compared against multiple daily injections (MDI) plus intermittently scanned continuous glucose monitor (isCGM).
Methods:
A cost-utility analysis was conducted, simulating lifetime outcomes for 1000 T1D individuals, with baseline hemoglobin A1c of 8.4%, using the IQVIA Core Diabetes Model (CDM) v9.5. A Singapore health care payer perspective was taken with 2023 costs applied. Treatment effects were taken from the ADAPT study and treatment-related events from a combination of sources. T1D complication costs were derived from local literature, and health state utilities and disutilities from published literature. Scenario analyses and probabilistic sensitivity analyses (PSAs) explored uncertainty. Cost-effectiveness was assessed based on willingness-to-pay (WTP) thresholds set to Singapore Dollars (SGD) 45,000 (United States Dollars [USD] 33,087) per quality-adjusted life year (QALY) and Singapore's gross domestic product (GDP) per capita of SGD 114,165 (USD 83,941) per QALY.
Results:
A switch from MDI plus isCGM to MM780G resulted in expected gains in life-years (+0.78) and QALYs (+1.45). Cost savings through reduction in T1D complications (SGD 25,465; USD 18,723) partially offset the higher treatment costs in the AHCL arm (+SGD 74,538; +USD 54,805), resulting in an estimated incremental cost-effectiveness ratio of SGD 33,797 (USD 24,850) per QALY gained. Findings were robust, with PSA outputs indicating 81% and 99% probabilities of cost-effectiveness at the stated WTP thresholds.
Conclusion:
MM780G is a cost-effective option for people with T1D managed in a Singapore setting.
Introduction
Diabetes mellitus (DM), consisting of type 1 (T1D) and type 2 diabetes, comes with a significant global disease burden and is a growing problem in Singapore. Poorly controlled diabetes places a high burden on health systems, with health care expenditure and productivity losses expected to grow substantially over the next decade, along with increased incidence of diabetes complications. 1
People with T1D have a significant burden of self-care due to the need for insulin replacement from diagnosis and high glucose variability. 2 According to the SingHealth Diabetes Registry (SDR), the number of people being treated for T1D saw a 2.3-fold increase in less than 10 years, from 2013 to 2020, with the number of adult patients reaching 1300 in 2020 in SDR. 3 The SDR gathers data from 193,000 people with diabetes across the SingHealth health cluster in Singapore. In 2016, a “War on Diabetes” policy was declared by Singapore's Ministry of Health (MOH), emphasizing diabetes as a priority condition. 4 This policy aimed to reduce the growing burden of diabetes and was accompanied by value-based approaches to improve access to and address gaps in care. These included the prioritization of therapies and devices that have demonstrated improved outcomes. 5
For people living with T1D, ideal glycemia is defined not just according to hemoglobin A1c (HbA1c) (which only describes mean glycemia) but also stable glycemia with minimal hypoglycemia (or time-below-range). 6 This is encapsulated within the international consensus guidelines on time-in-range (TIR) using continuous glucose monitoring data, with >70% time spent in TIR of 3.9–10 mmol/L being considered an optimal target and <4% of the time spent below range (TBR) of 3.9 mmol/L. Self-management in T1D involves using real-time glucose data to make decisions about insulin dosing. These decisions consider factors such as carbohydrate intake and physical activity. In addition, they may also be influenced by factors like being sick or the menstrual cycle. 7
Advancements in glucose testing platforms from point-of-care capillary blood glucose meters toward intermittently scanned continuous glucose monitor (isCGM) or real-time CGM now offer accurate, real-time, easily accessible glucose data, and have been demonstrated to improve outcomes. 8 –10 Similarly, insulin delivery systems have progressed from multiple daily injections (MDI) toward continuous subcutaneous insulin infusion (CSII), demonstrating improved glycemic control in those who continue to use them. 11 –13 However, with open-loop systems, where insulin delivery and glucose detection work independently, the burden of insulin dose titration still falls upon the person living with T1D and their health care professionals. Even when considering the various factors that influence insulin requirements, there remains much variability in daily insulin requirements in T1D, underpinning the challenges in sustaining stable and optimal mean glycemia.
Therefore, despite education in T1D self-management, 14,15 advances in insulin pump technology, and rapid growth of CGM use, glycemic outcomes among people with T1D remain suboptimal. The US T1D Exchange registry (n = 25,529) showed that mean HbA1c did not improve over an 8-year period, with only 17% of adolescents and 21% of adults achieving the American Diabetes Association's (ADA) target HbA1c goal of <7%. 12 The UK NHS National Diabetes Audit reported in July 2022 revealed that >65% of UK adults still had an HbA1c of >7.5%. 16 In addition, a 2021 study involving Singaporean T1D individuals reported even less favorable findings, with suboptimal glycemic levels in 75% of young adults included in the study. 17 Similarly, many individuals struggled to achieve recommended TIR targets of >70%. 9
Hybrid closed-loop (HCL) technology integrates a CGM with an insulin pump and an algorithm to automatically adjust basal insulin in response to sensor glucose changes and predictively adjust insulin delivery to minimize hypoglycemia and hyperglycemia. Meal announcements with manual boluses for meals are still required. The effectiveness of these systems in targeting hyperglycemia, while preventing hypoglycemia, has been proven in both clinical trials and real-world evaluations. 18,19 The latest iteration, the MiniMed 780G system (MM780G), is an advanced HCL (AHCL) system, which integrates an additional function of automated correctional boluses to further address hyperglycemia (>6.7 mmol/L) and may be delivered every 5 min. The safety and efficacy of MM780G have been demonstrated in a variety of clinical studies; perhaps more importantly, its continued use in real-world setting points toward user acceptability. 20 –23
In users already using isCGM plus MDI, yet not achieving HbA1c targets, the use of MM780G resulted in a substantial improvement in HbA1c (1.54%, standard deviation [SD]: 0.73) from a baseline of 9%. 19,24 These improvements in glycemia were achieved without increased TBR. Indeed, in those with impaired awareness of hypoglycemia, despite using a form of diabetes technology (isCGM/CSII or sensor-augmented pump), the use of MM780G led to both improvement of hypoglycemia awareness and HbA1c levels. 25 The benefit of automated insulin delivery systems notwithstanding, access to these systems remains limited in many parts of the world. This is particularly pertinent in countries where there are limited reimbursement policies for those living with diabetes, with all costs for devices borne by the person with T1D.
In Singapore, the Healthier SG movement aims to prioritize preventative health care to reduce the downstream burden from complications related to suboptimal management of noncommunicable diseases. 26 Accordingly, it is important to consider the costs of adopting this technology versus the benefits over a protracted time trajectory. The cost-effectiveness of MM780G has been assessed in two European countries, 27,28 but the health care structure of these settings and the tariffs applied in these analyses limit the ability to generalize these findings to other settings. This study explores the cost-effectiveness of MM780G for the management of T1D in a Singapore setting, which offers no reimbursement for CGM or AHCL systems, akin to neighboring countries in the region. Currently, available data are applied to a localized analysis setting.
Methods
We conducted a cost-utility analysis, simulating lifetime outcomes for 1000 people with T1D using the IQVIA Core Diabetes Model v9.5 (CDM) 29 –31 to estimate the cost-effectiveness of MM780G compared to MDI plus isCGM in a Singapore setting.
The economic model
The CDM T1D model consists of a series of Markov submodules that simulate the expected management pathway of people with T1D based on the likelihood of T1D complications (the model health states). Each health state is assigned an annual cost and a weighted utility to enable the estimation of an incremental cost-effectiveness ratio (ICER). Progression through the model (i.e., the T1D pathway) is defined according to the baseline characteristics of the cohort and the expected treatment impact of the modeled management strategies (based on extrapolation of the short-term impact on HbA1c).
Model outcomes are estimated for each management strategy (e.g., usual care vs. a novel intervention) and include projected lifetime direct treatment costs arising from diabetes-related complications and T1D management, indirect costs from lost workplace productivity, and health benefits. Health benefits are based on the estimated rates of T1D complications and the expected impact on both total life years (life expectancy) and quality-adjusted life years (QALYs). Cost-effectiveness is assessed based on a calculation of the ICER associated with a switch in management strategy (i.e., the difference in costs divided by the difference in QALYs).
Perspective, time horizon, and discounting
The analysis was conducted from the Singapore health care payer perspective with 2023 costs applied. 32 A lifetime time horizon was applied with an annual discount rate of 3% applied to both costs and outcomes, following Singapore's Agency for Care Effectiveness (ACE)'s technical guidance. 33 Scenario analysis explored the inclusion of costs related to productivity losses from T1D morbidity and complications, alongside the baseline perspective of direct medical costs alone, as a proxy for a societal perspective.
Patient population
The patient population comprised adults with T1D suitable for MM780G. Cohort characteristics including gender (76.0% female), baseline HbA1c (mean: 8.4%, SD: 1.84%), and mean age (32.2 years, SD: 15.9 years) were taken from the Singapore General Hospital (SGH) database, a main center for management of T1D in Singapore and the only center in Asia that is DAFNE accredited, 34 a structured group education program for people with T1D, globally recognized as a gold standard of care.
Intervention and comparator
MM780G was compared against MDI plus isCGM, a commonly used care method for people with T1D with a mean HbA1c of 8.4%. 35
Treatment effects and treatment-related events
Treatment effects were taken from the ADAPT trial 19 and treatment-related events were estimated from a combination of sources. 19,27,36,37 In MM780G, HbA1c was reduced by 1.54%, while in the MDI plus isCGM, the reduction was 0.2%. 19 The baseline rates of severe hypoglycemia events (SHE) requiring another person's assistance, but not requiring medical assistance (SHE 1), SHE requiring medical assistance (SHE 2), and diabetic ketoacidosis (DKA) were estimated at 64.6, 13.0, and 2.93 per 100 patient-years for MDI plus isCGM, respectively, based on an average of reported values. 37 –39 We assumed a conservative 50% reduction 40 in SHE and DKA with the use of AHCL versus MDI plus isCGM, noting that ADAPT recorded zero SHE and DKA events. Inputs were validated by the clinical authors based on local experience of T1D management (Table 1).
Treatment Effects and Treatment-Related Events
MM780 event rates assume a 50% reduction in SHE/DKA for AHCL.
AHCL, advanced hybrid closed loop; DKA, diabetic ketoacidosis; HbA1c, hemoglobin A1c; MDI, multiple daily injections; NICE, National Institute for Health and Care Excellence; NSHE, nonsevere hypoglycemia event; SHE 1, severe hypoglycemia event not requiring medical assistance; SHE 2, severe hypoglycemia event requiring medical assistance.
Treatment costs
Treatment costs were estimated based on local clinical practice with the application of direct costs to the Singapore health care system. 33 The annual cost of MDI plus isCGM was estimated at Singapore Dollars (SGD) 3829 (United States Dollars [USD] 2815), including insulin and insulin administration-related costs of SGD 1071 (USD 787). Insulin costs were estimated based on a total daily dose (TDD) of 0.71 per IU/kg (15,549 IU annual dose), based on a clinical expectation of T1D management in Singapore. 2 Insulin use was assumed to be split 50/50 between rapid-acting [SGD 9.27 (USD 6.82) per 300 U] and long-acting insulin [SGD 7.99 (USD 5.87) per 300 U] formulations, with consumable costs based on an average of 5 CBG strips and lancets per week, based on local clinical practice.
The annual cost of AHCL was estimated at SGD 7566 (USD 5563), including insulin and insulin administration-related costs of SGD 1487 (USD 1093). AHCL costs were based on a 4-year use of a pump, 3 days use per reservoir, 7 days use per sensor, and no additional cost for transmitter kits. Insulin costs were estimated based on a 20% reduction in total insulin use with AHCL compared to MDI plus isCGM (a TDD of 0.57 IU/kg) 41 –44 and 100% use of rapid-acting insulin; consumable costs were based on 3-day use per infusion set, 5 CBG strips, and lancets per week. Priming-related insulin wastage was not considered in the analysis. A scenario analysis explored the impact of applying retail pharmacy costs for insulins and consumables (as opposed to public institution prices charged to patients).
T1D complications costs
T1D complication costs (the model health state costs) were derived from local literature based on searches of the Tufts Cost-Effectiveness Analysis (CEA) Registry (
The cost of days off work (DOW) was estimated based on a mean monthly wage of SGD 5070 (USD 3728) for males in 2022 and SGD 4388 (USD 3226) for females in 2022. 50 The DOW was conservatively approximated with inpatient length of stay for diabetes-related complications taken from published MOH data. 49 These do not include DOW required for subsequent follow-up visits, home recovery, or rehabilitation, and hence underestimate the total DOW. Costs were inflated to 2023 SGD using the gross domestic product (GDP) implicit price deflator published by the International Monetary Fund. 32 For November 2023, exchange rates for SGD and USD were set at SGD 1.36 to 1 USD, 51 and additional currency conversions are provided in Supplementary Table S1. All cost inputs were validated by the clinical authors (Table 2).
Type 1 Diabetes Complication Costs (Health State Costs)
All costs are inflated to SGD 2023 values.
November 2023 exchange rates for SGD and USD were set at SGD 1.36 to 1 USD. 52
Estimated DOW limited to mean days recorded for complication-related inpatient days. 51
Where local data were not available, DOW entries were assumed to be zero following a very conservative approach.
DOW, days of work; NA, not applicable where the model did not require inputs; PVD, peripheral vascular disease; SGD, Singapore Dollars; USD, United States Dollars.
Health state and treatment-related utilities
Health state utilities and the disutilities associated with adverse events were derived from the published literature, based on established practice for CDM analyses. In addition, following previous analyses, 22 a utility benefit of 0.0432 was applied to the AHCL arm based on a reduction in fear of hypoglycemia (FoH). This was estimated based on ADAPT 6-month study findings that demonstrated a 5.4-point decrease in the hypoglycemic fear survey (HFS) for people using the AHCL therapy 22 compared against people using MDI plus isCGM. ADAPT study 6-month findings were also used for the MDI plus isCGM arm that reported a 2.0-point decrease in the HFS. 22 A 1-U increase in HFS score corresponded to a utility decrement of 0.016. An earlier study reported that a 1-U increase in HFS score corresponded to a utility decrement of 0.008. 52
Analysis outcomes
Model outcomes comprised lifetime cohort and per-person costs, outcomes, and QALYs plus estimations of ICERs. Cost-effectiveness was assessed against two willingness-to-pay (WTP) thresholds—one at SGD 45,000 (USD 33,087) per QALY, which is a threshold for medical technologies with positive subsidy recommendations in previous HTA evaluations, 50 and the other is an inferred WTP threshold of SGD 114,165 (USD 83,941) per QALY gained since Singapore does not have an explicit WTP threshold (equivalent to one-time Singapore GDP per capita, 53 a conservative marker of a GDP-based WTP threshold). This is also a commonly used WTP threshold in local cost-effectiveness studies. 46,54 –56
Treatment of uncertainty
The IQVIA CDM projects lifetime outcomes based on short-term treatment effects and is subject to uncertainty. A series of sensitivity analyses was undertaken to explore the impact of uncertainty on model outcomes. Specifically, we assessed the impact of adjusting the AHCL treatment effect on HbA1c down to the lower limit of effect in the ADAPT study (−0.8%), 19 adjusting the rates of expected SHE and DKA in the comparator arm to reflect locally expected rates of events (8.74 and 8.46 per 100 patient-years, respectively) with a conservative 50% reduction applied to estimate the comparative rates for AHCL, in line with our base case assumptions, modeling the impact of zero SHE or DKA events, in line with evidence from the ADAPT study and finally, assuming no insulin dose reduction in the AHCL cohort (i.e., applying a TDD insulin of 0.71 IU/kg across both management arms).
Alongside these analyses, we explored the impact of alternate cohort characteristics by changing the baseline HbA1c to simulate cohorts with higher baseline HbA1c levels (HbA1c of 9.0% and 9.5%). We ran an alternate combination scenario to simulate a cohort with a lower HbA1c baseline (8.0%) and an AHCL treatment effect (−1.0%). This scenario was based on an ad hoc analysis of the SGH data conducted by the clinical authors to represent the exploration of a plausible T1D subgroup. We also ran a scenario analysis based on ADAPT 12-month study findings in which we used the FoH utility of 0.0528 for AHCL and 0.0024 for MDI+isCGM.
Probabilistic sensitivity analysis (PSA) was conducted with input parameters varied between published limits (where data were available) and by ±20% for unit cost inputs (and where data were unavailable). PSA outputs were used to generate a scatterplot, plotting incremental cost and effectiveness pairs on the cost-effectiveness plane, and a cost-effectiveness acceptability curve (CEAC), plotting the probability of cost-effectiveness across a range of WTP thresholds, allowing a graphical representation of uncertainty.
Results
Base case analysis
In the base case analysis, a management switch for individuals with T1D from MDI plus isCGM to MM780G resulted in expected gains in life expectancy (+0.78 LYs) and quality of life (+1.45 QALYs), alongside an expected increase in costs (+SGD 49,073; USD 36,081), resulting in an estimated ICER of SGD 33,797 (USD 24,850) per QALY gained. Analysis outputs are reported in Table 3. Cost savings through reduction in T1D complications (SGD 25,465; USD 18,723) partially offset the higher treatment costs in the AHCL arm (+SGD 74,538; +USD 54,805).
Summary of Cost-Effectiveness Analysis Comparing Advanced Hybrid Closed-Loop Versus Multiple Daily Injections Plus Intermittently Scanned Continuous Glucose Monitor
November 2023 exchange rates for SGD and USD were set at SGD 1.36 to 1 USD. 52
CI, confidence interval; QALY, quality-adjusted life-year.
Sensitivity and scenario analysis
In sensitivity analyses, ICERs ranged from SGD 27,548 (USD 20,255) per QALY (a higher baseline HbA1c with the same treatment effect as the base case) to SGD 46,909 (USD 34,490) per QALY (a conservative estimate using a lower limit of treatment effect from the ADAPT study), compared against a base case ICER of SGD 33,797 (USD 24,850) per QALY gained. Full results are reported in Table 4.
Sensitivity and Scenario Analyses of Cost-Effectiveness Analysis Comparing Advanced Hybrid Closed-Loop Versus Multiple Daily Injections Plus Intermittently Scanned Continuous Glucose Monitor
Base case: Baseline HbA1c: 8.4% AHCL treatment effect −1.5%, MDI/isCGM treatment effect −0.2%, DKA: 2.93/100 patient-years; MDI/isCGM SHE2: 13.00/100 patient-years; SA 1: AHCL treatment effect −0.8%; SA 2: Baseline HbA1c: 9.0%; SA 3: Baseline HbA1c: 9.5%; SA 4: MDI/isCGM SHE2: 8.74/100 patient-years; SA 5: MDI/isCGM DKA: 8.46/100 patient-years; SA 6: AHCL SHE2 set to zero patient-years; SA 7: AHCL no reduction in insulin; SA 8: FoH utility from 12-month data 0.0528 for AHCL and 0.0024 for MDI+isCGM.
November 2023 exchange rates for SGD and USD were set at SGD 1.36 to 1 USD. 52
HbA1c baseline (8.0%) and AHCL treatment effect (−1.0%).
FoH, fear of hypoglycemia; ICER, incremental cost-effectiveness ratio.
When considering those scenarios that explored the impact of alternate baseline levels of HbA1c or AHCL treatment effect, ICERs ranged between the lower limit of SGD 27,548 (USD 20,255) (baseline HbA1c of 9.5%) and an upper limit of SGD 46,909 (USD 34,490). An approximation of a conservative subgroup where individuals had a lower baseline HbA1c of 8.0% at the initiation of the AHCL system with lower treatment effect (HbA1c −1.0%) led to the ICER at SGD 44,474 (USD 32,700) per QALY gained.
Alternate assumptions of SHE2 and DKA event rates resulted in ICERs ranging from SGD 27,906 (USD 20,518) (AHCL SHE2: zero patient-years) to SGD 35,686 (USD 26,238) (MDI/isCGM SHE2: 8.74/100 patient-years and AHCL SHE2: 4.37/100 patient-years).
When the costs of DOW due to T1D morbidity and complications were included in the analysis (to approximate a societal perspective), the cost difference between arms was reduced from +SGD 49,073 to +SGD 26,971 (+USD 36,081 to +USD 19,831) and the ICER re-estimated at SGD 18,575 (USD 13,657) per QALY gained. Using the FoH utilities from the combined data source of 12-month observation period led to the ICER at SGD 26,010 (USD 19,124).
PSA outputs are reported in figure legends
The PSA results showed an average QALY gain of 1.35 and a cost increase of SGD 42,779 (USD 31,454), close to the base case results (Figs. 1 and 2). The scatter plot and CEAC indicated that in the substantial majority (81% and 99%, respectively, at the WTP thresholds of SGD 45,000 (USD 33,087) per QALY and SGD 114,165 (USD 83,941) per QALY gained) of simulations, AHCL would be considered cost-effective. The CEAC indicated that AHCL would already be considered a cost-effective treatment when the threshold was above SGD 32,740/QALY (USD 24,072/QALY).
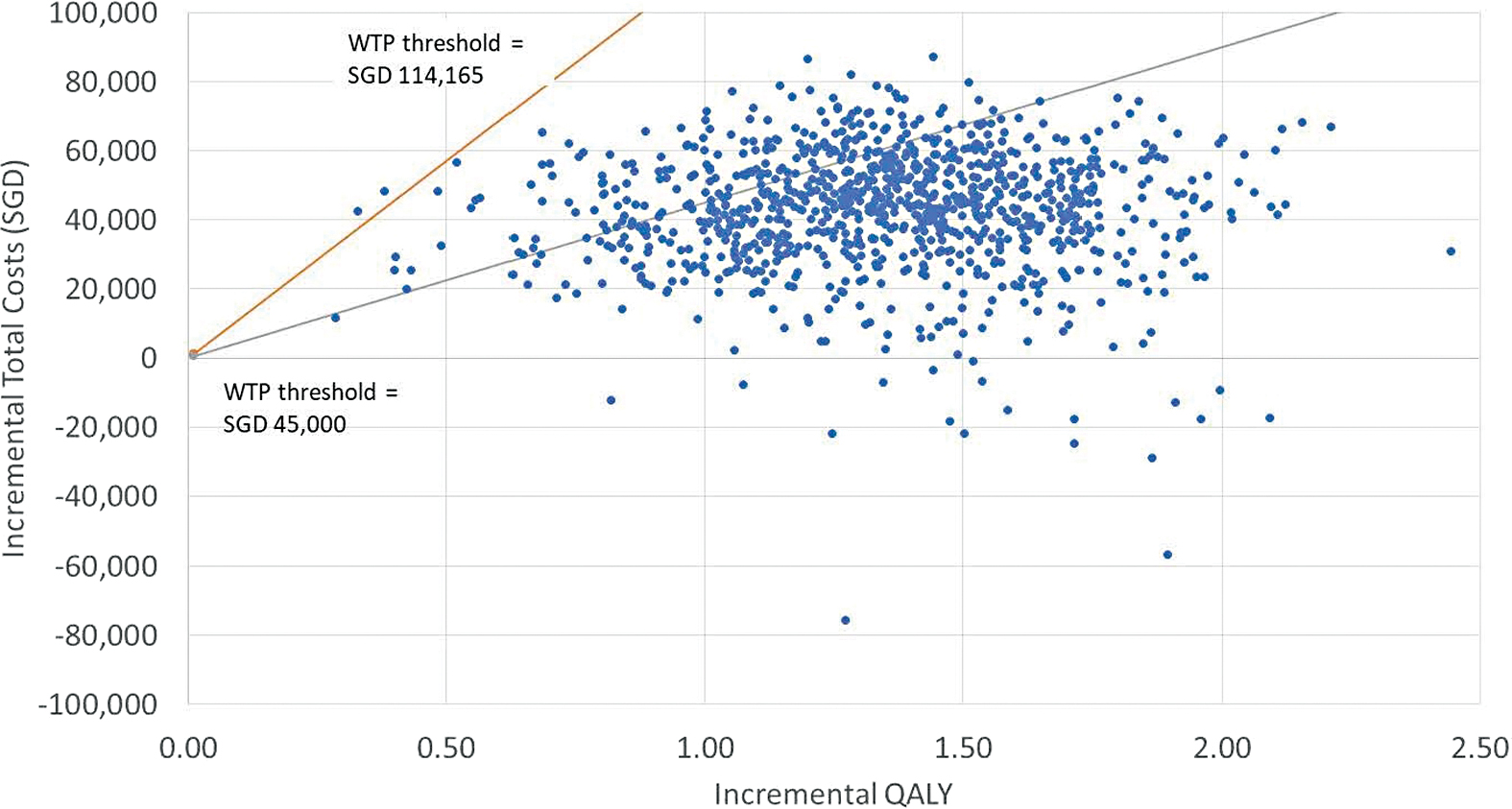
Probabilistic sensitivity analysis of CEA comparing AHCL versus MDI plus isCGM at WTP of SGD 45,000 per QALY (gray line) and SGD 114,165 per QALY (orange line) gained. AHCL, advanced hybrid closed loop; CEA, cost-effectiveness analysis; MDI, multiple daily injections; QALY, quality-adjusted life year; SGD, Singapore Dollars; WTP, willingness-to-pay. Color images are available online.
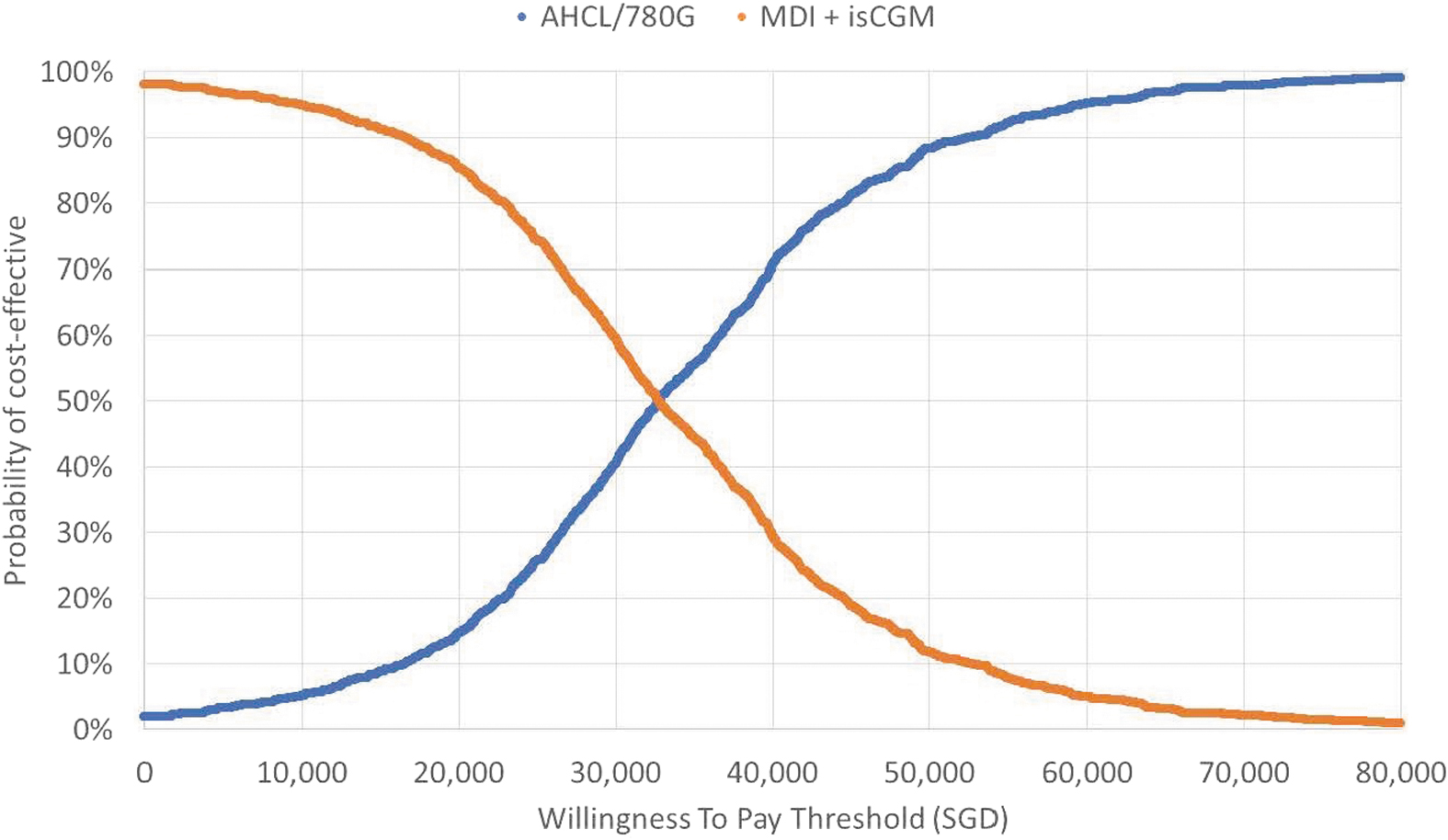
Cost-effectiveness acceptability curve comparing AHCL versus MDI plus isCGM. Cost-effectiveness was achieved at SGD 32,740/QALY (USD 24,072/QALY), where the lines cross each other. Where the line crosses for each therapy/system, is where the cost-effectiveness of one system (in this case the 780G) surpasses the comparator (MDI+isCGM). CEAC, cost-effectiveness acceptability curve; USD, United States Dollars. Color images are available online.
Discussion
Our analysis found that in people in Singapore living with T1D, who are not achieving glycemic targets with isCGM and MDI, the use of the MiniMed 780G system was projected to lead to an incremental gain in QALY expectancy and quality of life. The reductions in the occurrence and cost of T1D complications partially offset the additional management costs of AHCL compared to MDI and isCGM, resulting in an estimated ICER of SGD 33,797 (USD 24,850) per QALY gained. Model outcomes were robust to exploration of uncertainty and AHCL remained a cost-effective management choice when inputs were altered within plausible limits. Based on the two inferred thresholds of WTP, and under current model assumptions, AHCL is a cost-effective management option for similar cohorts of T1D individuals managed in a Singapore setting.
Extensive sensitivity analyses were conducted. Model outcomes were most sensitive to the exploration of a lower treatment effect, which is expected. 24 The difference between HbA1c improvements seen in the ADAPT study compared to other studies reflects the fact that the participants in ADAPT were specifically selected to reflect a cohort on MDI with high HbA1c, despite isCGM use, mapping well to the demographics of the SGH cohort from which baseline characteristics were modeled. 52,57 As reported by Choudhary et al., 19,24 these patients represent an underserved section of the T1D population with a high capacity to benefit from improved outcomes with advanced technology. In assuming a lower baseline HbA1c of 8.0% with a lower treatment effect of −1.0%, the ICER remains comfortably under WTP thresholds.
The baseline assumptions for SHE and DKA for the MDI plus isCGM were estimated using data from large observational studies with a long follow-up duration of 12 months. 38,39 Sensitivity analysis found that outcomes were not sensitive to alternate estimates of SHE and DKA rates obtained from the SDR database, which generated ICERs of SGD 35,686 (USD 26,238) per QALY and SGD 29,886 (USD 21,974) per QALY, respectively. An exploration of lower SHE2 event rate in the AHCL arm resulted in an ICER of SGD 27,906 (USD 20,518) per QALY gained. Future evidence generation could focus on a prospective assessment of rates of SHE and DKA in a Singapore setting over longer periods to better reflect a lifetime perspective.
The inclusion of indirect costs (limited to the costs of DOW due to T1D complications) resulted in an estimated ICER of SGD 18,575 (USD 13,657) per QALY gained. While this metric is not explicitly included in current reimbursement decisions in Singapore, recent moves toward the inclusion of patient perspectives and an emphasis on opportunities for involvement of people with T1D in ACE's decision making 58 suggest that metrics such as DOW may start to play an increasing role in reflecting values that matter to people beyond the health care perspective.
It should also be noted that DOW estimation herein was limited to just inpatient episodes 40 and is hence a conservative estimate, warranting additional exploration. The Singapore health care financing system is anchored on the dual philosophies of individual responsibility and affordable health care for all, 59 and reductions in DOW are likely to benefit not only the individual but also society as a whole through enhancement of overall population-level productivity. The sensitivity analysis using 12-month data from ADAPT on FOH utility decrement also resulted in an ICER of SGD 26,010 (USD 19,124). This metric was within the implied thresholds of WTP and was robust to changes.
Costs of insulin treatment clearly influenced model outcomes. The base case included the insulin cost, which is subject to substantial group procurement discounts and is categorized under the Singapore standard drug list, and therefore eligible for subsidy and reimbursement. In those who are not eligible for subsidized health care within the Singapore setting, the retail costs of insulin are substantially higher by approximately two to five times. In this scenario, the differing cost in insulin pricing alone resulted in a lowering of the ICER to SGD 23,349 (USD 17,168) per QALY gained. This adds a further consideration for payers when evaluating the cost-effectiveness of AHCL versus MDI and isCGM therapy.
Previous analyses assessing the cost-effectiveness of AHCL (specifically, MM780G) are not directly comparable to this analysis as we specifically focused our analysis on people with high HbA1c. However, despite cohort differences, our findings are similar; a study conducted in Greece reported a direct cost ICER of EUR 29,869 (∼USD 31,028) based on a treatment effect of −0.8% HbA1c, 27 while a study conducted in Sweden reported direct and indirect ICERS of SEK 430,663 and SEK 373,700, respectively (∼USD 40,822 and USD 35,425), based on a treatment effect of −0.5% HbA1c. 28
In both cases, the authors concluded that AHCL was a cost-effective alternative to MDI plus isCGM 27 or CSII. 28 Evidence of cost-effectiveness is a key driver of national-level decision making around health care funding, and reimbursement of AHCL is currently expanding across European markets as the evidence base for use increases. 60 For example, in June 2023, The National Institute for Health and Care Excellence (NICE) in United Kingdom recommended the use of HCL systems for T1D. 61 In Singapore and neighboring countries, where the costs of CGM and AHCL systems are currently borne entirely by the person with T1D, due consideration could be given to increase access to these systems through reimbursement schemes, especially when the inclusion of indirect costs of complications is factored in.
There are some limitations to this analysis. Those which relate specifically to the use of long-term modeling in diabetes have been extensively addressed elsewhere, 62,63 and will not be addressed further here. The baseline estimates for the rates of SHE and DKA were based on disparate , which were assessed to be the best available evidence derived from large-scale studies with long follow-up periods. We have, however, applied a conservative approach to the relative benefit of AHCL and explored the impact of our baseline assumptions in sensitivity analysis.
We also note potential limitations of the ADAPT dataset concerning its generalizability to a Singapore setting. The ADAPT study was conducted across a range of European settings and did not include a significant proportion of Asian patients. 18 However, the study represented a robust head-to-head comparison of AHCL and MDI plus isCGM and represents the best available data for this patient population at the time of publication of this analysis. While future research could usefully generate data specific to regionally relevant populations, the use of ADAPT data, in combination with local costs, has allowed us to explore a robust and localized assessment of the cost-effectiveness of AHCL in a Singapore setting.
This study has attempted to make the most of available head-to-head data within the framework of an established and fully validated economic model and using, where possible, local input to tailor the model inputs to a Singapore T1D management setting. It is, however, useful to remind ourselves and emphasize the limitations of a CEA approach in capturing other “off-model” benefits of such AHCL systems, including the impact on self-care burden, the simplification of management complexity through the use of technology, user preferences for ease of management, and patient and carer anxiety associated with the potential for subsequent hypoglycemia events (as evidenced by improved hypoglycemia survey scores). 19,20,64 –67
The MM780G system addresses important unmet needs in T1D management, and the provision of a compelling economic argument for use is key to payer acceptance. Despite the limitations and caveats to the use of CEA, we provide here an analysis based on the iterative and collaborative involvement of local clinicians, using a robust and well-validated model, and providing strong support for the positive positioning of MM780G in the T1D management pathway in Singapore.
Conclusion
This is the first CEA of the MM780 AHCL system in Asia, which provides valuable insights on the system's clinical and economic values to inform health care resource allocation and technology adoption. The analysis projected the expected lifetime costs and benefits of alternate management strategies for a cohort of people with T1D, not currently achieving target glycemia on MDI plus isCGM, and estimated an ICER of SGD 33,797 (USD 24,850) per QALY gained. From this, we can conclude that MM780G is a cost-effective option for people with T1D, suboptimally managed in a Singapore setting. Future research collating and reviewing locally generated data would further support robust policy decisions in this area.
Footnotes
Acknowledgments
We would like to thank Julia Lowin for medical writing and advisory support, and Samuel Goh for medical writing support.
Authors' Contributions
All authors contributed equally toward the conception, design, conduct, analysis, critical review of the study, and article writing.
Author Disclosure Statement
D.G. and S.R.C. have served as advisory panels for Medtronic. M.L., Z.Q., Y.I., and K.W. are employees of Medtronic.
Funding Information
This study was funded by Medtronic.
Supplementary Material
Supplementary Table S1
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
